Summary
Bryophytes, and in particular the mosses, Physcomitrella patens and Ceratodon purpureus have been developed for the genetic study of development and metabolism. Their ease of culture and the haploidy of their gametophyte stage, allow essentially microbiological techniques to be used. The discovery that gene targeting occurs at high frequency in both species, allows genes to be inactivated or modified, providing a very powerful tool for the analysis of gene function. The sequence of the P. patens genome is available and sequencing of the C. purpureus genome is in hand. The full potential of these experimental systems is now being realized for the study of photosynthesis.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Root Hair Development
- Selection Cassette
- Marchantia Polymorpha
- Ceratodon Purpureus
- Efficient Gene Target
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
I. Introduction
Bryophytes were some of the earliest species to be used for genetic analysis in the post-Mendel era. Using natural variants and inter-specific crosses, von Wettstein analysed crosses and carried out some of the pioneering analyses to establish the basis of Mendelian genetics (reviewed in von Wettstein 1932). Bryophytes, both mosses and liverworts, were among the first species in which the utility of mutagenic treatments, both irradiation and chemicals, was established (Knapp 1935; Barthelmess 1953). Their utility for the genetic analysis of metabolism and development was also recognized early (Barthelmess 1941a). These studies had used a number of species, with Marchantia polymorpha and Physcomitrium pyriforme becoming preferred species. However, as a result of encouragement by Harold Whitehouse, an outstanding geneticist and enthusiastic “amateur” bryologist, Physcomitrella patens was used for mutant isolation and genetic analysis by Paulinus Engel (1968). Subsequently genetic methodology was extended and used for studying auxin and cytokinin action using chemically-induced mutants of P. patens (Ashton et al. 1979). Funaria hygrometrica continued to be used widely for physiological investigations, but the development of genetic technology for P. patens, including transformation, led to a steady expansion in the P. patens community. The discovery that in P. patens, transforming DNA containing genomic sequence was targeted to the corresponding genomic sequence at high frequencies, comparable to that in Saccharomyces cerevisiae, led to it becoming the preferred species for genetic studies and its choice as the first non-seed land plant to have its genome sequenced. Gene targeting has also been shown to occur in Ceratodon purpureus and genome sequencing of this species is now in progress. An application to sequence a Sphagnum genome is also in hand.
II. Propagation
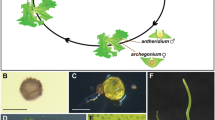
A. Life Cycle
In common with all bryophytes, the dominant stage of the life cycle of P. patens is the haploid gametophyte. Haploid spores germinate to produce protonemata, cell filaments growing by the serial division of the apical cell. Protonemal sub-apical cells can also divide to allow branching of the filament, or to give rise to gametophores, the leafy shoot that produce gametes by mitosis. P. patens is monoicous, producing both sperm and eggs on the same gametophore. Self fertilisation is common. The sporophytes produced are therefore homozygous, allowing the production of large numbers of genetically-identical haploid spores (following meiosis). Cross fertilization does occur, and recently a technique has been devised that identifies hybrid sporophytes, using fluorescent markers (Perroud et al. 2011). The haploidy of the gametophyte has considerable technical advantages; for example, much research has been carried out on a single genotype, Gransden, the strain, originally isolated by Harold Whitehouse from a single spore, and it was this strain that was chosen for genome sequencing. Haploidy removes the need for inbred lines and allows the identification of mutant phenotypes directly, following mutagenesis of either spore or protonemally-derived protoplasts. C. purpureus, in contrast, has separate male and female gametophytes (i.e., is dioicous), and currently a male and a female genome is being sequenced. Although, in this species, spores do not represent a uniform population of identical cells for mutagenesis, this is readily obtainable by way of protoplasting protonemal tissue.
B. Culture
P. patens, and many other moss species, are easily cultured on a simple medium containing only inorganic salts. Protonemal growth occurs on the surface of media solidified with agar, but culture in liquid media is also possible. As culture employs essentially microbiological techniques, it is not surprising that vitamin auxotrophs were among the first artificially-induced mutants to be isolated (Engel 1968; Ashton and Cove 1977). By overlaying solid medium with sterile cellophane, and inoculating with blended protonemal tissue, it is possible to obtain within 7 days from 90 mm Petri dish, a uniform mat of protonemal tissue weighing about 30 mg dry weight (200 mg fresh weight). This tissue may be used for the extraction of DNA, RNA or protein, and for the enzymatic isolation of protoplasts for mutagenesis or transformation. Such protonemal mats may also be used for biolistic transformation.
If greater amounts of protonemal tissue are required, growth in liquid media may be employed. Batch culture was used originally (Wang et al. 1980), but stirred or airlift bioreactors have since been developed (Boyd et al. 1988a, b; Hohe et al. 2002; Decker and Reski 2007). Growth rates are enhanced by passing an air/CO2 mix through the medium. In batch culture, development proceeds in a similar manner to growth on solid medium, but if the culture is diluted so that the tissue density remains more or less constant, development does not proceed beyond the protonemata. In the air lift bioreactor, the tissue is passed through a tissue homogeniser, restricting the size of tissue fragments to a maximum diameter of about 100 μm. Because of the strong regenerative capacity of protonemal tissue, growth is continuous and about 4 g dry weight (about 30 g fresh weight) can be harvested daily from a 6 l bioreactor (DJ Cove, unpublished data).
Protocols for most of the experimental manipulations that may be carried out with P. patens can be found in Cove et al. (2009).
C. Strain Storage
The storage of P. patens strains can be carried out in a number of ways. Where spores can be obtained, these can be kept for many years, but vegetative tissue can also be stored. Cryopreservation of protonemal tissue (Grimsley and Withers 1983) allows long term (at least 10 years) of some strains, but recovery from freezing is often unsuccessful. This leads to a need to freeze multiple samples of each strain. Alternatively, tissue can be maintained by storage at 10°–15° in a cycle of 1–2 h bright light per 24 h day. Either agar-grown cultures or harvested protonemal tissue, suspended in sterile distilled water, may be stored. Most strains survive well for at least 2 years when stored in this way C. purpureus can be stored by the same techniques, and is generally more robust than P. patens. We have successfully maintained fresh cultures of C. purpureus at low temperatures for 10 years.
III. Genetic Manipulation
A. Mutagenesis
The haploidy of bryophytes is a considerable advantage for the isolation of mutant strains. Providing the mutant phenotype is not lethal, mutant strains may be identified directly following mutagenesis. Although not yet employed extensively, the mutagenesis of diploid strains should allow the isolation of recessive-lethal mutants.
Successful mutagenesis of P. patens has been carried out using X-ray or UV-irradiation or chemicals to treat spores or protonemal tissue (Engel 1968; Ashton and Cove 1977; Boyd et al. 1988a, b). When protonemal tissue is treated, single-celled propagules can be obtained either by treating isolated protoplast directly or by protoplasting protonemal tissue after mutagenesis. Since C. purpureus, having separate sexes, is an obligate out-crosser, uniform samples of spores are not available, however somatic mutagenesis can readily be employed (Cove and Quatrano 2006). X-rays have been used to induce mutation in a number of other bryophytes including Brachythecium rutabulum (Moutschen 1954), Marchantia polymorpha (Miller et al. 1962), Physcomitrium pyriforme (Barthelmess 1941b) and Sphaero-carpus donnellii (Knapp 1935; Schieder 1973).
B. Sexual Crossing
P. patens produces male and female gametangia on the same gametophore and so self-fertilization is possible and usual. Co-inoculation of two strains to be crossed, allows hybrid sporophytes to be produced, but these may be rare. However, early studies established that pure cultures of vitamin-requiring strains were unable to produce sporophytes unless vitamin supplementation was greatly increased. However, when two different and complementary vitamin-requiring mutants were co-inoculated and grown with the standard (low) level of vitamin supplementation, sporophytes were produced, all of which were hybrid (Courtice et al. 1978). The use of strains having vitamin auxotrophies is not always convenient, so recently an alternative method of identifying hybrid sporophytes has been devised. This uses transgenic strains expressing a fluorescent protein. Since the fluorescent phenotype is dominant, fluorescing sporophytes borne by a non fluorescently-tagged strain, co-inoculated with a tagged strain, must have been produced following fertilization by the tagged strain. Although hybrid sporophytes may be uncommon, they are nevertheless very easily identified (Perroud et al. 2011). C. purpureus, having separate sexes, needs no such technology, all sporophytes being hybrid.
The haploidy of the gametophyte, makes genetic analysis straightforward. “Gametophytic” segregation ratios are observed, i.e., 1:1 segregation for single gene differences (Engel 1968; Ashton and Cove 1977; Courtice et al. 1978), and 1:1:1:1 segregations are observed for two different unlinked genes (Ashton and Cove 1977). Early P. patens crosses detected no linkage but with the limited number of marked genes available and the high chromosome number (27) this was not surprising. With the availability of molecular markers, a detailed linkage map, anchored against the genome sequence, is now available (Kamisugi et al. 2008). A similar map of molecular markers is also available for C. purpureus (McDaniel et al. 2007) and it should be relatable to the genome sequence, once this is available. This study involved crosses between geographically widely separated strains and marked deviations from expected 1:1 segregation ratios were detected. Some formal genetic analysis has also been carried out in Spaerocarpus donnellii (Schieder 1973).
C. Somatic Hybridisation
Although the haploidy of the gametophyte is usually seen as an advantage for genetic studies, it does not allow allelic dominance to be studied directly. The sexual sterility of some developmental mutants is also a bar to their conventional genetic analysis. Both these problems can be overcome by the use of somatic hybridisation. Early studies obtained hybrid diploid gametophytes by aposporous regeneration of sporophyte tissue (von Wettstein 1932), but experimental selection of hybrid gametophytes is possible by the use of protoplast fusion. This was first demonstrated for Sphaerocarpus donnellii (Schieder 1974) and P. patens (Grimsley et al. 1977a). Somatic hybrids allow not only dominance relationships to be established but also allow complementation to be analysed (Grimsley et al. 1977b). Complementation analysis by way of somatic hybrid production is especially valuable in C. purpureus, where hybrids can be made between two mutant strains of the same sex (Cove and Quatrano 2006).
D. Transformation
Genetic transformation of P. patens, was first achieved by the treatment of isolated protoplasts with naked DNA in the presence of polyethylene-glycol (Schaefer et al. 1991), using a technique based on the transformation of angiosperm mesophyll protoplasts. This technique has remained essentially unchanged and is also effective for the transformation of C. purpureus. A number of different selection cassettes are available, but geneticin- (G418) and hygromycin-resistance genes driven by a 35S promoter are most commonly used. Two classes of transformants are obtained. The most frequent only retains the transforming DNA sequence and the transformed phenotype, provided selection is maintained. Subculture onto, for example antibiotic-free medium, leads to loss of the transforming DNA. However, providing selection is maintained, the transforming DNA is replicated. The second class of transformant is stable and retains the transformed phenotype in the absence of selection. In this class, the transformed DNA is integrated into the genome and is inherited in a Mendelian manner. DNA containing no sequence homologous to genomic sequence is integrated randomly, but where the transforming DNA contains genomic sequence, integration is targeted to the corresponding genomic sequence (Schaefer and Zryd 1997).
E. Gene Targeting
The ability to deliver transforming DNA to a predetermined genetic locus provides a powerful tool for the analysis of gene function. Furthermore, this property is rare in eukaryotes, being straightforward only in the yeast, Saccharomyces cerevisiae, in which relatively short oligonucleotides can be delivered to cells and subsequently integrated at target loci by homologous recombination. Other eukaryotic systems in which gene targeting is routinely used include the chicken DT40 cell line (a terminally differentiated and cancerous B-cell-derived line in which the pathway for generation of antibody diversity by gene conversion is permanently switched on), and mouse embryonic stem cells, which can be transformed and regenerated, ultimately to generate “knockout mice” that can be used as models for the understanding of human inherited disorders. However, the distinguishing feature of targeted transgene integration in P. patens is its efficiency, which is several orders of magnitude greater than that in mouse ES cells: in some experiments, 100 % of transgenic moss plants have been found to have integrated the transgene at the designated locus (Kamisugi et al. 2005).
The parameters for efficient gene targeting in P. patens have been determined, and the principle is indicated in Fig. 11.1 Briefly, two targeting sequences, corresponding to the 5′- and 3′-ends of the gene to be targeted, are cloned on either side of a selectable marker cassette. The length of the targeting sequences (i.e. the extent of homology between the transforming DNA and the target gene) is the most crucial factor, with as little as 500 bp on either side of the selection cassette being sufficient to achieve a targeting frequency of 50 % (Kamisugi et al. 2005). For maximum efficiency, the construct is delivered as a linear fragment, either by recovery from the original cloning vector by restriction enzyme digestion, or, more conveniently, by PCR amplification of the targeting sequence. In a successful targeting experiment, the central sequence in the targeted gene is replaced by the selection cassette. The creation of multiply mutated strains by transformation with additional targeting cassettes can be achieved if the selection marker can be removed from the moss genome following targeted gene replacement. This is possible using an initial targeting vector in which the selection cassette is flanked by the bacteriophage lambda loxP sites. In this case, the selection cassette can be removed from the targeted locus by transient expression of the Cre recombinase gene, thereby allowing a further round of transformation and selection (Trouiller et al. 2006).
Targeted transformation in Physcomitrella (a): Targeted gene replacement. A fragment of transforming DNA comprising a selection cassette flanked by ca.500–800 bp of sequence derived from the 5′- and 3′-ends of the gene sequence to be replaced is delivered to Physcomitrella protoplasts by polyethylene glycol-mediated transfection. Homologous recombination between each end of the construct and the corresponding sequence of the target gene results in replacement of the endogenous sequence. (b): Targeted gene deletion. In this instance, the transforming DNA contains two direct repeats of the LoxP recombination sequence from bacteriophage lambda on either side of the selection cassette. Following the selection and regeneration of transformants in which targeted gene replacement has occurred, delivery of an expression construct to protoplasts with subsequent transient expression of the Cre recombinase results in removal of the selection cassette, by recombination between the two LoxP sites. The mutant strain can now be re-transformed with another targeting cassette containing the original selection marker, to create multiply targeted strains.
It is probable that other moss species share the ability to be mutagenised by gene targeting. Certainly, this has been achieved in Ceratodon purpureus: Brucker et al. (2005) generated specific point mutations in the C. purpureus heme oxygenase gene by delivery of linear fragments that had been specifically altered in a single base by site-directed mutagenesis. The targeted transformants were identified and selected by virtue of their acquisition of a mutant phenotype: the inability to respond phototropically to unilateral red light. Trouiller et al. (2007) compared targeting efficiencies in P. patens and C. purpureus and found the targeting frequency in the latter to be ca. 10-fold less efficient than in P. patens, but still two orders greater than that achievable in flowering plants.
The realisation that efficient gene targeting was possible in P. patens led to a significant burgeoning of interest in the use of this organism as a vehicle for gene function analysis. In particular, the evolutionary distance between the mosses and flowering plants (ca. 200 million years between the respective divergences of the bryophyte and angiosperm lineages) suggested that Physcomitrella might be most valuable as a model in which to study the evolution of gene function within the plant kingdom. However, in order to realize its potential, it was clearly important to develop resources that would enable any Physcomitrella gene to be cloned and manipulated without delay. The initiation of the Physcomitrella Genome Project met this requirement.
IV. Genomic Data and Applications
A. Genome Sequence
The first development of Physcomitrella genomic resources took the form of EST-based gene discovery projects in Freiburg, supported by substantial industrial funding from BASF, in Okazaki and in Leeds and St. Louis. Subsequently, at the annual moss conference, “Moss 2004” in Freiburg, a consortium was assembled to undertake the sequencing of the Physcomitrella genome, through the auspices of the Community Sequencing Program of the United States Department of the Energy’s Joint Genome Institute. Co-ordinated by Brent Mishler and Ralph Quatrano, the proposal was successful, and the sequencing of the genome commenced in 2005.
Using a whole-genome shotgun sequencing approach, the first genome assembly was completed in 2006 and made available through the Joint Genome Institute website (http://genome.jgi-psf.org/Phypa1_1/Phypa1_1.home.html). The salient features of the genome were published early in 2008, following a community-wide gene curation and annotation effort (Rensing et al. 2008).
The “Version 1” genome assembly comprised nearly 2000 sequence scaffolds, corresponding to a sequence length of 480 Mbp. This was in good accord with previous genome size estimates based on flow cytometry of 511 Mbp (Schween et al. 2003), and is about 3× larger than the Arabidopsis. As is the case in most multicellular eukaryotes, the genome size is correlated with the proportion of repetitive DNA, rather than the number of genes, and although the initial annotation indicated a gene content of nearly 40,000 genes, subsequent annotations reported in the “Cosmoss” database (http://www.cosmoss.org) and replicated through the “Phytozome” resource (http://www.phytozome.net/physcomitrella) have reduced this number toca. 32,000 protein coding loci.
Improvements to the genome assembly are continuing as additional genomic resources are developed. Importantly, the US Department of Energy has recognized the importance of the Physcomitrella genome in a commitment by the JGI to develop the genomic resources as one of its “flagship” plant genomes. Further enhancements to the genome assembly are expected to be released within the calendar year 2013, as a result of the integration of genetic linkage data in to the scaffold ordering process. Until recently, no genetic linkage map was available for P. patens, rendering “forward genetic” identification of the sequences underpinning mutant phenotypes problematic. However, a sequence-anchored linkage map based on molecular markers (Amplified Fragment Length Polymorphisms and Simple Sequence Repeats) was developed in 2008 (Kamisugi et al. 2008) and the development of a chromosome-scale genome assembly is being made possible through the high-density genotyping of SNP markers. These rapid improvements are being underpinned by next-generation sequencing technologies: data reported at the “Moss 2011” conference described the identification of nearly 600,000 SNPs between the standard “Gransden” laboratory strain and the genetically divergent “Villersexel” strain, collected by Michael Lüth, from the Haute Saône region of South-East France. This represents a SNP density of approximately 1/800 bp: a rich resource for linkage mapping (T Nishiyama, personal communication).
In addition to the nuclear genome, both organellar genomes have also been sequenced (Sugiura et al. 2003; Terasawa et al. 2007), as have those of a number of basal land plants, including the liverwort, Marchantia polymorpha (Ohyama et al. 1986; Oda et al. 1992) – the first plant chloroplast genome to be completely sequenced – and the hornwort, Anthoceros formosa (Kugita et al. 2003), together with organelle genomes from the charophyte algae (Turmel et al. 2002, 2006): a group believed to represent the immediate aquatic ancestors of the land plants.
With the rapid advance in genome sequencing technologies, Physcomitrella is no longer the only bryophyte with a genome sequencing programme. The genome sequence of the liverwort, Marchantia polymorpha, is currently being annotated prior to release by a JGI consortium led by John Bowman (Monash University). This will substantially enhance the potential for investigating the roots of the land plant lineage, since M. polymorpha resources include an efficient Agrobacterium-mediated transformation system (although without the ability to undertake gene targeting, as in mosses) and the potential to identify mutants through genetic map-based cloning methodologies. Additionally programmes to sequence the genomes of Ceratodon purpureus and Sphagnum are in progress, whilst EST collections are available for a number of aquatic algae (including Chara, Coleochaete and Spirogyra – Timme and Delwiche 2010).
B. Genomics
What questions have the availability of a moss genome sequence allowed us to address? Principally, the interesting questions are evolutionary. How has the form and function of land plants evolved, and what processes led to the domination of terrestrial ecosystems by green plants? – an event of fundamental importance to the natural history of our planet.
One of the defining features of plant evolution, in general, is the propensity for polyploidization to act as an engine for evolutionary change, by enabling immediate reproductive isolation. Evidence of polyploidy is widespread throughout the Plant Kingdom, and Physcomitrella is not excepted from this. Analysis of the genome sequence enabled the identification of a large-scale (probably whole-genome) duplication having occurred relatively recently in its evolutionary history (between 30 and 60 MYA). Originally predicted from the similarity of paralagous gene pairs identified in the EST collection (Rensing et al. 2007), the remnants of this event can be found in the genome assembly where syntenic regions of conserved genes are found interspersed with additions or deletions (Fig. 11.2)
Example of internal synteny within the Physcomitrella genome. This shows two screenshots from the JGI Physcomitrella genome browser, corresponding to ca. 300-kilobase regions in sequence scaffolds 181 (top) and 282 (bottom), respectively. Genes duplicated and in a syntenic relationship to one another are circled and identified by underlined annotations: “BiP” (ER-chaperone) “FZL” (Mitochondrial GTPase) CBF (Histone fold transcription factor CBF), “SPI” (serine proteinase inhibitor), “E3” (E3 ubiquitin ligase), “SnRNP” (Splicing factor). The intervening genes differ between the two scaffolds.
Since modern bryophytes are the direct descendants of the first plants to colonize terrestrial habitats, it is likely that a number of Physcomitrella genes encode “primitive” traits that were advantageous for this step-change in plant evolution. Comparative functional analysis of genes between Physcomitrella and angiosperms can highlight some of these traits.
A particularly elegant example of this derives from the identification of two Physcomitrella orthologs of the Arabidopsis gene, RDH6, encoding a transcription factor regulating root hair development. Targeted knockout of the Physcomitrella genes demonstrated that their function was to regulate rhizoid formation, and the conservation of a biological function necessary for interaction with the substratum was further demonstrated by the ability of one of the moss genes to cross-complement a mutant rdh6 allele and restore root hair development in A. thaliana (Menand et al. 2007).
The first plants to colonise the land must necessarily have developed adaptations to resist a wide range of abiotic stresses associated with the terrestrial environment. Chief among these are resistance to drought and enhanced radiation, and genes known to be required for resistance to both stresses in flowering plants play similar functions in Physcomitrella. Thus, the genes encoding the ABA-mediated drought stress response are present in the moss genome, and regulate a similar suite of responses as are seen in angiosperms. This includes the ABA-activated closure of stomata (present on the moss sporophyte) (Chater et al. 2011), ABA signalling using the ABI1-like protein phosphatases (Komatsu et al. 2009) and the transcriptional activation of LEA gene expression by ABA (Cuming et al. 2007) through the activity of ABI3 transcription factors (Khandelwal et al. 2010) – a function restricted to seed development in angiosperms. Vascular plants possess only a single ABI3 gene, whereas in Physcomitrella there are three ABI3 orthologs with redundant function in the initiation of the drought response. It is suggested that evolutionary loss of the additional copies in the vascular plants led to the sequestration of ABA-mediated desiccation tolerance in the seed development pathway (Rensing et al. 2008).
By contrast with the conservation of the ABA signalling pathway, the status of the gibberellin signalling pathway is less clearly defined. The Physcomitrella genome contains genes encoding enzymes of the gibberellin biosynthetic pathway (ent-kaurene synthetase, and GA20- and GA3-oxidases). It also contains genes encoding the vascular plant GA receptor (GID) protein and its interacting DELLA growth-repressive factor, but whilst knockout mutants of the ent-kaurene synthetase gene produce an abnormal protonemal phenotype (Hayashi et al. 2010), it is unclear whether this can be ascribed to a gibberellin-like activity, at least so far as this is understood in vascular plants. Analyses of the Physcomitrella GID-DELLA interaction and its participation in GA signalling indicate that the PpGID protein does not bind gibberellins, and that while the GID-DELLA interaction is fully functional in the lycophyte Selaginella, this is not the case in Physcomitrella, indicating that this hormone signalling pathway evolved subsequent to the divergence of the bryophytes, and is characteristic of the vascular plant lineages (Hirano et al. 2007; Yasumura et al. 2007).
V. Potential for Photosynthetic Studies
The availability of genomic tools and resources for mosses, together with the ease with which the model species can be grown and manipulated in culture provides new opportunities for photosynthetic research. Thus, whilst the parameters of photosynthesis established in both Physcomitrella and Ceratodon appear essentially similar to those in vascular plants, the ability to undertake a genetic analysis of these processes – either through “forward genetic” mutagenesis or “reverse genetic” approaches – offers considerable promise. Because Ceratodon is more amenable to heterotrophic growth than P. patens, this species has been used to generate a large number of mutants (via UV-irradiation) that are deficient in photosynthetic processes, exhibiting characteristic alterations in chlorophyll fluorescence, and in chlorophyll synthesis and accumulation (Thornton et al. 2005). The development of genomic resources for this species over the next 2 years should enable a map-based cloning pipeline to be established by which such mutants can be characterised.
The availability of genomic resources in Physcomitrella is already being exploited for photosynthetic studies. A bioinformatic interrogation of the photosynthetic gene set has been used to to determine the evolution of the photosynthetic process during the colonisation of land by green plants (Alboresi et al. 2008), and the capacity for gene targeting has enabled the interrogation of components of the photosynthetic light harvesting and electron transport chain under varying environmental conditions (Alboresi et al. 2010, 2011; Gerotto et al. 2011).
Other aspects of chloroplast function addressed in Physcomitrella patens include the mechanisms of chloroplast division and of protein import into plastids. Indeed, the very first mutant phenotype to be obtained by gene targeting in P. patens was the formation of giant chloroplasts, that resulted from the disruption of the FtsZ gene (Strepp et al. 1998) – an ancient member of the tubulin family, subsequently shown to form cytoskeletal networks in chloroplasts (KIessling et al. 2000). Both plastid and cytosolic isoforms of FtsZ exist, and both isoforms are required for the correct specification of plastid division (Gremillon et al. 2007).
Import of proteins by plastids – an essential process in the establishment of the original endosymbiotic event that gave rise to the green plants – is (unsurprisingly) conserved between moss and angiosperms (Hoffman and Theg 2003), suggesting that the targeted mutagenesis of moss import proteins could provide a powerful means of identifying key residues in the protein uptake polypeptides.
In summary, the existing and future genomic resources available for the molecular analysis of life processes in bryophytes will aid us in the unravelling of the complexity of the evolution of gene function throughout the Plant Kingdom.
References
Albanese A, Caffarri S, Nogué F, Bassi R, Morosinotto T (2008) In silico and biochemical analysis of Physcomitrella patens photosynthetic antenna: identification of subunits which evolved upon land adaptation. PLoS ONE 3:e2033
Alboresi A, Gerotto C, Giacometti GM, Bassi R, Morosinotto T (2010) Physcomitrella patens mutants affected on heat dissipation clarify the evolution of photoprotection mechanisms upon land colonization. Proc Natl Acad Sci U S A 107:11128–11133
Alboresi A, Gerotto C, Cazzaniga S, Bassi R, Morosinotto T (2011) A red shifted antenna protein associated to photosystem II in Physcomitrella patens. J Biol Chem 286:28978–28987
Ashton NW, Cove DJ (1977) The isolation and preliminary characterisation of auxotrophic mutants of the moss, Physcomitrella patens. Mol Gen Genet 154:87–95
Ashton NW, Grimsley NH, Cove DJ (1979) Analysis of gametophytic development in the moss, Physcomitrella patens, using auxin and cytokinin resistant mutants. Planta 144:427–435
Barthelmess A (1941a) Über Beziehungen zwischen Genetik, Systematik, Morphologie und Entwicklungsphysiologie und die Möglichkeit einer Zusammenarbeit. Zeitschrift für die gesamte Naturwissenschaften H3(4):84–92
Barthelmess A (1941b) Mutationsversuche mit einem Laubmoos Physcomitrium piriforme II. Morphologische und physiologische analyse der univalenten und bivalenten protonemen einiger mutanten. Zeitschrift für induktive Abstammungs- und Vererbunglehre 79:153–170
Barthelmess A (1953) Mutationsauslösung mit chemikalien. Naturwissenschaften 22:583–584
Boyd PJ, Grimsley NH, Cove DJ (1988a) Somatic mutagenesis of the moss, Physcomitrella patens. Mol Gen Genet 211:545–546
Boyd PJ, Hall J, Cove DJ (1988b) An airlift fermenter for the culture of the moss Physcomitrella patens. In: Glime JM (ed) Methods in bryology. Proceedings of Bryological Methods Workshop, Mainz. Hattori Botanical Laboratory, Nichinan, pp 41–45
Brucker G, Mittmann F, Hartmann E, Lamparter T (2005) Targeted site-directed mutagenesis of a heme oxygenase locus by gene replacement in the moss Ceratodon purpureus. Planta 220:864–874
Chater C, Kamisugi Y, Movahedi M, Fleming A, Cuming AC, Gray JE, Beerling JD (2011) Regulatory mechanism controlling stomatal behavior conserved across 400 million years of land plant evolution. Curr Biol 21:1025–1029
Courtice GRM, Ashton NW, Cove DJ (1978) Evidence for the restricted passage of metabolites into the sporophyte of the moss, Physcomitrella patens. J Bryol 10:191–198
Cove DJ, Quatrano RS (2006) Agravitropic mutants of the moss, Ceratodon purpureus, do not complement mutants having a reversed gravitropic response. Plant Cell Environ 29:1379–1387
Cove DJ, Perroud P-F, Charron J, McDaniel SF, Khandelwal A, Quatrano RS (2009) The Moss Physcomitrella patens: a novel model system for plant development and genomic studies. In: Behringer RR, Johnson AD, Krumlauf RE (eds) Emerging model organisms: a laboratory manual, vol 1. Cold Spring Harbor Laboratory Press, New York, pp 69–104
Cuming AC, Cho SH, Kamisugi Y, Graham H, Quatrano RS (2007) Microarray analysis of transcriptional responses to abscisic acid and osmotic, salt and drought stress in the moss, Physcomitrella patens. New Phytol 176:275–287
Decker EL, Reski R (2007) Moss bioreactors producing improved biopharmaceuticals. Curr Opin Biotechnol 18:393–398
Engel PP (1968) The induction of biochemical and morphological mutants in the moss Physcomitrella patens. Am J Bot 55:438–446
Gerotto C, Alborese A, Giacometti GM, Bassi R, Morosinotto T (2011) Role of PSBS and LHCSR in Physcomitrella patens acclimation to high light and low temperature. Plant Cell Environ 34:922–932
Gremillon L, Kiessling J, Hause B, Decker EL, Reski R, Sarnighausen E (2007) FtsZ isoforms specifically interact in the chloroplasts and in the cytosol of Physcomitrella patens. New Phytol 176:299–310
Grimsley NH, Withers LA (1983) Cryopreservation in the moss, Physcomitrella patens. Cryo-Lett 4:251–258
Grimsley NH, Ashton NW, Cove DJ (1977a) The production of somatic hybrids by protoplast fusion in the moss, Physcomitrella patens. Mol Gen Genet 154:97–100
Grimsley NH, Ashton NW, Cove DJ (1977b) Complementation analysis of auxotrophic mutants of the moss, Physcomitrella patens, using protoplast fusion. Mol Gen Genet 155:103–107
Hayashi K, Horie K, Hiwatashi Y, Kawaide H, Yamaguchi S, Hanada A, Nakashima T, Nakajima M, Mander LM, Yamane H, Hasebe M, Nozaki H (2010) Endogenous diterpenes derived from ent–kaurene, a common gibberellin precursor, regulate protonemal differentiation of the moss Physcomitrella patens. Plant Physiol 153:1085–1097
Hirano K, Nakajima M, Asano K, Nishiyama T, Sakakibara H, Kojima M, Katoh E, Xiang H, Tanahashi T, Hasebe M, Banks JA, Ashikari M, Kitano H, Ueguchi-Tanaka M, Matusoka M (2007) The GID1-mediated gibberellin perception mechanism is conserved in the lycophyte Selaginella moellendorffii but not in the bryophyte Physcomitrella patens. Plant Cell 19:3058–3079
Hoffman NR, Theg SM (2003) Chloroplast protein transport in the moss, Physcomitrella patens: conserved machineries between vascular and non-vascular plants. Plant Mol Biol 53:643–654
Hohe A, Decker EL, Gorr G, Schween G, Reski R (2002) Tight control of growth and cell differentiation in photoautotrophic growing moss (Physcomitrella patens) bioreactor cultures. Plant Cell Res 20:1135–1140
Kamisugi Y, Cuming AC, Cove DJ (2005) Parameters determining the efficiency of gene targeting in the moss Physcomitrella patens. Nucleic Acids Res 33:e173
Kamisugi Y, von Stackelberg M, Lang D, Care M, Reski R, Rensing SA, Cuming AC (2008) A sequence-anchored genetic linkage map for the moss, Physcomitrella patens. Plant J 56:855–866
Khandelwal A, Cho SH, Marella H, Sakata Y, Perroud P-F, Pan A, Quatrano RS (2010) Role of ABA and ABI3 in desiccation tolerance. Science 327:546
Kiessling J, Kruse S, Rensing SA, Harter K, Decker EL, Reski R (2000) Visualisation of a cytoskeleton-like FtsZ network in chloroplasts. J Cell Biol 151:945–950
Knapp E (1935) Untersuchungen über die Wirkung von Röntgenstrahlen an dem Lebermoos, Sphaerocarpus, mit Hilfe der Tetraden-Analyse. Zeitschrift für induktive Abstammungs- und Vererbungslehre 70:309–349
Komatsu K, Nishikawa Y, Ohtsuka T, Taji T, Quatrano RS, Tanaka S, Sakata Y (2009) Functional analyses of the ABI1-related protein phosphatase type 2C reveal evolutionarily conserved regulation of abscisic acid signaling between Arabidopsis and the moss Physcomitrella patens. Plant Mol Biol 70:327–340
Kugita M, Kaneko A, Yamamoto Y, Takeya Y, Matsumoto T, Yoshinaga K (2003) The complete nucleotide sequence of the hornwort (Anthoceros formosae) chloroplast genome: insight into the earliest land plants. Nucleic Acids Res 31:716–721
McDaniel SF, Willis JH, Shaw J (2007) A linkage map reveals a complex basis for segregation distortion in an interpopulation cross in the moss Ceratodon purpureus. Genetics 176:2489–2500
Menand B, Yi K, Jouannic S, Hoffmann L, Ryan E, Linstead P, Schaefer DG, Dolan L (2007) An ancient mechanism controls the development of cells with a rooting function in land plants. Science 316:1477–1480
Miller MW, Gauber ED, Voth PD (1962) Nutritionally deficient mutants of Marchantia polymorpha induced by X-rays. Bot Gaz 124:94–102
Moutschen J (1954) Contribution a l’étude de la génétique des mousses. Action des rayons X et des rayons gamma. La Cellule 46:181–210
Oda K, Yamato K, Ohta E, Nakamura Y, Takemura M, Nozato N, Akashi K, Kanegae T, Ogura Y, Kohchi T, Ohyama K (1992) Gene organization deduced from the complete sequence of liverwort Marchantia polymorpha mitochondrial DNA: a primitive form of plant mitochondrial genome. J Mol Biol 223:1–7
Ohyama K, Fukuzawa H, Kohchi T, Shirai H, Sano T, Sano S, Umezono K, Shiki Y, Takeuchi M, Chang Z, Aota S-I, Inokuchi H, Ozeki H (1986) Chloroplast gene organization deduced from complete sequence of liverwort Marchantia polymorpha chloroplast DNA. Nature 322:572–574
Perroud P-F, Cove DJ, Quatrano RS, McDaniel SF (2011) An experimental method to facilitate the identification of hybrid sporophytes in the moss Physcomitrella patens using fluorescent tagged lines. New Phytol 191:301–306
Rensing SA, Ick J, Fawcett JA, Lang D, Zimmer A, Van de Peer Y, Reski R (2007) An ancient genome duplication contributed to the abundance of metabolic genes in the moss Physcomitrella patens. BMC Evol Biol 7:130
Rensing SA, Lang D, Zimmer AD, Terry A, Salamov A, Shapiro H, Nishiyama T, Perroud P-F, Lindquist E, Kamisugi Y, Tanahashi T, Sakakibara K, Fujita T, Oishi K, Shin-I T, Kuroki Y, Toyoda A, Suzuki Y, Hashimoto S, Yamaguchi K, Sugano S, Kohara Y, Fujiyama A, Anterola A, Aoki S, Ashton N, Barbazuk WB, Barker E, Bennetzen JL, Blankenship R, Cho SH, Dutcher SK, Estelle M, Fawcett JA, Gundlach H, Hanada K, Heyl A, Hicks KA, Hughes J, Lohr M, Mayer K, Melkozernov A, Murata T, Nelson D, Pils B, Prigge M, Reiss B, Renner T, Rombauts S, Rushton PJ, Sanderfoot A, Schween G, Shiu S, Stueber K, Theodoulou FL, Tu H, Van de Peer Y, Verrier PJ, Waters E, Wood A, Yang L, Cove D, Cuming AC, Hasebe M, Lucas S, Mishler BD, Reski R, Grigoriev IV, Quatrano RS, Boore JL (2008) The genome of the moss Physcomitrella patens reveals evolutionary insights into the conquest of land by plants. Science 319:64–69
Schaefer DG, Zryd J-P (1997) Efficient gene targeting in the moss Physcomitrella patens. Plant J 11:1195–1206
Schaefer DG, Zryd J-P, Knight CD, Cove DJ (1991) Stable transformation of the moss Physcomitrella patens. Mol Gen Genet 226:418–424
Schieder O (1973) Untersuchungen an Nicotinsaüre-auxotrophen Stämmen von Sphaerocarpus donnellii Aust. Zeitschrift für Planzenphysiologie 70:185–189
Schieder O (1974) Selektion einer somatischen Hybride nach Fusion von Protoplasten auxotropher Mutanten von Sphaerocarpus donnellii. Aust Zeitschrift für Planzenphysiologie 74:357–365
Schween G, Gorr G, Hohe A, Reski R (2003) Unique tissue-specific cell cycle in Physcomitrella. Plant Biol 5:50–58
Strepp R, Scholz S, Kruse S, Speth V, Reski R (1998) Plant nuclear gene knockout reveals a role in plastid division for the homolog of the bacterial cell division protein FtsZ, and ancestral tubulin. Proc Natl Acad Sci U S A 95:4368–4373
Sugiura C, Kobayashi Y, Aoki S, Sugita C, Sugita M (2003) Complete chloroplast DNA sequence of the moss Physcomitrella patens: evidence for the loss and relocation of rpoA from the chloroplast to the nucleus. Nucleic Acids Res 31:5324–5331
Terasawa K, Odahara M, Kabeya Y, Kikugawa T, Sekine Y, Fujhiwara M, Sato N (2007) The mitochondrial genome of the moss Physcomitrella patens sheds new light on mitochondrial evolution in land plants. Mol Biol Evol 24:699–709
Thornton LE, Keren N, Ohad I, Pakrasi HB (2005) Physcomitrella patens and Ceratodon purpureus, mosses as model organisms in photosynthesis studies. Photosynth Res 83:87–96
Timme RE, Delwiche CF (2010) Uncovering the evolutionary origin of plant molecular processes: comparison of Coleochaete (Coleochaetales) and Spirogyra (Zygnematales) transcriptomes. BMB Plant Biol 10:96
Trouiller B, Schaefer DG, Charlot F, Nogué F (2006) MSH2 is essential for the preservation of genome integrity and prevents homeologous recombination in the moss Physcomitrella patens. Nucleic Acids Res 34:232–242
Trouiller B, Charlot F, Choinard S, Schaefer DG, Nogué F (2007) Comparison of gene targeting efficiencies in two mosses suggests that it is a conserved feature of Bryophyte transformation. Biotechnol Lett 29:1591–1598
Turmel M, Otis C, Lemieux C (2002) The chloroplast and mitochondrial genome sequences of the charophyte Chaetosphaeridium globosum: insights into the timing of the events that restructured organelle DNAs within the green algal lineage that led to land plants. Proc Natl Acad Sci U S A 99:11275–11280
Turmel M, Otis C, Lemieux C (2006) The chloroplast genome sequence of Chara vulgaris sheds new light into the closest green algae relatives of land plants. Mol Biol Evol 23:1324–1338
von Wettstein F (1932) Genetik. In: Verdoorn F (ed) Manual of bryology. Martinus Nijhoff, The Hague, pp 232–272
Wang TL, Cove DJ, Beutelmann P, Hartmann E (1980) Isopentenyladenine from mutants of the moss, Physcomitrella patens. Phytochemistry 19:1103–1105
Yasumura Y, Crumpton-Taylor M, Fuentes S, Harberd NP (2007) Step-by-step acquisition of the gibberellin-DELLA growth-regulatory mechanism during land-plant evolution. Curr Biol 17:1225–1230
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Cove, D.J., Cuming, A.C. (2014). Genetics and Genomics of Moss Models: Physiology Enters the Twenty-first Century. In: Hanson, D., Rice, S. (eds) Photosynthesis in Bryophytes and Early Land Plants. Advances in Photosynthesis and Respiration, vol 37. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-6988-5_11
Download citation
DOI: https://doi.org/10.1007/978-94-007-6988-5_11
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-6987-8
Online ISBN: 978-94-007-6988-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)