Abstract
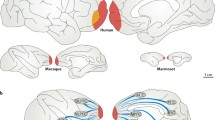
In humans and mammals with higher cognitive capabilities, the neocortex is a very prominent brain structure (Fig. 1). As such it seems to be crucially involved in the cognitive processes. The neocortex can be subdivided into a set of functionally different areas (Van Essen et al. 1992), and it communicates with most of the other brain systems. It is a structure with a high internal functional complexity and diversity which is involved in most aspects of cerebral processing. Various cortical areas represent and process different aspects of the environment and the subject’s internal states in a distributed way. In the visual modality for example, occipital to temporal regions of the brain are thought to mainly represent object identity-related sensory information, whereas occipital to parietal brain regions are thought to mainly represent and process spatial information and aspects preparing motor plans. The former is referred to as the “ventral stream” and the latter as the “dorsal stream” (Ungerleider and Haxby 1994 Lateral prefrontal areas are thought to store contextual information of the present and recent past, which can serve as a reference framework for the behavioral relevance of visual stimuli and motor plans, and can form a basis for decision- making processes (Leon and Shadlen 1998).

Illustration of the human neocortex. Gray-shaded regions group the cortical areas by functional similarity, black arrows schematically indicate inter-areal connectivity. Adapted from Statter (2002)
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
- Prefer Location
- Visual Working Memory
- Inattentional Blindness
- Attractor Network
- Attentional Facilitation
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Abeles A (1991) Corticonics. Cambridge University Press, New York
Almeida R, Deco G, Stetter M (2004) Modular biased-competition and cooperation: a candidate mechanism for selective working memory. Eur J Neurosci 20(10):2789–2803
Amit DJ, Brunel N (1997a) Dynamics of a recurrent network of spiking neurons before and following learning. Network Comput Neural Syst 8:373–404
Amit DJ, Brunel N (1997b) Model of global spontaneous activity and local structured (learned) delay activity during delay periods in cerebral cortex. Cereb Cortex 7:237–252
Amit DJ, Brunel N, Tsodyks M (1994) Correlations of cortical hebbian reverberations: experiment versus theory. J Neurosci 14:6435–6445
Bosking WH, Zhang Y, Schofield B, Fitzpatrick D (1997) Orientation selectivity and the arrangement of horizontal connections in tree shrew striate cortex. J Neurosci 17: 2112–2127
Brunel N, Wang XJ (2001) Effects of neuromodulation in a cortical network model of object working memory dominated by recurrent inhibition. Comput Neurosci 11:63–85
Chelazzi L (1998) Serial attention mechanisms in visual search: a critical look at the evidence. Psychol Res 62:195–219
Chelazzi L, Miller E, Duncan J, Desimone R (1993) A neural basis for visual search in inferior temporal cortex. Nature 363:345–347
Corchs S, Stetter M, Deco G (2003) System-level neuronal modeling of visual attentional mechanisms. Neuroimage 20:143–160
Deco G, Rolls ET (2003) Attention and working memory: a dynamical model of neuronal activity in the prefrontal cortex. Eur J Neurosci 18:2374–2390
Deco G, Rolls ET, Horwitz B (2004) “What” and “where” in visual working memory: a computational neurodynamical perspective for integrating fmri and single-neuron data. J Cogn Neurosci 16:683–701
Desimone R, Duncan J (1995) Neural mechanisms of selective visual attention. Annu Rev Neurosci 18:193–222
Everling S, Tinsley C, Gaffan D, Duncan J (2002) Filtering of neural signals by focused attention in the monkey prefrontal cortex. Nat Neurosci 5:671–676
Freedman DJ, Riesenhuber M, Poggio T, Miller EK (2003) A comparison of primate prefrontal and inferior temporal cortices during visual categorization. J Neurosci 23:5235–5246
Hebb DO (1949) The organization of behavior. Wiley, New York
Hopfield JJ (1982) Neural networks and physical systems with emergent collective computational abilities. Proc Natl Acad Sci U S A 79:2554–2558
Kandel ER, Schwartz JH, Jessel TM (1991) Principles of neural sciences. Prentice Hall International, London.
Kisvarday ZF, Toth E, Rausch M, Eysel UT (1997) Orientationspecific relationship between populations of excitatory and inhibitory lateral connections in the visual cortex of the cat. Cereb Cortex 7:605–618
Koch KW, Fuster JM (1989) Unit activity in monkey parietal cortex related to haptic perception and temporary memory. Exp Brain Res 76:292–306
Leon ML, Shadlen MN (1998) Exploring the neurophysiology of decisions. Neuron 21:669–672
Lund JS, Levitt JB, Wu Q (1994) Topography of excitatory and inhibitory connectional anatomy in monkey visual cortex. In: Lawton TB (Ed) Computational vision based on neurobiology. SPIE, Bellingham WA, pp 174–184
Malach R, Amir Y, Harel M, Grinvald A (1993) Relationship between intrinsic connections and functional architecture revealed by optical imaging and in vivo targeted biocytin injections in primate striate cortex. Proc Natl Acad Sci USA 90:10469–10473
Moran J, Desimone R (1985) Selective attention gates visual processing in the extrastriate cortex. Science 229:782–784
Reynolds J, Desimone R (1999) The role of neural mechanisms of attention in solving the binding problem. Neuron 24:19–29
Rolls ET, Deco G (2002) Computational neuroscience of vision. Oxford University Press, Oxford
Salin P, Bullier J (1995) Corticocortical connections in the visual system: structure and function. Physiol Rev 75:107–154
Sigala N, Logothetis N (2002) Visual categorization shapes feature selectivity in the primate temporal cortex. Nature 415:318–320
Simons DJ (2000) Attentional capture and inattentional blindness. Trends Cognit Sci 4:147–155
Somogyi P, Tamas G, Lujan R, Buhl EH (1998) Salient features of synaptic organization in the cerebral cortex. Brain Res Brain Res Rev 26:113–135
Stetter M (2002) Exploration of cortical function. Kluwer Academic Publishers, Dordrecht
Szabo M, Almeida R, Deco G, Stetter M (2004) Cooperation and biased competition model can explain attentional filtering in the prefrontal cortex. Eur J Neurosci 19:1969–1977
Szabo M, Almeida R, Deco G, Stetter M (2005) A neuronal model for the shaping of feature selectivity in it by visual categorization. Neurocomputing 65–66:195–201
Tomita H, Ohbayashi M, Nakahara K, Hasegawa I, Miyashita Y (1999) Top-down signal from prefrontal cortex in executive control of memory retrieval. Nature 401:699–703
Ungerleider LG, Haxby JV (1994) “What” and “where” in the human brain. Curr Opin Neurobiol 4:157–165
Van Essen DC, anderson CH, Felleman DJ (1992) Information processing in the primate visual system: an integrated systems perspective. Science 255:419–423
Wilson F, Scalaidhe S, Goldman-Rakic P (1994) Functional synergism between putative gamma-aminobutyrate-containing neurons and pyramidal neurons in prefrontal cortex. Proc Natl Acad Sci USA 91:4009–4013
Zeki S, Shipp S (1988) The functional logic of cortical connections. Nature 335:311–317
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2007 Springer
About this chapter
Cite this chapter
Deco, G., Stetter, M., Szabo, M. (2007). Biased Competition and Cooperation: A Mechanism of Mammalian Visual Recognition?. In: Osaka, N., Rentschler, I., Biederman, I. (eds) Object Recognition, Attention, and Action. Springer, Tokyo. https://doi.org/10.1007/978-4-431-73019-4_13
Download citation
DOI: https://doi.org/10.1007/978-4-431-73019-4_13
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-73018-7
Online ISBN: 978-4-431-73019-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)