Abstract
Since the discovery of microRNAs (miRNAS) in 1993, findings in gene regulation have increased in the biological scenery. miRNAs have shown an important role in the modulation of a wide collection of physiological and pathological processes, starting on embryonic evolution with carcinomas development. Nowadays, the high availability of gene expression data and the development of computational approaches that predicts miRNA targets are continuously increasing the knowledge about miRNA functions, as well as the sources and consequences of miRNA deregulation within a wide interaction network. In this chapter, knowledge of miRNA expression and its function on prostate cancer is presented.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
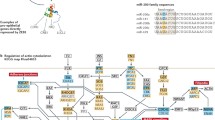
MicroRNAs (miRNAs) are a family of small regulatory RNAs that control the expression of specific genes in a post-transcriptional level by base-pairing typically to the 3′-untranslated regions (3′UTRs) of a target messenger RNAs (mRNAs) to direct a reduction in either translation or stability [1]. They are members of the family of small non-coding RNAs that are endogenous small interfering RNAs (endo-siRNAs), such as PIWI-single-stranded non-coding RNAs ofo 20–23 interacting RNAs (pi-RNAs) [2, 3]. MiRNAs are involved in a broad range of cellular functions, as developmental, cell differentiation and cell maintenance functions [4]. MiRNAs are distributed throughout the genome, they can be found as isolated transcript units or clustered and co-transcribed as polycistronic primary transcripts [5] and can either be encoded within protein coding genes in both introns and exons or transcribed from independent genes as intergenic regions. MiRNA transcription can be carried out by RNA polymerase III and II to produce the primary transcripts (primiRNAs) [6, 7], which are thousands nucleotide long capped and polyadenlylated hairpin-shaped transcripts. In the canonical miRNA genesis pathway, pri-miRNAs are recognized and cleaved in the nucleus then the mature miRNAs are generated by several processing steps of endonucleolytic cleavages in the cytoplasm. The cleave in the nucleus is carried out by an RNase III enzyme named Drosha, which acts with the microprocessor complex subunit DGCR8 (a molecular anchor necessary for the recognition of pri-miRNA at dsRNA-ssRNA junction) and cleaves 11 bp away from the junction to release hairpin-shaped pre-miRNAs that are subsequently cut by the cytoplasmic RNase III enzyme called DICER to generate mature miRNAs [8,9,10,11,12,13] (Fig. 1). An alternative miRNAs biogenesis pathway called Mitrons-pathway has been described, miRNAs is embedded in short mRNA introns, then pre-miRNAs are produced unending Drosha cleave, those are completed from splicing and debranching [14, 15]. In both miRNAs pathways, pre-miRNAs are transported into the cytoplasm by exportin 5 and Ran-GTP. In the cytoplasm Dicer joined to TRBP (Trans activated response [TAR] RNA Binding protein) cleaves the terminal loop end of the pre-miRNA producing a short double-stranded RNA duplex that measure around 20–23 bp length. The strand of the duplex with a less thermodynamically stable 5′ end is preferentially conjugated to an Argonaute protein forming the miRNA-induced silencing complex (RISC) [16], however in some cases the complementary strand can be used in the RISC complex [17, 18].
The mature miRNA joined to RISC binds to the mRNA in the 3′ UTR, or in less cases in the coding region, based on complementarity between the miRNA and the mRNA target. Seed sequence, nucleotides 2–8 (counted from the 5′ end) of the mature miRNA [1, 19] is critical for target recognition and hybridizes nearly perfectly with the target mRNA [5].
The understanding of miRNA functions into a network context and the signaling or metabolic pathways with the consequences that their deregulation can produce has been achieved by novelty advances, as the genome-wide identification of miRNA–target, the RNA sequencing.
In the last two decades, several strategies have been used to identify miRNA targets, physical association and/or the correlation of gene expression. Lists of target genes can then be examined collectively in the contexts of KEGG pathways (KEGG PATHWAY is a collection of manually drawn pathway maps representing our knowledge on the molecular interaction, reaction and relation networks for: (1) Metabolism, (2) Genetic information processing, (3) Environmental information processing, (4) Cellular processes, (5) Organismal systems, (6) Human diseases, and (7) Drug development) (http://www.genome.jp/kegg/pathway.html) [20,21,22], protein–protein interaction networks [23,24,25,26] and enrichment analysis for common gene ontology terms [27, 28]. Several computational tools for target enrichment analysis facilitate the identification of hierarchical functions of miRNAs in gene regulatory networks (Table 1). The involvement of miRNAs in signaling pathways and genetic networks provides a challenge to understanding their function. The level of evidence required to establish miRNA–target relationships is set too low that the genuine interactions are eclipsed by noise.
Recently it has been proposed five steps (or levels) to unravel the specific role of miRNA. In the level 1 the objective is to find or identify a specific microRNA (from the vast list of known microRNAs) and enlist pathways expected to be involved in a disease under study, increasing the probability to associate a miRNA with the disease and create a base to identify the pathway. The condition of a miRNA under a cellular condition can be achieved by the micro-array/transcriptome technique; level 2 The next step is to identify the status of the miRNA and the key pathway specific genes expression in the selected disease. This is achieved through a database search. Alternatively, the absence of enough published information, conducting a case-control or a pair (tumor and normal tissue) study in cancer could help to establish the status of the microRNA and the key genes, preferably representing the pathway specific markers; level 3 the predicted miRNA targets need to be experimentally validated. Many human miRNAs are released in the latest version of miRBase (Release 20), but yet many remain to be identified. In the last ten years several algorithms have been developed to identify targets for miRNAs, those are based on the conservation of seed region and binding energy, however, recently some of the algorithms (TargetScan, miRanda, RNA22, PICTAR, PITA, RNAhybrid, DIANA-microT, mirSVR, microInspector, mirTarget2) have incorporated expression profiles in their scoring function [29], helping to increase the accuracy of prediction. It has been observed that the stability of miRNA seed sequences was evolutionarily selected according to the adaptive temperature of each organism, however non-conserved binding sites also mediate repression in translation, so new algorithms should incorporate non-conserved binding sites to avoid false negatives predicted results. In the level 4, it is needed to co-relate the gene and the miRNA expression data. Those pathways should be regulated directly by the miRNA according to the results obtained in the first three levels. Bioinformatics tools as Central Pathway or Cytoscape could be used for this propose. In the level 5 it should be proved the association of miRNA with the identified pathway experimentally and preferably in a tissue specific manner in concordance with the settled in level 1 [30].
2 MiRNAs in Cancer
As it has been discussed, miRNAs play a main role in gene expression control at post-transcriptional level, when the miRNAs are deregulated several kinds of pathological diseases can occur, one of the diseases with a main interest in the study for miRNAs is the cancer. Key processes in cancer biology are related to the “hallmarks of Cancer” (HC). The HC comprise six biological capabilities acquired during the multi-step development of human tumors, and constitute an organizing principle for rationalizing the complexities of neoplastic disease. They include sustaining proliferate signaling, evading growth suppressors, resisting cell death, inducing angiogenesis, and activating invasion and metastasis. Underlying these hallmarks is genome instability, which generates the genetic diversity that expedites their acquisition, evasion of the immune system, deregulation of energy metabolism and inflammation, which fosters multiple hallmark functions [31].
Apoptosis plays a significant role in both animal development and disease, the deregulation of this process has been invariably linked to the progression of various neoplastic processes. miRNAs that regulate apoptosis, termed apoptomiRs, can be either pro- or antiapoptotic [32]. The first miRNA described as a regulator of apoptosis was the Drosophila bantam gene, which directly suppressed the proapoptotic factor hid, thus facilitating proliferation [33, 34]. Several miRNAs that play a role in modulating apoptosis have also been linked to the initiation and progression of various neoplastic processes. Approximately 50% of miRNAs are located at genomic sites that are disrupted or amplified in various cancers [35]. The first evidence of miRNAs playing a role in cancer development came to light in 2002 in a study [36] that attempted to find tumor suppressor genes at chromosome 13q14, which is frequently deleted in chronic lymphocytic leukemia (CLL) [37]. CLL is characterized by the presence of substantial increased numbers of predominantly non-dividing malignant B cells over-expressing the antiapoptotic B-cell lymphoma 2 (Bcl2) protein. In patients with CLL, the tumor suppressor locus on chromosome 13q14 was found to be frequently altered. However, instead of coding for a tumor suppressor protein, this region contained two miRNA genes, miR-15a and miR-16-1, which are overexpressed, regulating negatively the antiapoptotic bcl2 gene at posttranscriptional level. Later, many miRNAs with tumor suppressor roles were identified. The miR-34 family, for example, has been shown to exert significant tumor suppressor capabilities. Up-regulation of p53 (a potent tumor suppressor/cell cycle regulator) caused increased miR-34 expression that resulted in G1 arrest in a complementary and parallel fashion to mRNAs that are directly activated by P53 [35, 38]. Also, miR-34 was shown to inhibit the silent mating information regulator 1 (SIRT1) gene resulting in the upregulation of P53, P21, and PUMA (P53-upregulated modulator of apoptosis), thus regulating cell cycle and apoptosis and functioning as a tumor suppressor by modulating the SIRT1-P53 pathway [39]. Furthermore, miR-34 has mediated growth arrest via direct regulation of cell cycle regulatory factors, such as cyclin E2 (CCNE2), cyclin-dependent kinase 4 (CDK4), the transcription factor E2F3, and the hepatocyte growth factor receptor (c-MET), ultimately leading to increased caspase-dependent cell death [34]. In a separate study, miR-34 inhibited the proliferation/growth of human pancreatic tumor–initiating cells, and its overexpression in P53-deficient human pancreatic cancer cells partially restored the tumor-suppressing function of P53 [68]. MCL-1, a member of the BCL-2 family, was also demonstrated to be posttranscriptionally regulated by miR-29a, b, and c [40, 41]. Forced expression of miR-29b to induce tumor cell apoptosis by reducing MCL-1 expression may represent a novel intervention for cancer therapy. Along similar lines, let-7a exerts tumor suppressor functions by directly targeting the expression of ras and hmga2, 2 widely recognized oncogenes [42, 43]. Other examples of tumor suppressor miRNAs include miR-7, miR-124, miR-137, miR-146b, miR-15b, miR-128, and miR-326. Furthermore, knockdown of mature miRNAs by selectively targeting dicer1, rnasen, and its cofactor dgcr8 increased the oncogenic potential of transformed cell lines, resulting in the accelerated tumor formation in mouse models of K-RAS-driven lung cancer and Rb-driven retinoblastoma [44,45,46,47].
MiRNAs can also promote tumor development (oncomiRs) depending on the functions of the target protein(s) they regulate. These oncogenic miRNAs include miR-155 and the miR-17-92 cluster that accelerates the tumor development in B-cell lymphomas [48, 49]. Ectopic expression of miR-155 in transgenic mice resulted in pre-B-cell expansion, splenomegaly, and lymphopenia that preceded the development of lymphoblastic leukemia and lymphoma [50]. Now it is known that mir-155 plays a critical role in the development of lymphomas, the components of its regulatory pathways upstream and downstream of the targets remain unclear. It is interesting to note that even before the discovery of miRNAs in mammalian cells, Tam et al. [54] had reported that “bic” locus, the common retroviral integration site for the avian leukosis virus, generated a non-coding RNA. Later, after the discovery of miRNAs, it was found that this transcript harbored the mature miR-155 coding sequence, thus offering a potential explanation for the function of bic [55]. Members of the mir-17-92 cluster are potent activators of cell proliferation and are frequently overexpressed in several neoplasms, including lymphoma, multiple myeloma, medulloblastoma, and cancers of the lung, colon, breast, and prostate [44]. miR-21 is another commonly upregulated miRNA in cancers that include glioblastoma, lymphomas, and cancers of the breast, ovary, colon, rectum, pancreas, lung, liver, gallbladder, prostate, stomach, thyroid, and cervix [44, 51]. Increased expression of miR-21 was found in glioblastoma tumors and cell lines, and its inhibition resulted in increased cell death, suggesting that miR-21 could play the role of an oncogene that inhibited cell death in these tumors [85]. For example, in prostate cancer it has been found that miR21 controls the expression of many mRNA targets related to micro-vascular proliferation and tumor invasiveness, correlating with weak biochemical recurrence-free survival having a predictive value for biochemical recurrence risk in prostate cancer patients after radical prostatectomy [68], and also correlating with castration resistance and metastatic disease. Furthermore, in glioblastoma cells, knockdown of miR-21 induced the activation of caspase-3, transforming growth factor-β, P53, and mitochondrial apoptotic pathways mainly through upregulation of its validated targets, heterogeneous nuclear ribonucleoprotein K, P53-related TAP63, and PDCD4, acting in synergy with the aforementioned proteins [51,52,53].
3 miRNAs in Prostate Cancer
Prostate cancer is the second place occurring cancer in men worldwide and the first place in mortality [54]. Prostate cancer (PCa) occurrence and mortality are up to 20-fold higher in developed countries with respect to emerging ones, diet and lifestyle have been proposed as factors causing this discrepancy, both modify serum factors that slow down the growth and induce apoptosis in androgen-dependent PCa cells, while high body mass index, blood pressure, and several metabolic factors were correlated with high risk of prostate cancer death [54]. Mortality rates are clearly differentiated by ethnicity. Caribbean hold the highest rates in the world (26.3%), following sub-Saharan Africans (10%) and, at last, the Asians show the lowest (2.5%). In the United States of America it was found that the risk of developing prostate cancer is highest in African Americans, in compared to other populations[55].
As it was discussed above miRNAs are deregulated in several disorders also in cancer. Deregulation can increase or decrease their concentration into the affected cell, and according to their function in normal concentrations miRNAs can be classified as oncogenic miRNAs (oncomiR) and tumor suppressor miRNAs. It has been reported several miRNAs with their respective functions in prostate cancer (Table 2).
4 Circulating miRNAs as Possible Biomarkers in Prostate Cancer
Prostate cancer can be clinically diagnosed as local or advanced and different treatments including observation, radiotherapy, radical prostatectomy, and androgen-deprivation treatment are expected [56,57,58,59].
The main screening exams to diagnose prostate cancer include digital rectal exam (DRE), serum level of prostate-specific antigen (PSA), and transrectal ultrasound guided biopsy. A suspected DRE alone reveals prostate cancer, regardless of normal PSA level (in about 18% of all patients; moreover, a suspected digital rectal exam has a positive predictive value of 5–30% in patients who have a PSA level up to 2 ng/mL). Nowadays, PSA assessment is the most usual marker able to correlate with prostate cancer risk, aggressiveness, and outcome in order to detect prostate cancer [60]. Nevertheless, it has been found that several patients develop prostate cancer in spite of low PSA levels though a higher PSA level shows the presence of prostate cancer [61, 62]. High PSA levels could be the result of several factors as well as benign prostatic hyperplasia, gland inflammation, prostatitis, infection, and some drugs [63,64,65,66]. PSA screening is usually associated with over-diagnosis, although it has decreased the death rate of prostate cancer [67]. For this reason, efforts to find new biomarkers that enhance the accuracy of screening should be continued.
Circulating miRNas in blood were described by Mitchell et al. [70, 71]. miRNAs are released to the blood stream from the tumoral tissue through several ways, as free miRNAs, into exosomes, into microvesicles, into big oncosomes, joined to apoptotic bodies, associated with high density lipoproteic complexes or joined to proteins. The presence of miRNA in the extracellular environment ignited the hypotheses that cells selectively release miRNAs which mediate cell–cell signaling via paracrine or even endocrine routes [78,79,80]. Some research groups have demonstrated that extracellular miRNAs entrapped within apoptotic bodies and exosomes can be transferred to recipient cells, alter gene expression, and mediate functional effects [78, 81,82,83,84,85]. Patterns of mRNAs in exosomes and their donor cells correlate poorly, suggesting specific sorting of miRNA “for export” [78, 84,85,86]. The mechanism behind this sorting needs to be investigated in more detail.
The miRNAs released in the bloodstream are protected against degradation and readily detectable by PCR methods; mainly in cancer specific miRNAs levels have been used to correlate disease status, stage, aggressiveness, and response to therapy performing the miRNA profiling in plasma or serum. In the case of prostate cancer, cell-free miRNAs levels are able to differentiate between localized and metastatic prostate cancer or correlate with the risk score or Gleason grade, associating miRNAs with aggressiveness or indolent disease and may aid in tumor staging and treatment decisions at the time of diagnosis [72,73,74,75].
Agalogu et al. [69] assessed serum levels of three miRNAs in 51 patients with prostate cancer (subdivided into localized/locally advanced and metastatic) and 20 healthy controls. They identified increased levels of miR-21 and miR-221 in the prostate cancer group as a whole compared to the controls. miR-141 was not significantly elevated in the group as a whole, but when the metastatic group was considered in isolation, all three miRNAs were significantly elevated, with mir-141 being the most elevated of the three.
Lodes et al. [70] analyzed sera from patients with a number of malignancies using a micro-array hybridization technique, identifying 15 upregulated miRNAs in the sera of prostate cancer patients vs. healthy controls (miR-16, -92a, -103, -107, -197, -34b, -328, -485-3p, -486-5p, -92b, -574-3p, -636, -640, -766, -885-5p). This study did not confirm the finding of increased miR-141 expression; however, the group of prostate cancer patients was small (n = 6). Additionally, no qRT-PCR confirmation of the micro-array results was performed.
Several miRNAs have been found over-expressed in plasma/serum of patients with prostate cancer, some of them as miR-21, and miR-141 were validated about the cellular function respect prostate cancer context following the five levels assessment. Some studies [70, 76, 77] have reported an over-expression of the miRNA-107, which is encoded at 10p23.11, this miRNA has a fold change around 10; a study in a court of thirty-eight prostate cancer patients at different clinical stages (from I to IV) revealed that miR-107 is over-expressed at each clinical stage of prostate cancer relative to the control group (Fig. 2), there was not significative difference between media value at each clinical stage of cancer (t-test), however there is a shape showing an increasing of relative expression for miR-107 related to the clinical stage of cancer.
It should be necessary to increase the size of the assessed groups to find a clear correlation between the clinical stage of prostate cancer and the relative expression of miR-107.
5 Conclusion
MicroRNAs are small biomolecules with a great number of possible targets in the cellular functions, when those are deregulated several diseases can occur, one of the most studied is cancer, miRNAs can be relevant as therapeutic targets or biomarkers, in prostate cancer there are several miRNAs that have been validated as oncogenic miRNAs and tumor suppressor miRNAs, some of the over-expressed miRNAs in plasma/serum of patients with prostate cancer have been proposed as possible biomarkers of the disease as miR-21 and miR-141, recently miR107 has been relevant by their high relative over-expression in plasma/serum, it has been tested in a cohort of patients with prostate cancer at different clinical stages showing that it is possible to find a correlation between their relative expression in plasma and the clinical stage of cancer. It makes necessary to increase the efforts to find either miRNAs functions in prostate cancer or possible new biomarkers to shelter the current prostatic specific antigen test.
References
Filipowicz W, Bhattacharyya SN, Sonenberg N (2008) Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nat Rev Genet 9:102–114
Ghildiyal M, Zamore PD (2009) Small silencing RNAs: an expanding universe. Nat Rev Genet 10(2):94–108
Pauli A, Rinn JL, Schier AF (2011) Non-coding RNAs as regulators of embryogenesis. Nat Rev Genet 12(2):136–149
Bushati N, Cohen SM (2007) microRNA functions. Annu Rev Cell Dev Biol 23:175–205
Rodriguez A, Griffiths-Jones S, Ashurst JL, Bradley A (2004) Identification of mammalian microRNA host genes and transcription units. Genome Res 14(10A):1902–1910
Borchert GM, Lanier W, Davidson BL (2006) RNA polymerase III transcribes human microRNAs. Nat Struct Mol Biol 13(12):1097–1101
Lee Y, Kim M, Han J, Yeom KH et al (2004) MicroRNA genes are transcribed by RNA polymerase II. EMBO J 23(20):4051–4060
Carthew RW, Sontheimer EJ (2009) Origins and mechanisms of miRNAs and siRNAs. Cell 136:642–655
Kim VN, Han J, Siomi MC (2009) Biogenesis of small RNAs in animals. Nat Rev Mol Cell Biol 10:126–139
Siomi H, Siomi MC (2009) On the road to reading the RNA-interference code. Nature 457:396–404
Lee Y, Ahn C, Han J, Choi H et al (2003) The nuclear RNase III Drosha initiates microRNA processing. Nature 425(6956):415–419
Denli AM, Tops BB, Plasterk RH, Ketting RF et al (2004) Processing of primary microRNAs by the microprocessor complex. Nature 432(7014):231–235
Gregory RI, Yan KP, Amuthan G, Chendrimada T et al (2004) The microprocessor complex mediates the genesis of microRNAs. Nature 432(7014):235–240
Ruby JG, Jan CH, Bartel DP (2007) Intronic microRNA precursors that bypass Drosha processing. Nature 448(7149):83–86
Rasschaert P, Figueroa T, Dambrine G, Rasschaert D, Laurent S (2016) Alternative splicing of a viral mirtron differentially affects the expression of other microRNAs from its cluster and of the host transcript. RNA Biol 13(12):1310–1322
Schwarz DS, Hutvagner G, Du T, Xu Z et al (2003) Asymmetry in the assembly of the RNAi enzyme complex. Cell 115(2):199–208
Krol J, Loedige I, Filipowicz W (2010) The widespread regulation of microRNA biogenesis, function and decay. Nat Rev Genet 11(9):597–610
Cheloufi S, Dos Santos CO, Chong MM, Hannon GJ (2010) A dicer-independent miRNA biogenesis pathway that requires Ago catalysis. Nature 465(7298):584–589
Rigoutsos I (2009) New tricks for animal microRNAS: targeting of amino acid coding regions at conserved and nonconserved sites. Cancer Res 69(8):3245–3248
Gaidatzis D, van Nimwegen E, Hausser J, Zavolan M (2007) Inference of miRNA targets using evolutionary conservation and pathway analysis. BMC Bioinf 8:69
Maragkakis M et al (2009) DIANA-microT web server: elucidating microRNA functions through target prediction. Nucleic Acids Res 37:273–276
Shirdel EA, Xie W, Mak TW, Jurisica I (2011) NAViGaTing the micronome using multiple microRNA prediction databases to identify signalling pathwayassociated microRNAs. PLoS ONE 6:e17429
Liang H, Li W (2007) MicroRNA regulation of human protein—protein interaction network. RNA 13:1402–1408
Hsu CW, Juan HF, Huang HC (2008) Characterization of microRNA-regulated protein-protein interaction network. Proteomics 8:1975–1979
Yuan X et al (2009) Clustered microRNAs’ coordination in regulating protein-protein interaction network. BMC Syst Biol 3:65
Sass S et al (2011) MicroRNAs coordinately regulate protein complexes. BMC Syst Biol 5:136
Ashburner M et al (2000) Gene ontology: tool for the unification of biology. Nat Genet 25:25–29
Hung JH, Yang TH, Hu Z, Weng Z, DeLisi C (2011) Gene set enrichment analysis: performance evaluation and usage guidelines. Brief Bioinform 13:281–291
Betel D, Wilson M, Gabow A, Marks DS, Sander C (2008) The microRNA.org resource: targets and expression. Nucleic Acids Res 36(Database issue):D149–D153
Manvatia S, Mangalharab KC, Khana J, Pathaniab GL, Kaulc S, Kaushika M, Aroraa A, Dhara PK (2017) Deciphering the role of microRNA – a step by step guide. Gene Expr Patterns 25–26:59–65
Hanahan D et al (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674
Su Z, Yang Z, Xu Y, Chen Y, Yu Q (2015) MicroRNAs in apoptosis, autophagy and necroptosis. Oncotarget 6(11):8474–8490
Brennecke J, Hipfner DR, Stark A et al (2003) Bantam encodes a developmentally regulated microRNA that controls cell proliferation and regulates the proapoptotic gene hid in Drosophila. Cell 113:25–36
Schickel R, Boyerinas B, Park SM et al (2008) MicroRNAs: key players in the immune system, differentiation, tumorigenesis and cell death. Oncogene 27:5959–5974
Calin GA, Sevignani C, Dan Dumitru C et al (2004) Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci U S A 101:2999–3004
Calin GA, Dumitru CD, Shimizu M et al (2002) Frequent deletions and down-regulation of micro-RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci U S A 99:15524–15529
Iorio MV, Croce CM (2012) MicroRNA deregulation in cancer: diagnostics, monitoring and therapeutics. A comprehensive review. EMBO Mol Med 4:143–159
Bommer GT, Gerin I, Feng Y et al (2007) p53-mediated activation of miRNA34 candidate tumor-suppressor genes. Curr Biol 17:1298–1307
Yamakuchi M, Ferlito M, Lowenstein CJ (2008) miR-34a repression of SIRT1 regulates apoptosis. Proc Natl Acad Sci U S A 105:13421–13426
Welch C, Chen Y, Stallings RL (2007) MicroRNA-34a functions as a potential tumor suppressor by inducing apoptosis in neuroblastoma cells. Oncogene 26:5017–5022
Mott JL, Kobayashi S, Bronk SF et al (2007) mir-29 regulates Mcl-1 protein expression and apoptosis. Oncogene 26:6133–6140
Lee YS, Dutta A (2007) (2007) The tumor suppressor microRNA let-7 represses the HMGA2 oncogene. Genes Dev 21:1025–1030
Johnson SM, Grosshans H, Shingara J et al (2005) RAS is regulated by the let-7 microRNA family. Cell 120:635–647
Farazi TA, Spitzer JI, Morozov P et al (2011) miRNAs in human cancer. J Pathol 223:102–115
Kumar MS, Lu J, Mercer KL et al (2007) Impaired microRNA processing enhances cellular transformation and tumorigenesis. Nat Genet 39:673–677
Kumar MS, Pester RE, Chen CY et al (2009) Dicer1 functions as a haploinsufficient tumor suppressor. Genes Dev 23:2700–2704
Lambertz I, Nittner D, Mestdagh P et al (2010) Monoallelic but not biallelic loss of Dicer1 promotes tumorigenesis in vivo. Cell Death Differ 17:633–641
Eis PS, Tam W, Sun L et al (2005) Accumulation of miR-155 and BIC RNA in human B cell lymphomas. Proc Natl Acad Sci U S A 102:3627–3632
He L, Thomson JM, Hemann MT et al (2005) A microRNA polycistron as a potential human oncogene. Nature 435:828–833
Costinean S, Zanesi N, Pekarsky Y et al (2006) Pre–B cell proliferation and lymphoblastic leukemia/high-grade lymphoma in E mu-miR155 transgenic mice. Proc Natl Acad Sci U S A 103:7024–7029
Chan JA, Krichevsky AM, Kosik KS (2005) MicroRNA-21 is an antiapoptotic factor in human glioblastoma cells. Cancer Res 65:6029–6033
Chen Y, Liu W, Chao TF et al (2008) MicroRNA-21 down-regulates the expression of tumor suppressor PDCD4 in human glioblastoma cell T98G. Cancer Lett 272:197–205
Papagiannakopoulos T, Shapiro A, Kosik KS (2008) MicroRNA-21 targets a network of key tumor-suppressive pathways in glioblastoma cells. Cancer Res 68:8164–8172
GLOBOCAN (2012) Estimated cancer incidence, mortality and prevalence Worldwide in 2012: International Agency for Research on Cancer. http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx [consulted on August 2, 2016]
Gronberg H (2003) Prostate cancer epidemiology. Lancet 361:859–864
Di Franco R, Borzillo V, Ravo V, Ametrano G, Falivene S, Cammarota F, Rossetti S, D’Aniello C, Cavaliere C, Romano FJ, Malzone MG, Montanari M, Vanacore D et al (2016) Hypofractionated versus conventional radiotherapy in patients with low/intermediate-risk localized prostate cancer: review of rectal and urinary toxicity. WCRJ 3:e751
D’Aniello C, Cavaliere C, Fiorica F, Facchini G (2016) Letter to the editor: unenhanced whole-body MRI versus PET-CT for the detection of prostate cancer metastases after primary treatment. Eur Rev Med Pharmacol Sci 20:4626–4627
Nardone V, Botta C, Caraglia M, Martino EC, Ambrosio MR, Carfagno T, Tini P, Semeraro L, Misso G, Grimaldi A, Boccellino M, Facchini G, Berretta M et al (2016) Tumor infiltrating T lymphocytes expressing FoxP3, CCR7 or PD-1 predict the outcome of prostate cancer patients subjected to salvage radiotherapy after biochemical relapse. Cancer Biol Ther 17:1213–1220
Heidenreich A, Bellmunt J, Bolla M, Joniau S, Mason M, Matveev V et al (2011) EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and treatment of clinically localized disease. Eur Urol 59:61–71
Loeb S, Catalona WJ (2007) Prostate-specific antigen in clinical practice. Cancer Lett 249:30–39
Boccellino M, Alaia C, Misso G, Cossu AM, Facchini G, Piscitelli R, Quagliuolo L, Caraglia M (2015) Gene interference strategies as a new tool for the treatment of prostate cancer. Endocrine 49:588–605
Facchini, G. Caffo, O. Ortega, C. D’Aniello, C. Di Napoli, M. Cecere, S.C. Pepa, C.D. Crispo, A. Maines, F. Ruatta, F. Iovane, G. Pisconti, S. Montella, M. et al. (2016). Very early PSA response to abiraterone in mCRPC patients: a novel prognostic factor predicting overall survival. Front Pharmacol 7:123.
Wang G, Chan ES, Kwan BC, Li PK, Yip SK, Szeto CC, Ng CF (2012) Expression of microRNAs in the urine of patients with bladder cancer. Clin Genitourin Cancer 10:106–113
Lolli C, Caffo O, Scarpi E, Aieta M, Conteduca V, Maines F, Bianchi E, Massari F, Veccia A, Chiuri VE, Facchini G, De Giorgi U (2016) Systemic immune-inflammation index predicts the clinical outcome in patients with mCRPC treated with abiraterone. Front Pharmacol 7:376
Verzoni E, De Giorgi U, Derosa L, Caffo O, Boccardo F, Facchini G, Porcu L, De Vincenzo F, Zaniboni A, Chiuri VE, Fratino L, Santini D, Adamo V et al (2016) Predictors of longterm response to abiraterone in patients with metastastic castration-resistant prostate cancer: a retrospective cohort study. Oncotarget 7:40085–40094
Caffo O, Lo Re G, Sava T, Buti S, Sacco C, Basso U, Zustovich F, Lodde M, Perin A, Facchini G, Veccia A, Maines F, Barile C et al (2015) Intermittent docetaxel chemotherapy as first-line treatment for metastatic castration-resistant prostate cancer patients. Future Oncol 11:965–973
Li T, Li RS, Li YH, Zhong S, Chen YY, Zhang CM, MM H, Shen ZJ (2012) miR-21 as an independent biochemical recurrence predictor and potential therapeutic target for prostate cancer. J Urol 187:1466–1472
Zhang HL, Qin XJ, Cao DL, Zhu Y, Yao XD, Zhang SL, Dai B, Ye DW (2013) An elevated serum miR-141 level in patients with bone-metastatic prostate cancer is correlated with more bone lesions. Asian J Androl 15:231–235
Agaoglu FY, Kovancilar M, Dizdar Y, Darendeliler E, Holdenrieder S, Dalay N, Gezer U (2011) Investigation of miR-21, miR-141, and miR-221 in blood circulation of patients with prostate cancer. Tumor Biol 32(3):583–588
Lodes MJ, Caraballo M, Suciu D, Munro S, Kumar A, Anderson B (2009) Detection of cancer with serum miRNAs on an oligonucleotide microarray. PLoS One 4:7
Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL et al (2008) Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci U S A 105:10513–10518
D’Amico AV, Whittington R, Malkowicz SB, Cote K, Loffredo M, Schultz D et al (2002) Biochemical outcome after radical prostatectomy or external beam radiation therapy for patients with clinically localized prostate carcinoma in the prostate specific antigen era. Cancer 95:281–286
Lian F, Sharma NV, Moran JD, Moreno CS (2015) The biology of castration-resistant prostate cancer. Curr Probl Cancer 39:17–28
James ND, Spears MR, Clarke NW, Dearnaley DP, De Bono JS, Gale J et al (2015) Survival with newly diagnosed metastatic prostate cancer in the “Docetaxel Era”: data from 917 patients in the control arm of the STAMPEDE Trial (MRCPR08, CRUK/06/019). Eur Urol 67:1028–1038
Sweeney CJ, Chen YH, Carducci M, Liu G, Jarrard DF, Eisenberger M et al (2015) Chemohormonal therapy in metastatic hormone-sensitive prostate cancer. N Engl J Med 373:737–476
Bryant RJ, Pawlowski T, Catto JWF, Marsden G, Vessella RL, Rhees B, Kuslich C, Visakorpi T, Hamdy FC (2012) Changes in circulating microRNA levels associated with prostate cancer. Br J Cancer 106(4):768–774
Mihelich BL, Maranville JC, Nolley R, Peehl DM, Nonn L (2015) Elevated serum microRNA levels associate with absence of high-grade prostate cancer in a retrospective cohort. PLoS ONE 10(4):e0124245
Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, Lötvall JO (2007) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9(6):654–659
Cortez MA, Bueso-Ramos C, Ferdin J, Lopez-Berestein G, Sood AK, Calin GA (2011) MicroRNAs in body fluids–the mix of hormones and biomarkers. Nat Rev Clin Oncol 8(8):467–477
Chendrimada TP, Gregory RI, Kumaraswamy E, Norman J, Cooch N, Nishikura K et al (2005) TRBP recruits the Dicer complex to Ago2 for microRNA processing and gene silencing. Nature 436:740–744
Skog J, Würdinger T, van Rijn S, Meijer DH, Gainche L, Sena-Esteves M, Curry WT Jr, Carter BS, Krichevsky AM, Breakefield XO (2008) Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell Biol 10(12):1470–1476
Kosaka N, Iguchi H, Yoshioka Y, Takeshita F, Matsuki Y, Ochiya T (2010) Secretory mechanisms and intercellular transfer of microRNAs in living cells. J Biol Chem 285(23):17442–17452
Pegtel DM, Cosmopoulos K, Thorley-Lawson DA, Van Eijndhoven MA, Hopmans ES, Lindenberg JL et al (2010) Functional delivery of viral miRNAs via exosomes. Proc Natl Acad Sci U S A 107:6328–6333
Pigati L, Yaddanapudi SC, Iyengar R, Kim DJ, Hearn SA, Danforth D, Hastings ML, Duelli DM (2010) Selective release of microRNA species from normal and malignant mammary epithelial cells. PLoS One 5(10):e13515
Mittelbrunn M, Gutiérrez-Vázquez C, Villarroya-Beltri C, González S, Sánchez-Cabo F, González MÁ, Bernad A, Sánchez-Madrid F (2011) Unidirectional transfer of microRNA-loaded exosomes from T cells to antigen-presenting cells. Nat Commun 2:282
Skog J, Würdinger T, van Rijn S, Meijer DH, Gainche L, Sena-Esteves M, Curry WT Jr, Carter BS, Krichevsky AM, Breakefield XO (2008) Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell 10(12):1470–1476
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
de la Rosa Pérez, D.A. et al. (2018). MicroRNAs, Gene’s Regulator in Prostate Cancer. In: Olivares-Quiroz, L., Resendis-Antonio, O. (eds) Quantitative Models for Microscopic to Macroscopic Biological Macromolecules and Tissues. Springer, Cham. https://doi.org/10.1007/978-3-319-73975-5_2
Download citation
DOI: https://doi.org/10.1007/978-3-319-73975-5_2
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-73974-8
Online ISBN: 978-3-319-73975-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)