Abstract
Purpose
The soil microbial community is an important bioactive component of terrestrial ecosystems. Its structural and functional diversity directly affects carbon and nitrogen processes. This study aimed to investigate the variations in the diversity and composition of soil bacterial communities in a wetland with different nitrogen deposition conditions.
Methods
A long-term simulated nitrogen deposition experiment was conducted in the Ecological Locating Research Station of the Institute of Nature and Ecology of Heilongjiang Academy of Sciences. Three different treatments were evaluated, including low nitrogen (LK; 40 kg N·hm-2·a-1), high nitrogen (HN; 80 kg N·hm-2·a-1), and control (CK; 0 kg N·hm-2·a-1). Bacterial 16S rDNA was then sequenced and analyzed using the next-generation sequencing technology.
Result
Higher levels of N deposition resulted in an α-diversity increase followed by a decrease, with significant reductions in the HN treatment. Simulated nitrogen deposition resulted in changes in the structure and abundance of bacterial communities in wetland soils. The dominant phyla in all three plots were Proteobacteria and Acidobacteria. Compared with CK, the relative abundance of Chloroflexi increased significantly under the HN treatment (P < 0.05), whereas the relative abundance of Firmicutes and Bacteroidetes decreased significantly (P < 0.05). Nitrogen input changed the composition and relative abundance of the bacterial community, which was possibly due to N-induced soil acidification.
Conclusion
This study thus provides a theoretical basis for predicting the effects of atmospheric nitrogen deposition on soil microorganisms, as well as changes in the wetland ecosystem in Sanjiang Plain.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Background
Nitrogen deposition has steadily increased since the twentieth century due to fossil fuel combustion, the use of large amounts of agricultural fertilizers, and the intensification of animal husbandry, all of which have had profound effects on the carbon and nitrogen cycle of terrestrial ecosystems. Particularly, the rapid economic development and urbanization of China have led to marked increases in nitrogen deposition. China has become the third-largest nitrogen deposition area in the world, after Europe and North America (Mo et al. 2006; Galloway et al. 2008). Nitrogen deposition is reportedly the most important driving factor for carbon sequestration in terrestrial ecosystems in China (Bai et al. 2010; Tian et al. 2011). The recent intensification of nitrogen deposition has resulted in soil acidification, changes in plant and microbial composition and diversity, and changes in the soil carbon-nitrogen cycle (Hiiesalu et al. 2014; Bassiri 2015), thus affecting ecosystem stability and health. Therefore, reducing nitrogen deposition and its impact on human health and the environment has become an important challenge.
The soil microbiota is an important part of the ecosystem, as it plays a critical role in mediating soil material cycles and energy flow. Further, soil microorganisms promote the mineralization and decomposition of soil organic matter and the circulation and transformation of soil nutrients such as C, N, P, and S, in addition to playing a crucial role in the global biogeochemical cycle (Bai et al. 2010; Liu et al. 2016). Nitrogen is an important limiting factor in most terrestrial ecosystems, and increased nitrogen deposition can cause changes in soil microbial activity and microbial community structure by affecting soil chemical properties. The structure of soil microbial communities can also indirectly affect the growth of aboveground plants (Lammel et al. 2018; Lu et al. 2020). Changes in soil microbial diversity can be used as a sensitive indicator of ecosystem function. Therefore, understanding the effects of nitrogen deposition on microbial diversity is one of the most important challenges in the field of ecology (Li et al. 2019a, 2019b; Wei et al. 2020). At present, a large number of studies have been carried out on the impact of nitrogen deposition on the structure and function of microbial communities (Yao et al. 2014; Wei et al. 2020; Liu et al. 2020). Yao et al. (2014) found that when the N deposition reached a certain threshold (5.25–10.5 g m-2 year-1) in typical grassland, it leads to decrease the soil pH < 6.0 and therefore significantly decrease the bacterial community diversity. Wei et al. (2020) found that N addition significantly reduced α-diversity and changed the bacterial communities across generally all months. Liu et al. (2020) found that the β-diversity of soil bacteria increased first and then decreased with nitrogen addition. However, most studies on the effects of nitrogen deposition on soil microorganisms have mainly focused on forest and grassland ecosystems, whereas the study of natural wetlands has remained largely neglected, particularly those with nitrogen applied for more than 10 years.
Sanjiang Plain is the most complete and well-preserved wetland in China and is also an important grain production center. Further, this region plays an irreplaceable role in ensuring national food security and participates in critical ecological processes. Deyeuxia angustifolia is a constructive and dominant plant in typical meadows and swampy meadows wetlands in the Sanjiang Plain, and a dominant or subdominant species in swamp vegetation (Li et al., 2019b). Many studies have been carried out in Deyeuxia angustifolia wetland in the Sanjiang Plain, mainly focusing on wetland carbon flux (Wang et al. 2017), plant diversity (Dai et al. 2010), and plant photosynthesis (Xu et al. 2020). However, the research on the response of microbial community structure and function to nitrogen deposition still poorly understands. Therefore, our study employed high-throughput sequencing technology to study changes in soil microbial community structure and its relationship with soil physical and chemical properties in the Sanjiang Plain wetland under different nitrogen deposition levels. Our study thus provides a scientific basis for the development of effective wetland management strategies in the Sanjiang Plain to ensure the sustainable development of this region.
Results
Changes in soil physical and chemical properties under different nitrogen deposition treatments
Table 1 summarizes the soil physical and chemical properties of the simulated nitrogen deposition treatments. Soil pH, TN, NH4, NO3, and AP changed significantly, whereas SOC and TP remained unaffected. The results of the soil physical and chemical property analyses of the simulated nitrogen deposition plots indicated that soil pH and available phosphorus decreased with increased nitrogen application (Table 1). In contrast, total nitrogen, nitrate-nitrogen, and ammonium nitrogen contents increased gradually with increasing nitrogen deposition.
Bacterial α- and β-diversities under different nitrogen deposition treatments
The Observed OTUs, Chao1 index, and Shannon diversity index of soil bacteria did not change significantly in the LN treatment, whereas the HN treatment did exhibit significant changes compared with CK (Table 2). According to the Table 2, the LN treatment significantly increased the OTUs, Shannon, and Chao1 indices of soil bacteria but HN treatment significantly decreased the OTUs, Shannon and Chao1 indices of soil bacteria.
Principal coordinates analysis (PCoA) illustrates the variations in beta-diversity of the bacterial communities (Fig. 1). The soil bacterial community of HN was significantly different from that of the CK and LN treatments, whereas the soil bacterial communities of CK and LN were similar. According to principal coordinates analysis (PCoA), PC2 and PC3 explained 35.10% and 8.66% of the variation in soil bacteria, respectively, accounting for a total of 43.76%. The bacterial community structure between CK, LN, and HN changed significantly (permanova R2 = 0. 83, P < 0.05)
Variation of soil bacterial composition under different nitrogen deposition treatments
At the phylum level (Fig. 2), the microbial community of the CK, LN, and HN treatments was dominated by Proteobacteria (CK:21.57%, LN:21.34%, HN:21.75%) and Acidobacteria (CK:25.42%, LN:16.47%, HN:21.57%), followed by Firmicutes (CK:13.95%, LN:15.84%, HN:6.39%), Chloroflexi (CK:7.57%, LN:7.46%, HN:20.65%), Bacteroides (CK:12.31%, LN:12.66%, HN:7.18%), and Actinobacteria (CK:5.51%, LN:7.79%, HN:7.17%). Additionally, Verrucomicrobia and Gemmatimonadetes were also detected in the three nitrogen deposition treatments with relative abundances exceeding 2% (Fig. 2). According to the Fig. 2, the Acidobacteria, Proteobacteria, Firmicutes, Chloroflexi, Bacteroides, Actinobacteria, Verrucomicrobia, Gemmatimonadetes, and Cyanobacteria were all changed significantly under N deposition treatments. Addition, compared with CK, the N depositions decreased the abundance of Acidobacteria and Nitrospirae, while increased the abundance of Proteobacteria and Chloroflexi (Fig. 2). The abundance of Firmicutes increased in LN and then decreased significantly in the HN (P < 0.05). HN treatment significantly increased the relative abundance of Chloroflexi (P < 0.05) and decreased the relative abundance of Bacteroidetes (P < 0.001). Moreover, all the bacterial phyla (except unclassified species and Rokubacteria) differed significantly among the three nitrogen deposition treatments (P < 0.05) (Fig. 2).
At the genus level (Fig. 3), the bacterial community of the CK, LN, and HN treatments was dominated by Acinetobacter (CK:5.76%, LN:8.26%, HN:11.23%) and Nitrobacter (CK:3.17%, LN:4.69%, HN:5.78%), followed by Bryobacter (CK:2.31%, LN:3.02%, HN:2.76%) and Bradyrhizobium (CK:2.84%, LN:2.49%, HN:3.14%).
According to one-way ANOVA and compared with CK, the relative abundance of Acinetobacter, Bryobacter, and Nitrobacter significantly increased within LN and HN, but Bradyrhizobium decreased in the LN and increased significantly in the HN. In contrast, HA17, Variibacter, Acidothermus, Asticcacaulis, A21b, Geobacter, Subgroup-2, Caulobacter, RBG13, Erythrobacter, Pseudomonas, and Caditatus decreased significantly within LN and HN treatments compared with CK treatment.
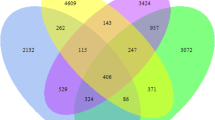
The structures of the bacterial communities, particularly the unique and shared OTUs among the different soil samples, were visualized using Venn diagrams. A total of 1381 OTUs (account for 82.01%) were shared between all the soil samples (Fig. 4). Among the remaining OTUs, 37 were only identified in CK, accounting for 2.20%, 39 in 99 in LN, accounting for 5.88%, 32 in HN, accounting for 1.90% (Fig. 4).
Heatmap analysis using the 30 most abundant bacterial genera indicated that the genera Eschericha-Shigella, Megamones, Bifidobaterium, Bacteroides, Holdemanella, and Subdoligranulum were dominant in the CK (Fig. 5). Furthermore, the LN was dominated by Bradyrhizobium, Bryobacter, and Geobacter (Fig. 5). Nicotiana_otophora, Sphingomonas, and RB 41 were the most dominant genera in the HN (Fig. 5).
Relationship between soil physicochemical properties and bacterial communities
Pearson correlation analysis was used to detect the correlation between soil bacteria α-diversity and soil physicochemical factors. The results indicated that soil Shannon diversity was significantly correlated with NO3 and NH4 (P < 0.05), whereas other soil physicochemical properties did not correlate with soil α-diversity (Table 3).
The cumulative variations in the first and second RDA axes were 35.52% and 29.07, respectively (Fig. 6), which demonstrated that the structure of the bacterial communities was significantly influenced by the physicochemical properties of the soil. As illustrated by the RDA plot (Fig. 6), HN was positively affected by soil TN, TP, NO3, and NH4, whereas LN was positively affected by soil pH, AP, and SOC, and the CK was positively affected by SOC, AP, and pH.
Discussion
Soil pH is one of the most important factors affecting soil microbial diversity and therefore plays an important role in the decomposition and synthesis of soil organic matter and the synthesis and transformation of N, P, and K (Ndour et al. 2007). Our findings indicated that the soil pH of the Deyeuxia angustifolia wetland decreased with increasing nitrogen concentrations, which was mainly due to acidification caused by increases in inorganic nitrogen content in the soil. Increased nitrogen deposition also affected soil nutrient composition. Soil organic carbon (SOC) and nitrogen increased significantly (Table 1) with higher nitrogen deposition, suggesting that nitrogen input can promote carbon sequestration, thus increasing soil organic carbon content. These findings were consistent with those of Chung et al. (2010) and Liao et al. (2020). Further, our results indicated that exogenous nitrogen input increased the available nitrogen of the wetland ecosystem and therefore has an important effect on the SOC content of wetland soil. Additionally, increases in exogenous nitrogen input also enhanced the nitrate and ammonium nitrogen content in the soil, promoted the mineralization rate of soil nitrogen, and increased the content of mineral nitrogen in the soil. These findings were consistent with those of previous studies, thus confirming that soil nitrate nitrogen and ammonium nitrogen increase with exogenous nitrogen input until soil nitrogen saturation is reached (Zhou et al. 2017; Yan et al. 2020). Other similar studies evaluated the early effects of simulated nitrogen deposition on available nitrogen content in south subtropical forest soils. These studies found that higher atmospheric nitrogen deposition rates proportionately increased the available N content in soil (Boot et al. 2016; Cheng et al. 2020).
Diversity indices are important indicators of the richness and diversity of soil microbial communities (Haidich and Ioannidis 2004; Sui et al. 2016). Our findings indicated that soil bacterial diversity changed significantly under different nitrogen deposition conditions. The Shannon index exhibited a slight increase in the LN treatment and a significant decrease in the HN treatment. Further, our results suggested that the effects of nitrogen deposition on bacterial diversity were largely dependent on the levels of nitrogen input, as demonstrated by the increases and decreases in the abundance of specific bacterial taxa depending on the nitrogen treatment (Freitag et al. 2005). This may be because little nitrogen deposition increases the soil nutrient content, and changes the soil nutrition supply, thus promoting bacterial growth. However, adding high concentration nitrogen will restrict the bacterial abundance and Shannon diversity because increasing nitrogen alters the availability of nutrients in the soil, which leads to the growth of some bacterial populations, but inhibits other populations, thus resulted in the Shannon diversity decreasing. Therefore, a certain concentration of nitrogen would promote the growth of bacteria, which is consistent with the findings of another study conducted in the Stipa baicalensis Steppe (Liu et al. 2019). Further, this decrease in diversity may be due to soil acidification resulting from long-term N deposition in the supersaturated state (Chung et al. 2010; Wang et al. 2018a, 2018b). A decrease in microbial diversity with high N deposition was also found in previous studies (Zhou et al. 2015; Zhang et al. 2018; Lu et al. 2020). Our findings indicated that soil pH was significantly and positively correlated with soil bacterial alpha diversity. Soil pH is a major modulator of soil microbial community diversity and composition in diverse ecosystems (Rousk et al. 2010; Zhang et al. 2015; Lammel et al. 2018). Soil microbial communities are directly affected by soil pH, as different soil microbial taxa have different optimal pH ranges that promote their growth (Rousk et al. 2010; Krulwich et al. 2011). In general, high nitrogen deposition in some areas of China will likely impact soil microbial community structures and further affect the stability of the ecosystem (Wang et al. 2018a, 2018b; Clare et al. 2019).
Soil physico-chemistry properties strongly influence the structure and diversity of bacterial communities. Here, soil bacterial community structure exhibited a positive correlation with TN, TP, NO3, NH4, and SOC, which was consistent with the findings of Frey et al. (2020) and Sui et al. (2021). Other studies have demonstrated that several physicochemical characteristics of soil including moisture (Bi et al. 2012), pH (Siles and Margesin 2016), organic carbon (Yang et al. 2018), and C/N ratio (Luo et al. 2017) substantially affect the composition of bacterial communities. Our findings also confirmed that the composition of soil bacteria at the OTU level was significantly correlated with soil nutrients (Fig. 6).
Nitrogen is one of the limiting factors of wetland ecosystems and increases in nitrogen deposition will likely change the relative abundance of dominant soil bacteria (Huang et al. 2020). In this study, Proteobacteria and Acidobacteria were the dominant species in the three treatments. Many other studies have also shown that the main bacterial communities in wetland soils are Acidobacteria and Proteobacteria (Turlapati et al. 2013; Cederlund et al. 2014). With increased nitrogen deposition, the relative abundance of Acidobacteria significantly increased in LN but significantly decreased in HN. The relative abundance of Chloroflexi increased gradually and significantly under both the LN and HN treatments. The relative abundance of Firmicutes and Bacteroidetes decreased gradually but was only significantly lower under the HN treatment. Previous studies suggest that exogenous nitrogen addition reduces bacterial diversity and changes bacterial community composition, which in turn changes the relative abundance of the dominant phyla (Jangid et al. 2008; Campbell et al. 2010; Freedman et al. 2015). The nutrient hypothesis proposed by Fierer et al. (2012) could explain the role of nitrogen addition in soil bacterial community dynamics. Given that the members of the Actinobacteria and Proteobacteria phyla are eutrophic microorganisms, their growth rate and relative abundance increase in eutrophic environments. However, Acidobacteria and Firmicutes are generally considered oligotrophic microorganisms, and therefore, their growth rate and relative abundance decrease in eutrophic environments, which is consistent with the results of this study. Our redundancy analyses indicated that the main factors affecting bacterial community structure were ammonium nitrogen, nitrate-nitrogen, and soil pH, which confirmed the critical role of soil physical and chemical properties in determining the structures of microbial communities (Lamb et al. 2011; Ren et al. 2018; Sui et al. 2021).
As decomposers, soil microorganisms play an extremely important role in the material cycle of wetland ecosystems, and the diversity of soil microorganisms is also a key factor affecting the function of terrestrial ecosystems (Copley 2000). Wetland ecosystem plays a very important role on control nitrogen deposition and global change. Depend on this study, long-term nitrogen deposition leads to changes in soil bacterial diversity and composition. With the increase of China’s population, the reduction of cultivated land, and the extensive use of chemical fertilizers, the functions of wetland ecosystems will change significantly. But at present, there are relatively few studies on wetlands, and the study on nitrogen deposition is even weaker. This is because wetlands, compared with ecosystems such as forests and grasslands, have special environments and seasonal or perennial water accumulation. Therefore, the study on wetlands in different locations and different seasons will vary greatly. Therefore, the study of soil microorganisms in wetland is a very complex and systematic project, and its research needs to be accumulated from all aspects, and should be infiltrated and joined by multiple disciplines. Under the background of increasing atmospheric nitrogen deposition, nitrogen deposition in Northeast of China will increase. Thus, carrying out research on the response of wetland soil bacterial community composition and structure to nitrogen deposition is helpful for studying the future climate change on wetland ecosystem pattern and functional stability.
Conclusions
In general, low nitrogen deposition can positively increase the α-diversity of soil bacteria and high nitrogen deposition can negatively decrease the α-diversity of soil bacteria in long-term nitrogen deposition in the Deyeuxia angustifolia wetland of the Sanjiang Plain. Soil NH4 and NO3 were key soil physicochemical properties affecting the soil Shannon diversity. PCoA indicated that LN significantly changed the structure of the soil bacterial community. Soil pH, SOC, TN, NH4, NO3, TP, and AP were significantly correlated with the structure of the soil bacterial communities of the HN and CK treatments. These results provide a theoretical basis that would facilitate the study of future climate change impacts on wetland ecosystem diversity and functional stability.
Materials and methods
Survey of the study area
The present study was conducted at the Sanjiang Plain Wetland Ecological Positioning Research Station of the Institute of Nature and Ecology of the Chinese Academy of Sciences in Heilongjiang (Fig. 5). The average annual temperature in this region is 1.9°C, the effective accumulated temperature above 10°C is 2165–2624°C, the annual precipitation is 585 mm (concentrated in 7–9 months), and the annual evaporation is 1166 mm. The wetland is dominated by Deyeuxia angustifolia, Glyceria spiculosa, Carex lasiocarpa, and Carex pseudocuraica. The main soil types are meadow marsh soil and peat marsh soil. The sample plot was located in a Deyeuxia angustifolia marsh meadow wetland. The water accumulated in the sample plot was concentrated in July and August, and the depth of the accumulated water was 10–20 cm (Fig. 7).
Experimental design
This study employed a long-term simulated nitrogen deposition test platform at the Sanjiang Plain Wetland Ecological Positioning Research Station of the Institute of Nature and Ecology, Heilongjiang Academy of Sciences. The experiment began in 2010 and nine plots were established. The plots (6 m × 4 m) were separated by a one-meter buffer zone to prevent increased horizontal movement and lateral loss of N. Three levels of N fertilization were set, including the control (CK; 0 kg N hm-2·a-1), low nitrogen (LN; 40 kg N hm-2·a-1), and high nitrogen (HN; 80 kg N hm-2·a-1). Each treatment consisted of three replicates that were used for microbial analysis. For each fertilization event, NH4NO3 was first dissolved in pure water and then sprayed evenly on the target area. The same amount of pure water without nitrogen was sprayed in the control area. The treatments were applied once a month from May to October every year.
Soil sample collection and analysis
In June 2019, nine sampling sites were selected randomly to remove the litter layer, after which 0–20 cm of the surface soil layer was sampled using a 7-cm diameter soil drill. Three samples were taken from each site, after which they were mixed and sifted to remove fine roots and stones. The soil samples were then divided into two equal parts. One portion was dried to determine the physical and chemical properties of the soil, and the other was stored in a −20°C freezer for analysis of soil microorganisms. A fresh soil sample equal to 20 g of dry soil was then placed in a shaker, and 50 ml of 2 mol/l potassium chloride solution was added. The preparation was then left undisturbed for 2 h. The filtrate was used for the determination of ammonium nitrogen (indophenol blue colorimetric method) and nitrate-nitrogen (UV photometer method). A fresh soil sample with a mass equal to 5 g of dry soil was placed in a jar, after which 50 ml of deionized water was added. The filtrate was then shaken for 30 min. Dissolved organic carbon (DOC) was determined using a Type Liqui-TOC II TOC analyzer, and the soil total carbon and total nitrogen were determined using a Vario MACRO CUBE analyzer. The air-dried soil was then passed through a 2-mm sieve and mixed with 25 ml of distilled water in a 50-ml beaker. The soil was stirred with a magnetic stirrer for 2 min and left undisturbed for 30 min. The pH value of the soil was measured with a Sartorius PB-10 pH meter. Total phosphorus was determined via sulfuric acid-perchloric acid dissolution anti-Mo-Sb colorimetry. Available phosphorus was determined by colorimetry using a 0.5 mol/l sodium bicarbonate antimony extract.
Soil DNA extraction
The total DNA of soil microorganisms was extracted using the QIAGEN DNeasy PowerSoil Pro Kit according to the manufacturer’s instructions. The bacterial 16S rRNA region was amplified using the 338F and 806R universal primers for the V3–V4 region of the bacterial 16S rRNA gene. Each sample had a different 6 bp barcode primer at the 5′ end of primer to distinguish the different samples. The PCR amplification protocol consisted of an initial denaturation stage at 94°C for 10 min, followed by 30 cycles of 90°C for 60 s, 55°C for 60 s, and 72°C for 60 s, and finally 72°C for 10 min. The product was purified using the QIAquick PCR Purification Kit (QIAGEN). The PCR products of different samples were mixed and sequenced by BMC Biotechnology Co., Ltd., using an Illumina MiSeq sequencer.
Illumina high-throughput data processing
The raw sequences were processed using QIIME2. Forward and reverse reads were merged by using FLASH software (Caporaso et al. 2010). “Trim” was used to remove low-quality sequences with length less than 150 bp as well as a mean quality score less than 20 and sequences containing anonymous nucleotides (Bolger and Usadel 2014). The chimeric sequences were identified and removed with Uchime algorithm to obtain high-quality sequences (Edgar et al. 2011). The 97% similarity optimization sequence is divided into operable taxonomic units (OTU, Optional Taxonomic Units) through UPARSE (V.7.0.1090) for OTU cluster analysis. Operational taxonomic identities were determined using QIIME by executing the BLAST algorithm against sequences in the SILVA database (https: //www.arb-silva) (Schloss 2017). “alpha_diversity.py” was used to calculate the Observed OTUs, Chao1, and Shannon index to reflect the α diversity of the bacterial community, then used “beta_diversity.py” to calculate the Bray-Curtis distance matrix to reflect the β diversity of the bacterial community.
Statistical analysis
One-way ANOVA was used to analyze the effects of different nitrogen deposition treatments on soil physical and chemical properties and microbial community structure and α diversity indices by using SPSS (19.0). Correlation analysis between soil physical and chemical properties and bacterial alpha diversity was conducted using SPSS (19.0). The results were considered statistically significant when P < 0.05. α and β diversity were performed using the “vegan” package in R. Principal coordinates analysis (PCoA) based on Bray-Curtis matrices was carried out to characterize the composition of the soil bacterial community using the “vegan” R package. PERMANOVA was used to evaluate the varieties in the community structure of soil bacteria with the function adonis in the “vegan” package in R (version 3.1; R Development Core and Team, 2015). Venn and Heatmap were performed in the “vegan” package in R based on OTU level (version 3.1; R Development Core and Team, 2015). Redundancy analysis (RDA) was used to elucidate the relationship between soil physical and chemical properties and microbial community composition.
Availability of data and materials
The original data is recorded in an Excel named “Data record sheet” and has been attached to this article.
Abbreviations
- CK:
-
Control
- LN:
-
Low nitrogen treatment
- HN:
-
High nitrogen treatment
- RDA:
-
Redundancy analysis
- SOC:
-
Soil organic carbon
- TN:
-
Total nitrogen
- NH4 :
-
Ammonium nitrogen
- NO3 :
-
Nitrate nitrogen
- TP:
-
Total phosphorus
- AP:
-
Available phosphorus
References
Bai YF, Wu JG, Clark CM, Naeem S, Pan QM, Huang JH, Zhang LX, Han XG (2010) Tradeoffs and thresholds in the effects of nitrogen addition on biodiversity and ecosystem functioning: evidence from inner Mongolia Grasslands. Glob Chang Biol 16:358–372. https://doi.org/10.1111/j.1365-2486.2009.02142.x
Bassiri RH (2015) Consequences of atmospheric nitrogen deposition in terrestrial ecosystems, old questions, new perspectives. Oecologia 177(1):1–3. https://doi.org/10.1007/s00442-014-3116-2
Bi J, Zhang N, Liang Y, Yang H, Ma K (2012) Interactive effects of water and nitrogen addition on soil microbial communities in a semiarid steppe. J Plant Ecol 5(3):320–329
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illuminasequence data. Bioinformatics 30(15):2114–2120. https://doi.org/10.1111/j.1365-2486.2009.02142.x
Boot CM, Hall EK, Denef K, Baron JS (2016) Long-term reactive nitrogen loading alters soil carbon and microbial community properties in a subalpine forest ecosystem. Soil Biol Biochem 92:211–220. https://doi.org/10.1016/j.soilbio.2015.10.002
Campbell BJ, Polson SW, Hanson TE, Mack MC, Schuur EAG (2010) The effect of nutrient deposition on bacterial communities in Arctic tundra soil. Environ Microbiol 12(7):1842–1854. https://doi.org/10.1111/j.1462-2920.2010.02189.x
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Peña AG, Goodrich JK, Gordon JI, Huttley GA, Kelley ST, Knights D, Koenig JE, Ley RE, Lozupone CA, McDonald D, Muegge BD, Meg P, Reeder J, Sevinsky JR, Turnbaugh PJ, Walters WA, Widmann J, Yatsunenko T, Zaneveld J, Knight R (2010) QIIME allows analysis of high-throughput community se-quencing data. Nat Methods 7(5):335–336. https://doi.org/10.1038/nmeth.f.303
Cederlund H, Wessén E, Enwall K, Jones CM, Juhanson J, Pell M, Philippot L, Hallin S (2014) Soil carbon quality and nitrogen fertilization structure bacterial communities with predictable responses of major bacterial phyla. Appl Soil Ecol 84:62–68. https://doi.org/10.1016/j.apsoil.2014.06.003
Cheng Y, Wang J, Wang JY, Wang SQ, Chang SX, Cai ZC, Zhang JB, Niu SL, Hu SJ (2020) Nitrogen deposition differentially affects soil gross nitrogen transformations in organic and mineral horizons. Earth Sci Rev 201. https://doi.org/10.1016/j.earscirev.2019.103033
Chung H, Zak DR, Reich PB, Ellsworth DS (2010) Plant species richness, elevated CO2, and atmospheric nitrogen deposition alter soil microbial community composition and function. Glob Chang Biol 13(5):980–989. https://doi.org/10.1111/j.1365-2486.2007.01313.x
Clare EK, Charlotte ER, Peter BR, Sarah EH (2019) Long-term nitrogen addition does not increase soil carbon storage or cycling across eight temperate forest and grassland sites on a sandy outwash plain. Ecosystems 22(7):1592–1605. https://doi.org/10.1007/s10021-019-00357-x
Copley J (2000) Ecology goes underground. Nature 406(6795):452–455. https://doi.org/10.1038/35020131
Dai HY, Sui X, Fu XY, Zhang RT (2010) Effect of community diversity of Deyeuxiaangustifolia and CO2 emission flux under nitrogen deposition conditions in Sanjiang plain. Northern Horticult 08:123–128. https://doi.org/10.1371/journal.pone.0066563
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27(16):2194–2200. https://doi.org/10.1093/bioinformatics/btr381
Fierer N, Lauber CL, Ramirez KS, Zaneveld J, Bradford MA, Knight R (2012) Comparative metagenomic, phylogenetic and physiological analyses of soil microbial communities across nitrogen gradients. ISME J 6(5):1007–1017. https://doi.org/10.1038/ismej.2011.159
Freedman ZB, Romanowicz KJ, Upchurch RA, Zak DR (2015) Differential responses of total and active soil microbial communities to long-term experimental N deposition. Soil Biol Biochem 90:275–282. https://doi.org/10.1016/j.soilbio.2015.08.014
Freitag TE, Chang L, Clegg CD, Prosser JI (2005) Influence of inorganic nitrogen management regime on the diversity of nitrite-oxidizing bacteria in agricultural grassland soils. Appl Environ Microbiol 71(12):8323–8334. https://doi.org/10.1128/AEM.71.12.8323-8334.2005
Frey B, Carnol M, Dharmarajah A, Brunner I, Schleppi P (2020) Only minor changes in the soil microbiome of a sub-alpine forest after 20 years of moderately increased nitrogen loads. Front Forests Global Change 3:77. https://doi.org/10.3389/ffgc.2020.00077
Galloway JN, Townsend AR, Erisman JW, Bekunda M, Cai ZC, Freney JR, Martinelli LA, Seitzinger SP, Sutton MA (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320:889–892. https://doi.org/10.1126/science.1136674
Haidich AB, Ioannidis JPA (2004) The Gini coefficient as a measure for understanding accrual inequalities in multicenter clinical studies. J Clin Epidemiol 57(4):341–348. https://doi.org/10.1016/j.jclinepi.2003.09.011
Hiiesalu I, Paertel M, Davison J, Gerhold P, Metsis M, Moora M, Öpik M, Vasar M, Zobel M, Wilson SD (2014) Species richness of arbuscular mycorrhizal fungi: associations with grassland plant richness and biomass. New Phytol 203(1):233–244. https://doi.org/10.1111/nph.12765
Huang LB, Bai JH, Wen XJ, Zhang GL, Zhang CD, Cui BS, Liu XH (2020) Microbial resistance and resilience in response to environmental changes under the higher intensity of human activities than global average level. Glob Chang Biol 26:2377–2389. https://doi.org/10.1111/gcb.14995
Jangid K, Williams MA, Franzluebbers AJ, Sanderlin JS, Reeves JH, Jenkins MB, Endale DM, Coleman DC, Whitman WB (2008) Relative impacts of land-use, management intensity and fertilization upon soil microbial community structure in agricultural systems. Soil Biol Biochem 40(11):2843–2853. https://doi.org/10.1016/j.soilbio.2008.07.030
Krulwich TA, Sachs G, Padan E (2011) Molecular aspects of bacterial pH sensing and homeostasis. Nat Rev Microbiol 9:330–343. https://doi.org/10.1038/nrmicro2549
Lamb EG, Kennedy N, Siciliano SD (2011) Effects of plant spe-cies richness and evenness on soil microbial community diversity and function. Plant Soil 338:483–495. https://doi.org/10.1007/s11104-010-0560-6
Lammel DR, BarthG OO, Cruz LM, Zanatta JA, Ryo M, Souza EM, Pedrosa FO (2018) Direct and indirect effects of a pH gradient bring insights into the mechanisms driving prokaryotic community structures. Microbiome 6(1):106. https://doi.org/10.1186/s40168-018-0482-8
Li YB, Bezemer TM, Yang JJ, Lü XT, Li XY, Liang WJ, Han XG, Li Q (2019a) Changes in litter quality induced by N deposition alter soil microbial communities. Soil Biol Biochem 130:33–42. https://doi.org/10.1016/j.soilbio.2018.11.025
Li ZF, Lu XG, Yang Q (2019b) Soil-surface CO2 fluxes in a Deyeuxia angustifolia wetland in Sanjiang Plain, China. Wetl Ecol Manag 13(1):35–41. https://doi.org/10.1007/s11273-003-5041-8
Liao S, Tan SY, Peng Y, Wang DY, Yang YS (2020) Increased microbial sequestration of soil organic carbon under nitrogen deposition over China’s terrestrial ecosystems. Ecol Process 9(1):271–281. https://doi.org/10.1186/s13717-020-00260-7
Liu CX, Dong YH, Sun QW, Jiao RZ (2016) Soil bacterial community response to short-term manipulation of the nitrogen deposition form and dose in a Chinese fir plantation in Southern China. Water Air Soil Pollut 227(12):447. https://doi.org/10.1007/s11270-016-3152-0
Liu HM, Yang DL, Zhang HF, Zhao JN, Wang H, Zhang NQ (2019) Effects of nitrogen addition on the soil bacterial community structure of Stipa baicalensis steppe. Acta Pratacul Sin 28(09):23–32 https://doi.org/CNKI:SUN:CYXB.0.2019-09-003
Liu WX, Jiang L, Yang S, Wang Z, Tian R, Peng ZY, Chen YL, Zhang XX, KuangJL LN, Wang SP, Liu LL (2020) Critical transition of soil bacterial diversity and composition triggered by nitrogen enrichment. Ecology 101(8):e03053. https://doi.org/10.1002/ecy.3053
Lu GR, Xie BH, Cagle GA, Wang XH, Han GX, Wang XJ, Hou AX, Guan B (2020) Effects of simulated nitrogen deposition on soil microbial community diversity in coastal wetland of the Yellow River Delta. Sci Total Environ 757. https://doi.org/10.1016/J.SCITOTENV.2020.143825
Luo S, Wang S, Tian L, Li S, Li X, Shen Y, Tian C (2017) Long-term biochar application influences soil microbial community and its potential roles in semiarid farmland. Appl Soil Ecol 117:10–15. https://doi.org/10.1016/j.apsoil.2017.04.024
Mo JM, Brown S, Xue JH, Fang YT, Li ZA (2006) Response of litter decomposition to simulated N deposition in disturbed, rehabilitated and mature forests in subtropical China. Plant Soil 282:135–151. https://doi.org/10.1007/s11104-005-5446-7
Ndour NYB, Achouak W, Christen R, Heulin T, Brauman A, Chotte JL (2007) Characteristics of microbial habitats in a tropical soil subject to different fallow management. Appl Soil Ecol 38(1):51–61. https://doi.org/10.1016/j.apsoil.2007.09.001
Ren BH, Hu YM, Chen BD, Zhang Y, Thiele J, Shi RJ, Liu M, Bu RC (2018) Soil pH and plant diversity shape soil bacterial community structure in the active layer across the latitudinal gradi-ents in continuous permafrost region of northeastern china. Sci Rep 8(1):5619. https://doi.org/10.1038/s41598-018-24040-8
Rousk J, Baath E, Brookes PC, Lauber CL, Lozupone C, Caporaso JG, Knight R, Fierer N (2010) Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J 4:1340–1351. https://doi.org/10.1038/ismej.2010.58
Schloss PD (2017) The effects of alignment quality, distance calculation method, sequence filtering, and region on the analysis of 16S rRNA gene-based studies. PLoS Comput Biol 6(7):e1000844. https://doi.org/10.1371/journal.pcbi.1000844
Siles JA, Margesin R (2016) Abundance and diversity of bacterial, archaeal, and fungal communities along an altitudinal gradient in alpine forest soils: what are the driving factors? Microb Ecol 72(1):207–220. https://doi.org/10.1007/s00248-016-0748-2
Sui X, Zhang RT, Frey B, Yang LB, Liu YN, Ni HW, Li MH (2021) Soil physicochemical properties drive the variation in soil microbial communities along a forest successional series in a degraded wetland in northeastern China. Ecol Evol 11(5):2194–2208. https://doi.org/10.11829/j.issn.1001-0629.2015-0444
Sui X, Zhang RT, Yang LB, Xu N, Chai CR, Wang JF, Fu XL, Zhong HX, Xing JH, Zhang Y, Ni HW (2016) Effect of simulation nitrogen depositions on bacterial diversity of Deyeuxia angustifolia in wetland of Sanjiang Plain. Pratacult Sci 33(4):589–598. https://doi.org/10.11829/j.issn.1001-0629.2015-0444
Tian HQ, Melillo J, Lu CQ, Kicklighter D, Liu ML, Ren W, Xu XF, Chen GS, Zhang C, Pan SF, Liu JY, Running S (2011) China’s terrestrial carbon balance: contributions from multiple global change factors. Glob Biogeochem Cycles 25(1):GB1007. https://doi.org/10.1029/2010GB003838
Turlapati SA, Minocha R, Bhiravarasa PS, Tisa LS, Thomas WK, Minocha SC (2013) Chronic N-amended soils exhibit an altered bacterial community structure in Harvard Forest, MA, USA. FEMS Microbiol Ecol 83(2):478–493. https://doi.org/10.1111/1574-6941.12009
Wang C, Liu DW, Bai E (2018a) Decreasing soil microbial diversity is associated with de-creasing microbial biomass under nitrogen addition. Soil Biol Biochem 120:126–133. https://doi.org/10.1016/j.soilbio.2018.02.003
Wang C, Lu XK, Mori T, Mao QG, Zhou GY, Nie YX, Mo JM (2018b) Responses of soil microbial community to continuous experimental nitrogen additions for 13 years in a nitrogen-rich tropical forest. Soil Biol Biochem 121:103–112. https://doi.org/10.1016/j.soilbio.2018.03.009
Wang JB, Zhu TC, Ni HW, Zhong HX, Fu XL, Wang JF (2017) Effects of elevated CO2 and nitrogen deposition on ecosystem carbon fluxes on the Sanjiang plain wetland in Northeast China. PLoS One 8(6):e66563. https://doi.org/10.1371/journal.pone.0066563
Wei HW, Wang XG, Li YB, Yang JJ, Wang JF, Lü XT, Han XG (2020) Simulated nitrogen deposition decreases of soil microbial diversity in a semiarid grassland, with little mediation of this effect by mowing. Pedobiologia 8. https://doi.org/10.1016/j.pedobi.2020.150644
Xu N, Ding JN, Wu YN, Li JB, Wang JB, Zhong HX, Ni HW, Hou F, Yu SP, Zhang HH (2020) Systemic signaling in the photosynthetic induction phase of Calamagrostis angustifolia on the Sanjiang plain wetland of Northeast China. Int J Agricult Biol 23(2):493–500. https://doi.org/10.17957/IJAB/15.1313
Yan GY, Xing YJ, Han SJ, Zhang JH, Wang QG, Mu CC (2020) Long-time precipitation reduction and nitrogen deposition increase alter soil nitrogen dynamic by influencing soil bacterial communities and functional groups. Pedosphere 30(3):363–377. https://doi.org/10.1016/S1002-0160(19)60834-9
Yang LB, Liu YN, Sui X, Zhang T, Xu N, Ni HW, Wu YN (2018) Effects of different land uses on community structure of soil arbuscular mycorrhizal fungi in Sanjiang Wetlands. Bangladesh J Bot 47:689–697
Yao MJ, Rui JP, Li JB, Dai YM, Bai YF, Heděnec P, Wang JM, Zhang SH, Pei KQ, Liu C, Wang YF, He ZL, Frouz J, Li XZ (2014) Rate-specific responses of prokaryotic diversity and structure to nitrogen deposition in the Leymus chinensis steppe. Soil Biol Biochem 79:81–90. https://doi.org/10.1016/j.soilbio.2014.09.009
Zhang TA, Chen HYH, Ruan HH (2018) Global negative effects of nitrogen deposition on soil microbes. ISME J 12(7). https://doi.org/10.1038/s41396-018-0096-y
Zhang XM, Liu W, Zhang GM, Jiang L, Han XG (2015) Mechanisms of soil acidification reducing bacterial diversity. Soil Biol Biochem 81:275–281. https://doi.org/10.1016/j.soilbio.2014.11.004
Zhou J, Guan DW, Zhou BK, Zhao BS, Ma MC, Qin J, Jiang X, Chen SF, Cao FM, Shen DL, Li J (2015) Influence of 34-years of fertilization on bacterial communities in an intensively cultivated black soil in northeast China. Soil Biol Biochem 90:42–51. https://doi.org/10.1016/j.soilbio.2015.07.005
Zhou ZH, Wang CK, Zheng MH, Jiang LF, Luo YQ (2017) Patterns and mechanisms of responses by soil microbial communities to nitrogen addition. Soil Biol Biochem 115:433–441. https://doi.org/10.1016/j.soilbio.2017.09.015
Acknowledgements
We are grateful to reviewer to give many considerable comments on this manuscript and to “Scientific Manuscript Editing” for help with linguistics processing.
Funding
This work was funded by the Natural Sciences Foundation of Heilongjiang Province (LH2019C076, LH2020C088), the Special Project Foundation of the Heilongjiang Academy of Sciences (KY2021ZR01), and the Outstanding Youth Foundation of the Heilongjiang Academy of Sciences (CXJQ2019ZR01).
Author information
Authors and Affiliations
Contributions
Zhang Rongtao and Ying-Nan Liu performed this experiment, analyzed the data, and wrote this MS, and Hai-Xiu Zhong performed this experiment and help to write this MS; Xiang-Wei Chen and Xin Sui designed this experiment and revised this MS. The author(s) read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study did not violate ethics, and all participants agreed to publish the paper.
Consent for publication
Not applicable.
Competing interests
The authors declare that that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, RT., Liu, YN., Zhong, HX. et al. Effects of simulated nitrogen deposition on the soil microbial community diversity of a Deyeuxia angustifolia wetland in the Sanjiang Plain, Northeastern China. Ann Microbiol 72, 11 (2022). https://doi.org/10.1186/s13213-022-01666-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13213-022-01666-8