Abstract
The paper presents the results of the development of a unit for calculating mass transport in a membrane gas separation apparatus in the Aspen Plus environment. The created user model has been verified by comparing the results of the calculation of separation characteristics with available experimental data for two cases. In the first case, the calculation results were compared with experimental data on the separation of the ternary H2O/EtOH/N2 (13.3/5.5/81.2 mol %) vapor–gas mixture simulating the stream from the stripping column of a laboratory membrane vapor separation unit with a plate-and-frame membrane module for ethanol recovery from fermentation broth. It has been shown that the calculated concentrations of the components in the retentate and permeate streams are close to the experimental values obtained in the separation of the ternary vapor–gas mixture. In the second case, the calculation results were compared with experimental data obtained upon the separation of a binary N2/CH4 gas mixture (99/1 mol %) simulating a stream with a low content of an easily penetrating impurity component using a radial membrane module. It has been found that the calculated concentration of CH4 in the retentate is noticeably less than in the experiment, especially at high stage cuts, a difference that is associated with the increasing influence of the longitudinal mixing of components at low speeds of the mixture in the channels of the membrane module, which deteriorates the separation characteristics of the apparatus with respect to the removal of impurities and requires additional consideration of the deviation from the plug-flow mode in computational mathematical models used for this case.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
INTRODUCTION
Mathematical modeling is a powerful tool in the development, study, and optimization of real technological processes. There are a large number of available software products for the simulation of technological processes that make it possible to build and explore various process flow diagrams. Today, the Aspen Plus software (developed by AspenTech©) is a leader in the field of modeling technological processes of various industries.
Currently observed tightening of requirements for the quality of substances produced by the chemical industry creates a trend towards the development of new technologies and the optimization of existing technologies [1]. In the field of gas separation, especially in the production of high-purity gases, a technology based on membrane gas separation can be considered as an attractive alternative to conventional, energy-intensive processes [2]. The separation of gas mixtures with the use of membranes is provided by the physicochemical properties of the material, the process conditions, and the technological parameters. This process can be carried out at room temperature in the absence of phase transitions, and the hardware design is characterized by simplicity and easy scalability. A variety of studies devoted to the development of new materials and processes have been reported, which show the promise held by this approach to solution of the problems of natural gas purification [3], ammonia recovery [4], and deep gas purification [5] and to invention of new nonstationary membrane separation processes [6]. In recent years, membranes have also been used in the developing area of recovery of bioalcohols (bioethanol, biobutanol). The energy-saving membrane vapor separation method provides concentration of bioalcohols to a high level from dilute aqueous alcohol solutions formed in the fermentation process [7]. In this regard, the implementation of a multiparameter mathematical model that takes into account the engineering and physicochemical aspects of the process is an important task.
To fulfill the task, the Aspen Plus software package was chosen, which has broad functionality and is capable to simulate various chemical engineering schemes and complex technological processes, as well as to calculate energy costs and carry out economic calculations. Aspen Plus contains a voluminous database of physicochemical properties and thermodynamic models, which allow prediction of properties for both gaseous mixtures and liquids. With the use of the latest versions of Aspen Plus, it is possible to integrate design schemes created by the user in Excel [8], thereby greatly expanding the possibilities of modeling nonstandard blocks of the process scheme. Also, custom models made in other software products can be integrated into Aspen Plus. There are examples in the literature of using the software platforms MATLAB [9] and FORTRAN [10] to create a custom model with its subsequent export to Aspen Plus.
Currently, Aspen Plus does not contain a ready solution for modeling mass transport processes in membrane gas separation devices; however, the software company (AspenTech©) provides, among other things, the Aspen Custom Modeler (ACM) modeling tool that focused on creating mathematical models of its own, which can be used to design a custom membrane block. The advantage of the model created in ACM is the ease of exporting and binding it to Aspen Plus, after which it can be used in the same way as other models already available in the Aspen Plus library.
In this study, a gas separation membrane module was created in the ACM program and exported to Aspen Plus for further work. The created block can simulate the operation of a gas separation membrane apparatus in three modes: crossflow, direct flow, and countercurrent flow. For each of the modes, a mathematical model of its own is used. The membrane block is connected to all Aspen Plus thermodynamic packages and can be used not only to calculate the membrane process, but also to work in combination with other processes.
MATHEMATICAL MODEL OF MEMBRANE MODULE
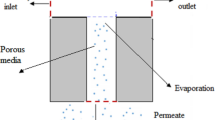
The transfer of gases and vapors through nonporous polymeric materials is described in terms of the solution–diffusion mechanism [11]. To calculate mass transport in the membrane module (Fig. 1), the following equations were used:
—change in a component flux upstream of the membrane:
—change in a component flux downstream the membrane:
—component flux through the membrane:
—driving force for the transport of a component across the membrane:
—mole fraction of the component upstream of the membrane:
—mole fraction of the component downstream of the membrane:
—boundary conditions:
—outlet fluxes:
In this study, a plate-and-frame membrane module was chosen as the object of simulation. This type of module was designed by Stern [12] and was intended for the recovery of helium from natural gas on an industrial scale. The design of the module is similar in configuration to that with flat sheet membranes, which are often used in laboratory experiments in primary research. The module consists of flat sheet membranes sandwiching a spacer made from drainage material [13]. With this design of the module and the feed stream composition, the crossflow rate becomes the main parameter that determines the mass transport in the module.
In the study we considered three operating modes of the membrane module: direct flow, countercurrent flow, and crossflow. Mathematical models of mass transport for all operation modes are based on the following assumptions: isothermal conditions, the absence of a pressure gradient in the channel along the membrane, ideal gas behavior of components, and constancy of membrane permeability. In the cases of direct and countercurrent flows, the plug-flow mode is realized in the feed and permeate channels; whereas the plug-flow mode for crossflow operation is realized in the feed channel and the flow direction is perpendicular to the membrane in the permeate channel.
CREATING A MODEL OF MEMBRANE GAS SEPARATION BLOCK IN ASPEN CUSTOM MODELER
The principle of building a model in ACM is based on writing equations describing the behavior of components or their characteristics in the system under consideration, for example, material balance equations, and any necessary additional conditions (for example, boundary conditions). In the course of building a model, a certain sequence of actions is observed. Work on model building begins with the definition of all parameters and variables involved in the model. Aspen Custom Modeler has its own extensive database of parameters and variables that can be selected in the user interface. Next, mathematical equations are written, which, as a rule, describe the material and heat balances. The order of the description of a variable or parameter can be studied in the ACM assistant. To solve the equations, ACM uses metric units (basic units of measurement); therefore, the user must ensure that the variables, parameters, and coefficients in the model equations are written in basic units. However, the user can select the units for the variables that are displayed in the graphical user interface. This means that the values displayed in graphs and tables are automatically displayed in the current unit of measurement, and the values input into forms are automatically converted to basic units of measurement. After entering the information about the parameters and variables and spelling out mathematical equations, streams are connected to the model; in the given case, the block has three streams: feed stream as the inlet stream and the retentate and permeate as outlet streams. In ACM, streams are denoted through ports in which parameters and variables that carry information, such as flow temperature, pressure, stream component composition, and energy characteristics (not considered in this paper), are built. After the complete information is defined, the model can be tested. To do this, the compiler built into ACM is run, which starts the calculation according to the described model. At this stage, ACM can identify errors resulting from building the model. Table 1 lists the parameters necessary for the calculation of the custom model.
To build a process flowchart using the created block, the user must have the ACM file in which the model was created. To export a model from ACM to Aspen Plus, it is also necessary to have a C++ compiler (Visual C ++ package from Visual Studio©) [14]. In order to add the custom block (Fig. 2) to the Aspen Plus library, it is necessary to enable the option of export to ACM and then run the import option for this model in Aspen Plus. Further, the custom model can be used in the same way as any other elements already available in the Aspen Plus library.
COMPARISON OF CALCULATION RESULTS IN ASPEN PLUS WITH EXPERIMENTAL RESULTS
The calculation block developed was tested on the basis of comparison of the calculated separation characteristics with available experimental data for the following two cases:
—comparison of the calculation results with the experimental data obtained upon the separation of the ternary H2O/EtOH/N2 (13.3/5.5/81.2 mol %) vapor–gas mixture [15] simulating the stream from the stripping column used in ethanol recovery from fermentation broth by membrane vapor permeation in a laboratory plate-and-frame membrane module, with the module operating in the countercurrent mode (the experimental part is detailed in [16]);
—comparison of the calculation results with experimental data obtained upon the separation of a binary N2/CH4 (99/1 mol %) [6] gas mixture simulating a stream with a low concentration of easily penetrating impurity component using a radial membrane module, with the module operating in the countercurrent mode.
The comparison showed that in the case of separation of the ternary vapor–gas mixture, the calculated values for the concentrations of the components in the retentate and permeate streams are basically close to the experimental values (Table 2). However, a noticeable deviation is observed for some of them, which can primarily be due to the influence of longitudinal mixing in the membrane module channels (due to the presence of the net spacer) and is not taken into account in mathematical models used for calculation.
In the case of the removal of the easily permeable impurity component from the binary gas mixture, it was found that the calculated CH4 concentration in the retentate is noticeably lower than the experimental one, especially at low feed flow rates (high stage cuts, Figs. 3, 4). This is apparently due to the influence of the longitudinal mixing of components in the membrane module channels because of a long residence time of the feed upstream of the membrane (tens of seconds), since the diffusion reduces the separation characteristics of the membrane apparatus, a change that is especially noticeable upon the removal of impurities and is not taken into account in mathematical models used for the calculation.
In general, the obtained give evidence for the adequacy of the mathematical models used in the gas separation membrane block developed; the block can be used to build process flowcharts and analyze the characteristics of separation processes.
CONCLUSIONS
A membrane gas separation unit operating in various modes has been developed in the Aspen Plus environment for calculating membrane gas separation processes. The model built is convenient to export to the Aspen Plus library, thereby greatly expanding the capabilities for modeling various processes. The developed unit makes it possible to perform calculations on both gas separation and vapor separation membrane processes. The user is given the opportunity to select the module operation mode for solving a particular separation task. A comparison of the results has shown that the calculated data are close to the experimental data, confirming that the block can be used for modeling membrane gas separation processes.
LIST OF SYMBOLS
B membrane module width, m
J flow rate upstream of membrane, mol/s
J ' flow rate downstream of membrane, mol/s
L membrane module length, m
p pressure upstream of membrane, kPa
p' pressure downstream of membrane, kPa
Q permeability, mol/(m2 s kPa)
с mole fraction
x longitudinal coordinate
SUB/SUPERSCRIPTS
F feed
i component
M membrane
P permeate
R retentate
REFERENCES
V. Abetz, T. Brinkmann, M. Dijkstra, et al., Adv. Eng. Mater. 8, 328 (2006).
V. M. Vorotyntsev, Pet. Chem. 55, 259 (2015).
I. V. Vorotyntsev, A. A. Atlaskin, M. M. Trubyanov, et al., Desalin. Water Treat. 75, 305 (2017).
I. V. Vorotyntsev, D. N. Shablykin, P. N. Drozdov, et al., Pet. Chem. 57, 172 (2017).
A. A. Atlaskin, M. M. Trubyanov, N. R. Yanbikov, et al., Pet. Chem. 58, 508 (2018).
M. M. Trubyanov, P. N. Drozdov, A. A. Atlaskin, et al., J. Membr. Sci. 530, 53 (2017).
M. G. Shalygin, A. A. Kozlova, A. I. Netrusov, and V. V. Teplyakov, Pet. Chem. 56, 984 (2016).
H.-J. Huang, W. Lin, S. Ramaswamy, and U. Tschirner, Appl. Biochem. Biotechnol. 154, 26 (2009).
P. Moein, M. Sarmad, H. Ebrahimi, et al., J. Nat. Gas Sci. Eng. 26, 470 (2015).
G. Ye, D. Xie, W. Qiao, et al., Int. J. Hydrogen Energy 34, 4755 (2009).
J. G. Wijmans and R. W. Baker, Materials Science of Membranes for Gas and Vapor Separation, Ed. by B. Freeman, Yu. Yampolskii, and I. Pinnau (Wiley, Chichester, 2006), p. 159.
R. W. Baker, Membrane Technology and Applications (Wiley, Chichester, 2004), 2nd Ed., Ch. 3, p. 139.
Encyclopedia of Membranes, Ed. by E. Drioli and L. Giorno (Springer, Berlin, 2016), p. 1578.
W. Hensen, Embedding equation oriented models of process unit operations in a sequential modular flowsheet simulator, Master Thesis (Delft, 2005).
V. V. Teplyakov, M. G. Shalygin, A. A. Kozlova, et al., Pet. Chem. 57, 747 (2017).
V. V. Teplyakov, M. G. Shalygin, A. A. Kozlova, and A. I. Netrusov, Pet. Chem. 58, 949 (2018).
ACKNOWLEDGMENTS
The work was supported by the Russian Foundation for Basic Research, project no. 17-38-50100 mol_nr, and the President of the Russian Federation, grant no. MK-2924.2017.8.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by S. Zatonsky
Rights and permissions
About this article
Cite this article
Kozlova, A.A., Trubyanov, M.M., Atlaskin, A.A. et al. Modeling Membrane Gas and Vapor Separation in the Aspen Plus Environment. Membr. Membr. Technol. 1, 1–5 (2019). https://doi.org/10.1134/S2517751619010049
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2517751619010049