Abstract
The use and conservation of agrobiodiversity have become critical to face the actual and future challenges imposed by climate change. Collecting phytogenetic resources is a first step for their conservation; however, the genetic material must be analysed to understand their potential to improve agricultural resilience and adaptation to the new climatic conditions. We have selected nine Phaseolus vulgaris, one P. lunatus and two Vigna unguiculata landraces from two different climatic backgrounds of the Andean region of South Ecuador and one P. vulgaris commercial cultivar, and we grew them under two different conditions of temperature and humidity (open field and greenhouse). Then, we recorded data for 32 characters of plant architecture, flower and fruit characteristics and yield, and 17 events in the phenology of the plants. We analysed the impact of treatment on species, climatic background, and each of the landraces, and identified both characters and landraces that are mostly affected by changes in their environmental conditions. Overall, higher temperatures were benign for all materials except for two P. vulgaris landraces from cold background, which performed better or developed faster under cold conditions. Finally, we calculated a climate resilience landrace index, which allowed us to classify the landraces by their plasticity to new environmental conditions, and found heterogeneous landrace susceptibility to warmer conditions. Two P. vulgaris landraces were highlighted as critical targets for conservation.
Similar content being viewed by others
Introduction
Improving agricultural resilience and adaptation to climate change effects is a priority to ensure crop production and food security in the upcoming years1. Climate change modifies the trends in temperature and precipitation, thus affecting the response to environmental factors of many species at different geographical levels, with drastic effects and, often, a negative impact on crops2,3. However, there is a consensus among different projections that indicate negative impacts of the increase in temperatures on the main crops in many agricultural regions4. Moreover, the more updated models with improved and diverse scenarios produce more pessimistic projections for yield responses for maize, rice, and soy bean, although wheat could benefit from higher CO2 concentrations5. Supporting the latter predictions, the analysis of combined published results of different analytical methods highlights the vulnerability of agriculture to climate change, suggesting a yield reduction of around 3.0 to 6.0% for each degree increase in temperature6. Besides the direct effects of temperature and rainfall on crops and plant diseases7, climate change has important economic consequences on agriculture8. However, the impact of climate change effects on crops depends on both the crop identity and its geographic location9.
Plants exhibit the ability to cope with changes in their environment via phenotypic plasticity10. Therefore, prior to developing crop improvement or conservation programmes, either to face climate change challenges or to choose appropriate crop varieties that might perform well under certain local conditions, it is indispensable to gain knowledge on the plant plasticity and its potential adaptability to abiotic factors11. Moreover, the study of crop phenotypic plasticity must include the analysis of phenology, in addition to other critical traits related to plant morphology and reproduction, because phenology is highly susceptible to changes in the environmental factors12. Thus, the information gathered in this way might allow: (1) to select those varieties suitable for a particular range of environmental conditions, with important consequences for local community development, sustainable agriculture and food security; (2) it might help to identify those varieties more prone to suffer negative effects, and thus, allowing their conservation before their effective loss due to the abandoning of low yield landraces13; (3) the overall information might provide useful to develop specific indexes aimed at quantifying the resilience potential of either one species or landrace or a particular character.
Beans are a major food resource grown worldwide and represent the main source of protein for many societies, thus playing a vital role in the human diet of developing societies14. Beans (Phaseolus spp) originated in the New World. Common bean (P. vulgaris) had two main domestication centres at Middle and Andean South America, with four major genetic groups in Mesoamerica, Colombia, Northern Andes of Ecuador and north Peru, and the southern Andes; and exhibits both a wide morphological variability (> 40,000 varieties), and adaptation to a broad array of environments15,16,17,18. Lima beans (P. lunatus), distributed from northern Mexico to northern Argentina, have three major genetic groups, two in Mesoamerica and one in the Andes (southern Ecuador and northern Peru), which is the most likely origin of the species19,20,21. Moreover, there exists great diversity of wild bean species with potential to improve the resistance to environmental factors in common bean crops22. In addition, Latin America represents about the 50% of the bean world production, followed by Africa; per capita consumption of beans in these regions can oscillate between 12 to 60 kg per year and represent a significant source of protein14,23. In Ecuador, beans, which are commonly named fréjol, fríjol and poroto (P. vulgaris) and torta (P. lunatus), belong to the genus Phaseolus spp., while the cowpeas, named vaina, are Vigna spp., originated in Africa24. Both species represent the main leguminous crops with a soil surface around 32,817 ha devoted for their culture, with an overall production of 27,492 t25. Moreover, as in many other countries, most producers are smallholder farmers that grow beans mainly for self-consume, whether as crop rotation or associated to maize, and thus, beans are an important contribution to Ecuador food sovereignty26.
There are robust projections predicting a generalized decline in crops yield due to the impact of climate change, and highlighting the urgency of further research on the effects of high temperatures and other factors on crops to gain a better understanding on the uncertainties of production impacts5,9,27. We lack specific predictions; however, for the impact of climate change effects on the production of legumes for the Andean region, where a negative impact on the production of cereals is expected9. The effects of climate change on the acceleration of phenological aspects of crops can be counteracted by shifting existing varieties into different regions28. Thus, it is pivotal to promote the investigation of the impact of abiotic stresses on landraces; defined by Casañas et al.29 as those “cultivated varieties that have evolved and may continue evolving, using conventional or modern breeding techniques, in traditional or new agricultural environments within a defined ecogeographical area and under the influence of the local human culture”.
Several studies have focused on the effects that different aspects of climate change and abiotic stress factors have on the common bean. For example, drought, the most extensively studied factor, has drastic effects on legumes because it accelerates plant maturation in Phaseolus spp. and Vigna spp., among other effects, and reduces yield components and biomass14,30,31; elevated CO2 concentrations have direct positive effects on stem mass, and a strong genotype × CO2 interaction for pod number, seed mass and yield on P. vulgaris32; and high temperatures negatively affect reproduction, fertilization, and post-fertilization33; Lima beans (P. lunatus); however, are more tolerant to heat than P. vulgaris14.
In the present work, we have investigated the effects of two different environmental conditions on the architecture, reproduction, yield, and phenology by using standard agromorphologic and phenological descriptors on 12 landraces of P. vulgaris, P. lunatus, and Vigna unguiculata sampled from different localities at the Andes of south Ecuador, and a commercial P. vulgaris cultivar (Supporting Tables S1 and S2; Supporting Fig. S1). Moreover, to test for potential adaptation or conditioning to local environmental factors, four P. vulgaris and the P. lunatus landraces came from cold background locations, and five P. vulgaris and the two V. unguiculata landraces, came from warm background locations. Then, to understand the implications of the results better, we calculated an index, the climate resilience landrace index, with potential application in decision-making.
Results
A summary describing all plant architecture, flower, fruit, and yield, and phenological traits for each of the thirteen Phaseolus sp. and Vigna sp. landraces in the open field and the greenhouse conditions is provided in Supporting Tables S3, S4 and S5. Main effects Kruskal–Wallis tests are summarised in Table 1, and the interactions between treatment conditions (open field and greenhouse) and species, and landrace and climatic background are summarised in Table 2.
I. Plant architecture
Plants under high temperatures and low humidity in the greenhouse exhibited significant higher overall mean rank values than field plants for stem diameter, the degree of branch orientation, composite sheet length and width, and the terminal leaflet length. The size of the angle of the base of the terminal leaflet, however, was bigger in the field (Supporting Tables S3 and Table 1). There were overall significant differences for species and landrace for all studied characters (Table 1). The Kruskal–Wallis analyses of the interactions between treatment (open field vs greenhouse conditions) and species, climatic background, and landrace were significant for all the traits (p-value < 0.001; Table 2).
Post hoc pairwise comparisons for treatment × species interaction (Table 3), found that P. vulgaris plants produced significant higher mean rank values for branch orientation angle in the greenhouse than in the field (median values: 140.00° vs 133.33°). Similarly, P. lunatus plants exhibited significant higher values in the greenhouse for composite sheet length and width and terminal leaflet width (median values: 238.28, 209.95 and 115.26 mm, respectively) than in the field (median values: 208.34, 169.27 and 93.76 mm, respectively); but the terminal leaflet length performed better in the field compared to greenhouse (medians: 62.36 and 52.02 mm). Post hoc pairwise comparisons for treatment × climatic background highlighted that cold background landraces had higher values for branch orientation angle, composite leaf length and width, and terminal leaflet length in the greenhouse than in the field. Cold background landraces produced wider terminal leaflet widths in the greenhouse while warm background landraces did it in the field (Table 3). Post hoc analysis for the treatment × landrace (Table 4) found that P. lunatus had higher mean rank values in the greenhouse than in the open field for composite sheet length (238.28 vs 208.34 mm) and width (209.95 vs 169.27 mm), and terminal leaflet length (115.26 vs 93.76 mm). P. vulgaris landrace 8 failed to grow in the field. Plant architectural traits were only affected in one P. vulgaris (8) and the P. lunatus (13) landraces. P. vulgaris landrace 1, from cold background, performed better in the field than in the greenhouse. Landrace 8, from cold background as well; however, performed better in the greenhouse than in the field, suggesting a wrong identification of its real origin.
II. Flower and fruit characteristics and yield
Greenhouse plants exhibited significant higher overall mean rank values than field plants for the total number of flowers per plant (medians: 57 vs 11 flowers), the number of pods per plant (medians: 34.5 vs 2.0 pods), the number of pods per infructescence (medians: 1.6 vs 1.4 pods), the number of grains per pod (medians: 4.2 vs 0.0 grains), the dry weight of 100 seeds (medians: 37.46 vs 0.0 g), the gross weight of seeds per plant (medians: 24.92 vs 0.0 g), the net weight of seeds per plant (medians: 24.01 vs 0.0 g), and the number of seeds (medians: 60.0 vs 0.0). On the contrary, the sheath width was larger in the field (12.48 mm) than in the greenhouse (11.01 mm; see Supporting Table S4 and Table 1). There were overall significant differences for species and landrace for all characters (Supporting Table S4 and Table 1). For the climatic background of the landraces, all characters exhibited significant differences except for the peduncle length, the number of pods per plant and the number of pods per infructescence.
The Kruskal–Wallis analyses of the treatment × species, treatment × climatic background (except for sheath length) and treatment × landrace interactions were significant for all the traits (Table 2). Post hoc pairwise comparisons for treatment × species interaction found that P. vulgaris, V. unguiculata and P. lunatus produced higher mean rank values in the greenhouse than in the field (Table 3) for: the number of pods per plant (median values: 31.0, 20.5 and 96.0, respectively vs 3.5, 0.0 and 47.5 pods), the gross weight of seeds per plant (medians: 24.04, 17.11 and 251.90 vs 0.00, 0.00 and 112.12 g, respectively), the net weight of seeds per plant (medians: 22.32, 15.25 and 238.78 vs 0.00, 0.00 and 104.80 g, respectively), and the total number of seeds (medians: 36.5,93.5 and 205 vs 0.0, 0.0 and 93.5 seeds, respectively). The sheath width was significantly higher in the field than in the greenhouse (medians: 12.8 and 11.4 mm, respectively), and the scar length mean ranks were higher in the greenhouse than in the field only for P. vulgaris. Then, P. vulgaris and V. unguiculata, produced higher mean rank values for the following characters in the greenhouse than in the field (Table 3) for the number of flowers per plant (medians: 48.0 and 28.0 vs 11 and 7.5 flowers, respectively) and the number of grains per pod (medians: 4.2 and 10.0 vs 0.0 grains, respectively). Similarly, P. vulgaris and P. lunatus exhibited higher mean rank values in the greenhouse than in the field for the weight of 100 seeds (medians: 40.51 and 116.39 vs 0.00 and 109.43 g, respectively).
In the treatment × climatic background, post hoc pairwise comparisons (Table 3) found significant higher mean rank values in landraces from cold background growing in the field than in the greenhouse for chalice length. Warm background landraces produced higher mean rank values in the greenhouse for the number of flowers per plant, the number of pods per plant and infructescence, the number of grains per pod, the 100 seeds weight, the gross and net weight of seeds per plant and the number of seeds. The commercial cultivar exhibited higher median peduncle length in the field than in the greenhouse, and higher median values in the greenhouse than in the field for the number of flowers per plant, the 100 seeds weight, the gross and net weight of seeds per plant and the number of seeds.
In the treatment × landrace, post hoc analysis (Table 4) found that the number of flowers per plant was significantly higher in P. vulgaris landraces 2, 4, 5, 8 and 9, and in V. unguiculata 10. Analogously, more pods per plant were produced in the greenhouse for P. vulgaris landraces 2, 4, 5 and 8, and in V. unguiculata 10. The total number of seeds produced in the greenhouse was higher than in the field for P. vulgaris landraces 2, 4, 5, 6 and 8, both V. unguiculata 10 and 11, and P. lunatus 13. P. vulgaris 3 produced higher mean rank values for the 100 seeds weight in the greenhouse than in the field. P. vulgaris landraces 5 and 6 (both from warm background), however, failed to produce enough flowers, pods, or seeds. Landrace 1, from cold background, failed to prosper in the greenhouse, and landrace 8 did not grow in the field. Otherwise, all other significant values highlighted the positive effects of warmer conditions compared to the field acting on flower, fruit, and yield characteristics (Table 4).
III. Phenology
The treatment had significant overall effects for all the studied characters except for the emergence of hypocotyl, the full flowering when the 50% of the flowers are open, and the 50% of pods ripe (Supporting Tables S5 and Table 1; Fig. 1). Moreover, growing in the open field under colder and more humid conditions than in the greenhouse, delayed the development of the characters except for the finishing of the flowering, which was delayed in the greenhouse (medians: 144 and 123 days, respectively), and the time when pods are fully ripe (medians: 169 and 146 days, respectively). All the three species showed significant differences in mean ranks for all their phenological characters except for the first side shoot visible, while for landrace, there were significant differences for all the characters (Table 1). However, the climatic background of the landrace had no effects on the first side shoot visible, the full flowering and the end of flowering.
The Kruskal–Wallis analyses of the treatment × species, treatment × climatic background and treatment × landrace were significant for all traits (Table 2). Post hoc analysis for treatment × species (Table 3) found that V. unguiculata developed the first phenological traits (P08–P12) faster than P. vulgaris and P. lunatus. However, V. unguiculata was the slowest species producing the flowering period (median: 121–169 days; P. vulgaris 71–116 days; P. lunatus 107–169 days), and produced more seeds per pod (9 seeds) than P. vulgaris (3.6 seeds) or P. lunatus (2.1 seeds), for other yield related traits produced lower values than the Phaseolus sp. The presence of the first shoot visible had higher mean rank values (appearance of the character delayed in time) in the field than in the greenhouse for all the species. In P. vulgaris, all significant comparisons confirmed that field conditions delayed the development of such traits, except for the fully ripe pods, which was delayed in the greenhouse for P. vulgaris and V. unguiculata (medians: 156.0 and 189.0 vs 136.5 and 149.0 days, respectively). In addition, P. lunatus produced nine or more leaves unfolded later in the field than in the greenhouse (medians: 60 vs 51 days).
Post hoc analysis for the treatment × climatic background (Table 3), found significant differences in mean rank values for the development of nine or more leaves unfolded, the first side shoot visible, the first flower buds visible and enlarged, and the first petals visible independently of the background origin of the landraces, which happened later in the field than in the greenhouse. In landraces from cold or warm background, the third true leaf developed later in the field compared to greenhouse but the fully ripe pods developed later in the greenhouse (Table 3). In warm background landraces, the flowering finishing was delayed in the greenhouse but the presence of first pods visible and the occurrence of 10% of pods ripe was delayed in the field.
Post hoc analysis for the treatment × landrace (Table 4) found that the number of pods per plant exhibited significant higher values in the field than in the greenhouse for P. vulgaris landraces 1, 4, 6, 7 and 9, P. lunatus 13 and both V. unguiculatus 10 and 11. P. vulgaris landrace 2 finished the flowering and the fully ripe pods later in the greenhouse than in the field; and landrace 8 failed to grow under field conditions. The development of nine or more leaves for P. vulgaris landraces 3, 7 and 9 and P. lunatus, and the end of flowering for P. vulgaris landraces 1, 3, 4 and 7 were delayed in the field. Other significant differences in character expression had higher values in the field (Table 4).
IV. Climate resilience landrace index and clustering
Across all landraces, the following morphological and reproductive characters were the strongly affected by changing the environmental conditions of the plants (when three or more landraces exhibited significant changes in the expression of the character): the number of flowers per plant, the number of pods per plant, the number grains per pod, the grain length, width and thickness, the scar length, the 100 seeds weight, the gross and net weight of seeds/plant and the number of seeds. Phenologically, the most affected traits were: cotyledons completely unfolded, two full leaves unfolded, the unfolding of nine or more leaves, the emergence of the first shoot, first flower buds visible and enlarged, first petals visible, the end of flowering and the pods fully ripe.
The climate resilience landrace index (CRLI, Table 4) found that P. vulgaris landrace 8 (0.688) was highly susceptible, and landraces 1 (0.396) and 6 (0.250) and V. unguiculata landrace 11 (0.354) were very susceptible to changes in their environmental conditions. P. vulgaris landrace 12 and the commercial variety 9 were the most resilient to environmental conditions (0.000 and 0.063, respectively). When using this index for species, P. vulgaris was the most susceptible (0.479) and P. lunatus the most resilient (0.146) to treatment. Warm background landraces were more prone to accumulate significant differences in their traits (0.404) compared to cold background (0.298) or the commercial cultivar (0.234).
The clustering of the mean ranks for the 48 characters expressed by the 12 landraces in both treatments produced a heatmap (Fig. 2) that identified the groups of morphological and phenological characters based on the components of the PCA, thus highlighting the differences induced by the treatment. All phenological characters were grouped in two clusters.
Heatmap of the studied landraces based on the mean rank values for all morphological and phenological characters. Hierarchical clustering of the heatmap for all the studied characters (columns) in the 13 landraces (rows). Columns are clustered using Euclidean distance and complete linkage. Then, we stablished seven groups of characters after the results of the PCA and the parallel analysis that suggested seven factors.
Discussion
Coping with climate change impact on crops might strongly benefit from those landraces that had been bred locally34. The analysis of the phenotypic plasticity of the intra- and interspecific diversity of local bean landraces from the Andes of South Ecuador found that climate change effects might accelerate the loss of certain landraces while having benign or none effects on others.
Changing environmental conditions had a direct impact on more than half (60.4%) of the 48 analysed characters. The most informative results; however, are those related to the interaction of treatment with species, climatic background, and landraces. The treatment affected the three species differently. Moreover, some characters exhibited higher levels of variation across species and landraces than others in response to the treatment: the number of flowers and seeds per plant, the gross and net weight of seeds and the emergence of the first side shoot visible were the most plastic traits across species. In addition, we found three P. vulgaris landraces from cold background with very different behaviours: landrace 1 was very susceptible to warmer conditions; landrace 8 performed very poorly in general, although better under warmer conditions than in the field; and landrace 12 was the most resilient material to any environmental change. Thus, we identified the former two landraces as critical targets for conservation and the latter as a potential landrace to replace locally those beans crops more affected by environmental conditions. Similar results can be found in maize landraces from Chiapas, where climate change may have strong impact on farmers that depend on locally adapted landraces35. Moreover, we identified suitable plant material for greenhouse cultivation at high altitudes in Andean communities. The later have three nonexclusive advantages that promote sustainable agriculture: (1) boosting natural soil conservation (maintenance of covered soils) and fertilisation with legume crops during fallow or crop rotation periods, either in the field or in the greenhouse; (2) in situ conservation of landraces; and (3) extra benefits for small farmers (production of fodder, food surplus production, and extra income in markets).
P. lunatus was the more tolerant species and P. vulgaris the more susceptible to changes in environmental conditions, confirming previous findings14. While warm conditions affected one architectural trait in P. vulgaris and four traits in P. lunatus, it had not effects on V. unguiculata.
The major effects of the treatment on characters related to reproduction and yield happened in P. vulgaris and V. unguiculata, and most of the phenological differences were found in P. vulgaris. Cold background landraces produced larger chalice lengths, thus suggesting either a potential adaption of cold background landraces to cold environmental conditions, related to the protection of the reproductive organs or facilitating the accumulation of heat trapped within the flower structure with micro greenhouse-like effects36. Warm conditions boosted seed and yield traits in the warm background and the commercial cultivars, supporting that warm background landraces are better adapted to temperatures higher than in the field and that the commercial cultivar performs better in warmer conditions. All the phenological characters involving flower development were delayed in the field, independently of the climatic background of the landrace. The fully ripe pods happened later in the greenhouse than in the field for both cold and warm background landraces, suggesting that warmer conditions might impose higher demands of water and storage of products in the grains, thus, prolonging the time for maturation.
We identified, through using the CRLI, three P. vulgaris landraces that are sensible (1, 6 and 8) and one landrace more resilient (12) to environmental changes. This could lead to developing specific in situ and ex situ conservation strategies. While many landraces are grown locally by the farmers, others might be present as the result of seed exchange activities (e.g., seed fairs) or migration of people. Flower and fruit characteristics and yield were improved in the greenhouse conditions, except for P. vulgaris landrace 1. Colder conditions delayed the emergence of many phenological traits except for the end of flowering of P. vulgaris landrace 2, from cold background, supporting its cold climatic background origin.
The first side shoot visible was the most widely affected trait by the environmental changes across landraces, highlighting the overall delay in vegetative growth in the field, and suggesting potential trade-offs in resource allocation. Warmer conditions increased stem diameter, the composite sheet length and width, and the terminal leaflet length. The latter might highlight both that some landraces had higher optimal temperatures of growth that the ones faced in the field, and that the temperatures reached in the greenhouse (day, night, their difference, and soil temperatures) are not high enough to promote plant respiration over photosynthesis. The former hypothesis seems plausible when interpreting the results after analysing the interaction between treatment and climatic background of the landraces. Moreover, that some cold background landraces performed better at higher temperatures than warm background landraces suggests that the later have reached an adaptive plateau that might not be surpassed. A potential explanation is that cold background materials could have a warm background origin, thus suggesting the later migration of the landraces by farmers through seed exchange and commercialization.
As expected, we found differences in performance and phenology among species supporting previous findings37. The values found for P. vulgaris traits, are within the expected variation among previously studied landraces and cultivars38. However, differences in the timing of flowering traits among P. vulgaris landraces might alter gene flow and promote reproductive isolation; thus, affecting their evolutionary dynamics39. P. lunatus was the most productive species; moreover, P. lunatus might be considered as a potential species to grow in greenhouses and replace other crops, economically less attractive, thus, adding a value to food security and soil fertilisation.
The greenhouse conditions had a positive effect on the production of flowers and on many important components related to pods and yield. We did not expect these positive effects after previous findings in P. vulgaris, where an increase of temperature affected negatively the number of seeds per pod, seed size and yield40,41. Moreover, the higher production of flowers and, thus, seeds under greenhouse conditions might be partially explained by the protection against winds that affect flower survival in the open field. Previous work; however, found that very high temperatures reduced the values for many reproductive traits33; but the latter was tested in a climate chamber a range of temperatures much higher than the ones reached in our greenhouse. The lower median values for the dry weight of the seeds in the open field; however, might be due to trade-offs between investment in reproduction and resistance against harsh abiotic or biotic conditions. Furthermore, the positive relation between temperature and seed weight was previously observed in V. unguiculata42. It is noteworthy to mention, that our results highlighted the importance of the climatic background from where the landraces come from as a fixed factor because it allowed us to better interpret the actual impact of temperature on crops and their landraces. Thus, we found that cold background plants produced the highest number of flowers and seeds and that warm background landraces performed better in the greenhouse than in the field. The behaviour of the commercial cultivar, like cold background landraces, suggests its potential cold background origin.
Warmer conditions accelerated the phenological development of plants for many of the analysed traits, although the day when the pods are fully ripped, was delayed in the greenhouse. This trend has been reported in other crop species but exhibiting a negative effect on crop yield43,44. Thus, global warming might be critical in several Andean crops, such as potato, which is highly sensible to high temperature45. Moreover, in addition to a negative impact on yield and quality, the rise of global temperatures will reduce the areas suitable for other crop species, such as coffee (Coffea arabica)46. In P. vulgaris, the advance of the flowering stage in the field due to warmer conditions highlighted the critical problem of flower-pollinator coupling47,48. Early flowering might respond to different environmental pressures and, for example, in oat Mediterranean landraces, it represents a potential mechanism to scape terminal drought44. However, we found that warm background landraces developed faster in the greenhouse than in the field for some characters, which might represent important adaptive characteristics (e.g., faster acquisition of energy) suggesting a fine-tuned genotype × environment interaction. Notwithstanding, most of the phenological traits developed later in the field for both the cold and warm background landraces. These results make us think of colder conditions imposing more costs to the plant, and then, architectural and reproductive traits develop slower in colder than in warmer conditions, where the latter facilitates fruit and seed maturation.
The ecological background from where the genetic material comes from (Table 3) plays a key role in the potential tolerance to high temperatures. Moreover, the hereby-proposed climate resilience landrace index (CRLI) might work as a proxy to identify landraces that are more prone to suffer the effects of climate change, such as P. vulgaris landrace 1, which seems to be very sensible to heat, or P. vulgaris landraces 6 and 8, more sensible to colder conditions than the other landraces. Moreover, this CRLI index highlighted P. vulgaris landrace 12 as the most resilient against environmental changes. The CRLI index, then, might be a powerful preliminary approach before identifying potential QTLs or traits for new breeds or genetic improvement, as well as a help for decision-making institutions or management tools23.
The hierarchical clustering of the heatmap for the 13 landraces in both treatments pointed out both that the level of expression of some characters are landrace or species specific, and that many plant morphological characters are very plastic, depending on the environmental conditions in which those traits are measured (Fig. 2). This highlights the relevance of the in situ morphological characterization of any plant population, landrace, or species. In addition, these plant genetic resources might be opportune as tools for generating resilience against climate change effects, and boost sustainable agriculture practices, strengthening food security. Moreover, the use of landraces to tackle climate challenges can fulfil specific climatic needs and might represent a source of germplasm for plant breeders49. Our work further emphasizes the importance of identifying the ecological antecedents of the landraces or crop populations because these data, being part of the characterization and later analysis, might become a robust tool for their conservation. Furthermore, the identification of appropriate standardised agromorphologic descriptors and phenological scales for each species might improve the use of plant passport data50,51,52,53. Finally, the characterization of the agromorphological and physiological profiles of a collection of local plant materials can generate resilience against other disasters in addition to climate change, such as the COVID-19 pandemic; since it has caused the shortage of seeds of commercial varieties in developing communities54.
Material and methods
Plant material, location, and cultivation
We selected nine Phaseolus vulgaris, one P. lunatus and two V. unguiculata landraces collected from small farmers from the Andean region of South Ecuador during 2017 and conserved in the UTPL germplasm bank, and one commercial P. vulgaris cultivar. All UTPL landraces were collected from sites with different climatic background conditions from Loja province (Ecuador) except the UTPL-PGR-0798 landrace, collected from El Oro province (Supporting Table S1, Supporting Fig. S1). We selected these landraces to test whether their climatic background had an effect on their performance against two different environmental conditions. Thus, depending on the altitude at which the material was found, which has a direct correlation in Ecuador with climatic conditions, we assigned each landrace to cold (above 2000 m.a.s.l.), warm (between 900 and 2000 m.a.s.l), or commercial (unknown) background. P. vulgaris landraces are numbered as 1–9 and 12 (UTPL germplasm bank codes: UTPL-PGR-0156, UTPL-PGR-0168, UTPL-PGR-0311, UTPL-PGR-0314, UTPL-PGR-0316, UTPL-PGR-0318, UTPL-PGR-0344, UTPL-PGR-0345 and UTPL-PGR-0798, respectively), V. unguiculata as 10 and 11 (UTPL-PGR-0313 and UTPL-PGR-0317), and P. lunatus as 13 (UTPL-PGR-0230). Cold background landraces are 1, 2, 8, 12and 13; the warm background landraces are 3–7, 10 and 11. The number 9 is the commercial cultivar.
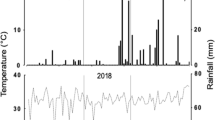
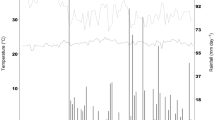
We started the experiments on July 19th 2018, and lasted until the end of March 2019 at a field located in the Universidad Técnica Particular de Loja (UTPL, Loja, Ecuador. Coordinates: 4°0′1.59" S and 79°10′48.46" W). The site has an altitude of 2160 m.a.s.l., an average minimum/maximum temperature across the year of 12.9 °C and 22.6 °C (annual mean around 16.7 °C), an average annual precipitation of 780 mm and a relative humidity of 81.07%. In the same location, we settled a greenhouse with a monthly average temperature of 23.5 °C (21 °C minimum and 26 °C maximum) and relative humidity of 57.62%. Loja corresponds to the low dry montane forest (bs-MB) ecological formation55. Then, we first seeded an excess of seed for each landrace in a tunnel nursery at 25 °C and 45% relative humidity for 18 days. For each landrace we transplanted 72 healthy seedlings in each of the two environments (open field and greenhouse), at 50 cm between plants and one meter between rows, and drip irrigation in the greenhouse (up to twice a week to avoid hydric stress). Each environment represented one climatic condition. According to soil analysis performed in the Agrocalidad laboratory (Agencia de Regulación y Control Fito y Zoosanitario), a fertilization program was applied with mineral fertilization based on 12:36:12 (N-P2O5-K2O) as basic fertilization on both environments.
Morphoagronomic characterisation
We quantified the development and production of each plant by using 49 descriptors based on Bioversity International (http://www.bioversityinternational.org/publications), while integrating the particularity of each studied species: P. vulgaris50, P. lunatus51 and V. unguiculata52, and evaluated 32 morphological characters of plant architecture, inflorescence and fruit characters, and yield. Then, we used the BBCH codifications53 to register 17 phenological stages (Supporting Table S2). Plant architecture characters: stem length (Q1), stem diameter (Q2), number of main branches (Q3), branch orientation (Q4), composite sheet length (Q6), composite sheet width (Q7), apex angle of terminal leaflet (Q8), terminal leaflet length (Q9), terminal leaflet width (Q10) and angle of the base of the terminal leaflet (Q11). Flower and fruit characteristics and yield characters: number of flowers per plant (Q12), peduncle length (Q15), left wing length (Q16), right wing length (Q17), banner length (Q18), style length (Q19), chalice length (Q20), number of pods per plant (Q21), number of pods per infructescence (Q22), sheath length (Q23), sheath width (Q24), sheath thickness (Q25), number of loculi per pod (Q26), number of grains per pod (Q27), grain length (Q28), grain width (Q29), scar length (Q30), grain thickness (Q31), 100 seed weight (Q32), gross weight of seeds/plant (Q33), net weight of seed/plant (Q34) and number of seeds (Q35). Phenological characters: Hypocotyl reaches the soil surface (P08), hypocotyl with cotyledons break through soil surface (P09), cotyledons completely unfolded (P10), 2 full leaves (P12), 3rd true leaf (P13), 9 or more leaves unfolded (P19), first side shoot visible (P21), first flower buds visible (P51), first flower buds enlarged (P55), first petals visible, flowers still closed (P59), beginning of flowering (P61), full flowering: 50% of flowers open (P65), flowering finishing (P67), end of flowering (P69), 10% of pods ripe (P81), 50% of pods ripe (P85) and fully ripe pods (P89).For most descriptors, we gathered up measures directly in the field, while other characters were measured in the laboratory by using the image-processing tool ImageJ56 (Supporting Table S2).
Data analysis
We calculated an explorative correlation matrix between all pairs of the 49 variables and found that Q3 was uncorrelated with any other factor (all values below 0.3). All other 48 variables accomplished the assumptions for the PCA analysis. The Kaiser–Meyer–Olkin Measure of Sampling Adequacy (KMO) produced a meritorious value of 0.897, after Kaiser’s evaluation57. Moreover, the Bartlett's Test of Sphericity strongly supported that our data is suitable for a factor reduction technique (χ2 = 15,722.84, df = 1128; p < 0.001). Overall, the extracted communalities represented well our variables, with values between 0.500 and 0.966, except for Q1 (0.401), Q4 (0.431) and Q8 (0.336). Eight components had eigenvalue values above 1.000 accounting for a cumulative 80.230% of the variability of our original 48 variables. To know how many factors retain we conducted a parallel analysis58 that suggested seven factors (77.648%).
We analysed for differences in plant performance between both climatic conditions (open field vs greenhouse), among species (P. vulgaris, P. lunatus and V. unguiculata), the 13 landraces, the climatic background (cold, warm, and commercial), and for the interactions between treatment × species, treatment × landrace, and treatment × climatic background using the nonparametric independent samples Kruskal–Wallis test, because the data did not meet the assumptions of the ANOVA. We further conducted post-hoc pairwise comparisons adjusted after Bonferroni. All statistical analyses were conducted in IBM® SPSS® Statistics v. 24.
The information gathered up in the post-hoc pairwise comparisons about each landrace performance in both climatic conditions allowed us to create and calculate the climate resilience landrace index (CRLI). First, we quantified how many characters exhibited significant post hoc p-values for each of the 13 landraces (total landrace significance, TLS), and divide each value by the number of traits investigated (48 morphological and phenological traits). This index highlights which landraces are more likely to be affected by different aspects of the climate change effects and, thus, will provide important information about both conservation and agricultural strategies to build up resilience against climate change effects. The closer the index is to zero the lower the impact on the landrace, and the maximum impact is one. Thus, we suggest three categories to interpret these indexes: (1) weak or none effects (strong resilience) for indexes values between 0.00 and 0.33; (2) moderate effects for values between 0.33 and 0.66; and (3) strong or drastic effects (e.g., highly susceptible landraces) for values between 0.66 and 1.00.
We conducted a Principal Component Analysis (PCA) and clustering of the 13 landraces based on the mean values of the agromorphological characters using the ClustVis webtool59. During the pre-processing of the data, we applied unit variance scaling as variance normalization method because we are analysing variables of different units and intensity ranges. Afterward, we followed the Nipals (Nonlinear Iterative Partial Least Squares) PCA method. In order to understanding the effects of the treatment (open field vs greenhouse) on agromorphological and phenological variation better, we further constructed a clustered heatmap with the 13 landraces in both environmental conditions. We produced the heatmap using Euclidean distance and complete linkage methods.
Ethics declaration
Experimental research and the field study on plants complied with Ecuadorian research normative (Ley Orgánica de Agrobiodiversidad, Semillas y Fomento de la Agricultura Sustentable, 2017).
References
FAO. Coping with Climate Change—The Roles of Genetic Resources for Food and Agroculture. 110. (FAO, 2015).
Batisti, D. S. & Naylor, R. L. Historical warnings of future food insecurity with unprecedented seasonal heat. Science 323, 240–244. https://doi.org/10.1126/science.1164363 (2009).
IPCC. Global Warming of 1.5 °C. An IPCC Special Report on the Impacts of Global Warming of 1.5 °C Above Pre-Industrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change, Sustainable Development, and Efforts to Eradicate Poverty (eds. Masson-Delmotte, V. et al.) (2018).
Rosenzweig, C. et al. Assessing agricultural risks of climate change in the 21st century in a global gridded crop model intercomparison. Proc. Natl. Acad. Sci. USA 111, 3268–3273. https://doi.org/10.1073/pnas.1222463110 (2014).
Jägermeyr, J. et al. Climate impacts on global agriculture emerge earlier in new generation of climate and crop models. Nat. Food 2, 873–885. https://doi.org/10.1038/s43016-021-00400-y (2021).
Zhao, C. et al. Temperature increase reduces global yields of major crops in four independent estimates. PNAS 114, 9326–9331. https://doi.org/10.1073/pnas.1701762114 (2017).
Coakley, S. M., Scherm, H. & Chakraborty, S. Climate change and plant disease management. Annu. Rev. Phytopathol. 37, 399–426. https://doi.org/10.1146/annurev.phyto.37.1.399 (1999).
Fróna, D., Szenderák, J. & Harangi-Rákos, M. Economic effects of climate change on global agricultural production. Nat. Conserv. 44, 117–139. https://doi.org/10.3897/natureconservation.44.64296 (2021).
Lobell, D. B. et al. Prioritizing climate change adaptation needs for food security in 2030. Science 319, 607–610. https://doi.org/10.1126/science.1152339 (2008).
Schlichting, C. D. The evolution of phenotypic plasticity in plants. Ann. Rev. Ecol. Syst. 17, 667–693. https://doi.org/10.1146/annurev.es.17.110186.003315 (1986).
Gratani, L. Plant phenotypic plasticity in response to environmental factors. Adv. Bot. 2014, 208747. https://doi.org/10.1155/2014/208747 (2014).
Richardson, A. D. et al. Climate change, phenology, and phenological control of vegetation feedbacks to the climate system. Agric. For. Meteorol. 169, 156–173. https://doi.org/10.1016/j.agrformet.2012.09.012 (2013).
Mall, R. K., Gupta, A. & Sonkar, G. Effect of climate change on agricultural crops. in Current Developments in Biotechnology and Bioengineering—Crop Modification, Nutrition, and Food Production (eds. Dubey, S. K., Pandey, A. & Sangwan, R. S.). 23–46. (Elsevier, 2017).
Beebe, S. E., Rao, I. M., Blair, M. W. & Acosta-Gallegos, J. A. Phenotyping common beans for adaptation to drought. Front. Physiol. 4, 1–20. https://doi.org/10.3389/fphys.2013.00035 (2013).
Broughton, W. J. et al. Beans (Phaseolus spp.)—Model food legumes. Plant Soil 252, 55–128. https://doi.org/10.1023/a:1024146710611 (2003).
Gepts, P. Origin and evolution of common bean: Past events and recent trends. Hortic. Sci. 33, 1124–1130. https://doi.org/10.1071/FP11102 (1998).
Singh, S. P., Gepts, P. & Debouck, D. G. Races of common bean (Phaseolus vulgaris, Fabaceae). Econ. Bot. 45, 379–396. https://doi.org/10.1007/BF02887079 (1991).
Tohme, J., Gonzalez, D. O., Beebe, S. & Duque, M. C. AFLP analysis of gene pools of a wild bean core collection. Crop Sci. 36, 1375–1384. https://doi.org/10.2135/cropsci1996.0011183x00360005004 (1996).
Serrano-Serrano, M. L., Hernández-Torres, J., Castillo-Villamizar, G., Debouck, D. G. & Chacón Sánchez, M. I. Gene pools in wild Lima bean (Phaseolus lunatus L.) from the Americas: Evidences for an Andean origin and past migrations. Mol. Phylogenet. Evol. 54, 76–87. https://doi.org/10.1016/j.ympev.2009.08.028 (2010).
García, T. et al. Comprehensive genomic resources related to domestication and crop improvement traits in Lima bean. Nat. Commun. 12, 702. https://doi.org/10.1038/s41467-021-20921-1 (2021).
Chacón-Sánchez, M. I. & Martínez-Castillo, J. Testing domestication scenarios of lima bean (Phaseolus lunatus L.) in Mesoamerica: Insights from genome-wide genetic markers. Front. Plant Sci. 8, 1551. https://doi.org/10.3389/fpls.2017.01551 (2017).
Cerda-Hurtado, I. M. et al. Climatic adaptation and ecological descriptors of wild beans from Mexico. Ecol. Evolut. 8, 6492–6504. https://doi.org/10.1002/ece3.4106 (2018).
McClean, P. E., Burridge, J., Beebe, S., Rao, I. M. & Porch, T. G. Crop improvement in the era of climate change: An integrated, multi-disciplinary approach for common bean (Phaseolus vulgaris). Funct. Plant Biol. 38, 927–933. https://doi.org/10.1071/FP11102 (2011).
Lush, W. M. & Evans, L. T. The domestication and improvement of cowpeas (Vigna unguiculata (L.) Walp.). Euphytica 30, 579–587. https://doi.org/10.1007/BF00038783 (1981).
INEC. Encuesta de Superficie y Producción Agropecuaria Continua—ESPAC. https://www.ecuadorencifras.gob.ec/estadisticas-agropecuarias-2/ (2019).
Garcés-Fiallos, F. R. & Olmedo-Zamora, I. M. Potencial agronómico de 18 líneas de fréjol F6 en Ecuador. IDESIA 33, 107–118 (2015).
Wheeler, T. & von Braun, J. Climate change impacts on global food security. Science 341, 508–513. https://doi.org/10.1126/science.1239402 (2013).
Zabel, F. et al. Large potential for crop production adaptation depends on available future varieties. Global Change Biol. 27, 3870–3882. https://doi.org/10.1111/gcb.1564 (2021).
Casañas, F., Simó, J., Casals, J. & Prohens, J. Toward an evolved concept of landrace. Front. Plant Sci. 8, 145. https://doi.org/10.3389/fpls.2017.00145 (2017).
Araújo, S. S. et al. Abiotic stress responses in legumes: Strategies used to cope with environmental challenges. Crit. Rev. Plant Sci. 34, 237–280. https://doi.org/10.1080/07352689.2014.898450 (2014).
Diaz, L. M. et al. QTL analyses for tolerance to abiotic stresses in a common bean (Phaseolus vulgaris L.) population. PLoS ONE 13, e0202342. https://doi.org/10.1371/journal.pone.0202342 (2018).
Bunce, J. A. Contrasting responses of seed yield to elevated carbon dioxide under field conditions within Phaseolus vulgaris. Agr. Ecosyst. Environ. 128, 219–224. https://doi.org/10.1016/j.agee.2008.06.003 (2008).
Da Silva, D. A. et al. Influence of high temperature on the reproductive biology of dry edible bean (Phaseolus vulgaris L.). Sci. Agric. 77, e20180233. https://doi.org/10.1590/1678-992x-2018-0233 (2020).
Snowdon, R. J., Wittkop, B., Chen, T.-W. & Stahl, A. Crop adaptation to climate change as a consequence of long-term breeding. Theor. Appl. Genet. 134, 1613–1623. https://doi.org/10.1007/s00122-020-03729-3 (2021).
Mercer, K. L. & Perales, H. R. Evolutionary response of landraces to climate change in centers of crop diversity. Evol. Appl. 3, 480–493. https://doi.org/10.1111/j.1752-4571.2010.00137.x (2010).
van der Kooi, C. J., Kevan, P. G. & Koski, M. H. The thermal ecology of flowers. Ann. Bot. 124, 343–353. https://doi.org/10.1093/aob/mcz073 (2019).
Stoilova, T. & Berova, M. Differences in growth and productivity between genotypes of common bean (Phaseolus vulgaris L.) and cowpea (Vigna unguiculata L.) in the Sadovo region of Bulgaria. Folia Hortic. 24, 53–59. https://doi.org/10.2478/v10245-012-0006-0 (2012).
Rana, J. C. et al. Characterisation of 4274 accessions of common bean (Phaseolus vulgaris L.) germplasm conserved in the Indian gene bank for phenological, morphological and agricultural traits. Euphytica 205, 441–457. https://doi.org/10.1007/s10681-015-1406-3 (2015).
Franks, S. J. & Weis, A. E. Climate change alters reproductive isolation and potential gene flow in an annual plant. Evol. Appl. 2, 481–488. https://doi.org/10.1111/j.1752-4571.2009.00073.x (2009).
Prasad, P. V. V., Boote, K. J., Allen, L. H. Jr. & Thomas, J. M. G. Effects of elevated temperature and carbon dioxide on seed-set and yield of kidney bean (Phaseolus vulgaris L.). Glob. Change Biol. 8, 710–721. https://doi.org/10.1046/j.1365-2486.2002.00508.x (2002).
Soltani, A., Weraduwage, S. M., Sharkey, T. D. & Lowry, D. B. Elevated temperatures cause loss of seed set in common bean (Phaseolus vulgaris L.) potentially through the disruption of source-sink relationships. BMC Genomics 20, 312. https://doi.org/10.1186/s12864-019-5669-2 (2019).
Angelotti, F., Guerra Barbosa, L., Alves Barros, J. R. & Fernandes dos Santos, C. A. Cowpea development under different temperatures and carbon dioxide concentrations. Pesq. Agropec. Trop. Goiânia 50, e59377. https://doi.org/10.1590/1983-40632020v5059377 (2020).
Hatfield, J. L. & Prueger, J. H. Temperature extremes: Effect on plant growth and development. Weather Clim. Condit. 10, 4–10. https://doi.org/10.1016/j.wace.2015.08.001 (2015).
Canales, F. J. et al. Deciphering main climate and edaphic components driving oat adaptation to mediterranean environments. Front. Plant Sci. 12, 780562. https://doi.org/10.3389/fpls.2021.780562 (2021).
Handayani, T., Gilani, S. A. & Watanabe, K. N. Climatic changes and potatoes: How can we cope with the abiotic stresses?. Breed. Sci. 69, 545–563. https://doi.org/10.1270/jsbbs.19070 (2019).
Ovalle-Rivera, O., Läderach, P., Bunn, C., Obersteiner, M. & Schroth, G. Projected shifts in Coffea arabica suitability among major global producing regions due to climate change. PLoS ONE 10, e0124155. https://doi.org/10.1371/journal.pone.0124155 (2015).
Kehrberger, S. & Holzschuh, A. Warmer temperatures advance flowering in a spring plant more strongly than emergence of two solitary spring bee species. PLoS ONE 14, e0218824. https://doi.org/10.1371/journal.pone.0218824 (2019).
Elisante, F. et al. Insect pollination is important in a smallholder bean farming system. Peer J 8, e10102. https://doi.org/10.7717/peerj.10102 (2020).
Galluzzi, G., Seyoum, A., Halewood, M., López Noriega, I. & Welch, E. W. The role of genetic resources in breeding for climate change: The case of public breeding programmes in eighteen developing countries. Plants 9, 1129. https://doi.org/10.3390/plants9091129 (2020).
IPGRI. Descritores para Phaseolus vulgaris. (International Plant Genetic Resources Institute, 2001).
IBPGR. Lima Bean Descriptors. (International Board for Plant Genetic Resources, 1982).
IBPGR. Descriptors for Cowpea. (International Board for Plant Genetic Resources, 1983).
Feller, C. et al. Phänologische Entwicklungsstadien von Gemüsepflanzen: II. Fruchtgemüse und Hülsenfrüchte. Nachrichtenbl. Deut. Pflanzenschutzd. 47, 217–232 (1995).
Nchanji, E. B. et al. Immediate impacts of COVID-19 pandemic on bean value chain in selected countries in sub-Saharan Africa. Agric. Syst. 188, 103034. https://doi.org/10.1016/j.agsy.2020.103034 (2021).
Holdridge, L. R. Life Zone Ecology. (Tropical Science Center, 1967)
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675. https://doi.org/10.1038/nmeth.2089 (2012).
Kaiser, H. F. An index of factorial simplicity. Psychometrika 39, 31–36 (1974).
Patil V. H., Singh, S. N., Mishra, S. & Donavan, D. T. Parallel Analysis Engine to Aid in Determining Number of Factors to Retain using R [Computer software]. https://analytics.gonzaga.edu/parallelengine/ (2017).
Metsalu, T. & Vilo, J. Clustvis: A web tool for visualizing clustering of multivariate data using Principal Component Analysis and heatmap. Nucleic Acids Res. 43(W1), W566–W570. https://doi.org/10.1093/nar/gkv468 (2015).
Acknowledgements
Víctor Gavidia Morales for technical support.
Author information
Authors and Affiliations
Contributions
P.G.A.-Q. & M.X.R.-G. designed the experiments, analysed the data, and wrote the manuscript. E.H.V.-S. and J.M.M.-C. collected the data. E.J.-V. collected the landrace from El Oro province and produced several preliminary experiments. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Acosta-Quezada, P.G., Valladolid-Salinas, E.H., Murquincho-Chuncho, J.M. et al. Heterogeneous effects of climatic conditions on Andean bean landraces and cowpeas highlight alternatives for crop management and conservation. Sci Rep 12, 6586 (2022). https://doi.org/10.1038/s41598-022-10277-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-10277-x
- Springer Nature Limited
This article is cited by
-
International Lima Bean Network: from the origin of the species to modern plant breeding
Genetic Resources and Crop Evolution (2023)