Abstract
Background
Oral cancer is a major public health challenge, along with other common diseases worldwide. Risk factors like tobacco usage and alcohol consumption have contributed towards the rise in the development of oral cancer cases. The low survival rate of oral cancer creates a necessity for early detection. At present, clinical examination followed by biopsy and histopathological-based assessment is considered the gold standard in the early diagnosis of oral cancer. However, it also holds drawbacks like invasiveness and time consumption in confirming a diagnosis, which have raised the need for other non-invasive diagnostic techniques. Spectroscopic techniques used as diagnostic tools are to collect the spectral signatures generated by endogenous fluorophore molecules in abnormal and healthy tissues upon excitation with a suitable wavelength.
Objective
The present review aimed to highlightthe potential application of non-invasive spectroscopic tools such as fluorescence spectroscopy in oral cancer detection and diagnosis and with a brief standpoint of the conventional techniques used in oral cancer diagnosis.
Conclusion
The review emphasizes the potentiality of spectroscopic techniques to replace conventional diagnostic procedures, given their minimally invasive or non-invasive approach, cost-effective nature, and ability to provide a diagnosis in real time.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cancer is one of the major causes of mortality among adults worldwide. A rise in cancer-related deaths is expected, primarily because of an increase in the risk factors for cancer [1]. Oral cancer is a significant health concern, especially in Asian countries, as it shows a great deal of variation worldwide in terms of its incidence rate owing to racial and ethnic differences among the people as well as associated lifestyles [2]. Countries in South and South-East Asia present the most worrisome rate of incidence, according to the International Agency for Research on Cancer, which includes Sri Lanka, India, and Pakistan, among others [3]. These countries, which are said to be developing, lower-income countries, are often more affected by this burden compared with developed countries and are responsible for two-thirds of the oral malignancies worldwide. The Asian population is more prone to oral cancer compared with other communities due to the habit of chewing tobacco and areca nut in betel quid being rampant in these groups of people. The incidence of oral cancer found to occur more commonly in males than in females [4, 5]. With the increase in age, the risk of developing oral cancer increases. Further, the 5-year incidence and mortality rate as per GLOBOCAN (2018) report indicate oral cancer incidence and mortality are higher in males compared with females [6].
The majority of oral cancer incidence is found to occur in people age 50 years and above. In high-risk countries, oral cancer cases among people below the age of 40 have also been reported. A study conducted by Dikshit R et al. has highlighted oral cancer as one of the fatal diseases in Indian men belonging to the age group of 30–69 years, accounting for 22.9% of the cases, which included cancers of the lip, oral cavity, and pharynx, while in women belonging to the same age group, oral cancer accounted for 9.8% of the cancer cases after cervical (17.1%), stomach (14.1%), and breast (10.2%) [1].
Oral cancer could be defined as a non-homogenous group of diseases of the oral cavity, involving the lips, tongue, labial mucosa, buccal mucosa, palate, the floor of the mouth, gingiva, and other anatomical sites. However, literature does not adopt a clear cut definition for the same. Oral cancer has a multifactorial etiology, with tobacco considered a significant risk factor. Apart from smoking or chewing any form of tobacco, along with or without betel quid, excess consumption of alcohol, infection by human papilloma virus (HPV), poor oral hygiene, and poor dentition and even genetic susceptibility are associated with risk factors [7,8,9]. A considerable minority of the cases have also reported that oral cancer can be caused by factors other than the traditional risk factors. Lower economic status and lack of awareness about oral health are also related to increased risk of developing the disease [10]. The level of education is associated with knowledge about oral cancer, and its risk factors were looked into by Elango JK et al. in a survey conducted among a high-risk population. They reported that higher education level, more significant, was the awareness for both oral cancer and its risk factors (p < 0.001), while oral cancer-causing risk habits, which included smoking, chewing pan with/without tobacco, and alcohol, were significantly less prevalent among those with a better level of education [11].
Oral cancers show a low survival rate (less than 50% in most countries) since half of the diagnosed cases would have progressed to an advanced stage. The above progression of the diseases to advanced stage attributed due to the patient’s ignorance or reluctance in obtaining proper medical care and poor acceptance of the treatment [10]. Also, the lack of access to cancer treatment centers may further delay and also result from misdiagnosis and inappropriate treatment. On the contrary, in diagnosis at the stage where the primary tumor is localized, the survival rate is better, suggesting that early diagnosis could lead to better chances of survival. Therefore, detection of oral cancer at an early stage or a precancerous lesion before it becomes malignant is the key to ensure effective treatment and reduced morbidity [12].
In the present review, we have attempted to highlight the potentiality of fluorescence-based spectroscopic tools in the early oral cancer detection/diagnosis, while we also tried to summarize some of the conventional techniques used in oral cancer diagnosis with their limitations, which is routinely used in oral oncology screening.
Conventional diagnostic techniques
The fact is that the oral cavity is accessible for the visual and tactile examination and can be viewed to detect oral cancer or even for its precancerous change [9]. Presently, the assessment of the oral cavity is carried out either by oral examination, invasive approach (oral biopsy), and vital staining techniques (toluidine blue, Lugol’s iodine), or by light-based optical tools such as chemiluminescence techniques.
Oral examination
Conventional oral examination (COE) is carried out with the help of an incandescent light, which is used routinely in the screening of oral cancer. Visual examination to screen patients is advantageous as it is non-invasive and causes no pain and is also well accepted among the clinicians socially. However, studies have highlighted that the method is a bit obscure for screening premalignant or early lesions. On the contrary, few studies report that the COE method has better sensitivity and specificity. Presently there is hardly any test that can reveal an accurate detection of oral lesions when it comes to the screening program; the only method used for screening in the oral malignant or lesion is a conventional oral examination. Some of the earlier studies have reported the COE-based screening for the detection of potential oral malignant lesions or cancers performed by health experts [13, 14].
A hospital-based study was carried out by Chang et al. where a total of 13,878 participants were included in the study. The study aimed to demonstrate the effectiveness of oral visual screening in determining the potential risk of oral cavity cancer in chronic smokers, alcohol consumption, and betel quid chewers. The results clearly show that oral screening is effective and should be used regularly as a routine screening procedure to detect oral cancer at the earliest [15]. In another study, Walsh et al. have also highlighted the diagnostic methods, such as COE, vital stain rinsing, optical tools, and biomarker applications in oral cancer detection. The review mainly includes the sensitivity and specificity of the methods. The study shows that COE has better sensitivity, which was higher than 70%, and the specificity was more than 90% compared with other techniques used in oral cancer screening [16]. Table 1 below highlights the results of a few studies where COE is used as a screening tool in oral cancer.
Thus, based on earlier studies, it has been reported that COE could be used to screen the oral lesions and cancers. However, it is not useful in differentiating between the lesions that could progress to cancer and which would be in a benign state. Also, chromosomal aberrations may exist in areas of the mouth appearing to be normal, which could later lead to the development of cancer [22]. Also, certain precancerous lesions get hidden beneath normal-appearing mucosa and which is difficult to detect during COE and could lead to oral malignancies. Therefore, all suspicious lesions should be evaluated histologically to determine their potential in turning into a malignant condition.
Oral biopsy
Oral biopsy is considered an essential clinical tool in the diagnosis of oral lesions ranging from simple lesions such as precancerous lesions to cancerous lesions. The biopsy procedure helps the clinicians to determine the proper diagnosis of the diseases, which eventually will help patients in the timely treatment of the diseases. The suspicious oral lesions are evaluated either by oral brush cytology or by surgical biopsy.
The scalpel or punch biopsy, followed by its histopathological assessment, is considered the gold standard for the diagnosis of oral lesions [23]. However, it is not conceivable to perform a biopsy in all cases, as some of the patients might not give consent as their lesion may be asymptomatic. In some cases, the patients are critically ill due to which the procedure could not be possible to carry out. An alternative to the above, the clinicians go for an oral cytology approach, where they monitor changes in cytology of oral cells by performing a procedure such as brush cytology. The brush cytological study is minimally invasive and well accepted among the patients and gives consent to undergo the procedure, due to which it has been considered an alternative diagnostic option to a more invasive and painful biopsy approach [24]. The technique holds the potential in determining the pathological changes in the oral lesion. The technique mainly involves the collection of oral cells by rinsing the oral cavity after scraping the affected oral cavity area using an oral brush.
Oral computer-assisted brush cytology (OralCDx) is the most standard and routinely brush cytology procedure for assessing the oral lesions [25]. Several studies have reported encouraging results with oral brush cytology in evaluating cancerous lesions. Table 2 illustrates the utilization of oral brush cytology methods highlighting the sensitivity and specificity of the technique.
At present, a biopsy followed by a histopathological assessment of a tissue sample suspected of malignancy is considered the gold standard for the diagnosis of oral cancer. Histopathological evaluation of tissues involves a visual analysis of tissues at the cellular level, which enables the clinicians to make decisions on whether the tissue section under evaluation is malignant or not, based on the features like shape and distribution of the cells and the nuclei [31, 32]. Further, during the procedure, the site of the mouth from where the tissue specimen obtained during biopsy should be such that it should be able to represent the most severe changes in the oral cavity. Therefore, the accuracy of diagnosis depends on the clinician’s choice of an appropriate biopsy site and the pathologist’s interpretation based on the histopathological findings [33]. Due to the above problem with visually identifying sites for biopsy, which represents the most malignant transformation, it is subjective and depends entirely on the examiner and, this might lead to selecting an inappropriate lesion which in turn often requires redoing the biopsy, adding to the patient’s distress [34].
Further, the interpretation of microscopic sections includes the presence of artifacts that are common in biopsy specimens of the oral mucosa, which can result in a change in morphology and cytology, which will render the specimen useless and their presence depends on the type of biopsy technique used.
Staining techniques
The conventional vital staining techniques such as toluidine blue, Lugol’s iodine staining, and Rose Bengal staining are commonly used in oral cancer diagnosis [35]. The toluidine blue technique is the most accepted staining technique and coupled with other modalities to visualize the oral lesions. The staining technique helps in the diagnosis of oral cancer and has been used to detect both premalignant and malignant conditions. Toluidine blue is a vital stain used to visualize changes in the mucosa of the oral cavity as it is capable of staining nucleic acids. The dye binds DNA and RNA, which is seen to be higher side during the onset of malignancy [36], while Lugol’s iodine binds to the glycogen content present in the epithelium cells. The stain has been used in discriminating against the inflammatory or cancerous oral lesion from normal epithelium as they contain lower glycogen contents [37].
Various studies have reported the application of these stains in oral cancer detection; some of these studies are highlighted in Table 3 above. A study conducted by Chainani W et al. has reported the sensitivity and specificity of toluidine blue to the detection of high-risk lesions (carcinoma in situ or OSCCA). Of the 100 lesions evaluated from 43 patients, 77 lesions were biopsied and included for analysis. The results of the toluidine blue technique showed a sensitivity of 94% in the case of high-risk lesions, while that for OSCCA was 100%. However, the authors pointed out the limitations in the study where they found the specificity of 45% and 39% for the above cases, respectively [43]. To increase the sensitivity and specificity and overcome the limitations of the staining techniques, some authors have combined two different staining techniques to detect abnormal oral lesions. A study was carried out by Nagaraju K et al. where the authors determined the diagnostic applicability and reliability of toluidine blue with Lugol’s staining. The results show the combined staining technique can assess and discriminate between the premalignant and malignant oral lesions with a sensitivity of 92.7% and specificity of 60%, respectively [41]. The staining of the oral lesion by toluidine blue is shown in Fig. 1.
TBlue staining of leukoplakia, a before staining and b retention of TBlue dye by the white patches, reprinted from Awan K. et al. [36] with permission of John Wiley and Sons
Although the staining techniques are quite a sensitive tool, however, they possess some limitations such as erroneous retention of stain in regions that show inflammation and ulcers and inability to detect deeper-lying dysplasia in the epithelium and the hindrance in dye-uptake lesion area caused by the mucous [38].
Chemiluminescence
Chemiluminescence is a light-based screening technique used to examine the oral cavity. The method principally depends on alterations in the refractive index of the mucosa corresponding to the alterations in the metabolism and structure occurring due to malignancy. Normal mucosa absorbs the blue-white light, while the cells which are dysplastic and neoplastic reflect it. The chemiluminescence-based technique is marketed under the names of Vizilite plus and MicroLux DL. Vizilite Plus is a disposable light packet, while MicroLux DL is reusable. Vizilite Plus tool is a combination of chemiluminescence and toluidine blue (TB). The toluidine blue (TB) stain specifically stains the DNA. The procedure mainly involves 1-min rinsing with a 1% acetic acid solution, followed by the clinician observation of the rinsed lesion sites using the tool which emits light in a wavelength from 430 to 580 nm (as shown in Fig. 2) [44, 45]. The sensitivity of Vizilite was reported by Chaudhry A et al. where the authors claimed Vizilite-based detection showed better sensitivity in identifying leukoplakia [46]. Further, several studies have reported the application of the chemiluminescence tool in screening the oral lesions by clinicians as shown in Table 4 below.
Oral lesion at buccal mucosa under a incandescent light b stained with toluidine blue and c Aceto-white delineation of oral lesion on Vizilite plus, reprinted from Jain N. et al. [44] with permission of Elsevier
Although the above technique possesses better application compared with other conventional methods, however, they possess some limitations such as interference of saliva in visualizing lesions, inability to identify erythroplakia, and to differentiate non-dysplasia from dysplasia has been reported.
Non-invasive fluorescence-based tools in oral diagnosis
When an experienced practitioner performs a visual examination, he limits to differentiate between premalignant lesions from the conditions that are inflammatory and one that can lead to malignancy. Also, in many of the cases, the patient would get diagnosed at a later stage of cancer by the time they reach the clinician, followed by their histopathological assessment, which is very subjective. Impediments such as these, presented by conventional diagnostic methods in early detection of oral cancer, have led to the need for the development of alternative diagnostic modalities. The optical tools based on autofluorescence are effective in discriminating dysplastic lesions from early neoplastic and also provide information regarding changes at the biochemical and molecular levels. Autofluorescence is a phenomenon that takes place when an intrinsic fluorophore molecule gets excited by a light source of a particular wavelength, and emits fluorescence upon relaxation. On absorbing incident light of suitable wavelength, the electrons in the fluorophores get excited from the ground vibrational states to the higher energy states. The electrons then relax to the 1st excited state of the fluorophore through non-radiative relaxations followed by its transition to the ground vibrational levels through radiative relaxations known as fluorescence and thereby releasing the excitation energy of the fluorophores at higher wavelength compared with the excitation wavelength of the fluorophore as shown in Fig. 3 [52, 53].
Tissues possess endogenous fluorophores, which include aromatic amino acids, collagen, elastin, porphyrins, and metabolic products such as nicotinamide adenine dinucleotide (NADH) and flavin adenine dinucleotides (FAD). During the neoplastic and pre-neoplastic stages, these endogenous fluorophores demonstrate alterations in their metabolism and structures. Thus, a light of a particular wavelength is excited upon these endogenous fluorophores present in the tissues. They display different optical properties (absorbance, reflectance, and fluorescence) which are sensitive to their microenvironment changes, which in turn get reflected in their corresponding wavelength and emission profiles [54]. The observed fluorescence from tissues under pathological conditions truly reflects the change in biochemical and tissue architecture. Abnormal conditions such as dysplasia may lead to disturbance in the fluorophore distribution and quantity, showing corresponding loss of tissue autofluorescence as compared with the healthy tissues. The NADH and FAD are important fluorophores in the epithelium of the oral mucosa, while in the stroma, it is the collagen that is dominant. Any change in the levels of these fluorophores is indicative of the metabolic status of the cells. Hence, change in spectral intensity from disease tissues could be contributed by a change in the concentrations of fluorophores [55,56,57].
Autofluorescence-based tools are non-invasive, easy to handle, and also minimize unnecessary surgical biopsy interventions. Due to these properties, the techniques can be used as a potential tool to monitor the malignant area in medically compromised patients. The autofluorescence-based diagnostic tools for oral cancer include the following:
-
Visual autofluorescence (visually enhanced lesion scope) VELscope
-
Autofluorescence spectroscopy
VELscope
Over the last few decades, tissue fluorescence has been exploited much to detect oral lesions by involving a variety of devices. VELscope is one such device that is based on fluorescence emitted by the natural fluorophores of the oral tissues upon suitable light excitations in the region 400–600 nm. It is a handheld device that consists of an arc lamp as an excitation light source to shine into the oral cavity and a series of reflectors and filters to view the emitted fluorescence through them. The device avoids procedures like rinsing of the oral cavity with chemical agents [57, 58]. The diagnostic ability of VELscope depends on the differences in the fluorescence emitted by the diseased and healthy tissues. Pale green is seen in the case of normal mucosa, while dark coloration is seen in dysplastic tissues, as shown in Fig. 4 [59]. Various studies have reported using VELscope in the diagnosis of oral lesions which are listed in Table 5. Further, some studies have highlighted the limitations of using VELscope alone in the diagnosis of potentially malignant disorders or oral squamous cell carcinoma [65].
a Clinical oral examination of the tongue and b loss of fluorescence at the region of oral lesion observed under VELscope, reprinted from Cicciù M. et al. [59] with creative copyright Licensee MDPI, Basel, Switzerland
Autofluorescence spectroscopy
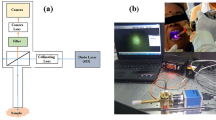
Fluorescence spectroscopy is a very sensitive technique that can detect minor metabolic or pathological changes taking place in the cells and tissues by illuminating the concerned fluorophores with appropriate monochromatic sources of light [66]. The fluorescence properties can differentiate abnormal tissues from the normal with higher sensitivity and specificity and thus providing an opportunity to use in diagnostic decision-making, including the detection of premalignant conditions and thereby bringing down the chances of repeated biopsies [67]. There are several reports of using fluorescence spectroscopy in the past to distinguish malignant tissues of different organs from their normal healthy counterparts in vitro/in vivo. The technique has shown its ability to detect even benign tissues from normal along with malignant tissues [68, 69]. The application of this technique in oral cancer detection is very well documented, and some studies are listed in Table 6. The fluorescence spectroscopy, when used along with suitable statistical analyses, has shown very high sensitivity and specificity in the discrimination of pathological conditions. The study by Jayanthi J L et al. applied linear discriminate analysis on the autofluorescence spectra recorded from 15 healthy people and 34 oral cancer patients. The results demonstrated clear differentiation of premalignant dysplasia from squamous cell carcinoma, as well as benign hyperplasia and hyperplasia from normal with a sensitivity of 86%, 78%, and 92%, and specificities of 90%, 100%, and 100%, respectively [75]. Similarly, in another study carried out by Sudha K D et al. the authors performed MATLAB-based PCA and k-means nearest neighbor (k-NN) analysis on 142 fluorescence spectra obtained from pathologically clinical samples recorded at 325-nm excitation using laser-induced fluorescence as a tool. The various features such as mean, median, maximum intensity, energy, spectral residuals, and standard deviation extracted from the fluorescence spectra of the normal, premalignant, and malignant oral tissue samples. In the study, the authors were able to show a robust application of PCA and k-NN analysis by which it can discriminate and also able to classify the unknown sample into a diagnostic group, which helps in reducing the misclassification as shown in Fig. 5. The authors were able to achieve sensitivity, specificity, and accuracy of 100, 94.5, and 96.17%, respectively. Based on the above study, the authors have tried to show that the laser-induced fluorescence technique can effectively be used to discriminate between normal, premalignant, and malignant conditions. Further, the time required to acquire and analyze the data is very less, and thus it holds very potentially as an alternative tool in the diagnosis of diseases [76].
The normalized fluorescence spectra of oral tissue. a Normal and b malignant and c scattered plot of 143 spectra from normal, premalignant, and malignant samples (NC, normal calibration; MC, malignant calibration; PC, premalignant calibration; NT, normal test; MT, malignant test; and PT, premalignant test), reprinted from Sudha KD. et al. [76] with permission of SPIE
While a study carried out by Yuvaraj M et al. monitored the spectral variation of porphyrin fluorophore in saliva samples collected using autofluorescence spectroscopy from oral squamous cell carcinoma (OSCC) patients and healthy individuals, the results demonstrate predominant emission patterns in the case of cancer patients as compared with the normal at 405-nm excitations. They observed additional spectral peaks at 625 and 687 nm in the cancer samples as compared with control due to the porphyrins. However, both of these peaks were absent in normal saliva samples [77]. The above observations have clearly demonstrated the potential of identifying porphyrins in saliva samples as a biomarker for oral cancer diagnosis using autofluorescence non-invasively. The above study reflects the potential use of fluorescence spectroscopy in early detection of premalignant lesions, even before when there are no visible clinical symptoms. The technique records fluorescence spectra qualitatively and is relatively inexpensive, portable, and easy to operate even by a technician with minimum training. These impeccable qualities make the fluorescence spectroscopy a promising tool to screen people with minimal complexity. They can provide a powerful and robust diagnostic platform for disease screening and subsequent treatment opportunities.
Conclusion
Oral cancer is preventable, and yet the present-day incidence and mortality rates remain to be high. Despite the existence of numerous conventional diagnostic techniques, many of which are invasive, and most of the cases are detected after they would have progressed to an advanced stage. The prevention entails in averting the progression of a premalignant disorder into full-blown cancer, by focusing on early detection strategies by using fluorescence-based spectroscopic tools, highlighted in this article. The fluorescence technique, explicitly fluorescence spectroscopy, holds the potential to identify oral precancerous and cancerous lesions with high sensitivity at an early stage, even when external morphological changes may not be visible. Besides, their cost-effectiveness, speed, and ability to provide real-time analysis of diagnosis objectively make them ideal diagnostic tools. Further, the technique holds immense potential in providing the molecular information generated from a fluorophore upon excitation. Therefore, it could increase the chances of survival, decrease morbidity, reduce the mortality rates, and ensure effective treatment, post-treatment, and a better quality of life for the patient.
References
Dikshit R, Gupta PC, Ramasundarahettige C, Gajalakshmi V, Aleksandrowicz L, Badwe R, Kumar R, Roy S, Suraweera W, Bray F, Mallath M, Singh PK, Sinha DN, Shet AS, Gelband H, Jha P (2012) Cancer mortality in India: a nationally representative survey. Lancet 379(9828):1807–1816
Sharma S, Satyanarayana L, Asthana S, Shivalingesh KK, Goutham BS, Ramachandra S (2018) Oral cancer statistics in India on the basis of first report of 29 population-based cancer registries. J Oral Maxillofac Pathol 22(1):18–26
Cheong SC, Vatanasapt P, Yi-Hsin Y, Zain RB, Kerr AR, Johnson NW (2017) Oral cancer in South East Asia: current status and future directions. Transl Res Oral Oncol 2:1–9
Johnson NW, Warnakulasuriya S, Gupta PC, Dimba E, Chindia M, Otoh EC, Sankaranarayanan R, Califano J, Kowalski L (2011) Global oral health inequalities in incidence and outcomes for oral cancer: causes and solutions. Adv Dent Res 23(2):237–246
Gupta N, Gupta R, Acharya AK, Patthi B, Goud V, Reddy S, Garg A, Singla A (2016) Changing trends in oral cancer-a global scenario. Nepal J Epidemiol 6(4):613–619
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Ray JG, Ganguly M, Rao BS, Mukherjee S, Mahato B, Chaudhuri K (2013) Clinico-epidemiological profile of oral potentially malignant and malignant conditions among areca nut, tobacco and alcohol users in Eastern India: a hospital based study. J Oral Maxillofac Pathol 17(1):45–50
Priya M, Lando HA (2014) Tobacco control: an issue twinned with oral cancer control. Int Dent J 64(5):229–232
Natarajan E, Eisenberg E (2011) Contemporary concepts in the diagnosis of oral cancer and precancer. Dent Clin N Am 55(1):63–88
Gupta B, Ariyawardana A, Johnson NW (2013) Oral cancer in India continues in epidemic proportions: evidence base and policy initiatives. Int Dent J 63(1):12–25
Elango JK, Sundaram KR, Gangadharan P, Subhas P, Peter S, Pulayath C, Kuriakose MA (2009) Factors affecting oral cancer awareness in a high-risk population in India. Asian Pac J Cancer Prev 10(4):627–630
Mes Brocklehurst P, Kujan O, O’Malley LA, Ogden G, Shepherd S, Glenny AM (2013) Screening programmes for the early detection and prevention of oral cancer. The Cochrane Database Syst Rev 19(3):CD004150
Walsh T, Liu JL, Brocklehurst P, Glenny AM, Lingen M, Kerr AR, Ogden G, Warnakulasuriya S, Scully C (2018) Screening for mouth cancer: the pros and cons of a national programme. Br Dent J 225:815–819
Messadi DV (2013) Diagnostic aids for detection of oral precancerous conditions. Int J Oral Sci 5(2):59–65
Chang IH, Rong SJ, Wong YK, Wu SH, Chen FJ, Liu SA (2011) Visual screening of oral cavity cancer in a male population: experience from a medical center. J Chin Med Assoc 74(12):561–566
Walsh T, Liu JL, Brocklehurst P, Glenny AM, Lingen M, Kerr AR, Ogden G, Warnakulasuriya S, Scully C (2013) Clinical assessment to screen for the detection of oral cavity cancer and potentially malignant disorders in apparently healthy adults. Cochrane Database Syst Rev 21(11):CD010173
Monteiro LS, Salazar F, Pacheco JJ, Martins M, Warnakulasuriya S (2015) Outcomes of invitational and opportunistic oral cancer screening initiatives in Oporto, Portugal. J Oral Pathol Med 44(2):145–152
Scott SE, Rizvi K, Grunfeld EA, McGurk M (2010) Pilot study to estimate the accuracy of mouth self-examination in an at-risk group. Head Neck 32(10):1393–1401
Mathew B, Sankaranarayanan R, Sunilkumar KB, Kuruvila B, Pisani P, Nair MK (1997) Reproducibility and validity of oral visual inspection by trained health workers in the detection of oral precancer and cancer. Br J Cancer 76(3):390–394
Jullien JA, Downer MC, Zakrzewska JM, Speight PM (1995) Evaluation of a screening test for the early detection of oral cancer and precancer. Community Dent Health 12(1):3–7
Downer MC, Evans AW, Hughes Hallett CM, Jullien JA, Speight PM, Zakrzewska JM (1995) Evaluation of screening for oral cancer and precancer in a company headquarters. Community Dent Oral Epidemiol 23(2):84–88
Mehrotra R, Yadav S (2006) Oral squamous cell carcinoma: etiology, pathogenesis and prognostic value of genomic alterations. Indian J Cancer 43(2):60–66
Masthan KM, Sankari SL, Babu NA, Rajguru JP (2013) How to help the oral pathologist in making an accurate diagnosis. J Clin Diagn Res 7(1):181–184
Woolgar JA, Triantafyllou A (2009) Pitfalls and procedures in the histopathological diagnosis of oral and oropharyngeal squamous cell carcinoma and a review of the role of pathology in prognosis. Oral Oncol 45(4–5):361–385
Masthan KM, Babu NA, Dash KC, Elumalai M (2012) Advanced diagnostic aids in oral cancer. Asian Pac J Cancer Prev 13(8):3573–3576
Goodson ML, Smith DR, Thomson PJ (2017) Efficacy of oral brush biopsy in potentially malignant disorder management. J Oral Pathol Med 46(10):896–901
Kaur M, Handa U, Mohan H, Dass A (2016) Evaluation of brush cytology and DNA image cytometry for the detection of cancer of the oral cavity. Diagn Cytopathol 44(3):201–205
Gupta S, Shah JS, Parikh S, Limbdiwala P, Goel S (2014) Clinical correlative study on early detection of oral cancer and precancerous lesions by modified oral brush biopsy and cytology followed by histopathology. J Cancer Res Ther 10(2):232–238
Reddy SG, Kanala S, Chigurupati A, Kumar SR, Poosarla CS, Venkata Ramana RB (2012) The sensitivity and specificity of computerized brush biopsy and scalpel biopsy in diagnosing oral premalignant lesions: a comparative study. J Oral Maxillofac Pathol 16:349–353
Delavarian Z, Mohtasham N, Mosannen-Mozafari P, Pakfetrat A, Shakeri MT, Ghafoorian-Maddah R (2010) Evaluation of the diagnostic value of a modified liquid-based cytology using OralCDx brush in early detection of oral potentially malignant lesions and oral cancer. Med Oral Patol Oral Cir Bucal 15(5):e671–e676
Babshet M, Nandimath K, Pervatikar S, Naikmasur V (2011) Efficacy of oral brush cytology in the evaluation of the oral premalignant and malignant lesions. J Cytol 28(4):165–172
Lindenblatt RC, Martinez GL, Silva LE, Faria PS, Camisasca DR, Lourenco Sde Q (2012) Oral squamous cell carcinoma grading systems-analysis of the best survival predictor. J Oral Pathol Med 41(2):34–39
Akhter M, Hossain S, Rahman QB, Molla MR (2011) A study on histological grading of oral squamous cell carcinoma and its co-relationship with regional metastasis. J Oral Maxillofac Pathol 15(2):168–176
He L, Long LR, Antani S, Thoma GR (2012) Histology image analysis for carcinoma detection and grading. Comput Methods Prog Biomed 107(3):538–556
Singh D, Shukla RK (2015) Utility of toluidine blue test in accessing and detecting intra-oral malignancies. Indian J Otolaryngol Head Neck Surg 67(Suppl 1):47–50
Awan K, Yang Y, Morgan P, Warnakulasuriya S (2012) Utility of toluidine blue as a diagnostic adjunct in the detection of potentially malignant disorders of the oral cavity-a clinical and histological assessment. Oral Dis 18(8):728–733
Petruzzi M, Lucchese A, Baldoni E, Grassi FR, Serpico R (2010) Use of Lugol’s iodine in oral cancer diagnosis: an overview. Oral Oncol 46(11):811–813
Vijayakumar V, Reghunathan D, Edacheriyan B, Aswin M (2019) Role of toluidine blue staining in suspicious lesions of oral cavity and oropharynx. Indian J Otolaryngol Head Neck Surg 71:142–146
Chaudhari A, Hegde-Shetiya S, Shirahatti R, Agrawal D (2013) Comparison of different screening methods in estimating the prevalence of precancer and cancer amongst male inmates of a jail in Maharashtra, India. Asian Pac J Cancer Prev 14(2):859–864
Cancela-Rodriguez P, Cerero-Lapiedra R, Esparza-Gomez G, Llamas-Martinez S, Warnakulasuriya S (2011) The use of toluidine blue in the detection of pre-malignant and malignant oral lesions. J Oral Pathol Med 40:300–304
Nagaraju K, Prasad S, Ashok L (2010) Diagnostic efficiency of toluidine blue with Lugol’s iodine in oral premalignant and malignant lesions. Indian J Dent Res 21(2):218–223
Allegra E, Lombardo N, Puzzo L, Garozzo A (2009) The usefulness of toluidine staining as a diagnostic tool for precancerous and cancerous oropharyngeal and oral cavity lesions. Acta Otorhinolaryngol Ital 29(4):187–190
Chainani-Wu N, Madden E, Cox D, Sroussi H, Epstein J, Silverman SJ (2015) Toluidine blue aids in detection of dysplasia and carcinoma in suspicious oral lesions. Oral Dis 21(7):879–885
Jain N, Nagarajappa AK, Bhasin M, Pandya D, Tripathi KP (2018) Role of chemiluminescence examination as non-invasive diagnostic tool in early detection of leukoplakia. J Oral Biol Craniofac Res 8(3):177–181
Liu D, Zhao X, Zeng X, Dan H, Chen Q (2016) Non-invasive techniques for detection and diagnosis of oral potentially malignant disorders. Tohoku J Exp Med 238(2):165–177
Chaudhry A, Manjunath M, Ashwatappa D, Krishna S, Krishna AG (2016) Comparison of chemiluminescence and toluidine blue in the diagnosis of dysplasia in leukoplakia: a cross-sectional study. J Investig Clin Dent 7(2):132–140
Vashisht N, Ravikiran A, Samatha Y, Rao PC, Naik R, Vashisht D (2014) Chemiluminescence and toluidine blue as diagnostic tools for detecting early stages of oral cancer: an in-vivo study. J Clin Diagn Res 8(4):ZC35–ZC38
Rajmohan M, Rao UK, Joshua E, Rajasekaran ST, Kannan R (2012) Assessment of oral mucosa in normal, precancer and cancer using chemiluminescent illumination, toluidine blue supravital staining and oral exfoliative cytology. J Oral Maxillofac Pathol 16(3):325–329
Shweta U, Mukta BM, Shirish D, Vijay W, Prajakta Z, Minal C, Vinay H, Tushar PT, Manju M (2012) Evaluation of chemiluminescence, toluidine blue and histopathology for detection of high risk oral precancerous lesions: a cross-sectional study. BMC Clin Pathol 12:6
Awan KH, Morgan PR, Warnakulasuriya S (2011) Utility of chemiluminescence (ViziLite) in the detection of oral potentially malignant disorders and benign keratoses. J Oral Pathol Med 40(7):541–544
Epstein JB, Gorsky M, Lonky S, Silverman S Jr, Epstein JD, Bride M (2006) The efficacy of oral lumenoscopy (ViziLite) in visualizing oral mucosal lesions. Spec Care Dentist 26(4):171–174
Nagi R, Reddy-Kantharaj YB, Rakesh N, Janardhan-Reddy S, Sahu S (2016) Efficacy of light based detection systems for early detection of oral cancer and oral potentially malignant disorders: systematic review. Med Oral Patol Oral Cir Bucal 21(4):e447–e455
Balasubramaniam AM, Sriraman R, Sindhuja P, Mohideen K, Parameswar RA, Muhamed Haris KT (2015) Autofluorescence based diagnostic techniques for oral cancer. J Pharm Bioallied Sci 7(Suppl 2):S374–S377
Liu W, Zhang X, Liu K, Zhang S, Duan Y (2013) Laser-induced fluorescence: progress and prospective for in vivo cancer diagnosis. Chin Sci Bull 58:2003–2016
Rathnakar B, Rao BSS, Prabhu V, Chandra S, Mahato KK (2018) Laser-induced autofluorescence-based objective evaluation of burn tissue repair in mice. Lasers Med Sci 33(4):699–707
Nazeer Shaiju S, Ariya S, Asish R, Salim Haris P, Anita B, Arun Kumar G, Jayasree RS (2011) Habits with killer instincts: in vivo analysis on the severity of oral mucosal alterations using autofluorescence spectroscopy. J Biomed Opt 16(8):087006
Prabhu V, Rao SB, Fernandes EM, Rao AC, Prasad K, Mahato KK (2014) Objective assessment of endogenous collagen in vivo during tissue repair by laser induced fluorescence. PLoS One 9(5):e98609
Mc-Namara KK, Martin BD, Evans EW, Kalmar JR (2012) The role of direct visual fluorescent examination (VELscope) in routine screening for potentially malignant oral mucosal lesions. Oral Surg Oral Med Oral Pathol Oral Radiol 114(5):636–643
Cicciù M, Cervino G, Fiorillo L, D’Amico C, Oteri G, Troiano G, Zhurakivska K, Lo Muzio L, Herford AS, Crimi S, Bianchi A, Di Stasio D, Rullo R, Laino G, Laino L (2019) Early diagnosis on oral and potentially oral malignant lesions: a systematic review on the VELscope® fluorescence method. Dent J (Basel) 7(3):93
Ganga RS, Gundre D, Bansal S, Shirsat PM, Prasad P, Desai RS (2017) Evaluation of the diagnostic efficacy and spectrum of autofluorescence of benign, dysplastic and malignant lesions of the oral cavity using VELscope. Oral Oncol 75:67–74
Huang TT, Huang JS, Wang YY, Chen KC, Wong TY, Chen YC, Wu CW, Chan LP, Lin YC, Kao YH, Nioka S, Yuan SF, Chung PC (2017) Novel quantitative analysis of autofluorescence images for oral cancer screening. Oral Oncol 68:20–26
Hanken H, Kraatz J, Smeets R, Heiland M, Assaf AT, Blessmann M, Eichhorn W, Clauditz TS, Gröbe A, Kolk A, Rana M (2013) The detection of oral premalignant lesions with an autofluorescence based imaging system (VELscope™)-a single blinded clinical evaluation. Head Face Med 9:23
Rana M, Zapf A, Kuehle M, Gellrich NC, Eckardt AM (2012) Clinical evaluation of an autofluorescence diagnostic device for oral cancer detection: a prospective randomized diagnostic study. Eur J Cancer Prev 21(5):460–466
Scheer M, Neugebauer J, Derman A, Fuss J, Drebber U, Zoeller JE (2011) Autofluorescence imaging of potentially malignant mucosa lesions. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 111(5):568–577
Mascitti M, Orsini G, Tosco V, Monterubbianesi R, Balercia A, Putignano A, Procaccini M, Santarelli A (2018) An overview on current non-invasive diagnostic devices in oral oncology. Front Physiol 9:1510
Jayaprakash V, Sullivan M, Merzianu M, Rigual NR, Loree TR, Popat SR, Moysich KB, Ramananda S, Johnson T, Marshall JR, Hutson AD, Mang TS, Wilson BC, Gill SR, Frustino J, Bogaards A, Reid ME (2009) Autofluorescence-guided surveillance for oral cancer. Cancer Prev Res (Phila) 2(11):966–974
Paderni C, Compilato D, Carinci F, Nardi G, Rodolico V, Lo Muzio L, Spinelli G, Mazzotta M, Campisi G (2011) Direct visualization of oral-cavity tissue fluorescence as novel aid for early oral cancer diagnosis and potentially malignant disorders monitoring. Int J Immunopathol Pharmacol 24(2Suppl):121–128
Francisco ALN, Correr WR, Azevedo LH, Galletta VK, Pinto CAL, Kowalski LP, Kurachi C (2014) Fluorescence spectroscopy for the detection of potentially malignant disorders and squamous cell carcinoma of the oral cavity. Photodiagn Photodyn Ther 11(2):82–90
Udayakumar K, Yuvaraj M, Awad F, Jayanth V, Aruna PR, Koteeswaran D, David MB, Ganesan S (2014) Native fluorescence and time-resolved fluorescence spectroscopic characterization of normal and malignant oral tissues under UV excitation—an in vitro study. J Fluoresc 24:613–623
Betsy J, Prasanth CS, Jayanthi JL, Presanthila J, Subhash N (2015) Detection and quantification of dental plaque based on laser-induced autofluorescence intensity ratio values. J Biomed Opt 20(4):048001
Francisco ALN, Correr WR, Azevedo LH, Galletta VK, Pinto CAL, Kowalski LP, Kurachi C (2014) Fluorescence spectroscopy for the detection of potentially malignant disorders of the oral cavity: analysis of 30 cases. Laser Phys 24:015701
Chaturvedi P, Majumder SK, Krishna H, Muttagi S, Gupta PK (2010) Fluorescence spectroscopy for noninvasive early diagnosis of oral mucosal malignant and potentially malignant lesions. J Cancer Res Ther 6(4):497–502
Kurachi C, Fontana CR, Rosa LE, Bagnato VS (2008) Fluorescence spectroscopy for the detection of tongue carcinoma-validation in an animal model. J Biomed Opt 13(3):034018
Majumder SK, Ghosh N, Kataria S, Gupta PK (2003) Nonlinear pattern recognition for laser-induced fluorescence diagnosis of cancer. Lasers Surg Med 33:48–56
Jayanthi JL, Mallia RJ, Shiny ST, Baiju KV, Mathews A, Kumar R, Sebastian P, Madhavan J, Aparna GN, Subhash N (2009) Discriminant analysis of autofluorescence spectra for classification of oral lesions in vivo. Lasers Surg Med 41(5):345–352
Sudha KD, Mahato KK (2007) Optical pathology using oral tissue fluorescence spectra: classification by principal component analysis and k-means nearest neighbour analysis. J Biomed Opt 12(1):014028
Yuvaraj M, Udayakumar K, Jayanth V, Prakasa RA, Bharanidharan G, Koteeswaran D, Munusamy BD, Murali KC, Ganesan S (2014) Fluorescence spectroscopic characterization of salivary metabolites of oral cancer patients. J Photochem Photobiol B 130:153–160
Acknowledgments
The authors thank Manipal Academy of Higher Education (MAHE), Manipal, India and TIFAC-CORE in Pharmacogenomics at Manipal School of Life Sciences for developing infrastructure and facilities to carry out this study. One of the authors, CRR would also like to thank MAHE for providing financial support under Dr. TMA Pai Structurted PhD Scheme and Indian Council of Medical research, Govt. of India, for Senior Research Fellowship.
Funding
Indian Council of Medical Research, Government of India, New Delhi (Sanction no.5/3/8/ITRF/2018-ITR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent to participation
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Raghushaker, C.R., D’Souza, M., Urala, A.S. et al. An overview of conventional and fluorescence spectroscopy tools in oral cancer diagnosis. Laser Dent Sci 4, 167–179 (2020). https://doi.org/10.1007/s41547-020-00104-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41547-020-00104-9