Abstract
Seroquel drug was studied for corrosion inhibition of low carbon steel in 1 M HCl solution by using experimentally with optimized concentrations at an elevated temperature range of 303–333 K. The inhibition efficiency was studied in the absence and presence of Seroquel by weight loss, electrochemical impedance spectroscopy and potentiodynamic polarization measurements. The inhibition effect of the inhibitor increases with the increasing concentration of Seroquel. Tafel polarization studies revealed that the inhibitor acts as mixed type inhibitor. The inhibition effect was attributed due to the adsorption process on low carbon steel and it obeys Temkin adsorption isotherm. The effect of temperature with the inhibition effect of inhibitor from the solution was examined by activation and thermodynamic parameters. Surface morphology of the corroded specimens were studied by SEM technique and IR spectrum of the corrosion product shows the influence of the Seroquel for corrosion inhibition.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Low carbon steel is an alloy of iron, and finds huge applications in industrial and structural field because of its superior mechanical properties. When it comes in contact with acidic media it looses its properties and undergoes dissolution. Several corrosion control approaches are used to avoid metal dissolution in acid media. But, the use of corrosion inhibitor is the most efficient and practical method to control the corrosion of metals. Corrosion inhibitors are heterocyclic molecules having heteroatoms such as nitrogen, phosphorous, sulfur, oxygen, and π electrons in their heterocyclic ring system [1]. The toxicity of widely used corrosion inhibitors intends to replace them by non-toxic chemical substances (medicinal drugs or plant products) [2]. Due to non-poisonous, extensive use, and harmless effects on environment, medicinal compounds are extensively used instead of toxic organic corrosion inhibitors [3, 4]. The reduction in the rate of corrosion is due to the adsorption of organic compounds on the metal surface which blocks the active cites on the surface [5]. Presence of heteroatoms such as nitrogen, oxygen, phosphorous, sulfur, and aromatic rings makes the organic molecule to adsorb through them and blocks the active corrosion sites on the metal surface [6, 7]. The influence of inhibitors on the kinetics of electrochemical reactions modifies the dissolution of metals in acid solution [8]. Some of the medicinal drugs such as Floctafenine [9], Ziprasidone [10, 11], Clozapine [12], Cefuroxime axetil [13], Amoxicillin [14], and Modazar [15] have been investigated as good corrosion inhibitors in acid solution.
The present work is attributed to the adsorption of inhibitor molecules on low carbon steel from bulk of the solution. The adsorption phenomenon may occur due to the presence of electron-rich heteroatoms and π bonds which makes to study and to prove it as an effective corrosion inhibitor. The scope of this work is to investigate the influence of Seroquel drug as an effective corrosion inhibitor for low carbon steel in HCl solution. The molecular structure of Seroquel clearly shows the presence of hetero atoms like N, O, and S in the aromatic rings, functional groups such as –C=N–, –OH, –NH in its structure, π electron-rich aromatic rings, fair molecular size, and quite good planarity to wrap sufficient metal surface which might be supportive of its adsorption on the metal surface, giving importance to its adsorption on the metal surface.
From the literature, it has been found that, several researchers worked with drug molecule as corrosion inhibitor having properties like antifungal, antibacterial [9,10,11,12], etc., since only few literatures are available on the use of antipsychotic drug as corrosion inhibitor, like Floctafenine [9], Ziprasidone [10, 11], Clozapine [12], which set off the use of Seroquel, an antipsychotic drug to check its inhibition action for corrosion of steel.
2 Experimental
2.1 Material
The commercially available low carbon steel strips (composition: 0.35% C, 0.032% Mn, 0.028% P, 0.03% S, and remaining Fe) with a dimension of 4 cm × 1 cm × 0.1 cm were used for the present study. The surface of the steel was polished by using SiC emery papers of grade number 80 up to 2000. Weight loss method was carried out in 1 M HCl solution by these same strips and electrochemical studies were carried out by strips with 1 cm2 exposed area (remaining portion was covered by epoxy resin). Aggressive corrosive media was prepared by AR grade HCl.
2.2 Inhibitor Solution
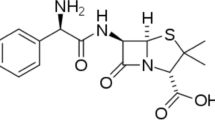
Seroquel is an antipsychotic medicine to treat schizophrenia and bipolar disorder. The IUPAC name of this drug is (2-[2-(4-dibenzo [b,f] [1, 4] thiazepin-11-yl-1-piperazinyl) ethoxy]-ethanol). The molecular structure of Seroquel is shown in Fig. 1. Different concentrations of the inhibitor in 1 M HCl were prepared and used for all corrosion experiments.
2.3 Weight Loss Measurements
Weight loss measurements were carried out by immersing steel sheets in 100 cm3 of 1 M HCl solution containing different concentrations of inhibitor. The steel sheets were taken out after 4 h of immersion time and washed with tap water then with distilled water, dried in acetone, and accurately weighed. All the tests were done in triplicate and an average value was considered.
2.4 Electrochemical Measurements
The electrochemical measurements were carried out by using CHI-608D electrochemical work station with three electrode system. The electrode system consists of working electrode (low carbon steel), reference electrode (calomel), and an auxiliary electrode (platinum). The electrochemical parameters were recorded at different temperatures from 303 to 333 K. The working electrode was kept in contact with test solution for 30 min to attain steady-state open circuit potential. Potential–current curves were recorded by scanning at a rate of 0.01 V/s in the potential range of − 0.2 –+ 0.2 V to the OCP values. The impedance data were obtained using AC signal with 0.005 V amplitude in the frequency range of 1 Hz to 10 kHz.
2.5 Adsorption Isotherm
Corrosion inhibition of inhibitors on steel in acid media is due to their adsorption on the steel surface. The adsorption property of Seroquel on low carbon steel at different temperatures in 1 M HCl was studied using several adsorption isotherms and the thermodynamic data were determined using standard equations.
2.6 Activation Parameters
The variation in inhibition efficiency at different temperatures was studied by activation parameters [16]. Using the data obtained for corrosion rate at different temperatures, graphs were plotted and calculations were made using standard equations.
2.7 Scanning Electron Microscopic Studies
Low carbon steel samples were kept in contact with 1 M HCl solution in the absence and presence of optimum concentration of inhibitors over a period of 4 h of immersion time. The samples were taken out and then washed with distilled water, dried, and analyzed by SEM technique. The SEM analysis was carried out using VEGA3 TESCAN SEM instrument at an accelerating beam of 25 kV.
2.8 FTIR Spectral Studies
FTIR Spectrum of the pure compound and the compound scarped from the metal surface after corrosion was carried out by using Frontier Perkin Elmer spectrometer.
3 Results and Discussion
3.1 FTIR Spectral Studies
FTIR spectral studies gives the valuable information about the coordination of inhibitor molecule with the metal surface through the hetero atoms. Specific to the inhibitor used in the present study, i.e., Seroquel, adsorption may be through the heteroatoms like, N, O, and S and is confirmed by the shift of absorption peak in the IR spectrum of scrapped compound compared to IR spectrum of pure compound.
FTIR spectrum of pure Seroquel is shown in Fig. 2a. The presence of broad band at 3312 cm− 1 indicates the presence of hydroxyl group stretching vibration. The peak at 2876 cm− 1 indicates the presence of carboxylic acid O–H stretching vibration. The peak at 1595 cm− 1 shows the presence of aromatic C=C bending vibration. The narrow peak at 1381 cm− 1 is due to C–H absorption. The presence of narrow band at 1140 cm− 1 is due to the C–O group. The peak at 763 cm− 1 shows the presence of aromatic C–H bending vibration.
Figure 2b shows the FTIR spectrum of the compound scrapped from the metal surface after corrosion in 1 M HCl solution. The appearance of absorption peak at 3452 cm− 1, compared to 3312 cm− 1 in the pure compound indicates the adsorption of molecule with the metal surface through –OH group of the Seroquel molecule. The band at 1115 cm− 1, compared to 1140 cm− 1 for the pure compound, corresponding to C–O–C stretching vibration also indicates the coordination of molecule through O heteroatom. The peak at 1020 cm− 1 is due to the presence of aliphatic amines, which also supports the bonding of Seroquel through N atom. Slight shift in the wave number of the absorption peaks in the scrapped compound confirms the interaction of the Seroquel with steel and the presence of inhibitor molecule on the surface of the metal.
3.2 Weight Loss Method
The study was carried out by immersing mild steel strips in 1 M HCl solution containing different concentrations of the inhibitor at different temperatures for maximum of 4 h. The weights of each steel strip were measured before and after immersion using an analytical balance. The percentage inhibition efficiency, ηw was calculated using the formula,
where W and Wo represent weight loss of steel in the presence and absence of corrosion inhibitor, respectively.
Rate of corrosion is determined using the following equation,
where Δm is weight loss in absence of inhibitor (m0) − weight loss in presence of inhibitor (m1), S is surface area, T is time of exposure in hours.
Calculated values of corrosion rates and inhibition efficiency are listed in Table 1. It is evident from the values obtained that the corrosion rate of steel decreases with increase in the concentration of Seroquel against blank. As shown in Table 1, it is clear that inhibition efficiency of Seroquel increases with increase in concentration up to 1000 ppm. So 1000 ppm is considered as the critical concentration for maximum inhibition efficiency to be achieved. The decrease in corrosion rate with the increase in inhibitor concentration is due to adsorption of inhibitor molecule on metal surface [17].
3.3 Electrochemical Measurements
3.3.1 Polarization Studies
Polarization studies were carried out to obtain the knowledge of kinetics of electrode reactions (both anodic and cathodic) [18]. Electrochemical corrosion parameters, i.e., corrosion current (icorr), corrosion potential (Ecorr), Tafel slope (βa and βc) from the extrapolation of the polarization curves, and ηp (inhibition efficiency according to polarization data) are given in Table 2. The polarization plots of low carbon steel in 1 M HCl in the presence and absence of different concentrations of Seroquel at different temperatures are shown in Fig. 3. The ηp was calculated using the following equation,
where \(i_{{{\text{corr}}}}^{{\text{o}}}\) and icorr are the current densities in the absence and presence of corrosion inhibitor, respectively. As shown in Fig. 3, it can be observed that the addition of Seroquel shifts both the anodic and cathodic curves toward lower values of current densities. This is due to the adsorption of Seroquel on steel surface which inhibits metal dissolution at anode and hydrogen liberation at cathode causing a remarkable decrease in the corrosion rate. As reported in many literatures, the inhibitor can be classified as anodic or cathodic type, if the displacement of corrosion potential is greater than ± 85 mV compared to the corrosion potential of the blank [19]. In our present work, since corrosion potential value is lesser than ± 85 mV, the inhibitor acts as mixed type inhibitor [20,21,22,23].
The effect of temperature on corrosion of steel in the absence and presence of Seroquel was studied in the temperature ranging from 303 to 333 K and the results are tabulated in Table 2. From the values obtained it is clear that the corrosion rate of low carbon steel increased as the temperature increases in the absence of the inhibitor. But in the presence of the inhibitor, the corrosion rate diminished with the rise in temperature; this also says that the increased adsorption of the inhibitor on the surface of metal thereby blocks metal sites which are prone for corrosion at high temperature. The inhibitor attained maximum inhibition efficiency in the range of 70–84% at an optimum concentration of 1000 ppm of Seroquel.
3.3.2 Electrochemical Impedance Spectroscopy Measurements
Figure 4 represents EIS spectra for low carbon steel in 1 M HCl at different temperatures in the presence and absence of different concentrations of Seroquel inhibitor which are presented as Nyquist plots. The equivalent circuit model is shown in Fig. 5 which was used to analyze the current response as a function of frequency. Calculated EIS experimental parameters such as charge transfer resistance which is equivalent to polarization resistance (Rp), double layer capacitance (Cdl), inhibition efficiency (ηz), and surface coverage (Θ) are listed in Table 3. It is clear from Table 3 that Rp value increases and Cdl value decreases with increase in inhibitor concentration. This is due to the adsorption of inhibitor molecules on the metal surface thereby decreasing the extent of metal dissolution [24]. The decrease in Cdl values with increase in inhibitor concentration is probably due to the decrease in dielectric constant and/or due to the increase in the thickness of the double layer on the metal surface [25]. The deviations from perfect semicircles are involved with the frequency dispersion of interfacial impedance which is due to many physical factors like active sites, surface roughness, and non-homogeneity of the mild steel. The increase in diameter of the semicircle with increase in the inhibitor concentration indicates that the charge transfer process is the main factor in controlling the corrosion of mild steel, which is mainly due to increase in charge transfer resistance and formation of more compact monolayer on metal surface [26].
Impedance study was also carried out in the temperature range of 303–333 K, and the results are shown in Table 3. It has been found that the value of Rp decreases as the temperature increases in the absence of the inhibitor, which indicates the increase of corrosion at high temperature. But, value of Rp increased significantly by the addition of Seroquel at high temperature which indicates that the increase of adsorption thereby decreases the corrosion rate.
The ηz values were calculated using the following relation,
where Rp and \(R_{{\text{p}}}^{{\text{o}}}\) are polarization resistance values in the presence and absence of inhibitor.
3.4 Adsorption Isotherm
Adsorption isotherm is the most important topic in the field of corrosion because it provides some fundamental information about interactions between adsorbate (inhibitor) and adsorbent (metallic surface). Depending on the electronic structure of inhibitor molecule, nature of metal and electrolyte, solution temperature, etc., adsorption may be chemisorption, physisorption, or both. Physical adsorption is associated with weak van der Waals or electrostatic interactions between the adsorbent and the adsorbate in the neutral or ionic form, respectively.
Corrosion inhibition tendency of Seroquel on steel surface is due to the adsorption phenomenon which decreases the corrosion rate and this is due to formation of protective film by adsorbed inhibitor molecule [27]. The adsorption process on the surface of corroding metal always tends to attain an adsorption steady state rather than equilibrium state. But when the rate of corrosion decreases in the presence of an inhibitor, the steady adsorption state tends to attain quasi-equilibrium state. Hence, it is required to consider quasi-equilibrium state in a thermodynamic way by proper adsorption isotherm. The adsorption isotherm study explains the interaction between mild steel surface and the inhibitor molecules [28]. For this, surface coverage (Θ) values were calculated at different concentrations of the inhibitor in 1 M HCl solution. Adsorption process can be explained by two types of interaction [29]: physisorption which involves electrostatic attraction between ionic charges, low heat of adsorption, and hence less stability at relatively low temperatures, and chemisorptions which involves charge transfer to the metal surface from the inhibitor molecules, stronger adsorption energy, and hence more stability at high temperatures.
The mode of adsorption of Seroquel on steel in 1 M HCl at different temperatures was studied by fitting the experimental data into different adsorption isotherms. In this study, several commonly used isotherms, namely, Langmuir, Temkin (Fig. 6), and Freundlich were tested. In each case, a function of the fractional surface coverage (θ) is plotted against a function of the concentration of inhibitor (C) and the associated adsorption parameters are deduced from slope or intercept or both. The routine involves fitting the surface coverage data into different adsorption models and the isotherm that best fits the data is used to describe the adsorption behavior. The best fit is usually the one that gives the highest regression coefficient (R2) value from the linear plots. Among the various isotherms, Temkin adsorption isotherm gave the best line of fit, with the values of regression coefficient (R2) very close to unity and is shown in Fig. 6. This suggests that the adsorption of inhibitor molecule on the metal surface obeys Temkin adsorption isotherm [30] and it is given by the following equation:
where Θ is the degree of surface coverage, C is the inhibitor concentration, f is the molecular interaction parameter, Kads is the adsorption equilibrium constant which indicates the interaction between adsorbent and adsorbate [31]. Kads is related to \(\Updelta G_{{{\text{ads}}}}^{{\text{o}}}\) by the following equation:
where R is the gas constant, T is the absolute temperature in K, and 55.5 is the molar concentration of water in the solution. Thermodynamic parameters for the adsorption of inhibitor at different temperatures derived from Temkin adsorption isotherms are given in Table 4. As reported in literature the higher values of Kads attribute to strong and stable adsorption of inhibitor layer on the metal surface [31, 32]. It is clear from Table 4 that the higher value of Kads contributes to strong interaction between the inhibitor molecule and the steel surface.
Literature also reveals that the negative value of \(\Updelta G_{{{\text{ads}}}}^{{\text{o}}}\) indicates the spontaneous adsorption of inhibitor molecule on the metal surface. It is evident from Table 4 that the reported \(\Updelta G_{{{\text{ads}}}}^{{\text{o}}}\) values at different temperatures are negative which indicates that the adsorption of Seroquel inhibitor molecule on the low carbon steel surface in 1 M HCl solution is a spontaneous process. Generally, the values of \(\Updelta G_{{{\text{ads}}}}^{{\text{o}}}\) lesser than or equal to − 20 kJ/mol represent the electrostatic interaction between the metal surface and the inhibitor molecules, i.e., physisorption, and the values greater than or equal to -40 kJ/mol represent the charge transfer between the metal surface and inhibitor molecules, i.e., chemisorptions [33]. In this work, \(\Updelta G_{{{\text{ads}}}}^{{\text{o}}}\) values were found to be ranging from − 30.77 to − 31.55 kJ/mol, which indicates that the adsorption process of the inhibitor on the steel was due to both physisorption and chemisorption (comprehensive adsorption) [34, 35].
A linear graph of \(\Updelta G_{{{\text{ads}}}}^{{\text{o}}}\)/T against 1000/T is shown in Fig. 7, and the calculated values of \(\Updelta H_{{{\text{ads}}}}^{{\text{o}}}\) and \(\Updelta S_{{{\text{ads}}}}^{{\text{o}}}\) are listed in Table 4. Slope of the straight line gives \(\Updelta H_{{{\text{ads}}}}^{{\text{o}}},\) and \(\Updelta S_{{{\text{ads}}}}^{{\text{o}}}\) values were calculated using the standard equation,
Generally, negative values of enthalpy of adsorption indicate physical adsorption, i.e., endothermic adsorption, whereas positive values indicate chemical adsorption, i.e., exothermic adsorption [36]. The values of enthalpy of adsorption lesser than − 40 kJ/mol involve physical adsorption, whereas enthalpy of adsorption equal to − 100 kJ/mol involves chemical adsorption [37]. In this work, the value of enthalpy of adsorption is − 34.15 kJ/mol which represents that the adsorption of the Seroquel inhibitor on the steel surface is endothermic and it suggests physical adsorption. The entropy is maximum when there is a free mobility of inhibitor molecule in the solution [37]. The negative value of the entropy of adsorption indicates that the adsorption is accompanied by a decrease in entropy and is due to orderly adsorption of the inhibitor molecules on the steel surface [38].
3.5 Activation Parameters
The corrosion rate of metal is largely influenced by the temperature as it modifies the inhibition property due to decomposition and/or rearrangements of inhibitor [39]. To evaluate thermodynamic parameters in order to explain inhibition and the adsorption process, electrochemical experiments were carried out in the absence and presence of different concentrations of the inhibitor in the temperature range of 303–333 K. The temperature dependence of corrosion rate can be explained by Arrhenius equation and transition state theory [40]. The Arrhenius equation and transition state equation can be written as,
where γcorr is the corrosion rate, Ea is the apparent activation energy, A is the Arrhenius pre-exponential factor, R is the gas constant, T is the temperature in Kelvin, h is Planck’s constant, N is the Avogadro number, ΔS* is the apparent entropy of activation, and ΔH* is the apparent enthalpy of activation. Arrhenius plot, obtained by plotting a graph of ln(γcorr) versus 1000/T, is shown in Fig. 8. The calculated values of Ea, A, ΔH*, and ΔS* are reported in Table 5. From the slope of each straight line shown in Fig. 8, Ea values were calculated using the relation Ea = (slope × R). Figure 9 represents a plot of ln(γcorr/T) versus 1000/T. From the straight line of the graph, ΔH* values were calculated by the slope of the straight line using the equation ΔH* = − slope × R and ΔS* values were calculated by the intercept using the equation,
From the Ea values reported in Table 5, it is clear that the higher value of Ea than that of the blank suggests that the metal dissolution is suppressed by the addition of inhibitor [41]. The increase in ΔH* value is an indication of the formation of protective layer on the steel surface in contact with the inhibited solution. The less negative value of ΔS* in the presence of inhibitor than compared to blank is due to adsorption of inhibitor on the steel surface (quasi substitution) [42, 43].
3.6 SEM Studies
SEM micrographs of the mild steel surface in 1 M HCl in the absence and presence of Seroquel inhibitor are shown in Fig. 10a, b. Surface morphology of the corroded steel surface in 1 M HCl solution in the absence of inhibitor for 4 h immersion time is shown in Fig. 10a, b, which shows the corroded steel surface in 1 M HCl solution in the presence of 1000 ppm of Seroquel inhibitor for the same immersion time. The SEM image of corroded metal sample clearly shows that the surface is rough and damaged in the absence of inhibitor compared to the surface in the presence of 1000 ppm of inhibitor due to decrease in the corrosion rate. This indicates that the surface damage was decreased considerably compared to the blank solution, indicating the formation of protective layer by the inhibitor on the mild steel surface.
4 Conclusions
Seroquel drug was found to be a good corrosion inhibitor for low carbon steel in 1 M HCl solution in the temperature range of 303–333 K. From the experimental evidence, it can be concluded that, Seroquel acts as better inhibitor even at high temperature and is confirmed by the values of surface coverage (Θ), icorr, and Rp values. Inhibition efficiency was found to increase with increase in inhibitor concentration in the range of 200–1000 ppm. Maximum inhibition efficiency was found to be around 83% at 333 K. Polarization studies revealed that the inhibitor behaves as mixed type. EIS parameters and activation parameters showed that the inhibition action is due to adsorption process and the corrosion process is due to endothermic effect. FTIR spectrum reveals the adsorption of Seroquel molecule on the metal surface through heteroatoms and SEM analysis showed that the corrosion rate is retarded due to the formation of the protective layer on the steel surface.
References
Thirumalairaj B, Jaganathan M (2016) Corrosion protection of mild steel by a new binary inhibitor system in hydrochloric acid solution. Egypt J Pet 25:423–432
Kolo AM, Sani UM, Kutama IU, Usman U (2016) Adsorption and inhibitive properties of Januvia for the corrosion of zinc in 0.1 M HCl. Pharm Chem J 3:109–119
Aziz RJ (2016) Study of some drugs as corrosion inhibitors for mild steel in 1 M H2SO4 solution. Int J Curr Pharm Sci 3:1–7
Fouda AAS, Rashwan SM, Abd El-Aal NF, Ramadan NH (2016) Unused augmentin drug as save corrosion inhibitor for α-brass in nitric acid solution. Zastita Mater 57:326–338
Andreani S, Znini M, Paolini J, Majidi L, Hammouti B, Costa J, Muselli A (2016) Study of corrosion inhibition for mild steel in hydrochloric acid solution by Limbarda crithmoides (L.) essential oil of Corsica. J Mater Environ Sci 7:187–195
Pavithra MK, Venkatesha TV, Punith Kumar MK, Anantha NS. Electrochemical, gravimetric and quantum chemical analysis of mild steel corrosion inhibition by colchicines in 1 M HCl medium. Res Chem Intermed. https://doi.org/10.1007/s11164-015-2158-3
Al-Shafey HI, Abdel Hameed RS, Ali FA, Aboul-Magd AE-AS, Salah M (2014) Effect of expired drugs as corrosion inhibitors for carbon steel in 1 M HCl solution. Int J Pharm Sci Rev Res 27:146–152
Wang J, Xu S-A (2016) The inhibition effect of a novel Mannich base on the corrosion of A3 mild steel in 1.0 M hydrochloric acid solution. Int J Electrochem Sci 1:2621–2637
Narayana Hebbar, Praveen BM, Prasanna BM, Sachin HP (2018) Anticorrosion potential of flectofenine on mild steel in hydrochloric acid media: experimental and theoretical study. J Fail Anal Prev 18:371–381
Shylesha BS, Venkatesha TV, Praveen BM (2011) Ziprasidone as a corrosion inhibitor for zinc in different acid medium. J Chem Pharm Res 3:501–507
Nataraja SE, Venkatesha TV, Tandon HC, Shylesha BS (2011) Quantum chemical and experimental characterization of the effect of ziprasidone on the corrosion inhibition of steel in acid media. Corros Sci 53:4109–4117
Lgaz H, Salghi R, Jodeh S, Hammouti B (2016) Effect of clozapine on inhibition of mild steel corrosion in 1.0 M HCl medium. J Mol Liq. https://doi.org/10.1016/j.molliq.2016.11.039
Ameh PO, Sani UM (2016) Cefuroxime axetil: a commercially available drug as corrosion inhibitor for aluminium in hydrochloric acid solution. Electrochim Acta 34:131–141
Fouda AS, Rashwan SM, Abd El-Aal NF, Ramadan NH (2016) Corrosion protection and adsorption properties of amoxicillin as save corrosion inhibitor for α- brass in nitric acid solution. J Chem Pharm Res 8:705–715
Fouda AS, El-Ewady G, Ali AH. Modazar as promising corrosion inhibitor of carbon steel in hydrochloric acid solution. Green Chem Lett Rev. https://doi.org/10.1080/17518253.2017.1299228
Prasanna BM, Praveen BM, Narayana Hebbar, Venkatesha TV, Tandon HC, Abd Hamid SB (2017) Electrochemical study on inhibition effect of Aspirin on mild steel in 1 M hydrochloric acid. J Assoc Arab Univ Basic Appl Sci 22:62–69
Prasanna BM, Praveen BM, Narayana Hebbar, Venkatesha TV (2016) Experimental and theoretical studies of hydralazine hydrochloride as corrosion inhibitor for mild steel. Anticorros Mater Methods 63:47–55
Praveen BM, Prasanna BM, Narayana Hebbar, Shivakeshava Kumar P, Jagadeesh MR (2018) Experimental and theoretical studies on inhibition effect of the Praziquantel on mild steel corrosion in 1 M HCl. J Bio Tribo-Corros 4:21
Narayana Hebbar, Prasanna BM, Venkatesha TV (2015) Corrosion inhibition behavior of ketosulfone for zinc in acidic medium. J Fundam Appl Sci 7:271–289
Naqvi I, Saleemi AR, Naveed S (2011) Cefixime: a drug as efficient corrosion inhibitor for mild steel in acidic media. Electrochemical and thermodynamic studies. Int J Electrochem Sci 6:146–161
Fouda AS, Mahmoud WM, Abdul Mageed HA (2016) Evaluation of an expired non toxic amoldipine besylate drug as a corrosion inhibitor for low carbon steel in hydrochloric acid solutions. J Bio Tribo Corros 7:2–11
Abdel Hameed RS, Ismail EA, Abu Nawwas AH, Al-Shafey HI (2015) Expired Voltaren drugs as corrosion inhibitor for aluminium in hydrochloric acid. Int J Electrochem Sci 10:2098–2109
Abdel Hameed RS (2011) Rantidine drugs as non toxic corrosion inhibitors for mild steel in hydrochloric acid medium. Port Electrochim Acta 29:273–285
Al-Amiery AA, Binti Kassim FA, Kadhum AA, Mohamad AB. Synthesis and characterization of a novel eco-friendly corrosion inhibition for mild steel in 1 M hydrochloric acid. Sci Rep. https://doi.org/10.1038/srep19890
Fouda AS, El Morsi MA, El Mogy T (2017) Studies on the inhibition of carbon steel corrosion in hydrochloric acid solution by expired Carvedilol drug. Green Chem Lett Rev 10:336–345
Zhao Q, Tang T, Tang P, Zhang Z, Wang F (2017) The corrosion inhibition effect of triazinedithiol inhibitors for aluminium alloy in a 1 M HCl. Solut Met 7:44
Prasanna BM, Praveen BM, Narayana Hebbar, Venkatesha TV (2015) Corrosion inhibitory action of mild steel in 1 M HCl by Chlorophenicol. Moroc J Chem 3:824–837
Karthi G, Sundaravadivelu M (2016) Studies on the inhibition of mild steel corrosion in hydrochloric acid solution by atenolol drug. Egypt J Pet 25:183–191
Obi-Egbedi NO, Obot IB (2011) Inhibitive properties, thermodynamic and quantum chemical studies of alloxazine on mild steel corrosion in H2SO4. Corros Sci 53:263–275
Obi-Egbedi NO, Essien KE, Obot IB (2011) Computational simulation and corrosion inhibitive potential of alloxazine for mild steel in 1 M HCl. J Comput Methods Mol Des 1:26–43
Singh AK, Quraishi MA (2010) The effect of some bis-thiadiazole derivatives on the corrosion of mild steel in hydrochloric acid. Corros Sci 52:1373–1385
Safak S, Duran B, Yurt A, Turkoglu G (2012) Schiff bases as corrosion inhibitor for aluminium in HCl solution. Corros Sci 54:251–259
Rodriguez-Torres A, Olivares-Xometl O, Valladares-Cisneros MG, Gonzalez-Rodriguez JG (2018) Effect of green corrosion inhibition by Prunus persica on AISI 1018 carbon steel in 0.5 M H2SO4. Int J Electrochem Sci 13:3023–3049
Wang K, Lai C, Tan B, Xie B, Zhu S, Zhu H, Liu K, Wei J (2018) Corrosion inhibition of mild steel by S-benzyl-O,O′-dialkyldithiophosphates in HCl solution. Int J Electrochem Sci 13:2627–2640
Abd El-Lateef HM (2015) Experimental and computational investigation on the corrosion inhibition characteristics of mild steel by some novel synthesized imines in hydrochloric acid solutions. Corros Sci 92:104–117
Manimegalai S, Manjula P (2015) Thermodynamic and adsorption studies for corrosion inhibition of mild steel in aqueous media by Sargasam swartzii (brown algae). J Mater Environ Sci 6:1629–1637
Narayana Hebbar, Praveen BM, Prasanna BM, Venkatesha TV (2015) The corrosion inhibition effect of Hydralazine·HCl on the zinc in acidic media. Moroc J Chem 3:496–506
Prasanna BM, Praveen BM, Narayana Hebbar, Venkatesha TV, Tandon HC (2014) Ketosulfone drug as a green corrosion inhibitor for mild steel in acidic medium. Ind Eng Chem Res 53:8436–8444
Idouhli R, N’Ait Ousidi A, Koumya Y, Abouelfida A, Benyaich A, Auhmani A, Itto MYA. Electrochemical studies of monoterpenic thiosemicarbazones as corrosion inhibitor for steel in 1 M HCl. Hindwai Int J Corros. https://doi.org/10.1155/2018/9212705
Aiad I, Shaban SM, Elged AH, Aljoboury OH. Cationic surfactant based on alignate as green corrosion inhibitors for the mild steel in 1.0 M HCl. https://doi.org/10.1016/j.ejpe.2018.01.003
Zhou L, Lv Y-L, Hu Y-X, Zhao J-H, Xia X, Li X (2018) Experimental and theoretical investigations of 1,3,5-tris(4-aminophenoxy) benzene as an effective corrosion inhibitor for mild steel in 1 M HCl. J Mol Liq 249:179–187
El Hafi M, Ezzanad A, Boulhaoua M, El Ouasif L, Saadouni M, El Aoufir Y, Ramli Y, Zarrouk A, Oudda H, Essassi EM (2018) Corrosion inhibition effect of novel pyrazolo [3, 4-d] pyramidine derivative on mild steel in 1 M HCl medium. Experimental and theoretical approach. J Mater Environ Sci 9:1234–1246
Prasanna BM, Praveen BM, Narayana Hebbar, Venkatesha TV, Tandon HC (2016) Inhibition study of mild steel corrosion in 1 M hydrochloric acid solution by 2-chloro 3-formyl quinolone. Int J Ind Chem 7:9–19
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Swetha, G.A., Sachin, H.P., Guruprasad, A.M. et al. Use of Seroquel as an Effective Corrosion Inhibitor for Low Carbon Steel in 1 M HCl. J Bio Tribo Corros 4, 57 (2018). https://doi.org/10.1007/s40735-018-0173-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40735-018-0173-9