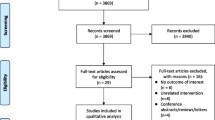
Abstract
Purpose of Review
The aim of this review is to synthesize the current evidence on obstructive sleep apnoea (OSA) in elderly subjects and to describe recent findings on the studies that have investigated the effects of CPAP treatment in elderly subjects.
Recent Findings
OSA is highly prevalent at advanced age, and its clinical presentation and impact in aged patients seem to differ from those in middle-aged subjects. Nevertheless, there are very few studies on the management of OSA in this age group, and indications of continuous positive airway pressure (CPAP) treatment are generally based on results extrapolated from studies performed on young adults. In recent years, some observational studies and clinical trials focused on elderly individuals have been published and shed light on the role of CPAP treatment in elderly individuals.
Summary
OSA is one of the most common respiratory disorders and is an important public health problem whose prevalence is expected to further increase in the coming years. Approximately one-third of the population over 65 years of age has moderate to severe OSA. Scientific evidence about OSA in the elderly and the effects of CPAP treatment are limited, there are still many questions that need to be answered.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The population pyramid is changing; it is expected that in the future decades, the percentage of older people will be higher and without severe disability [1]. Obstructive sleep apnoea (OSA) is one of the most common respiratory disorders, and it is well known that its prevalence increases with increasing age. Therefore, more than 24% of sleep studies are currently performed in elderly subjects. However, there is little knowledge on the impact of OSA in elderly individuals and the effects of treatment in this age group.
OSA prevalence has been found to be higher in the elderly than in the younger age groups, and existing studies suggest that clinical presentation and consequences could vary with age, being less symptomatic and with lower OSA impact at advanced age. However, there are few data available on this topic, and the results are not always conclusive. Moreover, there are other aspects that should be established in this age group concerning diagnosis and treatment. For example, OSA severity is classified according to the apnoea-hypopnea index (AHI) [2], and there is a debate on whether this parameter could be adequate for elderly individuals and whether the current cut-off values applied to younger patients correctly differentiate between physiological and pathological events in the older group. Moreover, OSA screening questionnaires remain to be validated in elderly patients. Regarding treatment, some studies indicate that it is well tolerated, and that adherence and compliance do not differ significantly from those of younger patients; nevertheless, the results are not always concordant. Moreover, despite the high number of elderly patients with OSA, there are few studies centred on this age group; recommendations have been extrapolated from studies performed in middle-aged adults.
In summary, there is no doubt that OSA is a prevalent disorder in the elderly, but there is scarce scientific evidence. Therefore, this article aimed to analyse the characteristics and impact of OSA in elderly individuals and to review the evidence on the effects of CPAP treatment in this age group.
Changes in Sleep and the Upper Airway with Ageing
Ageing decreases the minor negative pressures needed to produce upper airway collapse, increasing collapsibility [3] and therefore the probability of recurrent apnoeas and hypopneas. The aetiology of obstructive sleep apnoea (OSA) in elderly individuals is frequently multifactorial. On the one hand, there are anatomical factors (fat deposition in the upper airway or loss of teeth), neuromuscular factors (pharyngeal muscle dysfunction) and neuroventilatory factors (changes in sleep architecture) [3]. On the other hand, the aetiology may also be related to comorbidities such as stroke, dementia, oedema in the lower extremities or heart failure.
There are some physiological changes associated with ageing that can facilitate upper airway obstruction, such as an increase in the percentage of superficial sleep and a decrease in deep sleep [4] or an increase in the number of arousals. Some data suggest that an increase in OSA incidence in the elderly could be related to not only the physiological changes associated with ageing but also the increase in central apnoeas associated with greater respiratory instability during sleep. In addition, recent evidence has emerged that dysfunction of the genioglossal nerve and muscle in elderly subjects may have implications for OSA pathogenesis [5]. Moreover, it is important to consider the relevance of polypharmacy in sleep breathing disorders at advanced age and the hormonal changes and obesity (especially around the upper airway) [6] associated with menopause that could also facilitate airway obstruction [7, 8].
However, it is important to note that any of the factors described has been clearly identified as a main cause, and collapse probably depends on the interaction of several factors.
Prevalence of OSA in Older Adults
According to the American Academy of Sleep Medicine (AASM) criteria, in populations over 30 years old, the prevalence rates of mild and moderate-severe OSA are approximately 35.2% and 16.2%, respectively [9]. Higher values have been reported in certain population groups, such as those placed in nursing homes, home dwellings or hospitals [10, 11].
The prevalence of OSA in the elderly population is higher than that in the middle-aged population, and it could be related to the increased airway collapsibility observed in advanced age [12]. It has been described that the number of respiratory events increases with age [13], and OSA syndrome has been found in 18% of men and 7% of women aged 50–70 years. Moreover, a recent study reported that approximately 32% of subjects aged ≥ 60 years have moderate to severe OSA [14].
Male sex, obesity and old age are independent risk factors for OSA [7]. OSA in middle-aged individuals is more prevalent in males, but as the prevalence of OSA in females increases after menopause, the prevalence in both sexes is similar at advanced age.
Clinical Picture of OSA in the Elderly
The clinical presentation of OSA in older adults is different from that in younger adults. Compared to middle-aged adults, snoring and witnessed apnoeas are less frequent because the bed partner can be an older person or may sometimes be absent [3]. Moreover, sleepiness could be perceived as a normal symptom at advanced age, and frequently, there are other causes of somnolence that make it difficult to relate somnolence to OSA syndrome. Thus, in general, elderly individuals are less symptomatic, and anthropometric data have a more limited predictive value at advanced age.
It is important to note that OSA in the geriatric population could be associated with less typical symptoms, such as nocturia, neurocognitive symptoms, functional deterioration with frequent falls [15], sarcopenia [3] or the presence of an excess of cardiovascular events [16, 17].
For more detailed information regarding differences in OSA between age groups, see Table 1.
Screening and Diagnosis
The gold standard for diagnosing OSA is overnight polysomnography in a clinical sleep laboratory. Simpler diagnostic tools that can be performed at home, with easier management and lower costs, have been developed and validated [18] in middle-aged individuals. However, their use is more limited in elderly subjects because of the presence of comorbidities, ingestion of psychotropic medication or other sleep disorders such as insomnia or changes in sleep efficiency and architecture [4].
Moreover, screening seniors via questionnaires to detect OSA is problematic because they have not been validated in this age group. Recent studies have evaluated the diagnostic performance of the STOP-BANG and new modifications of this questionnaire called STOP-28 and STOP in older individuals [19]. The authors concluded that they have high sensitivity but low specificity. Moreover, some questionnaires, such as the Epworth Sleepiness Scale (ESS), have not been validated in elderly subjects [20]; therefore, they should be used with caution.
A controversial aspect in the diagnosis, given the high number of respiratory disorders observed in the elderly, is establishing the cut-off point for the apnoea-hypopnea index (AHI) in this age group. This threshold should distinguish between age-related respiratory events (probably physiological) and those that will have harmful consequences for the patient. In this sense, Bliwise et al. [21] suggested that a heuristic model of OSA could exist. On the one hand, there could be a type of OSA that appears at middle age and should be considered pathological. On the other hand, age-dependent OSA is more related to the ageing process, which could be less harmful. However, despite the debate in recent years, no specific diagnostic criteria are available for this population group.
Impact of OSA in the Elderly
Mortality
There are few studies evaluating the mortality of OSA specifically in elderly subjects, and the results are contradictory. Thus, Martinez-Garcia et al. [16] performed a prospective observational study in severe OSA in subjects older than 65 years and observed an increase in cardiovascular mortality. In contrast, Johansson P et al [22] and Mant A et al. [23] did not find an increase in mortality in elderly individuals with OSA.
If we look at studies that do not exclude subjects aged ≥ 65 years, it seems that in elderly subjects, OSA does not have the same impact on mortality as in young subjects. Lavie et al. [24] showed that moderate to severe OSA was associated with an excess mortality rate only in subjects aged < 50 years but not in elderly individuals. Moreover, Punjabi et al. [25] concluded that OSA is associated with mortality, particularly in men aged 40–70 years. In addition, Lavie et al [26] reported lower mortality than expected in the general population of the same age in elderly subjects with moderate OSA. The authors attributed this result to the presence of adaptive mechanisms and described the “hypoxic preconditioning hypothesis”. Nevertheless, it is important to note that differences in mortality should be related to methodological limitations of the studies or related to a survivor bias in patients with OSA and advanced age [27].
Cardiovascular Morbidity
The relationship between OSA and cardiovascular disease (CVD) in middle-aged subjects is well known; however, there are scarce data in older populations. Obstructive sleep apnoea has been associated with endothelial dysfunction [28], ventricular extrasystole [29] and hypertension [30, 31] in some studies, while no association with cardiovascular disease [22, 32] was found by other authors.
The major determinant of cardiovascular morbidity in relation to obstructive sleep apnoea is chronic intermittent hypoxia (CIH), and prior studies have shown that CIH induces changes in the structure of cardiovascular tissues, resulting in cardiovascular diseases [33, 34]. Some studies in animal models have evaluated whether there is a differential effect of OSA on cardiovascular tissues depending on age. A recent study by Castro-Grattoni AL et al. concluded that cardiovascular remodelling induced by severe chronic intermittent hypoxia is affected by age, with more pronounced deleterious cardiovascular effects in younger populations [31].
It is important to note that some of the patients included in studies evaluating OSA, hypertension and cardiovascular diseases are older than 60 years, and the associations observed appear to be greater in younger patients [33, 34]. Thus, the available evidence suggests that cardiovascular consequences could be less common in elderly individuals; nevertheless, evidence is scarce and not conclusive.
Cerebrovascular Morbidity
Clinical studies suggest a bidirectional relationship between cerebrovascular disease and OSA; therefore, suffering a stroke would make the presence of respiratory disorders more frequent during sleep, and having OSA could influence the risk and evolution of cerebrovascular disease [35]. OSA has been associated with a higher prevalence of lacunar or silent infarcts [36] and has been identified in elderly patients as an independent risk factor for incident stroke [37].
Neurocognitive Morbidity
OSA has been associated with negative effects at the cognitive level in experimental and clinical studies. The most frequent cognitive deficits are found in the attention and executive functions domains, while the effects observed in memory have been minor. Domains such as language or global intellectual function are relatively preserved.
The prevalence of OSA in Alzheimer’s patients is more than 45% [38], which is higher than that in populations of the same age with preserved cognitive function. Some authors have reported that mild cognitive impairment and Alzheimer’s disease are associated with breathing disorders [38, 39]. There is a plausible biological link between OSA and cognitive impairment [40,41,42], and it has been described that OSA could provoke disruptions in the amyloid process and the clearance of tau protein. It has been reported that increasing the apnoea-hypopnea index increases the risk of cognitive decline [43], and recent studies have shown a link between neurodegenerative biomarkers (Aβ40, Aβ42, total Aβ and P-tau 181) and OSA [44,45,46].
The results are contradictory regarding the effects of OSA on neurocognitive parameters and disorders. Longitudinal studies have suggested that OSA could be a risk factor for the development of cognitive impairment and dementia [47] due to its onset at an earlier age [48]. However, not all studies have reached such conclusions. Thus, a recent study showed that in patients with mild-moderate Alzheimer’s disease, OSA was not associated with greater cognitive decline after 12 months of follow-up [38].
Cancer
Multiple authors have described a higher incidence of cancer in patients with OSA [49, 50], although others have not found this association. It has been reported that OSA increases malignant tumour properties in breast, kidney and uterine cancers and melanoma [51]; nevertheless, the incidence rates of lung and colorectal cancer attributed to OSA are uncertain [49].
The mechanisms behind an increased risk of cancer and the prevalence of OSA are not completely understood, but intermittent hypoxia and sleep fragmentation have been proposed as key factors [52]. Studies in animal models have shown that intermittent hypoxia and sleep fragmentation can favour tumour growth and invasiveness. Nevertheless, it has been described that the effects of intermittent hypoxia could be modulated by age [53], with lower growth rates at advanced age. Some authors have reported that intermittent hypoxia can alter the immune response in elderly individuals and plays a pivotal role in lung adenocarcinoma progression and aggressiveness [51].
Several observational studies reported an association between OSA and cancer mortality [54], and data from experimental studies showed that there is biological plausibility to explain the link between both disorders and suggest that the effect seems lower at advanced age. However, the studies carried out have important limitations and are not conclusive.
CPAP Treatment in Elderly Patients with OSA
Even though there are a large number of elderly individuals under CPAP treatment, there is little evidence on the effectiveness of this treatment in elderly subjects. Treatment indications are mostly based on the results of studies performed in young adults or in studies including both young and old age groups, with very few studies centred on elderly subjects. Although scientific evidence on the effects of CPAP treatment focused exclusively on elderly individuals is scarce, some observational studies and clinical trials focused on elderly individuals have been published in recent years.
Observational Studies
There are few long-term observational studies performed exclusively in the elderly population, and they are frequently centred on disorders more prevalent at advanced age, such as cognitive disorders or cerebrovascular disease.
Cognitive Disorders
Most of the studies have been performed in subjects with cognitive deterioration or dementia, with results suggesting that CPAP treatment can improve cognitive function and even delay cognitive decline in elderly patients [48, 55, 56].
Studies performed in subjects with preserved cognitive function are much more limited. Aloia MS et al. [57] observed that CPAP treatment produces an improvement in neurocognitive variables after 3 months of CPAP treatment, and Crawford-Achour E. et al. [58] demonstrated that CPAP treatment is associated with the maintenance of memory performance.
Cardiovascular and Cerebrovascular Disease
Martinez-Garcia MA et al. [16] observed a decrease in cardiovascular mortality in patients with good CPAP compliance. Observational studies by López-Padilla et al. [59] (performed in subjects aged ≥80 years) and Ou et al. [60] (including patients with a mean age of 77.8 years) also presented better survival in those with good compliance with CPAP.
Moreover, studies carried out in subjects with stroke reported significant protection against the recurrence of new vascular events [61] with CPAP treatment.
Randomized Controlled Trials
Before 2014, data from only a few trials undertaken exclusively in subjects aged over 60-65 years were available, and those trials were mostly centred on evaluating the effect of CPAP treatment in cerebrovascular diseases.
Hsu CY et al [62] performed a study in 30 patients aged 65–81 years with stroke and severe OSA who were randomized to CPAP or conservative care for 8 weeks. The authors unveiled no effect of CPAP treatment on neurological performance, quality of life or sleepiness. Zhang X et al [61] studied 41 patients aged 60–74 years with moderate-severe OSA and did not find deterioration in analytic variables with respect to healthy controls. Parra O et al [63] conducted a study in 140 patients (aged 64.7 years) with ischaemic stroke and an AHI>20/h who were randomized to receive CPAP or conventional treatment. The authors concluded that nasal CPAP seems to accelerate neurological recovery and delay the appearance of cardiovascular events, although they did not find improvements in patient survival or quality of life.
Since 2014, four randomized clinical trials focusing exclusively on elderly subjects have been published. For more detailed information, see Table 2.
The first published study was the PREDICT trial by McMillan A et al. [64]. The PREDICT was a 12-month, multicentre randomized trial performed in the UK that included 278 patients older than 65 years with newly diagnosed OSA who were randomized to receive CPAP plus supportive care or supportive care alone. The authors concluded that CPAP reduces sleepiness over 12 months compared with supportive care alone. No differences were observed in mood, cognitive function, cardiovascular events or health care costs. It is important to note that subjects included in the PREDICT trial have low adherence, with a mean CPAP compliance lower than 2 h/night.
Dalmases M et al [65] performed a single-centre randomized pilot study in 33 patients aged ≥ 65 years old with severe OSA treated with either conservative care or CPAP for three months. They observed that patients treated with CPAP presented significant improvements in some cognitive domains (episodic and short-term memory, speed of mental processing and mental flexibility). Moreover, neuroimaging revealed an increase in connectivity in the right middle frontal gyrus after 3 months of CPAP treatment and a higher percentage of cortical thinning in the conservative care group. No association was seen between cognition and brain functional connectivity changes within the default mode network. The mean CPAP compliance was 6 h/night.
Martínez-García MA et al. [66] performed an open-label, multicentre clinical trial in a consecutive clinical cohort of 224 elderly patients (aged ≥70 years) with severe OSA who were randomized to receive CPAP or no CPAP for 3 months. The authors concluded that CPAP treatment was effective in improving quality of life (evaluated with the Quebec Sleep Questionnaire), sleep-related symptoms, anxiety, depression and some neurocognitive test scores (digit symbol and Trail making test A). The mean CPAP compliance was 4.9 h/night, and approximately 70% of the patients presented good adherence.
Recently, Ponce S et al. [67] performed an open-label, multicentre study in 145 elderly patients with moderate OSA (IAH>15/h) who were randomized to CPAP or no CPAP for 3 months. The authors concluded that in subjects with moderate OSA, CPAP improves diurnal hypersomnia and some sleep-related symptoms and quality of life domains without changes in neurocognitive tests and blood pressure [67].
Compliance with CPAP
Another important aspect of CPAP in elderly subjects is compliance with the treatment because it seems logical that elderly subjects could present some characteristics that could make CPAP adherence difficult, such as milder symptoms, the presence of cognitive disturbances, living alone, comorbidities or neurological deficits.
It is believed that adherence to CPAP is reduced by age (particularly over 75 years) [68], considering that the first week of adherence and socioeconomic variables are strong predictors of CPAP compliance [69].
Therefore, the indications for CPAP should be individualized in very elderly individuals and, when prescribed, closely monitored to address tolerance and compliance. Identifying predictive factors could be relevant to developing strategies to minimize their negative influence on adherence to CPAP treatment [69].
Current Situation and Future Perspectives
There are many elderly patients with OSA who are under CPAP treatment, and these numbers are expected to increase in future years; nevertheless, our knowledge on the impact of OSA and the benefits of treatment in this age group is very limited. Therefore, the study of OSA in the elderly is a challenge that needs to be addressed in the coming years. It is necessary to perform studies that clarify the impact of OSA in the elderly and the effectiveness of CPAP in this population group to establish specific recommendations for the elderly. Moreover, it could be important to develop new follow-up strategies and educational programs to improve adherence and to facilitate a closer follow-up in this age group, especially in those ages in which lower compliance has been observed. Future studies should also address the validation of specific questionnaires, the use of simplified diagnostic methods for elderly individuals and the evaluation of other therapeutic options for the golden age.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Christensen K, Doblhammer G, Rau R, Vaupel JW. Ageing populations: the challenges ahead. Lancet Respir Med. 2009;374:1196–208. https://doi.org/10.1016/S0140-6736(09)61460-4.
Black JK, Whittaker AC, Balanos GM. Undiagnosed obstructive sleep apnea and physical activity in older manual workers. J Aging Phys Act. 2019;27:293–9.
Morley JE, Sanford A, Bourey R. Sleep apnea: a geriatric syndrome. J Am Med Dir Assoc. 2017;18:899–904. https://doi.org/10.1016/j.jamda.2017.08.020.
Posadas T, Oscullo G, Zaldívar E, Garcia-Ortega A, Gómez-Olivas JD, Monteagudo M, et al. Treatment with CPAP in elderly patients with obstructive sleep apnoea. J Clin Med. 2020;9:546.
Saboisky JP, Stashuk DW, Hamilton-Wright A, Trinder J, Nandedkar S, Malhotra A. Effects of aging on genioglossus motor units in humans. PLoS One. 2014;9:e104572.
Perger E, Mattaliano P, Lombardi C. Menopause and Sleep Apnea. Maturitas. 2019;124:35–8.
Fietze I, Laharnar N, Obst A, Ewert R, Felix SB, Garcia C, et al. Prevalence and association analysis of obstructive sleep apnea with gender and age differences – results of SHIP-trend. J Sleep Res. 2019;28:1–9.
Huang T, Lin BM, Markt SC, Stampfer MJ, Laden F, Hu FB, et al. Sex differences in the associations of obstructive sleep apnoea with epidemiological factors. Eur Respir J. 2019;51:1–17.
Benjafield AV, Eastwood PR, Heinzer R, Morrell MJ, Federal U, Paulo DS, et al. Estimation of the global prevalence and burden of obstructive sleep apnoea: a literature-based analysis. Lancet Respir Med. 2020;7:687–98.
Senaratna CV, Perret JL, Lodge CJ, Lowe AJ, Campbell BE, Matheson MC, et al. Prevalence of obstructive sleep apnea in the general population: a systematic review. Sleep Med Rev. 2017;34:70–81. https://doi.org/10.1016/j.smrv.2016.07.002.
Academy A, Medicine S, Classifi I, Disorders S. Correspondence prevalence of sleep apnoea syndrome in the middle to old age. Lancet Respir Med. 2016;4:3–4.
Eikermann M, Jordan AS, Chamberlin NL, Gautam S, Wellman A, Lo YL, et al. The influence of aging on pharyngeal collapsibility during sleep. Chest. 2007;131:1702–9.
Young T, Shahar E, Nieto FJ, Redline S, Newman AB, Gottlieb DJ, et al. Predictors of sleep-disordered breathing in community-dwelling adults: the Sleep Heart Health Study. Arch Intern Med. 2002;162:893–900. https://doi.org/10.1001/archinte.162.8.893.
Heinzer R, Vat S, Marques-Vidal P, Marti-Soler H, Andries D, Tobback N, et al. Prevalence of sleep-disordered breathing in the general population: the HypnoLaus study. Lancet Respir Med. 2015;3:310–8.
Herrick JE, Bliwise DL, Puri S, Rogers S, Richards KC. Strength training and light physical activity reduces the apnea-hypopnea index in institutionalized older adults. J Am Med Dir Assoc. 2014;15:844–6. https://doi.org/10.1016/j.jamda.2014.08.006.
Martínez-García MA, Campos-Rodríguez F, Catalán-Serra P, Soler-Cataluña JJ, Almeida-Gonzalez C, De La Cruz MI, et al. Cardiovascular mortality in obstructive sleep apnea in the elderly: role of long-term continuous positive airway pressure treatment: a prospective observational study. Am J Respir Crit Care Med. 2012;186:909–16. https://doi.org/10.1164/rccm.201203-0448OC.
Collen J, Lettieri C, Wickwire E, Holley A. Obstructive sleep apnea and cardiovascular disease, a story of confounders! Sleep Breath. 2020;24:1299–313. https://doi.org/10.1007/s11325-019-01945-w.
Oliveira MG, Garbuio S, Treptow EC, Polese JF, Tufik S, Nery LE, et al. The use of portable monitoring for sleep apnea diagnosis in adults. Expert Rev Respir Med. 2014;8:123–32. https://doi.org/10.1586/17476348.2014.850421.
Martins EF, Martinez D, Cortes AL, Nascimento N, Brendler J. Exploring the STOP-BANG questionnaire for obstructive sleep apnea screening in seniors. J Clin Sleep Med. 2020;16:199–206.
Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14:540–5. https://doi.org/10.1093/sleep/14.6.540.
Bliwise DL. Epidemiology of age-dependence in Sleep Disordered Breathing (SDB) in old age: the Bay Area Sleep Cohort (BASC). Sleep Med Clin. 2019;4:57–64.
Johansson P, Alehagen U, Svanborg E, Dahlström U, Broström A. Clinical characteristics and mortality risk in relation to obstructive and central sleep apnoea in community-dwelling elderly individuals: a 7-year follow-up. Age Ageing. 2012;41:468–74.
Mant A, King M, Saunders NA, Pond CD, Goode E, Hewitt H. Four-year follow-up of mortality and sleep-related respiratory disturbance in non-demented seniors. Sleep. 1995;18:433–8. https://doi.org/10.1093/sleep/18.6.433.
Lavie P, Lavie L, Herer P. All-cause mortality in males with sleep apnoea syndrome: declining mortality rates with age. Eur Respir J. 2005;25:514–20. https://doi.org/10.1183/09031936.05.00051504.
Punjabi NM, Caffo BS, Goodwin JL, Gottlieb DJ, Newman AB, O’Connor GT, et al. Sleep-disordered breathing and mortality: a prospective cohort study. PLoS Med. 2009;6:e1000132. https://doi.org/10.1371/journal.pmed.1000132.
Lavie P, Lavie L. Unexpected survival advantage in elderly people with moderate sleep apnoea. J Sleep Res. 2009;18:397–403. https://doi.org/10.1111/j.1365-2869.2009.00754.x.
Mashaqi S, Gozal D. The impact of obstructive sleep apnea and PAP therapy on all-cause and cardiovascular mortality based on age and gender – a literature review. Respir Investig. 2020;58:7–20. https://doi.org/10.1016/j.resinv.2019.08.002.
Nieto FJ, Herrington DM, Redline S, Benjamin EJ, Robbins JA. Sleep apnea and markers of vascular endothelial function in a large community sample of older adults. Am J Respir Crit Care Med. 2004;169:354–60.
Mehra R, Stone KL, Varosy PD, Hoffman AR, Marcus GM, Blackwell T, et al. Nocturnal Arrhythmias across a spectrum of obstructive and central sleep-disordered breathing in older men: outcomes of sleep disorders in older men (MrOS sleep) study. Arch Intern Med. 2009;169:1147–55.
Endeshaw YW, White WB, Kutner M, Ouslander JG, Bliwise DL. Sleep-disordered breathing and 24-hour blood pressure pattern among older adults. J Gerontol A Biol Sci Med Sci. 2009;64:280–5.
Castro-Grattoni AL, Suarez-Giron M, Benitez I, Torres M, Almendros I, Farre R, et al. Effect of age on the cardiovascular remodelling induced by chronic intermittent hypoxia as a murine model of sleep apnoea. Respirology. 2020;25:312–20.
Haas DC, Foster GL, Nieto FJ, Redline S, Resnick HE, Robbins JA, et al. Age-dependent associations between sleep-disordered breathing and hypertension: importance of discriminating between systolic/diastolic hypertension and isolated systolic hypertension in the Sleep Heart Health Study. Circulation. 2005;111:614–21.
Hua-Huy T, Rouhani S, Nguyen X-Y, Luchon L, Meurice J-C, Dinh-Xuan AT. Cardiovascular comorbidities in obstructive sleep apnoea according to age: a sleep clinic population study. Aging Clin Exp Res. 2015;27:611–9.
Launois SH, Pépin J-L, Lévy P. Sleep apnea in the elderly: a specific entity? Sleep Med Rev. 2007;11:87–97.
Yaggi HK, Concato J, Kernan WN, Lichtman JH, Brass LM, Mohsenin V. Obstructive sleep apnea as a risk factor for stroke and death. N Engl J Med. 2005;353:2034–41. https://doi.org/10.1056/nejmoa043104.
Cho ER, Kim H, Seo HS, Suh S, Lee SK, Shin C. Obstructive sleep apnea as a risk factor for silent cerebral infarction. J Sleep Res. 2013;22:452–8. https://doi.org/10.1111/jsr.12034.
Catalan-Serra P, Campos-Rodriguez F, Reyes-Nuñez N, Selma-Ferrer MJ, Navarro-Soriano C, Ballester-Canelles M, et al. Increased incidence of stroke, but not coronary heart disease, in elderly patients with sleep apnea: role of continuous positive airway pressure treatment. Stroke. 2019;50:491–4.
Jorge C, Targa A, Benítez ID, Dakterzada F, Torres G, Minguez O, et al. Obstructive sleep apnoea and cognitive decline in mild-to-moderate Alzheimer’s disease. Eur Respir J. 2020;56:2000523.
Cross N, Lampit A, Pye J, Grunstein RR, Marshall N, Naismith SL. Is obstructive sleep apnoea related to neuropsychological function in healthy older adults? A systematic review and meta-analysis. Neuropsychol Rev. 2017;27:389–402. https://doi.org/10.1007/s11065-017-9344-6.
Blackwell T, Yaffe K, Laffan A, Redline S, Ancoli-Israel S, Ensrud KE, et al. Associations between sleep-disordered breathing, nocturnal hypoxemia, and subsequent cognitive decline in older community-dwelling men: the Osteoporotic Fractures in Men Sleep Study. J Am Geriatr Soc. 2015;63:453–61.
Magali Saint M, Sforza E, Roche F, Barthélémy JC, Thomas-Anterion C. Sleep breathing disorders and cognitive function in the elderly: an 8-year follow-up study. The Proof-Synapse Cohort Sleep. 2015;38:179–87.
Lutsey PL, Bengtson LGS, Punjabi NM, Shahar E, Mosley TH, Gottesman RF, et al. Obstructive sleep apnea and 15-year cognitive decline: the Atherosclerosis Risk in Communities (ARIC) Study. 2016;39:309–16.
Emamian F, Khazaie H, Tahmasian M, Leschziner GD, Morrell MJ, Hsiung G-YR, et al. The association between obstructive sleep apnea and Alzheimer’s disease: a meta-analysis perspective. Front Aging Neurosci. 2016;8. https://doi.org/10.3389/fnagi.2016.00078.
Bubu OM, Andrade AG, Umasabor-Bubu OQ, Hogan MM, Turner AD, de Leon MJ, et al. Obstructive sleep apnea, cognition and Alzheimer’s disease: a systematic review integrating three decades of multidisciplinary research. Sleep Med Rev. 2020;50:101250. https://doi.org/10.1016/j.smrv.2019.101250.
Sharma RA, Varga AW, Bubu OM, Pirraglia E, Kam K, Parekh A, et al. Obstructive sleep apnea severity affects amyloid burden in cognitively normal elderly. A Longitudinal Study. Am J Respir Crit Care Med. 2018;197:933–43.
Pase MP, Beiser AS, Himali JJ, Satizabal CL, Aparicio HJ, Decarli C, et al. Assessment of plasma total tau level as a predictive biomarker for dementia and related endophenotypes. JAMA Neurol. 2019;76:598–606. https://doi.org/10.1001/jamaneurol.2018.4666.
Yaffe K, Laffan AM, Harrison SL, Redline S, Spira AP, Ensrud KE, et al. Sleep-disordered breathing, hypoxia, and risk of mild cognitive impairment and dementia in older women. JAMA - J Am Med Assoc. 2011;306:613–9. https://doi.org/10.1001/jama.2011.1115.
Osorio RS, Gumb T, Pirraglia E, Varga AW, Lu SE, Lim J, et al. Sleep-disordered breathing advances cognitive decline in the elderly. Neurology. 2015;84:1964–71. https://doi.org/10.1212/WNL.0000000000001566.
Sillah A, Watson NF, Schwartz SM, Gozal D, Phipps AI. Sleep apnea and subsequent cancer incidence. Cancer Causes Control. 2018;29:987–94.
Fang HF, Miao NF, Chen CD, Sithole T, Chung MH. Risk of cancer in patients with insomnia, parasomnia, and obstructive sleep apnea: a nationwide nested case-control study. J Cancer. 2015;6:1140–7. https://doi.org/10.7150/jca.12490.
Li L, Ren F, Qi C, Xu L, Fang Y, Liang M, et al. Intermittent hypoxia promotes melanoma lung metastasis via oxidative stress and inflammation responses in a mouse model of obstructive sleep apnea. Respir Res. 2018;19:28. https://doi.org/10.1186/s12931-018-0727-x.
Campillo N, Torres M, Vilaseca A, Nonaka PN, Gozal D, Roca-Ferrer J, et al. Role of cyclooxygenase-2 on intermittent hypoxia-induced lung tumor malignancy in a mouse model of sleep apnea. Sci Rep. 2017;7. https://doi.org/10.1038/srep44693.
Torres M, Campillo N, Nonaka PN, Montserrat JM, Gozal D, Martínez-García MA, et al. Aging reduces intermittent hypoxia-induced lung carcinoma growth in a mouse model of sleep apnea. Am J Respir Crit Care Med. 2018;198:1234–6. https://doi.org/10.1164/rccm.201805-0892LE.
Nieto FJ, Peppard PE, Young T, Finn L, Mae Hla K, Farre R. Sleep-disordered breathing and cancer mortality results from the Wisconsin sleep cohort study. Am J Respir Crit Care Med. 2012;186:190–4. https://doi.org/10.1164/rccm.201201-0130OC.
Cooke JR, Ayalon L, Palmer BW, Loredo JS, Corey-Bloom J, Natarajan L, et al. Sustained use of CPAP slows deterioration of cognition, sleep, and mood in patients with Alzheimer’s disease and obstructive sleep apnea: a preliminary study. J Clin Sleep Med. 2009;5:305–9. https://doi.org/10.5664/jcsm.27538.
Ancoli-Israel S, Palmer BW, Cooke JR, Corey-Bloom J, Fiorentino L, Natarajan L, et al. Cognitive effects of treating obstructive sleep apnea in Alzheimer’s disease: a randomized controlled study. J Am Geriatr Soc. 2008;56:2076–81. https://doi.org/10.1111/j.1532-5415.2008.01934.x.
Aloia MS, Ilniczky N, Di Dio P, Perlis ML, Greenblatt DW, Giles DE. Neuropsychological changes and treatment compliance in older adults with sleep apnea. J Psychosom Res. 2003;54:71–6. https://doi.org/10.1016/S0022-3999(02)00548-2.
Crawford-Achour E, Dauphinot V, Saint MM, Tardy M, Gonthier R, Barthelemy JC, et al. Protective effect of long-term CPAP therapy on cognitive performance in elderly patients with severe OSA: the PROOF study. J Clin Sleep Med. 2015;11:519–24. https://doi.org/10.5664/jcsm.4694.
López-Padilla D, Alonso-Moralejo R, Martínez-García MÁ, De la Torre CS, Díaz de Atauri MJ. Continuous positive airway pressure and survival of very elderly persons with moderate to severe obstructive sleep apnea. Sleep Med. 2016;19:23–9.
Ou Q, Chen YC, Zhuo SQ, Tian XT, He CH, Lu XL, et al. Continuous positive airway pressure treatment reduces mortality in elderly patients with moderate to severe obstructive severe sleep apnea: a cohort study. PLoS One. 2015;10:e0127775.
Zhang X, Yin K, Wang H, Su M, Yang Y. Effect of continuous positive airway pressure treatment on elderly Chinese patients with obstructive sleep apnea in the prethrombotic state. Chin Med J. 2003;116:1426–8.
Hsu CY, Vennelle M, Li HY, Engleman HM, Dennis MS, Douglas NJ. Sleep-disordered breathing after stroke: a randomised controlled trial of continuous positive airway pressure. J Neurol Neurosurg Psychiatry. 2006;77:1143–9. https://doi.org/10.1136/jnnp.2005.086686.
Parra O, Sánchez-Armengol Á, Bonnin M, Arboix A, Campos-Rodríguez F, Pérez-Ronchel J, et al. Early treatment of obstructive apnoea and stroke outcome: a randomised controlled trial. Eur Respir J. 2011;37:1128–36. https://doi.org/10.1183/09031936.00034410.
McMillan A, Bratton DJ, Faria R, Laskawiec-Szkonter M, Griffin S, Davies RJ, et al. Continuous positive airway pressure in older people with obstructive sleep apnoea syndrome (PREDICT): a 12-month, multicentre, randomised trial. Lancet Respir Med. 2014;2:804–12.
Dalmases M, Solé-Padullés C, Torres M, Embid C, Nuñez MD, Martínez-Garcia MÁ, et al. Effect of CPAP on cognition, brain function, and structure among elderly patients with OSA a randomized pilot study. Chest. 2015;148:1214–23. https://doi.org/10.1378/chest.15-0171.
Martínez-García MÁ, Chiner E, Hernández L, Cortes JP, Catalán P, Ponce S, et al. Obstructive sleep apnoea in the elderly: role of continuous positive airway pressure treatment. Eur Respir J. 2015;46:142–51.
Ponce S, Pastor E, Orosa B, Oscullo G, Catalán P, Martinez A, et al. The role of CPAP treatment in elderly patients with moderate obstructive sleep apnoea: a multicentre randomised controlled trial. Eur Respir J. 2019;54:1900518.
Martinez-Garcia MA, Valero-Sánchez I, Reyes-Nuñez N, Oscullo G, Garcia-Ortega A, Gómez-Olivas JD, et al. Continuous positive airway pressure adherence declines with age in elderly obstructive sleep apnoea patients. ERJ Open Res. 2019;5:00178–2018. https://doi.org/10.1183/23120541.00178-2018.
May AM, Gharibeh T, Wang L, Hurley A, Walia H, Strohl KP, et al. CPAP Adherence predictors in a randomized trial of moderate-to-severe OSA enriched with women and minorities. Chest. 2018;154:567–78.
Availability of Data and Material
Not applicable
Code Availability
Not applicable
Funding
This work was supported by ISCIII y fondos FEDER “Una manera de hacer Europa” (PI19/00907).
Author information
Authors and Affiliations
Contributions
MZ, FB and MD participated in the critical revision of the literature and writing of the manuscript and approved the final version.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable
Consent to Participate
Not applicable
Consent for Publication
Not applicable
Conflict of Interest
The authors declare no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Sleep Apnea in the Golden Age
Rights and permissions
About this article
Cite this article
Zuil, M., Barbé, F. & Dalmases, M. OSA and CPAP in Older Patients—When to Treat?. Curr Sleep Medicine Rep 7, 97–104 (2021). https://doi.org/10.1007/s40675-021-00209-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40675-021-00209-y