Abstract
Objective
Obstructive sleep apnoea (OSA) is positively associated with cardiovascular diseases, and continuous positive airway pressure (CPAP) is a common treatment for such patients. This study aimed to explore the impact of CPAP on cardiovascular outcomes and prognosis in patients with OSA.
Methods
A search was conducted in the PubMed, Embase and CENTRAL databases for relevant studies published up to March 2024. Two independent reviewers screened the studies based on inclusion and exclusion criteria, and data were synthesised using RevMan 5.3 software. Heterogeneity was assessed using the Cochran Q test and the I2 statistic.
Results
A total of 10 randomised controlled trials and 3 observational studies, comprising 13,832 patients, were included. Compared with standard treatment, the use of CPAP did not significantly reduce the risk of major adverse cardiovascular events (MACE) (risk ratio [RR]: 0.73, 95% confidence interval [CI]: 0.52–1.03; p = 0.07; I2 = 66%), all-cause mortality (RR: 0.92, 95% CI: 0.72–1.16; p = 0.48; I2 = 0%), cardiovascular mortality (RR: 0.63, 95% CI: 0.33–1.19; p = 0.15; I2 = 70%) or non-cardiovascular mortality (RR: 0.81, 95% CI: 0.57–1.15; p = 0.23; I2 = 0%). Similarly, there were no significant differences in the incidence of myocardial infarction, stroke, hospitalisation due to unstable angina or heart failure or atrial fibrillation among those using CPAP. However, when CPAP adherence was ≥ 4 h, CPAP significantly reduced the risk of MACE and cardiovascular mortality.
Conclusion
Although CPAP’s cardiovascular benefits in patients with OSA are not confirmed, it may be that bias risks, CPAP adherence and characteristics of the study population may attenuate the perceived benefits of CPAP. Further research is needed to optimise CPAP therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive sleep apnoea (OSA) is a common sleep breathing disorder characterised by recurrent partial or complete obstruction of the upper airway during sleep, leading to pauses in breathing or reduced airflow, resulting in oxygen desaturation and decreased sleep quality [1]. Obstructive sleep apnoea is often accompanied by symptoms such as frequent nocturnal awakenings, snoring, breathing pauses and excessive daytime sleepiness, significantly affecting patients’ quality of life [2]. Previous studies have confirmed a close association between OSA and cardiovascular diseases. Patients with OSA often have an increased risk of developing hypertension, coronary artery disease and cardiac arrhythmias. Frequent nocturnal hypoxaemia and sympathetic nervous system activation during sleep may lead to abnormal cardiovascular responses, including elevated blood pressure and impaired endothelial function, thereby promoting the occurrence and progression of cardiovascular diseases [3]. Therefore, gaining a deeper understanding of the pathogenesis of cardiovascular diseases in patients with OSA and exploring effective treatment methods are crucial for improving patient outcomes.
Continuous positive airway pressure (CPAP) therapy is a commonly used and effective treatment for OSA [4]. This treatment method involves providing CPAP during sleep to prevent the upper airway from collapsing during exhalation, thereby maintaining airway patency and reducing the occurrence of breathing pauses. Continuous positive airway pressure is typically administered through a nasal or face mask connected to a ventilator, delivering positive pressure airflow to the patient’s airway to keep it open [5]. Past studies have shown that CPAP therapy can effectively alleviate symptoms in patients with OSA, including reducing nocturnal awakenings, improving sleep quality and decreasing excessive daytime sleepiness [6]. Additionally, some studies have found a slight reduction in blood pressure in some patients receiving CPAP therapy [7]. However, there is considerable controversy regarding the effects of CPAP therapy on cardiovascular diseases [8]. Although several meta-analyses have been conducted to evaluate the impact of CPAP therapy on cardiovascular diseases, inconsistencies in conclusions exist due to differences in study methods and included studies [9]. Furthermore, with the emergence of new research and evolving definitions of cardiovascular diseases and implementation methods of CPAP therapy, assessing its impact on cardiovascular diseases becomes increasingly complex.
Therefore, this updated meta-analysis aims to comprehensively assess the impact of CPAP therapy on the incidence and outcomes of cardiovascular diseases in patients with OSA. By including the latest studies and integrating different research findings, we aim to provide more reliable evidence for clinical practice and further clarify the role of CPAP therapy in improving cardiovascular health in patients.
Methods
Search strategy and literature selection
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines recommended by the Cochrane Collaboration were adopted in the execution of this study [10]. Three major electronic databases, namely PubMed, Embase and the Cochrane Library, were primarily searched between the inception of the database and 5 March 2024, with no language restrictions. A comprehensive search strategy combining ‘Medical Subject Headings’ and ‘Embase Subject Headings’ terms with free text terms was employed, covering topics such as OSA syndrome, CPAP therapy and randomised controlled trials (RCTs). Additionally, manual screening of potentially relevant references from related studies, reviews or meta-analyses was attempted. After the database search, retrieved records were imported into the Endnote reference management software for automatic and manual deduplication. Two independent reviewers initially screened potentially relevant records based on titles and abstracts, and full-text articles were then assessed to confirm study inclusion. Any discrepancies between the two reviewers were resolved by a third experienced researcher (Table 1).
Inclusion and exclusion criteria
The inclusion criteria for this study were established based on five elements: participants, intervention, comparison, outcome and study design. The inclusion criteria were as follows: 1) adult patients diagnosed with OSA; 2) intervention group receiving CPAP therapy; 3) control group receiving sham CPAP or standard treatment; 4) outcome measures including cardiovascular event incidence or prognosis; and 5) RCTs or observational studies. The exclusion criteria were as follows: 1) duplicate studies; 2) studies with intervention duration < 12 weeks or incorrect intervention methods; 3) studies with incomplete data or unreported predefined outcomes; and 4) irrelevant literature types, such as literature reviews and conference abstracts.
Data extraction and risk of bias assessment
After finalising the list of included studies, two independent reviewers extracted characteristics and data from them by reading full-text articles, including study details (e.g. first author, publication date, follow-up period, sample size, blinding method), baseline characteristics of participants, intervention methods, control settings and study outcomes. For RCTs, bias risk assessment was conducted by two independent reviewers according to the Cochrane Handbook for Systematic Reviews of Interventions 5.1 version, which mainly includes assessment of random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data and selective reporting. The Newcastle–Ottawa Scale (NOS) was used to assess the methodological quality of observational studies. The NOS is a commonly used tool for assessing the methodological quality of observational studies, including three major domains: selection, comparability and outcome.
Statistical analysis
This meta-analysis was performed using RevMan 5.3 (Nordic Cochrane Centre, Cochrane Collaboration, Copenhagen, Denmark) software. For dichotomous variables, the risk ratio (RR) and its corresponding 95% confidence interval (CI) were used as the statistical analysis indicators for effect size. The primary predefined study outcomes included cardiovascular prognosis, such as major adverse cardiovascular events (MACE), all-cause mortality, cardiovascular mortality and non-cardiovascular mortality and cardiovascular outcomes, such as myocardial infarction, stroke, hospitalisation due to unstable angina or heart failure and atrial fibrillation. The Cochran Q test combined with the I2 statistic was used to assess the degree of heterogeneity among the included study results. When the statistical heterogeneity among the included study results was low (p > 0.1 or I2 < 50%), the analysis was performed using a fixed-effect model; when there was statistical heterogeneity among the included study results (p < 0.1 or I2 ≥ 50%), a random-effects model for meta-analysis was used. The significance level of the meta-analysis was set at α = 0.05. Publication bias was assessed by plotting a funnel plot. Sensitivity analysis was performed to evaluate the influence of individual studies on the overall effect by observing changes in effect size after excluding individual studies. In addition, subgroup analyses were conducted to examine the effect of CPAP on the occurrence of cardiovascular diseases or prognosis in patients with OSA under different conditions (study design, CPAP adherence and duration of follow-up).
Results
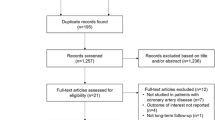
According to the search strategy, a total of 5,004 electronic records were retrieved, with 1,527 from PubMed, 2,854 from Embase, 623 from the Cochrane Library and 1 manually extracted from other sources. After removing 1,135 duplicate records, an initial screening of titles and abstracts resulted in the exclusion of 3,840 irrelevant articles. Following full-text assessment, 16 articles were further excluded for not meeting the inclusion criteria, leaving a final inclusion of 13 articles [11,12,13,14,15,16,17,18,19,20,21,22,23] for meta-analysis. The literature selection process is illustrated in Fig. 1.
Characteristics of included studies
The basic characteristics of the 13 included studies are summarised in Table 2. Among them, the majority of studies (61.5%) were conducted in Europe, comprising 10 RCTs and 3 observational studies, and the majority (69.2%) were multicentre studies. The total sample size of patients receiving CPAP intervention was 6,864, while the control group comprised 6,968 patients. Only RCTs reported daily CPAP usage duration, ranging from a mean or median of 1.4 to 6.6 h/day. Regarding the diagnosis of sleep apnoea, seven studies used cardiorespiratory or respiratory polygraphy, whereas four studies used polysomnography. In the process of including patients with OSA, eight studies used the apnoea–hypopnoea index for discrimination. The median follow-up period ranged from 6 to 90 months.
The characteristics of the included patients are presented in Table 3. The mean age ranged from 51.5 to 71.1 years, with a male proportion ranging from 64 to 97%, body mass index (BMI) ranging from 27 to 33.8 kg/m2, current smoking status ranging from 5 to 58%, type 2 diabetes prevalence ranging from 16.1% to 37.8%, apnoea–hypopnoea index ranging from 23 to 40.1 events/h and the Epworth Sleepiness Scale score ranging from 5.3 to 11.6. Adherence to CPAP was set at 4 h in five studies.
Risk of bias assessment
Detailed information regarding the risk of bias for RCTs is shown in Fig. 2. Due to the open-label design, all studies may have a high risk of performance bias. Additionally, two studies had unclear risks due to factors such as allocation concealment and outcome assessment blinding. None of the RCTs had evidence of incomplete outcome data, selective reporting or other biases. For observational studies, detailed information on the risk of bias is provided in Table S1. Two studies scored 8 points, while one study scored 7 points.
Meta-analysis
In assessing the relationship between CPAP and cardiovascular prognosis, after synthesising the data, it was found that CPAP use did not significantly reduce the risk of MACE, all-cause mortality, cardiovascular mortality or non-cardiovascular mortality compared with conventional treatment. The pooled RR values were 0.73 (95% CI: 0.52–1.03; p = 0.07; I2 = 66%), 0.92 (95% CI: 0.72–1.16; p = 0.48; I2 = 0%), 0.63 (95% CI: 0.33–1.19; p = 0.15; I2 = 70%) and 0.81 (95% CI: 0.57–1.15; p = 0.23; I2 = 0%), respectively (Fig. 3).
Moreover, similar results were obtained for the relationship between CPAP and cardiovascular outcomes. Relative to conventional treatment, there was no significant difference in the likelihood of myocardial infarction, stroke, hospitalisation due to unstable angina or heart failure or atrial fibrillation in the CPAP group. The pooled RR values were 0.94 (95% CI: 0.74–1.20; p = 0.42; I2 = 2%), 0.88 (95% CI: 0.70–1.10; p = 0.60; I2 = 0%), 1.12 (95% CI: 0.90–1.40; p = 0.49; I2 = 0%), 0.92 (95% CI: 0.69–1.24; p = 0.92; I2 = 0%) and 1.01 (95% CI: 0.96–1.07; p = 0.12; I2 = 45%), respectively (Fig. 4).
Subgroup analysis
Subgroup analyses were conducted for study design, CPAP adherence and follow-up duration to evaluate the effect of CPAP on cardiovascular diseases. The results showed that CPAP contributed to a reduction in MACE and cardiovascular mortality in the combined observational studies but not in RCTs (Fig. 3). For other outcomes, no significant differences were observed between CPAP and conventional control groups in both study types (Fig. 4). As shown in Table 4, studies with CPAP adherence ≥ 4 h demonstrated a significant reduction in the risk of MACE and cardiovascular mortality, while non-significant effects were mainly observed in studies with CPAP adherence < 4 h. However, no effect of different follow-up durations on CPAP efficacy was observed, as CPAP did not reduce cardiovascular events in studies with median follow-up durations greater or less than 48 months.
Publication bias and sensitivity analysis
Funnel plot analysis for cardiovascular prognosis and outcomes revealed a symmetric distribution in most studies, except for MACE and hospitalisation due to heart failure, suggesting no significant publication bias (Figures S1 and S2). Additionally, no substantial changes in the summary results were observed after excluding any single study, indicating a limited influence of individual studies on the overall results.
Discussion
In this meta-analysis, we assessed the impact of CPAP therapy on the incidence and outcomes of cardiovascular diseases in patients with OSA. The main findings of this study are as follows: 1) Compared with the control group, patients with OSA receiving CPAP did not show a significant difference in the incidence of MACE, all-cause mortality, cardiovascular mortality or non-cardiovascular mortality events; 2) CPAP use did not reduce the likelihood of myocardial infarction, stroke, hospitalisation due to unstable angina or heart failure or atrial fibrillation in OSA populations; 3) CPAP adherence ≥ 4 h may be a key factor for CPAP to achieve cardiovascular benefits, significantly reducing the risk of MACE and cardiovascular mortality. This study is expected to provide the latest evidence-based medicine evidence for the use of CPAP in preventing future cardiovascular diseases in patients with OSA and provide a reference for subsequent related research.
Existing studies have shown that patients with OSA are more prone to cardiovascular diseases, such as hypertension, diabetes and dyslipidaemia. This connection may be due to the repeated hypoxaemia and sympathetic activation caused by OSA, leading to abnormal reactions in the cardiovascular system, thereby promoting the occurrence and development of cardiovascular diseases [24, 25]. However, previous studies and meta-analyses have not supported the cardiovascular benefits of CPAP therapy for patients with OSA [4, 9, 26, 27]. In our study, CPAP treatment consistently did not significantly reduce the incidence or mortality of cardiovascular diseases in patients with OSA. However, this result needs to be interpreted with caution and further explored. First, we focused on CPAP treatment compliance. Our subgroup analysis found that in studies with compliance > 4 h, the summary results showed that CPAP significantly reduced the risk of MACE and cardiovascular mortality in patients with OSA, while this effect was not observed in studies with compliance < 4 h. A recent meta-analysis of three large RCTs also supported this finding, suggesting that patients with good CPAP treatment compliance have a lower risk of MACE, indicating that CPAP may play a crucial role in preventing OSA-related cardiovascular events [9]. Second, we considered the impact of follow-up duration. Unfortunately, our subgroup analysis did not find a significant effect of follow-up duration on the results, which may be due to insufficient sample size or too short a follow-up duration. More studies are needed to explore this issue further to clarify the impact of follow-up duration on the efficacy of CPAP treatment. Finally, we need to investigate how to improve CPAP treatment compliance. Various strategies can be adopted to address this issue, including improving the comfort and portability of CPAP devices, providing positive feedback, remote monitoring, strengthening patient education and psychological support and developing personalised treatment plans [28, 29]. These measures are expected to increase patient acceptance and compliance with CPAP treatment, thereby more effectively preventing OSA-related cardiovascular events.
This meta-analysis has several notable strengths. First, the number of studies included in our analysis is the most comprehensive among all existing meta-analyses, including not only RCTs but also additional observational studies. This approach makes our analysis more comprehensive, integrating the results of different types of studies, thereby improving the representativeness and credibility of the study. In the subgroup analysis, although the quality of observational studies was lower, the combined data suggested that CPAP could effectively reduce the risk of MACE and cardiovascular mortality in patients with OSA. However, this effect was not observed in RCTs, which may be due to biases in observational studies and small sample sizes. Therefore, we are more cautious in our neutral judgment of the overall results, emphasising the need for further research. Additionally, we specifically included the latest large RCT ISAACC [20], which makes our study more comprehensive compared with other meta-analyses, avoiding the possibility of missing important studies. Meanwhile, considering previous studies suggesting that CPAP may reduce incident atrial fibrillation, we also explored the effect of CPAP on atrial fibrillation outcomes. Although the number of studies reporting this outcome was small, our analysis provided a new perspective. Despite not finding a significant effect of CPAP on the risk of atrial fibrillation in patients with OSA, the importance of this disease outcome cannot be ignored as the population ages, warranting further investigation in the future.
However, this study also has some limitations to note. First, although we included a comprehensive range of studies, we relied on published study results and thus could not access individual patient data (IPD), which prevented us from conducting more detailed analyses. Obtaining IPD would allow us to explore potential factors underlying the lack of significant effects of CPAP treatment, such as baseline characteristics of participants, treatment compliance and airway pressure during treatment, thus finding new ways to improve future treatments. Second, the proportion of women included in the studies was low, which may limit the generalisability of our findings regarding the efficacy and safety of CPAP treatment. Due to the lower proportion of women among patients with OSA, we were unable to fully assess the effects and safety of CPAP treatment in female patients. Therefore, future studies need to pay special attention to female patients and conduct gender-specific analyses. Another limitation of our study is that most included studies set CPAP adherence at 4 h per day, which may not capture the full potential benefits of longer adherence durations. While our results suggest benefits at this threshold, it remains unclear if adherence of ≥ 8 h could yield different outcomes. Future research should explore the impact of longer CPAP usage durations to provide more comprehensive insights. Additionally, this study may suffer from some biases. Lack of blinding of the intervention process may affect the participants and thus affect the reliability of the study results. Therefore, future studies should adopt more rigorous study designs, including blinding of the intervention process, to reduce the impact of bias on study results. Most importantly, our analysis did not observe a potential correlation between BMI and the risk of major adverse cardiovascular events. Recent studies suggest that BMI, more so than the severity of OSA, may be a better marker for evaluating major cardiovascular risks in these patients [30, 31]. This is an important bias that should be considered. Carratù et al. [32] found that cardiovascular risk was more strongly related to obesity than to the apnoea–hypopnoea index in a population of southern Italy. Therefore, future research should investigate the role of BMI in assessing cardiovascular risk in patients with OSA. Another important aspect not examined in this paper is the concomitant presence of OSA and hypertension, which together have an additive role in the progression of carotid atherosclerosis and in increasing blood levels of inflammatory markers for atherosclerosis, such as interleukin-6 and pentraxin-3 [33]. Damiani et al. [34] highlight that the coexistence of OSA and hypertension significantly increases carotid intima-media thickness and levels of these inflammatory markers, suggesting a higher cardiovascular risk. Future research should address the combined impact of OSA and hypertension to better understand their effects on cardiovascular outcomes.
Conclusion
Although there is currently no clear evidence supporting CPAP as a treatment to prevent cardiovascular events in patients with OSA, factors such as study design, methodological quality, CPAP compliance and the characteristics of the study population may affect the evidence of CPAP cardiovascular benefits. Further exploration of factors affecting CPAP efficacy is needed to enhance its evidence for patients with OSA.
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Jordan AS, McSharry DG, Malhotra A (2014) Adult obstructive sleep apnoea. Lancet 383(9918):736–747. https://doi.org/10.1016/S0140-6736(13)60734-5
Batool-Anwar S, Goodwin JL, Kushida CA et al (2016) Impact of continuous positive airway pressure (CPAP) on quality of life in patients with obstructive sleep apnea (OSA). J Sleep Res 25(6):731–738. https://doi.org/10.1111/jsr.12430
Redline S, Azarbarzin A, Peker Y (2023) Obstructive sleep apnoea heterogeneity and cardiovascular disease. Nat Rev Cardiol 20(8):560–573. https://doi.org/10.1038/s41569-023-00846-6
Nokes B, Cooper J, Cao M (2022) Obstructive sleep apnea: personalizing CPAP alternative therapies to individual physiology. Expert Rev Respir Med 16(8):917–929. https://doi.org/10.1080/17476348.2022.2112669
Landry SA, Mann DL, Beare R et al (2023) Oronasal vs Nasal Masks: The Impact of Mask Type on CPAP Requirement, Pharyngeal Critical Closing Pressure (Pcrit), and Upper Airway Cross-Sectional Areas in Patients With OSA. Chest 164(3):747–756. https://doi.org/10.1016/j.chest.2023.03.025
Sánchez AI, Martínez P, Miró E, Bardwell WA, Buela-Casal G (2009) CPAP and behavioral therapies in patients with obstructive sleep apnea: effects on daytime sleepiness, mood, and cognitive function. Sleep Med Rev 13(3):223–233. https://doi.org/10.1016/j.smrv.2008.07.002
Hu X, Fan J, Chen S, Yin Y, Zrenner B (2015) The role of continuous positive airway pressure in blood pressure control for patients with obstructive sleep apnea and hypertension: a meta-analysis of randomized controlled trials. J Clin Hypertens (Greenwich) 17(3):215–222. https://doi.org/10.1111/jch.12472
Bock JM, Vungarala S, Karim S, Somers VK (2021) Obstructive sleep apnea as a cardiovascular risk Factor—Beyond CPAP. Can J Cardiol 37(5):756–765. https://doi.org/10.1016/j.cjca.2021.01.027
Sánchez-de-la-Torre M, Gracia-Lavedan E, Benitez ID et al (2023) Adherence to CPAP treatment and the risk of recurrent cardiovascular events: a meta-analysis. JAMA 330(13):1255–1265. https://doi.org/10.1001/jama.2023.17465
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339:b2700. https://doi.org/10.1136/bmj.b2700
Barbé F, Durán-Cantolla J, Sánchez-de-la-Torre M et al (2012) Effect of continuous positive airway pressure on the incidence of hypertension and cardiovascular events in nonsleepy patients with obstructive sleep apnea: a randomized controlled trial. JAMA 307(20):2161–2168. https://doi.org/10.1001/jama.2012.4366
Bradley TD, Logan AG, Kimoff RJ et al (2005) Continuous positive airway pressure for central sleep apnea and heart failure. N Engl J Med 353(19):2025–2033. https://doi.org/10.1056/NEJMoa051001
Craig SE, Kohler M, Nicoll D et al (2012) Continuous positive airway pressure improves sleepiness but not calculated vascular risk in patients with minimally symptomatic obstructive sleep apnoea: the MOSAIC randomised controlled trial. Thorax 67(12):1090–1096. https://doi.org/10.1136/thoraxjnl-2012-202178
Huang Z, Liu Z, Luo Q et al (2015) Long-term effects of continuous positive airway pressure on blood pressure and prognosis in hypertensive patients with coronary heart disease and obstructive sleep apnea: a randomized controlled trial. Am J Hypertens 28(3):300–306. https://doi.org/10.1093/ajh/hpu147
Kushida CA, Nichols DA, Holmes TH et al (2012) Effects of continuous positive airway pressure on neurocognitive function in obstructive sleep apnea patients: The Apnea Positive Pressure Long-term Efficacy Study (APPLES) [published correction appears in Sleep. 2016 Jul 1;39(7):1483]. Sleep 35(12):1593–1602. https://doi.org/10.5665/sleep.2226
McEvoy RD, Antic NA, Heeley E et al (2016) CPAP for prevention of cardiovascular events in obstructive sleep apnea. N Engl J Med 375(10):919–931. https://doi.org/10.1056/NEJMoa1606599
McMillan A, Bratton DJ, Faria R et al (2014) Continuous positive airway pressure in older people with obstructive sleep apnoea syndrome (PREDICT): a 12-month, multicentre, randomised trial. Lancet Respir Med 2(10):804–812. https://doi.org/10.1016/S2213-2600(14)70172-9
Parra O, Sánchez-Armengol Á, Capote F et al (2015) Efficacy of continuous positive airway pressure treatment on 5-year survival in patients with ischaemic stroke and obstructive sleep apnea: a randomized controlled trial. J Sleep Res 24(1):47–53. https://doi.org/10.1111/jsr.12181
Peker Y, Glantz H, Eulenburg C, Wegscheider K, Herlitz J, Thunström E (2016) Effect of positive airway pressure on cardiovascular outcomes in coronary artery disease patients with nonsleepy obstructive sleep apnea. The RICCADSA randomized controlled trial. Am J Respir Crit Care Med 194(5):613–620. https://doi.org/10.1164/rccm.201601-0088OC
Sánchez-de-la-Torre M, Sánchez-de-la-Torre A, Bertran S et al (2020) Effect of obstructive sleep apnoea and its treatment with continuous positive airway pressure on the prevalence of cardiovascular events in patients with acute coronary syndrome (ISAACC study): a randomised controlled trial. Lancet Respir Med 8(4):359–367. https://doi.org/10.1016/S2213-2600(19)30271-1
Capodanno D, Milazzo G, Cumbo M et al (2014) Positive airway pressure in patients with coronary artery disease and obstructive sleep apnea syndrome. J Cardiovasc Med (Hagerstown) 15(5):402–406. https://doi.org/10.2459/JCM.0000000000000009
Gill J, Wu C (2022) In-hospital outcomes and arrhythmia burden in patients with obstructive sleep apnea and heart failure with preserved ejection fraction. J Innov Card Rhythm Manag 13(6):5033–5040. https://doi.org/10.19102/icrm.2022.130602
Myllylä M, Hammais A, Stepanov M, Anttalainen U, Saaresranta T, Laitinen T (2019) Nonfatal and fatal cardiovascular disease events in CPAP compliant obstructive sleep apnea patients. Sleep Breath 23(4):1209–1217. https://doi.org/10.1007/s11325-019-01808-4
Bouloukaki I, Grote L, McNicholas WT et al (2020) Mild obstructive sleep apnea increases hypertension risk, challenging traditional severity classification. J Clin Sleep Med 16(6):889–898. https://doi.org/10.5664/jcsm.8354
Paschou SA, Bletsa E, Saltiki K et al (2022) Sleep apnea and cardiovascular risk in patients with prediabetes and type 2 diabetes. Nutrients 14(23):4989. https://doi.org/10.3390/nu14234989
Yu J, Zhou Z, McEvoy RD et al (2017) Association of positive airway pressure with cardiovascular events and death in adults with sleep apnea: a systematic review and meta-analysis. JAMA 318(2):156–166. https://doi.org/10.1001/jama.2017.7967
Labarca G, Dreyse J, Drake L, Jorquera J, Barbe F (2020) Efficacy of continuous positive airway pressure (CPAP) in the prevention of cardiovascular events in patients with obstructive sleep apnea: Systematic review and meta-analysis. Sleep Med Rev 52:101312. https://doi.org/10.1016/j.smrv.2020.101312
Chernyak Y (2020) Improving CPAP adherence for obstructive sleep apnea: a practical application primer on CPAP desensitization. MedEdPORTAL 16:10963. https://doi.org/10.15766/mep_2374-8265.10963
Riachy M, Najem S, Iskandar M, Choucair J, Ibrahim I, Juvelikian G (2017) Factors predicting CPAP adherence in obstructive sleep apnea syndrome. Sleep Breath 21(2):295–302. https://doi.org/10.1007/s11325-016-1408-y
Yao L, Heizhati M, Lin M et al (2022) Elevated body mass index increases the risk of cardiovascular events in hypertensive patients accompanied with obstructive sleep apnea: A cohort study. Obes Res Clin Pract 16(6):491–499. https://doi.org/10.1016/j.orcp.2022.10.010
Zdravkovic M, Popadic V, Klasnja S et al (2022) Obstructive sleep apnea and cardiovascular risk: the role of dyslipidemia, inflammation, and obesity. Front Pharmacol 13:898072. https://doi.org/10.3389/fphar.2022.898072
Carratù P, Di Ciaula A, Dragonieri S et al (2021) Relationships between Obstructive Sleep Apnea Syndrome and cardiovascular risk in a naïve population of southern Italy. Int J Clin Pract 75(12):e14952. https://doi.org/10.1111/ijcp.14952
Drager LF, Bortolotto LA, Krieger EM, Lorenzi-Filho G (2009) Additive effects of obstructive sleep apnea and hypertension on early markers of carotid atherosclerosis. Hypertension 53(1):64–69. https://doi.org/10.1161/HYPERTENSIONAHA.108.119420
Damiani MF, Zito A, Carratù P et al (2015) Obstructive sleep apnea, hypertension, and their additive effects on atherosclerosis. Biochem Res Int 2015:984193. https://doi.org/10.1155/2015/984193
Funding
Jinhua Science and Technology Plan Project (Public Welfare Project): 2020–4-020.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of Jinhua Central Hospital.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, G., Zhuge, P., Zhang, Z. et al. The impact of continuous positive airway pressure therapy on cardiovascular events in patients with obstructive sleep apnoea: an updated systematic review and meta-analysis. Sleep Breath (2024). https://doi.org/10.1007/s11325-024-03107-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11325-024-03107-z