Abstract
Summary
With impressive economic development, obesity has emerged as a critical public health issue in China. Recently it was reported that obesity has taken an adverse effect on osteoporosis. Because there is different body mass index (BMI) for obesity globally, studies based on BMI levels on association of obesity with osteoporosis were quite few. Therefore, we discussed the relationship of body composition with skeletal BMD according to WHO BMI and BMI on Working Group on Obesity in China (WGOC).
Methods
A total of 502 adult men aged 20–89 were enrolled as healthy subjects for osteoporosis study at Qianfoshan Hospital, Shandong University between September 2008 and August 2010. According to WHO BMI, all subjects were divided into three groups: normal weight (18.5 ≤ BMI < 25 kg/m2, n = 202), overweight (25 ≤ BMI < 30 kg/m2, n = 242), and obesity (BMI ≥ 30 kg/m2, n = 58). According to WGOC BMI, normal weight (18.5 ≤ BMI < 24 kg/m2, n = 137), overweight (24 ≤ BMI < 28 kg/m2, n = 225), and obesity (BMI ≥ 28 kg/m2, n = 140). Total body and regional BMD, lean mass (LM), lean body mass index (LBMI), fat mass (FM), percent body fat (%BF) and fat mass index (FMI) were measured by dual-energy X-ray absorptiometry. Age-partial Pearson correlation analyses between body composition-related parameters and BMD. Multiple regression analyses were performed to explore the associations of BMD with LM, LBMI, FM, %BF and FMI.
Results
Fat mass (FM), %BF, FMI, LM and LBMI were positively correlated with BMD at almost sites (P < 0.001) in all subjects. However, the relationship was not different among groups. LM, LBMI, FM and FMI were positively correlated with BMD (P < 0.01) in normal weight. LM and LBMI appeared significantly positive with BMD in overweight and obesity according to WHO and WGOC criteria. %BF and FMI were negative significance with BMD at total body and some regional BMD according to WHO criteria in overweight (P < 0.05). In two obese groups, %BF appeared negatively significant with BMD (P < 0.05) according to WGOC criteria, and %BF and FMI appeared negatively significant with BMD (P < 0.05) according to WHO criteria. In regression of independent variables as FM and LM, LM showed statistically positively significant relations with BMD at almost sites (P < 0.05) in all groups. FM appeared positively significant with BMD in normal groups and overweight group according to WGOC criteria. In regression of independent variables as %BF and FMI, %BF and FMI appeared statistically negatively significant relations with BMD in overweight and obesity, but %BF and FMI were inconsistent in same site.
Conclusions
Lean mass (LM) and LBMI could help to determinant of BMD, and %BF and FMI were adverse to BMD in overweight and obesity. Comparing with two criteria, we found the differences in fat-related parameters and BMD according to WHO criteria were more obvious than that according to WGOC criteria. We also found that %BF and FMI were useful to research the relationship between osteoporosis and obesity at the same time.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With impressive economic development and rapid urbanization, Chinese people have experienced remarked changes in widespread westernization of lifestyle in recent decades [1–3]. Obesity has emerged as a critical public health issue in China, for obesity could lead to many serious weight-related disorders such as hypertension, diabetes mellitus, cardiovascular disease, certain forms of cancer [4–6]. Recently, Data from the Global Longitudinal Study of Osteoporosis in Women confirmed the increased risk of fracture in underweight women but showed no protective effect of obesity [7]. Even more obesity is a risk factor of osteoporosis and its related fracture: data from the Osteoporotic Fractures in Men Study showed obese men had a significantly higher risk fracture than normal weight men after adjusted for BMD, which was particularly obvious with hip fracture [8–10]. Therefore, when we discussed the relationship between obesity and osteoporosis, we should explore the effect of the body composition such as FM, %BF, FMI, LM and LBMI on BMD. As we all know, LM was a determinant to BMD, and FM appeared controversial. The reason could be due to BMI levels related to all skeletal BMD. Clinical data confirmed low BMI (<18.5 kg/m2) appeared to be one risk factor for osteoporosis and its related fraction [11–14], and FM could be a protective effect on BMD in underweight. Recent studies showed contradict viewpoint by showing an inverse relationship between FM and BMD in obesity [15–17].
Body mass index (BMI) can be used as a predictor for assessing obesity and related diseases, which shows good resolution among different ages, genders, races, nation, and economic development levels. Because of these reasons, the BMI categories for obesity are quite different globally. WHO international BMI categories for overweight and obesity are 25 and 30 kg/m2 respectively in Western countries [18]. A WHO expert consultation proposed that the proportion of Asian people with a high risk of type 2 diabetes and cardiovascular disease was substantial at BMIs lower than the existing WHO cut-off point for overweight (≥25 kg/m2) [19] The revised guidelines categorized overweight as a BMI of 23.0–24.9 kg/m2 and obesity as a BMI ≥ 25 kg/m2 for Asian Indians [20]. The ethnicity-specific BMI categories for overweight and obesity are 24 and 28 kg/m2 respectively in China by Working Group on Obesity in China (WGOC), which was based on surveys on 239,972 people, aged 20–70 in 1990s covering 21 provinces [21–24]. The BMI categories in different nation should be paid more attention, because they are optimal cut-off points to prevent some chronic diseases epidemic which could not compose osteoporosis. In order to explore the association of obesity with osteoporosis in Chinese, it is necessary to select WHO BMI and WGOC BMI respectively and compare them each other. Perhaps a potentially important target on prevention and treatment of osteoporosis and related fracture in Chinese adult men was found.
Materials and methods
Subjects
A total of 515 adult men who were 20–89 years old were enrolled as healthy subjects for bone studies at Qianfoshan Hospital, Shandong University between September 2008 and August 2010. Subjects were recruited from the participants in a community-based osteoporosis prevention study. The protocol was approved by the Ethics Committee, Qianfoshan Hospital, Shandong University and informed consent was obtained from each subjects.
Exclusion criteria included any history of diseases as follows: history of metabolic bone diseases such as chronic renal disease, liver disease, thyroid disease and rheumatoid arthritis; history of disease affecting body composition such as thyrotoxicosis and hypothyroidism; history of having any medications likely to influence BMD and body composition such as thyroid hormones, glucocorticosteroids, bisphosphonates and antiobesity drugs. Besides, five men were excluded due to prostatectomy, and seven men excluded due to BMD at any of regions (≥Mean ± 3 SD). One man was excluded due to incomplete BMD data. In the end, 502 men were included in the analysis.
Each subject was required to finish a detailed questionnaire on demographic and lifestyle information about alcohol consumption, smoking, exercise outside, diet and medical history consisting of past illness and current medication. In the questionnaire, there were two types of drinking: drinkers and nondrinkers. Drinkers used to take an alcoholic beverage about twice a week (alcohol intake <20 g/day), and nondrinkers used to take no beer, wine, or hard liquor. There were two types of smoking: smokers and nonsmokers. Smokers were defined as those who have smoked at least 6 months consistently or cumulative, and whose smoking index (amounts of cigarettes per day × years of smoking) was less than 200, and nonsmokers were defined as those who have never smoked.
Anthropometry and body composition measurement
Dual-energy X-ray absorptiometry (DXA); software version 11.40.004; GE-lunar, WI, USA) was used to measure LM, LBMI, FM, FMI, %BF, total body BMD (TB) and regional BMD through whole-body scans. Regional BMD consisted of arm leg, trunk, pelvis, spine, femoral neck (FN), total femur (TF), and lumbar Spine BMD (L1–L4). For ethical reasons, we did not make any further assessments of the precision error of this equipment. According to the manufacturer brochure, this is a standardized commercial machine with an in vivo precision (%coefficient of variation) of <1 % for anterior–posterior spinal, femoral, total body BMD and body composition. DXA was calibrated using a standard phantom provided by the manufacturer, performed daily and demonstrated long-term (<2 years) CVs of ≤0.8 %.
Weight was measured to the nearest 0.1 kg with a calibrated standard balance beam scale. Height was measured to the nearest 0.1 cm with a wall-mounted stadiometer. All subjects wore light clothing and no shoes, while measured. All values were recorded as the mean of three measures. BMI was calculated as weight (kg)/height (m2). FMI was calculated as fat mass (kg)/height (m2), and LBMI was calculated as lean mass (kg)/height (m2).
We selected WHO BMI and WGOC BMI for obesity to divide all subjects into three groups respectively. According to WHO criteria, all subjects were divided into normal weight (18.5 ≤ BMI < 25 kg/m2, n = 202), overweight (25 ≤ BMI < 30 kg/m2, n = 242), and obesity (BMI ≥ 30 kg/m2, n = 58). According to WGOC criteria, normal weight (18.5 ≤ BMI < 24 kg/m2, n = 137), overweight (24 ≤ BMI < 28 kg/m2, n = 225), and obesity (BMI ≥ 28 kg/m2, n = 140).
Statistical analysis
Statistical analysis was performed using SPSS (version 16.0 for Windows, SPSS Inc., Chicago, IL, USA). Differences in means of continuous variables such as baseline characteristics, body composition and BMD at total body and regional sites (arm, leg, trunk, pelvis, spine, FN, TF and L1–L4) were assessed by One ANOVA. Differences in frequencies (drinker, nondrinker, smoker and nonsmoker) for categorical variables in groups were assessed by Chi Square. Because age is a significantly determinant to BMD, Age-adjusted Pearson correlation coefficients were detected to assess linear relationships between body composition-related parameters (LM, LBMI, FM, FMI, %BF) and BMD at total body and regional sites among groups. Because of a significant interaction between body composition-related parameters and BMD, the regression models evaluated, respectively, the associations of BMD with FM and LM, and with %BF and FMI. Linear regression of BMD at sites evaluated the independent associations of FM, LM, age height, smoking and regular alcohol consumption in one model. Linear regression of BMD at sites evaluated the independent effects of %BF, FMI, age, weight, height, smoking and regular alcohol consumption in another model. Determination coefficients, SE of estimate, beta coefficients and P value were calculated among groups according to WHO BMI and WGOC.
Results
Descriptive statistics
The basic characteristics of the subjects are shown in Table 1. Age and height appeared no significant differences among groups. The percentages of normal, overweight and obesity were 27.2, 44.8 and 28 % in adult men according WGOC criteria, and 40.2, 48.2 and 11.6 % according WHO criteria. The numbers of normal, overweight and obesity appeared significantly different (P < 0.01). LM, LBMI, FM, %BF and FMI were increased with increases in BMI. According to WHO criteria, Comparisons of LM, LBMI, FM, %BF, FMI among normal, overweight, and obesity were significantly different (P < 0.001). Compared to normal weight, total body and regional BMD of overweight and obesity were significantly higher (all P < 0.001). When comparing overweight with obesity, it showed significant difference (P < 0.05) except BMD at arm, leg, FN and TF. According to WGOC criteria, Comparisons of LM, LBMI, FM, %BF, FMI among normal, overweight, and obesity were significantly different (P < 0.001). Compared to normal weight, total body and regional BMD of overweight and obesity were significantly higher (all P < 0.001). When comparing overweight with obesity, it showed no significant difference except BMD at total body (P < 0.001), trunk and pelvis (P < 0.01). The number of drinker and smoker appeared not significantly different in groups.
Age-adjusted partial correlation analysis
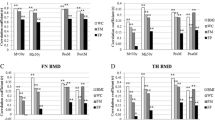
Age-adjusted partial correlation analyses body composition-related parameters and BMD are provided in Tables 2 and 3. LM, LBMI, FM, %BF, FMI, were positively correlated with BMD at almost sites (r = 0.193–0.504; P < 0.001) in all subjects.
In normal weight, LM was positively correlated with BMD at trunk, pelvis and spine sites (r = 0.258–0.282; P < 0.01), and FM was positively correlated with trunk BMD (r = 0.232; P < 0.01) according to WGOC criteria. LM and LBMI were positively correlated with BMD at almost sites (r = 0.159–0.350; P < 0.01). FM and FMI had positive associations with BMD at trunk, pelvis, spine and FN (r = 0.199–0.309; P < 0.01) according to WHO criteria.
In overweight, LM and LBMI were positively correlated with BMD at almost sites (r = 0.179–0.369; P < 0.01) according to WGOC criteria. LM and LBMI appeared significantly positive with BMD at almost sites (r = 0.173–0.432; P < 0.01), but %BF and FMI were negative significance with BMD at total body, arm, leg, spine and FN (r = negative 0.141–0.279; P < 0.05) according to WHO criteria.
In obesity, LM and LBMI were positively significant with BMD at total body and almost regional sites (r = 0.235–0.526; P < 0.01) by WHO and WGOC criteria. %BF appeared negatively significant with total body, arm and leg BMD (r = negative 0.105–0.227; P < 0.05) by WGOC criteria, however, %BF and FMI appeared negatively significant with BMD at arm, leg, FN and TF (r = negative 0.248–0.337; P < 0.05) by WHO criteria.
Multiple regression analysis
Tables 4 and 6 showed determination coefficients and regression coefficients for FM and LM. In normal weight, there was 16.5–42.1 % of BMD variability at almost sites according to WGOC criteria, 16.8–35.7 % according to WHO criteria. In overweight, there was 10.4–20.9 % of BMD variability at almost sites according to WGOC criteria, 12.1–29.5 % according to WHO criteria. In obesity, there was 14.3–36.7 % of BMD variability at almost sites in obesity according to WGOC criteria, 15.0–44.7 % according to WHO criteria.
Lean mass (LM) showed statistically positively significant relations with BMD at almost sites (all P < 0.05) in all groups. Only in normal weight and overweight by WGOC criteria, FM appeared positively significant with BMD at almost sites.
Tables 5 and 6 showed determination coefficients and regression coefficients for %BF and FMI. In overweight, there was 9.5–23.6 % of BMD variability at almost sites in overweight according to WGOC criteria, and 11.3–30.8 % according to WHO criteria. In obesity, there was 19.0–36.7 % of BMD variability at almost sites according to WGOC criteria and 16.5–44.1 % according to WHO criteria. %BF and FMI appeared insignificant related to BMD in normal groups. %BF and FMI appeared statistically negatively significant relations with BMD at almost sites in overweight and obesity. However, they were inconsistent in same site.
Discussion
The present results of our research showed that the percentage of obesity in adult men was different by two criteria for obesity: that of normal weight, overweight and obesity were 27.2, 44.8 and 28 % according to WGOC criteria, and 40.2, 48.2 and 11.6 % according to WHO criteria. The numbers of overweight were more than other groups. The results suggested the percentages of overweight and obesity were high, as Hou et al. [25] reported recently the prevalence of combined overweight and obesity was significantly increased in Chinese men. The body composition-related parameters (FM, %BF, FMI, LM and LBMI) and BMD at total body and regional sites gradually significantly increased among normal weight, overweight and obesity.
When the subjects were divided into normal weight, overweight and obesity, the body composition-related parameters appeared different effect on BMD. Not only by WHO BMI but also by WGOC BMI for obesity, LM and LBMI could always help to determinant of BMD at total body and regional sites in adult men, which means that LM and LBMI were strongest predictors of BMD. The results suggested that the future intervention studies to prevent loss of bone mass should focus on strategies to increase or maintain LM and LBMI. Many reports showed continent opinion [26].
According to WGOC criteria, FM appeared significantly positive with BMD at trunk, pelvis, spine, femur and L1–L4 but no significant with BMD at arm and leg. %BF appeared significantly negative with BMD in total body, arm and leg in overweight and obesity, but FMI showed no significance with BMD. According to WHO criteria, the relationships of %BF and FMI with BMD at trunk, pelvis, and femur were positive in normal weight, however, which appeared significantly negative with BMD at total body and arm, leg, femur in overweight and obesity. FM showed no significance. Therefore, %BF and FMI were adverse to overweight and obesity in adult Chinese men. It was consistent with the finding that the risks of osteoporosis, non-spine fractures were significantly higher for subjects with a higher percentage body fat in a study of a large cohort of Chinese subjects [27]. Other researches provided further evidence that FM may have no beneficial effect on bone [28–31].
So as to research the correlation of FM and LM to BMD, the regression model was established. The results showed LM was a determinant to BMD and FM was significantly positive to BMD in normal weight and overweight by WGOC criteria and only in normal weight by WHO criteria. Because there was inconsistent in fat-related parameters and BMD, we explored the effect of %BF and FMI on BMD. The results showed that %BF and FMI were not related to BMD in normal weight, and which were adverse to total body and regional BMD in overweight and obesity. Comparing with two criteria, we found the differences in fat-related parameters and BMD according to WHO criteria were more obvious than that according to WGOC criteria. We also found that %BF and FMI were useful to research the relationship between osteoporosis and obesity at the same time.
The outcomes based on the WHO and WGOC categories were interesting. Among northern Chinese men, Both LM and LBMI were strong predictors of BMD, but fat-related parameters showed significant differences: positively associations in normal, and negatively associations in overweight and obesity. It suggested that any of body composition was vital to conserve normal weight, and it was quite necessary to control overweight and obesity, especially %BF and FMI, and to increase and maintain LM and LBMI at the same time. The strength of this paper was that it was based on WHO and WGOC BMI categories, and based on adopted lean-related parameters such as LM and LBMI, and fat-related parameters such as FM, %BF and FMI. But it was under-estimated optimal cut-off values of parameters. Perhaps it would be a target to discuss relationship between obesity and osteoporosis and prevent osteoporosis and related fracture.
References
Wang GR, Li L, Pan YH, Tian GD, Lin WL, Li Z, Chen ZY, Gong YL, Kikano GE, Stange KC, Ni KL, Berger NA (2013) Prevalence of metabolic syndrome among urban community residents in China. BMC Public Health 13:599
Hu FB, Liu Y, Willett WC (2011) Preventing chronic diseases by promoting healthy diet and lifestyle: public policy implications for China. Obes Rev 12(7):552–559
Chung RY, Schooling CM, Cowling BJ, Leung GM (2010) How does socioeconomic development affect risk of mortality? An age-period-cohort analysis from a recently transitioned population in China. Am J Epidemiol 171(3):345–356
Messerli FH, Christie B, DeCarvalho JG, Aristimuno GG, Suarez DH, Dreslinski GR, Frohlich ED (1981) Obesity and essential hypertension. Hemodynamics, intravascular volume, sodium excretion, and plasma renin activity. Arch Intern Med 141(1):81–85
Kopelman PG (2000) Obesity as a medical problem. Nature 404(6778):635–643
Mokdad AH, Bowman BA, Ford ES, Vinicor F, Marks JS, Koplan JP (2001) The continuing epidemics of obesity and diabetes in the United States. JAMA 286(10):1195–1200
Compston JE, Watts NB, Chapurlat R, Cooper C, Boonen S, Greenspan S, Pfeilschifter J, Silverman S, Díez-Pérez A, Lindsay R, Saag KG, Netelenbos JC, Gehlbach S, Hooven FH, Flahive J, Adachi JD, Rossini M, Lacroix AZ, Roux C, Sambrook PN, Siris ES (2011) Obesity is not protective against fracture in postmenopausal women: glow. Am J Med 124(11):1043–1050
Nielson CM, Marshall LM, Adams AL, LeBlanc ES, Cawthon PM, Ensrud K, Stefanick ML, Barrett-Connor E, Orwoll ES, Osteoporotic Fractures in Men Study Research Group (2011) BMI and fracture risk in older men: the osteoporotic fractures in men study (MrOS). J Bone Miner Res 26:496–502
King CM, Hamilton GA, Cobb M, Carpenter D, Ford LA (2012) Association between ankle fractures and obesity. J Foot Ankle Surg 51(5):543–547
Premaor MO, Compston JE, Fina Avilés F, Pagès-Castellà A, Nogués X, Díez-Pérez A, Prieto-Alhambra D (2013) The association between fracture site and obesity in men: a population-based cohort study. J Bone Miner Res 28:1771–1777
De Laet C, Kanis JA, Odén A, Johanson H, Johnell O, Delmas P, Eisman JA, Kroger H, Fujiwara S, Garnero P, McCloskey EV, Mellstrom D, Melton LJ 3rd, Meunier PJ, Pols HA, Reeve J, Silman A, Tenenhouse A (2005) Body mass index as a predictor of fracture risk: a meta-analysis. Osteoporos Int 16(11):1330–1338
Holmberg AH, Johnell O, Nilsson PM, Nilsson J, Berglund G, Akesson K (2006) Risk factors for fragility fracture in middle age. A prospective population-based study of 33,000 men and women. Osteoporos Int 17(7):1065–1077
Sharma S, Fraser M, Lovell F, Reece A, McLellan AR (2008) Characteristics of males over 50 years who present with a fracture: epidemiology and underlying risk factors. J Bone Joint Surg Br 90(1):72–77
Lee SH, Khang YH, Lim KH, Kim BJ, Koh JM, Kim GS, Kim H, Cho NH (2010) Clinical risk factors for osteoporotic fracture: a population-based prospective cohort study in Korea. J Bone Miner Res 25(2):369–378
Zhao LJ, Liu YJ, Liu PY, Hamilton J, Recker RR, Deng HW (2007) Relationship of obesity with osteoporosis. J Clin Endocrinol Metab 92:1640–1646
Reid IR (2010) Fat and bone. Arch Biochem Biophys 503:20–27
Reid IR (2008) Relationship between fat and bone. Osteoporos Int 19:595–606
World Health Organization (2000) Obesity. Preventing and managing the global epidemic. Report on a WHO consultation on Obesity. Technical Report Series Number 894. Geneva. World Health Organization
World Health Organization (2004) Appropriate body mass index for Asian populations and its implications for policy and intervention strategies. Lancet 363:157–163
Misra A, Chowbey P, Makkar BM, Vikram NK, Wasir JS, Chadha D, Joshi SR, Sadikot S, Gupta R, Gulati S, Munjal YP; Consensus Group (2009) Consensus statement for diagnosis of obesity, abdominal obesity and the metabolic syndrome for Asian Indians and recommendations for physical activity, medical and surgical management. J Assoc Physicians India 57:163–170
Working Group on Obesity in China, Department of Disease Control, Minintry of Health (2004) The guideline a for prevention and control of overweight and obesity in Chinese adults. Biomed Environ Sci 17:1–35
Chen CM (2008) Overview of obesity in mainland China. Obes Rev 9(Suppl 1):14–21
Wang Y, Mi J, Shan XY, Wang QJ, Ge KY (2007) Is China facing an obesity epidemic and the consequences? The trends in obesity and chronic disease in China. Int J Obes 31(1):177–188
Zhou BF, Cooperative Meta-Analysis Group of the Working Group on Obesity in China (2002) Predictive values of body mass index and waist circumference for risk factors of certain related diseases in Chinese adults—study on optimal cut-off points of body mass index and waist circumference in Chinese adults. Biomed Environ Sci 15:83–96
Hou X, Liu Y, Lu H, Ma X, Hu C, Bao Y, Jia W (2013) Ten-year changes in the prevalence of overweight, obesity and central obesity among the Chinese adults in urban Shanghai, 1998–2007—comparison of two cross-sectional surveys. BMC Public Health 13(1):1064
Lu LJ, Nayeem F, Anderson KE, Grady JJ, Nagamani M (2009) Lean body mass, not estrogen or progesterone, predicts peak bone mineral density in premenopausal women. J Nutr 139(2):250–256
Hsu YH, Venners SA, Terwedow HA, Feng Y, Niu T, Li Z, Laird N, Brain JD, Cummings SR, Bouxsein ML, Rosen CJ, Xu X (2006) Relation of body composition, fat mass, and serum lipids to osteoporotic fractures and bone mineral density in Chinese men and women. Am J Clin Nutr 83(1):146–154
Sheng Z, Xu K, Ou Y, Dai R, Luo X, Liu S, Su X, Wu X, Xie H, Yuan L, Liao E (2011) Relationship of body composition with prevalance of osteoporosis in central south Chinese postmenopausal women. Clin Endocrinol 74(3):319–324
Ho-Pham LT, Nguyen ND, Lai TQ, Nguyen TV (2010) Contributions of lean mass and fat mass to bone mineral density: a study in postmenopausal women. BMC Muskuloskelet Disord 11:59
King CM, Hamilton GA, Cobb M, Carpenter D, Ford LA (2012) Association between ankle fractures and obesity. J Foot Ankle Surg 51(5):543–547
Moayyeri A, Luben RN, Wareham NJ, Khaw KT (2012) Body fat mass is a predictor of risk of osteoporotic fractures in women but not in men: a prospective population study. J Intern Med 271(5):472–480
Acknowledgments
This work was supported by the grants from Shandong Natural Science Foundation and Geriatrics and gerontology Research Programs of the Ministry of Civil Affairs during the 11th Five-Year Plan Period.
Conflict of interest
The authors D. H. Kang, L. F. Guo, T. Guo, Y. Wang, T. Liu, X. Y. Feng, and X. Q. Che declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, D.H., Guo, L.F., Guo, T. et al. Association of body composition with bone mineral density in northern Chinese men by different criteria for obesity. J Endocrinol Invest 38, 323–331 (2015). https://doi.org/10.1007/s40618-014-0167-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-014-0167-5