Abstract
Nowadays, commercial natural fibers cannot meet the increasing industrial demand. In order to meet this demand, recommending a new natural fiber for the composites industry is very important. In this paper, Carex panicea fibers were characterized for the first time and introduced as a potential natural fiber. Physical, chemical, thermal, mechanical, and morphological properties of the Carex panicea fibers were characterized using scanning electron microscopy, Fourier transform infrared spectroscopy, thermogravimetric analysis, X-ray photoelectron spectroscopy, and X-ray diffraction analysis. Carex panicea fibers consist of 65.70% cellulose and 27.8% hemicellulose content. The density and crystallinity index of the fiber were found as 1.247 g/cm3 and 56.42%, respectively. Tensile strength and Young’s modulus of fibers were determined as 143 ± 41 MPa and 5.5 ± 1.86 GPa, respectively. Carex panicea fibers are thermally stable up to 219.4 °C. Carex panicea fibers are potential bio-degradable reinforcement material for light-weight polymeric composites with relatively enhanced mechanical properties and decomposition temperature.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Recently, with the increasing environmental awareness and economical concerns, the utilization of renewable and eco-friendly resources became important [1,2,3,4]. Today, petroleum-based fibers are used as reinforcement for the manufacturing of polymer matrix composites, and these composites are utilized in many applications such as aerospace, automobile, and military [5,6,7,8,9]. While petroleum-based fibers possess high mechanical and physical properties, they have also negative impacts in terms of environmental and economic aspects [10,11,12,13]. Nowadays, the advancement of sustainable green technology has increased in the composite industry. However, ensuring high demand for natural fiber-reinforced composites by using commercial plant fibers is difficult. Therefore, the industry has sought a new plant fiber with desired thermal, physical, and mechanical properties [1, 14, 15]. To overcome the drawbacks of petroleum-based fibers and to meet the industrial reinforcement demand for composites, scientists try to replace man-made fibers with eco-friendlier natural fibers [16]. The utilization of natural fibers may help to protect the environment by reducing waste disposal, usage of hazardous material for the production of petroleum-based fibers, and increasing the usage of renewable sources [17].
Cellulose-based natural fibers can be extracted from different plants such as jute [18], kenaf [19], and hemp [20]. These fibers are considered to be a potential alternative to traditional petroleum-based and other synthetic fibers due to their attractive properties such as low density, biodegradability, renewability, cost-effectiveness, and abundancy around the world [3, 17, 21, 22]. Also, natural fibers can provide comparable mechanical properties to artificial fibers such as relatively high mechanical strength, stiffness, and modulus [10, 11, 23]. Researchers currently have focused on the identification and characterization of new natural fibers due to the increasing demand in the composite industry. Within this framework, new natural fibers such as Leucas aspera [24], Conium maculatum [25], Eleusine indica [2], Hierochloe odarata (Dalmis et al. 2020), Ellettaria cardamomum [26], Purple bauhinia [27], Trachelospermum jasminoides [28], Cordyline australis [29], Lavender stem [30], Atriplex halimus [31], Coccinia grandis L. [32], Lygeum spartum L. [33], Cissus vitiginea [34], Grewia damine [35], Grewia flavescens [35], Chrysanthemum morifolium [36], Vachellia farnesiana [37], and Althea officinalis L [38]. fibers were identified and characterized. In order to give desired properties to fibers, many modification and treatment studies have been carried out such as Inula viscosa [39], Symphirema involucratum [40], and Agave Americana [41].
Recently, researchers and experts have focused on the characterization of new cellulosic fibers because they represent alternatives to traditional reinforcements [42,43,44,45,46,47,48,49,50,51]. Carex panicea, also known as grass like plant, comes from Cyperaceae family. While Carex panicea is native to Europe, they have spread around Asia and America [52, 53]. The availability and abundancy of Carex panicea plants around the world make them attractive candidate as reinforcement for green composites. According to literature, Carex panicea fibers have not identified yet and within this framework, Carex panicea fibers were extracted and characterized for the first time as an alternative reinforcement material for composites. Physical, chemical, thermal, morphological, and mechanical properties of the Carex panicea fibers were characterized by the help of Archimedes density method, determination of chemical composition, Fourier transform infrared spectroscopy (FTIR), X-ray diffraction analysis (XRD), X-ray photoelectron spectroscopy (XPS), thermogravimetric analysis (TGA), scanning electron microscope (SEM), and tensile tests.
2 Materials and methods
2.1 Fiber extraction
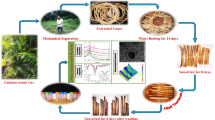
Carex panicea plants were harvested from Uzunalan, Çanakkale that is located in the west coast of Turkey. Images of Carex panicea plants and fiber extraction steps were given in the schematic diagram in Fig. 1. Conventional water fiber extraction technique is utilized to obtain fiber from the stem of Carex panicea plants. As a preparation for fiber retting, plants were cleaned and rinsed with distilled water for several times and cut into pieces to facilitate the extraction. For 6 weeks, the plants were placed in a plastic barrel filled with tap water and covered to enable microbial breakdown in order to facilitate fiber extraction from the plant. Fibers were then carefully removed from the stem, combed with a metal comb. Fiber separation process by metal comb was carried out under an aqueous medium. The obtained fibers about 200-µm diameter were washed until cleared completely and rinsed in distilled water. To remove excess moisture, the fibers were oven-dried for 24 h at temperature of 60 °C. Moisture content and moisture regain of the Carex panicea fiber were found to be 11.3% and 11.8%, respectively.
3 Characterization of fibers
3.1 Density measurement
The density of Carex panicea fibers was identified by using ASTM D8171-18 Method B (Eq. 1). The method is based on Archimedes law, and approximately 1 g of fiber specimen, coiled like a ball, with three replications according to standard was used for measurements. Specimens were dried at 40 °C for 24 h in a low temperature oven and weighed by using a precision balance (Mettler Toledo ME 204 with density Kit). After dry weights were taken, specimens were immersed in a baker for 24 h, which is containing boiling water. Then submerged weights of fibers were measured.
where d is the density of fibers, Wd and Ws are dry weight, and weight of samples submerged in water, respectively.
3.2 Chemical analysis
The chemical composition of Carex panicea fibers was determined by the method explained in details in related previous studies [36, 54]. Prior to chemical analysis, Carex panicea fibers were oven-dried at temperature of 105 °C for 4 h and then kept in desiccator to avoid humidity.
3.3 Fourier transform infrared (FTIR) analysis
ATR-FTIR spectra were recorded of fibers recorded using the Perkin Elmer Spectrum BX instrument. Measurement was conducted in the range of 650–4000 cm−1 wave number with a resolution of 2 cm− 1.
3.4 Thermogravimetric analysis (TGA)
To identify the thermal behavior of Carex panicea fiber, thermogravimetric analysis was utilized. Shimadzu DTG-60H instrument was utilized and analysis was recorded from room temperature to 800 °C under nitrogen atmosphere at a heating rate of 10 °C/min. Approximately 15 mg sample was implemented for measurement.
3.5 X-ray photoelectron spectroscopy (XPS) analysis
X-ray photoelectron spectroscopy (XPS) was conducted to determine the surface chemistry of Carex panicea fibers. Measurement was performed with Thermo Scientific instrument using Al-Ka X-ray source (1486.7 eV) between 1350 and 10 eV with a resolution of 1 eV. The surface of samples was cleaned with Ar gas prior to the analysis that was recorded with 20 scans.
3.6 X-ray diffraction analysis
X-ray diffraction method was performed to identify the crystallite index (CI) and crystallite size of Carex panicea fibers. A copper X-ray tube was used as the radiation source (λ-Cu-Kα1 = 1.54 Å) and power was kept at 40 kV–30 mA during scan. XRD pattern was obtained between 5 and 65° range with 2°/min scan rate.
Dried fibers were powdered in order to obtain sufficient intensity as a pattern, and CI was determined by using an empirical formula suggested by Segal et al. (1959) (Eq. 2). Three replications of XRD pattern were obtained and mean values of intensities were calculated to produce a more reliable pattern.
where I200 is the maximum intensity of cellulose crystal, which is (200) lattice plane, and Iam is the intensity of amorphous peak, which is located between 18 and 19°. The intensity of amorphous peak is determined by taking the minimum intensity between highest two peaks represented in the cellulose pattern [55].
3.7 Tensile test
A universal testing machine (Instron 4411) was used for the tensile test of Carex panicea fibers with 1 kgf load cell. The loading rate was 1 mm/min with a gauge length of 20 mm. Pneumatic grips which were used for clamping the fiber have of 0.5 MPa pressure. Tensile tests were conducted following ASTM D 3822 standard. Ten fiber specimens were tested to check repeatability.
3.8 Morphology analysis
Detailed morphological characterizations were carried out by scanning electron microscope (SEM) images taken from longitudinal and cross-sectional parts of the Carex panicea fiber. Observations were performed using a JEOL-JJM 6060 model SEM device. The surfaces of the Carex panicea fibers were coated by Au–Pd alloy using sputter coating before characterization in order to avoid the electron beam charging effect.
4 Results and discussion
4.1 Fiber density
The density of Carex panicea fibers was found as 1.247 g/cm3. As a candidate reinforcement, the low density of fibers triggers an increase in specific strength and results in lighter component production. When compared, the density of Carex panicea fibers is a little lighter than many common, and industrial natural fibers, such as kenaf (1.4 g/cm3), flax (1.50 g/cm3), hemp (1.48 g/cm3), jute (1.46 g/cm3), and sisal (1.45 g/cm3) [56] that can be advantageous. Fiber density plays a vital role in the design of lightweight components [3].
4.2 Chemical composition
Determination of fiber composition is significant due to its possible effects on chemical structure, physical properties, and mechanical performance of fiber and further reinforced composites. Cellulose contents of Carex panicea and other cellulosic natural fibers are listed in Table 1. The cellulose may be the most important component of the fiber which determines tensile strength and stiffness based on its high crystallinity. Carex panicea presents comparable cellulose content with recently characterized lignocellulosic fibers. Hemicellulose fraction (27.8%) is higher than that of many cellulosic fibers such as Acaicia niotica L. (14.14%) [1], Cajanus cajan (10.43%) [57], Acacia concinna (12.78%) [11], and purple bauhinia (9.17%) [27]. Hemicellulose can influence thermal resistance and water absorption of cellulosic fibers. The thermal resistance of fibers can be also affected by the lignin component. The fraction of lignin and the rest of the other constituents of Carex panicea was determined to be 6.5% which act as an adhesive to keep elementary cellulose cells together. However, lignin may deteriorate the interphase adhesion between components of polymeric composites [58].
4.3 FTIR analysis
To identify the main components of Carex panicea fibers (cellulose, hemicellulose, and lignin) and functional groups in fiber, FTIR spectroscopy was conducted. FTIR spectrum of Carex panicea fiber was recorded in the range of 4000–650 cm−1 and given in Fig. 2. The band located at 895 cm−1 is associated with the O-C-O stretching vibrations of cellulose [64,65,66]. The broad band at 1030 cm−1 indicates C-O and C-H stretching vibrations of fiber. The peak at 1160 cm−1 is related to the C–O antisymmetric bridge stretching of cellulose [36, 67]. The band at 1244 cm−1 can be assigned to C-O vibrations of acetyl groups in hemicellulose [68, 69]. The peaks at 1316 and 1371 cm−1 correspond to C-O and O–H bending vibrations of cellulose [67, 70, 71]. The bands located at 1422 and 1451 cm−1 correspond to CH2 bending vibrations and C-H deformation in groups of methyl, methoxyl, and methyl in lignin, respectively [54, 72, 73]. The band observed at 1506 cm−1 is associated with the C = C ring stretching of aromatic lignin in fiber [25, 74]. The absorption band at 1633 cm−1 corresponds to absorbed water in fiber [75, 76]. The band at 1730 cm−1 is related to the carbonyl groups of lignin [31, 77, 78]. The broad band at 3338 is related to the –OH, and 2920 cm−1 and 2853 cm−1 are related to the C-H stretching vibrations in cellulose and hemicellulose, respectively [79, 80]. FTIR results of Carex panicea agree with the most common natural fibers such as hemp [20], jute [81], and kenaf [19].
4.4 Thermogravimetric analysis
Thermogravimetry is a helpful characterization technique for understanding of the thermal behavior of cellulosic fibers. Because the production of polymer-based composites includes relatively high processing temperatures, the determination of the onset temperature of fibers is significant to benefit from fiber properties at a maximum rate [36, 82,83,84,85]. TG/DTG curves of Carex panicea fiber are shown in Fig. 3. As presented in Fig. 3, the first mass loss was observed between 25 and 100 °C with 5.36% due to the dehydration of water in fibers [86, 87]. The second mass loss occurred between 220 and 310 °C with 13.84% can be related to the degradation of hemicellulose in fibers [88]. The third and the major weight losses were recorded between 300 and 370 °C which indicates decomposition of cellulose in fiber with 36.53% [89, 90]. The temperatures of 219.4 °C and 351.59 °C were determined as onset decomposition temperature (Tonset) and maximum degradation temperature (Tmax), respectively. Comparison of Tonset and Tmax temperature of most utilized and some recent natural fibers were listed in Table 2. Compared to most utilized natural fibers such as kenaf (219 °C) and jute (205.1 °C), wood pine (234 °C), bagasse (222.3 °C), and cotton stalk (221.6 °C), Carex panicea is a proper reinforcement for polymeric composites due to high decomposition onset temperature [91]. The analysis was recorded up to 800 °C and after major decomposition, 28.905% mass loss was observed related to the residual content of fibers [92]. This weight loss may correspond to oxidative degradation of residues and lignin in the fiber [93].
4.5 XRD analysis
XRD pattern of Carex panicea fibers shows two main peaks (see Fig. 4.), first a broad peak at 15.96°, and a second peak at 22.21°. The first peak is formed by overlapping of two cellulose peaks that belongs to (110) and (\(1\overline{1 }0\)) lattice planes, and the second peak is formed by cellulose (200) lattice (French 2014; Kılınç et al. 2018).
The crystallinity index (CI) and crystallite size (L) of Carex panicea fibers were found as 56.42% and 7.6 nm, respectively. CI of Carex panicea fibers generally lower than many plant fibers (Table 3). The low crystallinity of fibers can be also a sign of a low-strength material. The decrease in the strength of fiber can be explained with the absence of crystalline microfibrils cellulosic and chains which cannot align regularly to form an ordered structure [99]. On the other hand, the crystallite size of Carex panicea fibers shows similarities with other natural fibers [36], which is a measure of coherency at the related XRD plane (200).
4.6 XPS analysis
Atomic concentrations for carbon and oxygen on the surface of Carex panicea were obtained to be 81.86 and 14.10%, respectively. The oxygen/carbon ratio of Carex panicea was determined as 0.17 which is in comparison with some lignocellulosic fibers (Table 4). It is clear that Carex panicea has a relatively hydrophobic surface character presenting dramatically lower O/C as compared with most of commonly used and recently characterized cellulosic fibers as tabulated in Table 4. This finding may indicate its potential as a reinforcement material in highly non-polar polymers considering possible surface compatibility. To determine the functional groups of Carex panicea, deconvolution of C1s and O1s peaks was conducted. C1s and O1s spectra of Carex panicea are depicted in Fig. 5a and b. The peaks located at 286.5 eV and 532.7 eV can be associated with C–O–C bonds [104]. The ratio of C–C/C-H (284.7 eV) and C–O–C (286.5 eV) groups was calculated as 69.44 and 30.56%, respectively. The major peak at 284.7 in C1s spectra representing C–C/C-H and also C–O–C bonds can affirm the presence of cellulose in fiber [104, 105].
4.7 Tensile properties
Stress–strain behavior of Carex panicea fibers is presented in Fig. 6. Tensile strength, Young’s modulus, and elongation at break data are listed and compared with the other natural fibers in Table 5. The standard deviation is quite large which is common for natural cellulosic fibers. As seen in the graph, fibers exhibit linear characteristic like other cellulosic natural fibers. On the other hand, a sudden load drop indicates the brittle nature of the fibers. It is clear that Carex panicea fiber has higher tensile strength than the Juncus effuses L. [110], Piassava [111], Chrysanthemum morifolium [36], and Phoenix dactylifera L. [112]. Young’s modulus of fiber (5.5 ± 1.86 GPa) is comparable with other natural fibers such as Juncus effuses L. [110], Phoenix dactylifera L. [112], and Piassava [111]. Due to its linear orientation and high degree of polymerization, cellulose can govern the tensile properties of the fibers (Kathirselvam et al., 2019).
4.8 Morphological characterizations
General and detailed scanning electron microscope images of the Carex panicea fiber are presented in Fig. 7 in order to interpret fiber morphology. Both longitudinal and cross-sectional SEM images are presented due to the fact that they provide different information. The fiber sample has about 200-µm diameter according to Fig. 7a. In addition, some impurity residues take attention on Carex panicea fiber surface (see Fig. 7a and b). It can be seen that these impurities on the surface cause the formation of an indented surface morphology (Fig. 7b). Thus, Carex panicea fiber endorses a high surface area for locking polymer matrix as an additive for composites.
As can be observed from cross-sectional images in Fig. 7c and d, Carex panicea fiber consists of many elementary fibers as other natural fibers [25]. The empty nutrition/water channels at the center of elementary fibers are called as lumen [113]. Lumen diameters of the elementary fibers of Carex panicea appear to be very variable according to the Fig. 7c and d. It can be concluded from SEM images that Carex panicea fibers have about 1–2 µm lumen wall thickness, low lumens about 5 µm lumen diameter while large lumens have a diameter of 50 µm. Combination of the large and small diameters of lumen causes low density, high insulation, and absorbance properties. Among these properties, Carex panicea fiber is striking for its very thin wall thickness. For example, Carex panicea fiber has lower wall thickness than curaua, jute, sisal, and Chrysanthemum morifolium fibers [36, 111].
5 Conclusion
In this study, Carex panicea fibers were characterized as new potential natural fiber reinforcement for polymeric composites. The obtained physicochemical characterization results of Carex panicea stem fibers have low density, comparable cellulose content and crystallinity index, better surface hydrophobicity, better thermal stability, relatively high tensile properties, and similar surface morphology. According to the results:
-
The density of fibers are calculated as 1.247 g/cm3 which is lower than the most utilized synthetic fibers and this indicate lighter composite production.
-
Thermal analysis shows that fibers thermally stable up to 219.4 °C without any degradation. Considering relatively high manufacturing temperature of polymer composites, thermal analysis indicates suitability of Carex panicea fibers for polymer composite production.
-
XPS results show that with very low O/C ratio (0.17), Carex panicea fibers have hydrophobic surface characteristics. Tensile strength and Young’s modulus of fibers were determined as 143 ± 41 MPa and 5.5 ± 1.86 GPa, respectively.
-
SEM images show that Carex panicea fibers consist of many elementary fibers bonded together. According to these results, Carex panicea fibers can be a potential natural fiber as an alternative to synthetic fibers, which can be utilized in polymer composites.
Data availability
Not applicable.
References
Kumar R, Sivaganesan S, Senthamaraikannan P et al (2020) Characterization of new cellulosic fiber from the bark of Acacia nilotica L. plant. J Nat Fibers. https://doi.org/10.1080/15440478.2020.1738305
Khan A, Vijay R, Singaravelu DL et al (2021) Extraction and characterization of natural fiber from Eleusine indica grass as reinforcement of sustainable fiber reinforced polymer composites. J Nat Fibers 18:1742–1750. https://doi.org/10.1080/15440478.2019.1697993
Narayanasamy P, Balasundar P, Senthil S et al (2020) Characterization of a novel natural cellulosic fiber from Calotropis gigantea fruit bunch for ecofriendly polymer composites. Int J Biol Macromol 150:793–801. https://doi.org/10.1016/j.ijbiomac.2020.02.134
Gholampour A, Ozbakkaloglu T (2020) A review of natural fiber composites: properties, modification and processing techniques, characterization, applications. J Mater Sci 55:829–892
Jiang S, Chen Y, Duan G et al (2018) Electrospun nanofiber reinforced composites: a review. Polym Chem 9:2685–2720
Linan LZ, Cidreira ACM, da Rocha CQ et al (2021) Utilization of acai berry residual biomass for extraction of lignocellulosic byproducts. J Bioresour Bioprod 6:323–337. https://doi.org/10.1016/j.jobab.2021.04.007
Fatima A, Yasir S, Khan MS et al (2021) Plant extract-loaded bacterial cellulose composite membrane for potential biomedical applications. J Bioresour Bioprod 6:26–32. https://doi.org/10.1016/j.jobab.2020.11.002
Venkatesh G, Vignesh V, Nagarajan KJ et al (2021) Extraction and characterization of agricultural discarded Sesbania aculeata stem waste as potential alternate for synthetic fibers in polymer composites. J Nat Fibers. https://doi.org/10.1080/15440478.2021.2002756
Babu NBK, Vignesh V, Nagaprasad N et al (2022) Studies on mechanical characterisation of bio-fibre reinforced polymer composites. Bio-Fiber reinforced composite materials. Springer, Singapore, pp 143–155
Vinod A, Vijay R, Singaravelu DL et al (2021) Extraction and characterization of natural fiber from stem of Cardiospermum halicababum. J Nat Fibers 18:898–908. https://doi.org/10.1080/15440478.2019.1669514
Amutha V, Senthilkumar B (2021) Physical, chemical, thermal, and surface morphological properties of the bark fiber extracted from Acacia concinna plant. J Nat Fibers 18:1661–1674. https://doi.org/10.1080/15440478.2019.1697986
Ding L, Han X, Cao L et al (2022) Characterization of natural fiber from manau rattan (Calamus manan) as a potential reinforcement for polymer-based composites. J Bioresour Bioprod 7:190–200. https://doi.org/10.1016/j.jobab.2021.11.002
Jamshaid H, Mishra R, Basra S et al (2020) Lignocellulosic natural fiber reinforced bisphenol F epoxy based bio-composites: characterization of mechanical electrical performance. J Nat Fibers 19:3317–3332. https://doi.org/10.1080/15440478.2020.1843586
Bijlwan PP, Prasad L, Sharma A (2021) Recent advancement in the fabrication and characterization of natural fiber reinforced composite: a review. In: Materials Today: Proceedings. Elsevier, pp 1718–1722
Vignesh V, Balaji AN, Mohamed Rabi BR et al (2021) Cellulosic fiber based hybrid composites: a comparative investigation into their structurally influencing mechanical properties. Constr Build Mater 271:121587. https://doi.org/10.1016/j.conbuildmat.2020.121587
Gaba EW, Asimeng BO, Kaufmann EE et al (2021) Mechanical and structural characterization of pineapple leaf fiber. Fibers 9:51. https://doi.org/10.3390/fib9080051
Kian LK, Saba N, Jawaid M, Fouad H (2020) Characterization of microcrystalline cellulose extracted from olive fiber. Int J Biol Macromol 156:347–353. https://doi.org/10.1016/j.ijbiomac.2020.04.015
Alvarez V, Rodriguez E, Vázquez A (2006) Thermal degradation and decomposition of jute/vinylester composites. J Therm Anal Calorim 85:383–389
Keshk S, Suwinarti W, Sameshima K (2006) Physicochemical characterization of different treatment sequences on kenaf bast fiber. Carbohydr Polym 65:202–206. https://doi.org/10.1016/j.carbpol.2006.01.005
Sawpan MA, Pickering KL, Fernyhough A (2011) Effect of various chemical treatments on the fibre structure and tensile properties of industrial hemp fibres. Compos Part A Appl Sci Manuf 42:888–895. https://doi.org/10.1016/j.compositesa.2011.03.008
Xu Y, Adekunle K, Ramamoorthy SK et al (2020) Methacrylated lignosulfonate as compatibilizer for flax fiber reinforced biocomposites with soybean-derived polyester matrix. Compos Commun 22:100536. https://doi.org/10.1016/J.COCO.2020.100536
Jagadeesh P, Puttegowda M, Mavinkere Rangappa S, Siengchin S (2021) A review on extraction, chemical treatment, characterization of natural fibers and its composites for potential applications. Polym Compos 42:6239–6264. https://doi.org/10.1002/PC.26312
Dawit JB, Regassa Y, Lemu HG (2021) Property characterization of acacia tortilis for natural fiber reinforced polymer composite. Mater Today Proc 44:1718–1722. https://doi.org/10.1016/j.rinma.2019.100054
Vijay R, Manoharan S, Arjun S et al (2020) Characterization of silane-treated and untreated natural fibers from stem of Leucas aspera. J Nat Fibers. https://doi.org/10.1080/15440478.2019.1710651
Kılınç AÇ, Köktaş S, Seki Y et al (2018) Extraction and investigation of lightweight and porous natural fiber from Conium maculatum as a potential reinforcement for composite materials in transportation. Compos Part B Eng 140:1–8. https://doi.org/10.1016/j.compositesb.2017.11.059
Ahmed J, Balaji MA, Saravanakumar SS, Senthamaraikannan P (2021) A comprehensive physical, chemical and morphological characterization of novel cellulosic fiber extracted from the stem of Elettaria cardamomum plant. J Nat Fibers 18:1460–1471. https://doi.org/10.1080/15440478.2019.1691121
Rajeshkumar G, Devnani GL, Maran JP et al (2021) Characterization of novel natural cellulosic fibers from purple bauhinia for potential reinforcement in polymer composites. Cellulose 28:5373–5385. https://doi.org/10.1007/s10570-021-03919-2
Gedik G (2021) Extraction of new natural cellulosic fiber from Trachelospermum jasminoides (star jasmine) and its characterization for textile and composite uses. Cellulose 28:6899–6915. https://doi.org/10.1007/s10570-021-03952-1
Sumihartati A, Wardiningsih W, Al Kautsar N et al (2021) Natural cellulosic fiber from Cordyline australis leaves for textile application: extraction and characterization. Res J Text Appar. https://doi.org/10.1108/RJTA-04-2021-0049
Eyupoglu S, Merdan N (2021) Physicochemical properties of new plant based fiber from lavender stem. J Nat Fibers: 1–11. https://doi.org/10.1080/15440478.2021.1982816
Belouadah Z, Toubal LM, Belhaneche-Bensemra N, Ati A (2021) Characterization of ligno-cellulosic fiber extracted from Atriplex halimus L. plant. Int J Biol Macromol 168:806–815. https://doi.org/10.1016/j.ijbiomac.2020.11.142
Senthamaraikannan P, Kathiresan M (2018) Characterization of raw and alkali treated new natural cellulosic fiber from Coccinia grandis.L. Carbohydr Polym 186:332–343. https://doi.org/10.1016/j.carbpol.2018.01.072
Belouadah Z, Ati A, Rokbi M (2015) Characterization of new natural cellulosic fiber from Lygeum spartum L. Carbohydr Polym 134:429–437. https://doi.org/10.1016/j.carbpol.2015.08.024
Chakravarthy KS, Madhu SM, Naga Raju JS, Shariff MdJ (2020) Characterization of novel natural cellulosic fiber extracted from the stem of Cissus vitiginea plant. Int J Biol Macromol 161:1358–1370. https://doi.org/10.1016/j.ijbiomac.2020.07.230
Arul Marcel Moshi A, Ravindran D, Sundara Bharathi SR et al (2020) Characterization of natural cellulosic fiber extracted from Grewia damine flowering plant’s stem. Int J Biol Macromol 164:1246–1255. https://doi.org/10.1016/j.ijbiomac.2020.07.225
Dalmis R, Kilic GB, Seki Y et al (2020) Characterization of a novel natural cellulosic fiber extracted from the stem of Chrysanthemum morifolium. Cellulose 27:8621–8634. https://doi.org/10.1007/s10570-020-03385-2
Vijay R, James Dhilip JD, Gowtham S et al (2022) Characterization of natural cellulose fiber from the barks of Vachellia farnesiana. J Nat Fibers 19:1343–1352. https://doi.org/10.1080/15440478.2020.1764457
KılınÇ AÇ, Köktaş S, Atagür M, Seydibeyoglu MÖ (2018) Effect of extraction methods on the properties of Althea officinalis L. fibers. J Nat Fibers 15:325–336. https://doi.org/10.1080/15440478.2017.1325813
Moussaoui N, Rokbi M, Osmani H et al (2021) Extraction and characterization of fiber treatment Inula viscosa fibers as potential polymer composite reinforcement. J Polym Environ 29:3779–3793. https://doi.org/10.1007/s10924-021-02147-w
Raju JSN, Depoures MV, Kumaran P (2021) Comprehensive characterization of raw and alkali (NaOH) treated natural fibers from Symphirema involucratum stem. Int J Biol Macromol 186:886–896. https://doi.org/10.1016/j.ijbiomac.2021.07.061
Mylsamy K, Rajendran I (2010) Investigation on physio-chemical and mechanical properties of raw and alkali-treated Agave americana fiber. J Reinf Plast Compos 29:2925–2935. https://doi.org/10.1177/0731684410362817
Kumar AM, Parameshwaran R, Kumar PS et al (2017) Effects of abaca fiber reinforcement on the dynamic mechanical behavior of vinyl ester composites. Mater Test 59:555–562. https://doi.org/10.3139/120.111044
Bhuvaneshwaran M, Subramani SP, Palaniappan SK et al (2021) Natural cellulosic fiber from Coccinia indica stem for polymer composites: extraction and characterization. J Nat Fibers 18:644–652. https://doi.org/10.1080/15440478.2019.1642826
Mylsamy B, Palaniappan SK, Subramani SP et al (2020) Innovative characterization and mechanical properties of natural cellulosic Coccinia indica fiber and its composites. Mater Test 62:61–67. https://doi.org/10.3139/120.111451
Mylsamy B, Palaniappan SK, Pavayee Subramani S et al (2019) Impact of nanoclay on mechanical and structural properties of treated Coccinia indica fibre reinforced epoxy composites. J Mater Res Technol 8:6021–6028. https://doi.org/10.1016/j.jmrt.2019.09.076
Aruchamy K, Pavayee Subramani S, Palaniappan SK et al (2020) Study on mechanical characteristics of woven cotton/bamboo hybrid reinforced composite laminates. J Mater Res Technol 9:718–726. https://doi.org/10.1016/j.jmrt.2019.11.013
Sethuraman B, Subramani SP, Palaniappan SK et al (2020) Experimental investigation on dynamic mechanical and thermal characteristics of Coccinia indica fiber reinforced polyester composites. J Eng Fiber Fabr 15:1–6. https://doi.org/10.1177/1558925020905831
Chinnasamy V, Subramani SP, Palaniappan SK et al (2020) Characterization on thermal properties of glass fiber and kevlar fiber with modified epoxy hybrid composites. J Mater Res Technol 9:3158–3167. https://doi.org/10.1016/j.jmrt.2020.01.061
Mylsamy B, Chinnasamy V, Palaniappan SK et al (2020) Effect of surface treatment on the tribological properties of Coccinia indica cellulosic fiber reinforced polymer composites. J Mater Res Technol 9:16423–16434. https://doi.org/10.1016/j.jmrt.2020.11.100
Aruchamy K, Subramani SP, Palaniappan SK et al (2022) Effect of blend ratio on the thermal comfort characteristics of cotton/bamboo blended fabrics. J Nat Fibers 19:105–114. https://doi.org/10.1080/15440478.2020.1731903
Nagappan S, Subramani SP, Palaniappan SK, Mylsamy B (2021) Impact of alkali treatment and fiber length on mechanical properties of new agro waste Lagenaria siceraria fiber reinforced epoxy composites. J Nat Fibers. https://doi.org/10.1080/15440478.2021.1932681
Rieley JO (1967) The ecology of carex flacca schreb and carex panicea L. Dissertation, Durham University
Vermeer JG (1983) Berendse F (1983) The relationship between nutrient availability, shoot biomass and species richness in grassland and wetland communities. Vegetatio 532(53):121–126. https://doi.org/10.1007/BF00043032
Keskin OY, Dalmis R, Balci Kilic G et al (2020) Extraction and characterization of cellulosic fiber from Centaurea solstitialis for composites. Cellulose 27:9963–9974
Ahvenainen P, Kontro I, Svedström K (2016) Comparison of sample crystallinity determination methods by X-ray diffraction for challenging cellulose I materials. Cellulose 23:1073–1086. https://doi.org/10.1007/s10570-016-0881-6
Sreenivas HT, Krishnamurthy N, Arpitha GR (2020) A comprehensive review on light weight kenaf fiber for automobiles. Int J Light Mater Manuf 3:328–337
Shyam Kumar R, Balasundar P, Al-Dhabi NA et al (2021) A new natural cellulosic pigeon pea (Cajanus cajan) pod fiber characterization for bio-degradable polymeric composites. J Nat Fibers 18:1285–1295. https://doi.org/10.1080/15440478.2019.1689887
Gapsari F, Purnowidodo A, Hidayatullah S, Suteja S (2021) Characterization of Timoho Fiber as a reinforcement in green composite. J Mater Res Technol 13:1305–1315. https://doi.org/10.1016/j.jmrt.2021.05.049
Senthamaraikannan P, Sanjay MR, Bhat KS et al (2019) Characterization of natural cellulosic fiber from bark of Albizia amara. J Nat Fibers 16:1124–1131. https://doi.org/10.1080/15440478.2018.1453432
Hyness NRJ, Vignesh NJ, Senthamaraikannan P et al (2018) Characterization of new natural cellulosic fiber from Heteropogon contortus plant. J Nat Fibers 15:146–153. https://doi.org/10.1080/15440478.2017.1321516
Manimaran P, Saravanan SP, Sanjay MR et al (2020) New lignocellulosic Aristida adscensionis fibers as novel reinforcement for composite materials: extraction, characterization and weibull distribution analysis. J Polym Environ 28:803–811. https://doi.org/10.1007/s10924-019-01640-7
Subramanian SG, Rajkumar R, Ramkumar T (2021) Characterization of natural cellulosic fiber from Cereus hildmannianus. J Nat Fibers 18:343–354. https://doi.org/10.1080/15440478.2019.1623744
Ramkumar R, Saravanan P (2021) Characterization of the cellulose fibers extracted from the bark of Piliostigma racemosa. J Nat Fibers. https://doi.org/10.1080/15440478.2021.1875356
Maepa CE, Jayaramudu J, Okonkwo JO et al (2015) Extraction and characterization of natural cellulose fibers from maize tassel. Int J Polym Anal Charact 20:99–109. https://doi.org/10.1080/1023666X.2014.961118
Seki Y, Köktaş S, Kilinc AC, Dalmis R (2019) Green alternative treatment for cellulosic fibers: ionic liquid modification of Abelmoschus esculentus fibers with methyl-tri-n-butyl ammonium methyl sulphate. Mater Res Express 6:8. https://doi.org/10.1088/2053-1591/AB2015
Seki Y, Kılınç AÇ, Dalmis R et al (2018) Surface modification of new cellulose fiber extracted from Conium maculatum plant: a comparative study. Cellulose 25:3267–3280. https://doi.org/10.1007/S10570-018-1797-0
Uma Maheswari C, Obi Reddy K, Muzenda E et al (2012) Extraction and characterization of cellulose microfibrils from agricultural residue - Cocos nucifera L. Biomass Bioenerg 46:555–563. https://doi.org/10.1016/j.biombioe.2012.06.039
Obi Reddy K, Guduri BR, Rajulu AV (2009) Structural characterization and tensile properties of Borassus fruit fibers. J Appl Polym Sci 114:603–611. https://doi.org/10.1002/app.30584
Mwaikambo LY, Ansell MP (2002) Chemical modification of hemp, sisal, jute, and kapok fibers by alkalization. J Appl Polym Sci 84:2222–2234. https://doi.org/10.1002/app.10460
Sreenivasan VS, Somasundaram S, Ravindran D et al (2011) Microstructural, physico-chemical and mechanical characterisation of Sansevieria cylindrica fibres - an exploratory investigation. Mater Des 32:453–461. https://doi.org/10.1016/j.matdes.2010.06.004
Jonoobi M, Harun J, Shakeri A et al (2009) Chemical composition, crystallinity, and thermal degradation of bleached and unbleached kenaf bast (Hibiscus cannabinus) pulp and nanofibers. BioResources 4:626–639. https://doi.org/10.15376/biores.4.2.626-639
Sgriccia N, Hawley MC, Misra M (2008) Characterization of natural fiber surfaces and natural fiber composites. Compos Part A Appl Sci Manuf 39:1632–1637. https://doi.org/10.1016/j.compositesa.2008.07.007
Dalmis R, Köktaş S, Seki Y, Kılınç AÇ (2020) Characterization of a new natural cellulose based fiber from Hierochloe odarata. Cellulose 27:127–139. https://doi.org/10.1007/s10570-019-02779-1
Fackler K, Stevanic JS, Ters T et al (2011) FT-IR imaging microscopy to localise and characterise simultaneous and selective white-rot decay within spruce wood cells. Holzforschung 65:411–420. https://doi.org/10.1515/HF.2011.048
De Rosa IM, Kenny JM, Puglia D et al (2010) Morphological, thermal and mechanical characterization of okra (Abelmoschus esculentus) fibres as potential reinforcement in polymer composites. Compos Sci Technol 70:116–122. https://doi.org/10.1016/j.compscitech.2009.09.013
De Rosa IM, Kenny JM, Maniruzzaman M et al (2011) Effect of chemical treatments on the mechanical and thermal behaviour of okra (Abelmoschus esculentus) fibres. Compos Sci Technol 71:246–254. https://doi.org/10.1016/j.compscitech.2010.11.023
Alemdar A, Sain M (2008) Isolation and characterization of nanofibers from agricultural residues - wheat straw and soy hulls. Bioresour Technol 99:1664–1671. https://doi.org/10.1016/j.biortech.2007.04.029
Xu F, Yu J, Tesso T et al (2013) Qualitative and quantitative analysis of lignocellulosic biomass using infrared techniques: a mini-review. Appl Energy 104:801–809
Oh SY, Dong IY, Shin Y et al (2005) Crystalline structure analysis of cellulose treated with sodium hydroxide and carbon dioxide by means of X-ray diffraction and FTIR spectroscopy. Carbohydr Res 340:2376–2391. https://doi.org/10.1016/j.carres.2005.08.007
Porras A, Maranon A, Ashcroft IA (2015) Characterization of a novel natural cellulose fabric from Manicaria saccifera palm as possible reinforcement of composite materials. Compos Part B Eng 74:66–73. https://doi.org/10.1016/j.compositesb.2014.12.033
Saha P, Manna S, Chowdhury SR et al (2010) Enhancement of tensile strength of lignocellulosic jute fibers by alkali-steam treatment. Bioresour Technol 101:3182–3187. https://doi.org/10.1016/j.biortech.2009.12.010
Sarikanat M, Seki Y, Sever K, Durmuşkahya C (2014) Determination of properties of Althaea officinalis L. (Marshmallow) fibres as a potential plant fibre in polymeric composite materials. Compos Part B Eng 57:180–186. https://doi.org/10.1016/j.compositesb.2013.09.041
Belaadi A, Amroune S, Seki Y et al (2022) Extraction and characterization of a new lignocellulosic fiber from Yucca treculeana L. leaf as potential reinforcement for industrial biocomposites. J Nat Fibers: 1–16. https://doi.org/10.1080/15440478.2022.2054895
Köktaş S, Keskin ÖY, Dalmiş R et al (2022) Extraction and characterization of natural cellulosic fiber from Taraxacum Sect. Ruderalia. J Nat Fibers. https://doi.org/10.1080/15440478.2022.2064389
Albayrak D, Seki Y, Balcı Kılıç G et al (2022) Exploration of alternative cellulosic natural fiber from the stem of Malva slyvestris. J Nat Fibers. https://doi.org/10.1080/15440478.2022.2073498
Kale RD, Getachew Alemayehu T, Gorade VG (2020) Extraction and characterization of lignocellulosic fibers from Girardinia bullosa (Steudel) Wedd. (Ethiopian Kusha Plant). J Nat Fibers 17:906–920. https://doi.org/10.1080/15440478.2018.1539940
Ridzuan MJM, Abdul Majid MS, Afendi M et al (2016) Characterisation of natural cellulosic fibre from Pennisetum purpureum stem as potential reinforcement of polymer composites. Mater Des 89:839–847. https://doi.org/10.1016/j.matdes.2015.10.052
Saravanakumar SS, Kumaravel A, Nagarajan T et al (2013) Characterization of a novel natural cellulosic fiber from Prosopis juliflora bark. Carbohydr Polym 92:1928–1933. https://doi.org/10.1016/j.carbpol.2012.11.064
Moniruzzaman M, Mahmood H, Yusup S, Akil HM (2017) Exploring ionic liquid assisted pretreatment of lignocellulosic biomass for fabrication of green composite. J Japan Inst Energy 96:376–379. https://doi.org/10.3775/jie.96.376
Baskaran PG, Kathiresan M, Senthamaraikannan P, Saravanakumar SS (2018) Characterization of new natural cellulosic fiber from the bark of Dichrostachys cinerea. J Nat Fibers 15:62–68. https://doi.org/10.1080/15440478.2017.1304314
Yao F, Wu Q, Lei Y et al (2008) Thermal decomposition kinetics of natural fibers: Activation energy with dynamic thermogravimetric analysis. Polym Degrad Stab 93:90–98. https://doi.org/10.1016/j.polymdegradstab.2007.10.012
Balasundar P, Narayanasamy P, Senthamaraikannan P et al (2018) Extraction and characterization of new natural cellulosic Chloris barbata fiber. J Nat Fibers 15:436–444. https://doi.org/10.1080/15440478.2017.1349015
Kathirselvam M, Kumaravel A, Arthanarieswaran VP, Saravanakumar SS (2019) Isolation and characterization of cellulose fibers from Thespesia populnea barks: a study on physicochemical and structural properties. Int J Biol Macromol 129:396–406. https://doi.org/10.1016/j.ijbiomac.2019.02.044
Poletto M, Ornaghi J H L, Zattera AJ (2015) Thermal decomposition of natural fibers: kinetics and degradation mechanisms. In: Reactions and mechanisms in thermal analysis of materials, 1st edn. Wiley, New Jersey, pp 515–545
Fu P, Hu S, Xiang J et al (2012) Study on the gas evolution and char structural change during pyrolysis of cotton stalk. J Anal Appl Pyrolysis 97:130–136. https://doi.org/10.1016/j.jaap.2012.05.012
Khan A, Vijay R, Singaravelu DL et al (2022) Extraction and characterization of natural fibers from Citrullus lanatus climber. J Nat Fibers 19:621–629. https://doi.org/10.1080/15440478.2020.1758281
Vijay R, Manoharan S, Arjun S et al (2021) Characterization of silane-treated and untreated natural fibers from stem of Leucas aspera. J Nat Fibers 18:1957–1973. https://doi.org/10.1080/15440478.2019.1710651
Raja K, Prabu B, Ganeshan P et al (2021) Characterization studies of natural cellulosic fibers extracted from Shwetark stem. J Nat Fibers 18:1934–1945. https://doi.org/10.1080/15440478.2019.1710650
Fan M (2010) Characterization and performance of elementary hemp fibres: factors influencing tensile strength. BioResources 5:2307–2322. https://doi.org/10.15376/biores.5.4.2307-2322
Seki Y, Kılınç AÇ, Dalmış R, Köktaş S (2019) Characterization of flax, jute, and sisal fibers after sodium perborate modification. AATCC J Res 6:25–31. https://doi.org/10.14504/ajr.6.6.4
Karimi S, Tahir PM, Karimi A et al (2014) Kenaf bast cellulosic fibers hierarchy: a comprehensive approach from micro to nano. Carbohydr Polym 101:878–885. https://doi.org/10.1016/j.carbpol.2013.09.106
Liu Y, Hu H (2008) X-ray diffraction study of bamboo fibers treated with NaOH. Fibers Polym 9:735–739. https://doi.org/10.1007/s12221-008-0115-0
Sunny T, Pickering KL, Lim SH (2020) Alkali treatment of hemp fibres for the production of aligned hemp fibre mats for composite reinforcement. Cellulose 27:2569–2582. https://doi.org/10.1007/s10570-019-02939-3
Chiang CL, Yang JM (2015) Flame retardance and thermal stability of polymer/graphene nanosheet oxide composites. In: Fillers and Reinforcements for Advanced Nanocomposites. Woodhead Publishing, pp 253–272
Pandey R, Sinha MK, Dubey A (2018) Cellulosic fibers from Lotus (Nelumbo nucifera) peduncle. J Nat Fibers 17:298–309. https://doi.org/10.1080/15440478.2018.1492486
Seki Y, Sarikanat M, Sever K, Durmuşkahya C (2013) Extraction and properties of Ferula communis (chakshir) fibers as novel reinforcement for composites materials. Compos Part B Eng 44:517–523. https://doi.org/10.1016/j.compositesb.2012.03.013
Tran LQN, Yuan XW, Bhattacharyya D, et al (2015) Fiber-matrix interfacial adhesion in natural fiber composites. In: International Journal of Modern Physics B. World Scientific Publishing Company
Mat Taib R, Ariawan D, Mohd Ishak ZA (2016) Surface characterization of alkali treated kenaf fibers by XPS and AFM. Key Eng Mater 94:29–33. https://doi.org/10.4028/www.scientific.net/KEM.694.29
Seki Y, Sever K, Erden S et al (2012) Characterization of Luffa cylindrica fibers and the effect of water aging on the mechanical properties of its composite with polyester. J Appl Polym Sci 123:2330–2337. https://doi.org/10.1002/app.34744
Maache M, Bezazi A, Amroune S et al (2017) Characterization of a novel natural cellulosic fiber from Juncus effusus L. Carbohydr Polym 171:163–172. https://doi.org/10.1016/j.carbpol.2017.04.096
Alves Fidelis ME, Pereira TVC, Gomes ODFM et al (2013) The effect of fiber morphology on the tensile strength of natural fibers. J Mater Res Technol 2:149–157. https://doi.org/10.1016/j.jmrt.2013.02.003
Amroune S, Bezazi A, Belaadi A et al (2015) Tensile mechanical properties and surface chemical sensitivity of technical fibres from date palm fruit branches (Phoenix dactylifera L.). Compos Part A Appl Sci Manuf 71:95–106. https://doi.org/10.1016/j.compositesa.2014.12.011
Sanjay MR, Madhu P, Jawaid M et al (2018) Characterization and properties of natural fiber polymer composites: a comprehensive review. J Clean Prod 172:566–581
Author information
Authors and Affiliations
Contributions
Ozgur Yasin Keskin: conceptualization, methodology, validation, investigation, writing — original draft, writing — review and editing, and visualization; Serhan Koktas: investigation and writing — review and editing; Yasemin Seki: conceptualization, methodology, validation, investigation, writing — original draft, and writing — review and editing; Ramazan Dalmis: investigation and writing — review and editing; Gonca Balci Kilic: investigation and writing — review and editing; Didem Albayrak: investigation and writing — review and editing.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Keskin, O.Y., Koktas, S., Seki, Y. et al. Natural cellulosic fiber from Carex panicea stem for polymer composites: extraction and characterization. Biomass Conv. Bioref. 14, 13901–13912 (2024). https://doi.org/10.1007/s13399-022-03458-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-03458-1