Abstract
One of the main mechanisms for salt marsh decline across the United States is the inability of the marsh surface to keep pace with sea level rise. The interior platform is especially vulnerable, leading to the encroachment of short form Spartina alterniflora pannes, pool formation, and ultimately runaway pool expansion if recovery is not possible. Coastal ecologists in New England have been implementing a restoration strategy of runnels, or shallow channels, to enhance drainage of oversaturated and ponding interior marshes. In 2015, runnels were constructed to drain two large and expanding pools in the Great Marsh System of Massachusetts, USA. Vegetation, elevation, and hydrology were monitored using field sampling and remote sensing analysis pre- and post-restoration over seven growing seasons to document the trajectory of the pools and adjacent salt marsh platforms. Pool drainage improved reflecting tidal cycles after three years. Substantial colonization of S. alterniflora and S. patens into the previously unvegetated pools required three growing seasons. In the adjacent platform, S. patens and Distichlis spicata increased in abundance with substantial declines in S. alterniflora. The runnel for one pool became blocked by vegetation after three years and inhibited drainage and recovery of the vegetation in the pool yet not the platform. Runnels may be a viable solution for restoring interior marshes following vegetation loss yet substantial improvements in vegetation and hydrology may require 3 – 5 years and complete recovery of the vegetation community in the regularly drained portion of the system for at least a decade.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Historically, salt marshes experienced a host of direct anthropogenic stressors ranging from modification of hydrology through intensive agriculture (Smith et al. 1989; Adamowicz et al. 2020) mosquito ditching and ditch-plugging practices (Meredith et al. 1985; Wolfe 1996; Vincent et al. 2014; Burdick et al. 2020a, b), fill-in for urbanization and infrastructure (Broomberg and Bertness 2005; Gedan et al. 2009), and development at the immediate upland edge (Bozek and Burdick 2005; Pontee 2013). Over the past thirty years, sea level rise (SLR) has been recognized to pose the largest threat to salt marsh systems on the East Coast in the United States (Kirwan and Megonigal 2013). Recent estimates of rapid SLR rates, however, may outpace salt marsh elevation gain (Crosby et al. 2016), which can further disrupt marsh building by increasing plant mortality and decreasing belowground biomass production (Payne et al. 2019). Increased flooding can also be exacerbated by limited sediment supplies (Ganju et al. 2017) and legacy effects of prior agricultural alterations (Mora and Burdick 2013; Adamowicz et al. 2020; Smith et al. 2021). Large loss of coastal wetlands from SLR has been documented across the northeastern US in Virginia (Burns et al. 2021), Maryland (Schepers et al. 2020), New York (Smith et al. 2021) and New England (Watson et al. 2017) as well as the Mississippi Delta (Day et al. 2011).
One of the main pathways for marsh loss to SLR is oversaturation, panne formation, and subsequent creation and expansion of pools on the interior marsh platform (Redfield 1972, Vincent et al. 2013, and Raposa et al. 2017). Compared to low marsh areas immediately along tidal creeks and shorelines, the higher elevation marsh platform is especially vulnerable to deterioration associated with reduced inorganic sediment inputs away from creeks (Temmerman et al. 2003, 2005, and Fagherazzi et al. 2013) and declines of belowground biomass inputs from dominant graminoids caused by increased flooding (Watson et al. 2016; Payne et al. 2019). Oversaturation of the interior marsh platform increases stressors on the vegetation community including anoxia, increased salinity, and build-up of hydrogen sulfide concentrations in the root zone (Van Huissteden and Van De Plassche 1998, Mendelssohn and Morris 2001, Berkowitz et al. 2018, Himmelstein et al. 2021). Over time high marsh graminoids (S. patens, Distichlis spicata, Juncus gerardii) die off and are replaced by more stress-tolerant short-form S. alterniflora and Salicornia spp. or open water (Warren and Niering 1993; Raposa et al. 2017) with less consolidated soils and shallow standing water (Berkowitz et al. 2018; Burdick et al. 2020a, b). Conversion of high marsh meadows to pannes and pools has been identified as a cause of population declines of endemic species such as Ammospiza caudacauta (Saltmarsh Sparrow) as well as A. nelsoni (Nelson’s Sparrow) (Gjerdum et al. 2005, Shriver et al. 2007).
Small pools with stable, vertical banks are natural features on the marsh interior (Redfield 1972; Adamowicz and Roman 2005). Coastal ecologists, however, have recently been documenting large swaths of interior platform being lost to pool expansion, especially at lower elevations and flat slopes (Wilson et al. 2014; Ganju et al. 2020). Continued vegetation die-off and decrease in elevation (e.g., surface erosion, subsidence, and organic matter decomposition) may “push” the platform over a tipping point to peat collapse and pool formation (DeLaune et al. 1994; Day et al. 2011; Chambers et al. 2019). Runaway pool collapse occurs when the inorganic sediment supply is insufficient for the marsh to keep pace with SLR and stressful biogeochemical conditions at pool edges causes further vegetation die-off and bank collapse (Mariotti 2016; Mariotti et al. 2020; Himmelstein et al. 2021). In addition, algal mats generated within pools can blanket and kill edge vegetation (Wasson et al. 2017). The interior marsh can convert to large areas of standing water as smaller pools expand and merge over time (Himmelstein et al. 2021). Although pool recovery has been well-documented after reconnection with a tidal creek (Wilson et al. 2014; Smith and Pellew 2021), coastal ecologists have identified insufficient sediment inputs, microtidal ranges (< 1 m), and increasing tidal prisms as factors that further exacerbate pool expansion after tidal reconnection (Ganju et al. 2017; Schepers et al. 2017, 2020; Vinent et al. 2021).
Coastal ecologists have implemented a restoration strategy, termed runnels, to improve drainage of oversaturated soils without the impacts of over-aeration of the peat soil column (Wigand et al. 2017). Runnels are shallow, often vegetated, swales (20 – 80 cm wide, 20 – 30 cm deep) constructed through areas of standing water or at the edge of pools which connect to the nearest hydrologic channel (ditch or creek). Improvements in drainage across the salt marsh platform is expected to decrease biogeochemical stressors and allow for eventual recolonization of high marsh graminoids. Besterman et al. (2022) documented a pattern of vegetation recovery over seven years after runnel construction in Rhode Island: (1) initial drainage of standing water and exposing bare ground, (2) colonization of S. alterniflora and Salicornia spp., and (3) replacement of low marsh vegetation with high marsh graminoids of S. patens, D. spicata, and J. gerardii. Additionally, Perry et al. (2021) observed revegetation across salt marsh platforms as well as no net increase in peat oxidation after runnel construction. On longer time scales, revegetation and drainage enhancement of the salt marsh surface may lead to gains in elevation.
Runnelling is an additional tool for coastal ecologists and land managers to conserve and improve the resiliency of salt marsh systems. Adapted from Open Marsh Water Management methods to enhance tidal flow of mosquito breeding depressions (Wolfe et al. 2021), runnels have only been recently implemented and monitored for the purpose of rebuilding and improving high marsh vegetation communities. Questions remain about the efficacy across different tidal ranges, sediment inputs, and tidal prisms as well as maintenance requirements over time (Besterman et al. 2022). Recent applications of runnels have focused on recently converted pannes, where elevation and vegetation losses are less severe than marshes converting to pools (see Perry et al. 2021; Besterman et al. 2022). We constructed runnels in Spring 2015 at two large pools in the Parker River National Wildlife Refuge (Newbury, MA), which have been present and relatively stable on the landscape as early as 1965 based on historic aerial imagery (United States Geologic Survey – Earth Explorer). Evidence of recent shoreline vegetation die-off and soil slumping since 2010 spurred concerns of pool expansion and loss of valuable high marsh habitat. Restoration goals for the runnels were (1) reconnect tidal exchange to the pools, (2) arrest pool expansion, (3) passively revegetate interior mudflats of pools, and (4) enhance the surrounding high marsh vegetation community. We monitored hydrology, vegetation, and elevation over seven growing seasons to document the efficacy of the runnels and the trajectory of the pool and adjacent marsh platform recovery. Additionally, we used remote sensing analysis of publicly available aerial imagery to document system-wide vegetation recovery over time.
Methods
Study Site & Study Design
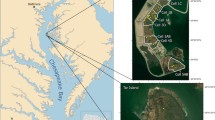
The study site, hereafter referred to as Pine Island Marsh, is located within the Great Marsh system and Parker River National Wildlife Refuge (Newbury, MA) between the Parker River and Pine Island Creek (Fig. 1—Left). Pine Island Marsh is a 191-ha salt marsh platform dissected by three major tidal creeks, numerous smaller order creeks, and artificial drainage ditches (density = 149 m ha−1). The system is mesotidal with a mean tidal range of 2.64 m (NOAA Gauge 8,441,241). The system is considered sediment deficient for long-term salt marsh stability (Cavatorta et al. 2003; Langston et al. 2020) with median suspended sediment concentrations of 15 mg L−1 and approaching 40 mg L−1 near the mouth of the estuary (Hopkinson et al. 2018). The three major rivers of the system (Rowley, Ipswich, and Parker Rivers) are dammed upstream, limiting the sediment supply. Average salt marsh elevation gain rates ranged from 3.5 mm yr−1 in the high marsh platform to 9.5 mm yr−1 in the low marsh compared to recorded SLR rate of 4.8 mm yr−1 (Langston et al. 2020). The vegetation community is comprised of low marsh on tidal creek and ditch banks of tall form S. alterniflora (> 35 cm height), high marsh platform dominated by S. patens, D. spicata, and J. gerardii mixed with pannes of low form S. alterniflora (< 35 cm), Salicornia depressa, and bare soil (Anderson and Treshow 1980; Millette et al. 2010).
North (42.7700, -70.8295) and south pools (42.7631, -70.8231) were selected for restoration using runnels based on recent dieback of halophyte graminoids, buildup of drifted algal mats, shoreline soil slumping, relict agricultural features (e.g., embankments and ditches), and proximity to hydrologic pathways for runnel construction (Fig. 1 – Right). In 2014, one year before runnel construction, the north and south pools had dimensions of 0.60 ha and 3.52 ha with perimeters of 327 m and 1536 m, respectively, and varying nearshore depths of 15 – 20 cm. Runnels approximately 30 cm width and 15 cm depth were constructed in the Spring of 2015 from the edge of the pool to the nearest hydrologic pathway with lengths ranging 60 – 200 cm (Fig. 2). A post driver and angle iron were used to compact the marsh to the appropriate dimensions. One runnel was constructed at the north pool and two at the south pool to compensate for its size and tidal prism (Fig. 3). We cut the runnels to set the pools on a drainage trajectory whereby the dimensions of the runnel would expand to eventually equalize with the pool’s tidal prism.
Left: Creation of runnel by compacting the marsh surface using a handheld tamping tool at the north pool site in 2015. Runnel connected an existing ditch to the expanding pool. Right: Runnel at the south pool site in 2015 after construction. Runnels were created with dimensions of approximately 30 cm width - 15 cm depth and lengths of 60 – 200 cm
Field Sampling
Restoration progress was monitored based on a before-after study design for vegetation and salt marsh surface elevation (Neckles et al. 2002). Pre-restoration data were collected for vegetation in Spring 2015 and follow-up monitoring was completed in Summer 2015, Summer 2016, Fall 2018, and Fall 2021. Elevation was monitored in Spring 2015, Summer 2016, and Fall 2021. Transects parallel to the pool edge were established in Spring of 2015 on four sides of each pool to monitor the recovery of the vegetation and elevation following runnel construction (Fig. 3). Five pairs of permanent vegetation plots were randomly located along each 30 m transect with at least 3 m between plots. Each location consisted of a pair of 0.5 m2 plots with one on the salt marsh platform edge and the second immediately adjacent in the pool (n = 40 per pool site)
.
All vascular and non-vascular species as well as abiotic categories (e.g., bare mud, dead vegetation, desiccated algal mat, etc.) were identified and estimated for visual cover to the nearest 1% by ocular method within two hours of low tide (Neckles et al. 2002). Species were classified as halophytes or submerged aquatic vegetation (i.e., Ruppia maritima) according to Tiner (2009). To better understand the encroachment of S. alterniflora in the high marsh zone, the High Marsh Health Ratio (HMH ratio) was calculated as:
where dominant high marsh graminoids is the aggregate cover of S. patens, D. spicata, and J. gerardii. The ratio ranges from zero to one, where values closer to one indicate a healthy high marsh dominated by high marsh graminoids. Values closer to zero indicate salt marsh platforms converting to low marsh or S. alterniflora pannes.
Surface elevations of the salt marsh platform and pool plots were monitored to estimate how runnel drainage would affect elevation change rates. In 2015, the relative elevation of the center of the vegetation plots was measured with a laser level (± 0.5 cm accuracy; Johnson Rotary Laser Level, Burnsville, MN). In 2016 and 2021, true plot elevations (NAVD88 m) were measured with an RTK-GPS (± 3.0 cm accuracy; Leia GSSN Rover model GS14, St. Gallen, Switzerland). The difference in elevation between the salt marsh platform and pool plot was calculated for each pair for all three years.
Hydrology was monitored post-restoration (June – North & September—South 2015, July 2016, October 2018, and October 2021) in the runnelled pools and adjacent natural hydrologic channels, which served to indicate the effectiveness of the hydrologic path. Odyssey capacitance water level loggers (2015 – 2018; Christchurch, New Zealand) and Hobo pressure transducers (2021; Onset, Inc., Bourne, MA) recorded every 10 min for one lunar tidal cycle (~ 30 days). Water level recorders were stored in PVC casings with 6.4 mm holes and attached to stakes with clamps. Stakes were driven into the sediment until the PVC casing touched the sediment surface. In 2021, water level elevations for pools were measured by placing water level recorders in perforated PVC tubes (6.4 mm holes every 10 cm for 0.6 m) set 60 cm below ground in the pools to monitor groundwater levels (Roman et al. 2001). Landscaping fabric was wrapped three times around perforations of PVC tubes to prevent buildup of sediment around HOBO loggers. An additional pressure transducer was attached to a nearby tree to compensate for atmospheric pressure. One logger was deployed 3 – 5 m within each pool to monitor pool hydrology. A second logger was placed in a nearby channel, located 15 – 50 m downstream of each runnel. Due to the clogged runnel, the channel water level recorder for the north pool was moved to Jericho Creek in 2021 (110 m from the pool). Elevations of water level recorders were measured with an RTK-GPS (NAVD88 m).
Statistical Analysis
Water level recorder and atmospheric pressure data were processed in Odyssey or Hoboware software. The average high tide, low tide, higher high tide, maximum water elevation, and flooding duration for the salt marsh platform and pool elevations was calculated for each water level recorder with the VulnToolKit package in R (R Core Team 2013, Hill and Anisfield 2021). The flooding duration metric, calculated as the percent of time of the monitoring period when tidal waters were greater than the elevation of the pool or marsh platform, serves as a proxy of how quickly the system can drain tidal waters after high tides. The average elevation of all platform and pool plots for each pool and season were used for the flooding duration analysis. The 2016 elevations were used for the 2015 and 2018 water level analyses since the true elevation of the plots were not recorded.
To assess broader community shifts in the vegetation community, non-metric dimensional scaling (NMDS) ordination was conducted for each pool separately (Bray–Curtis dissimilarity, square-root transformation). Rare species (< 5% occurrence) were removed prior to multivariate analysis. Species that were highly and significantly correlated (r2 > 20%, p < 0.05) were overlain on the ordination. Pairwise analysis of similarity (ANOSIM; Bray–Curtis dissimilarity) and Similarity Percentages (SIMPER) was conducted on the vegetation community to observe the trajectory of the platform and pool habitats (McCune and Grace 2002). ANOSIM, a multi-variate technique to compare a control group to treatments, determines whether the vegetation community shifted after runnel construction. ANOSIM provides a measure of dissimilarity, Global R, on a scale of 0 – 1, where zero is no dissimilarity, and statistical comparison between a control (Spring 2015) and treatments (Fall 2015 – 2021). The SIMPER analysis provides context to the ANOSIM by quantifying the species driving the dissimilarity between pre- and post-restoration comparisons. Cover types of similar functional groups were aggregated: (1) Bare, dead, desiccated algal mat, wrack, and Vaucheria algae as ‘non-live cover’, (2) Floating green algae and Fucus sp. as ‘algae’. Multivariate analyses were conducted with the vegan package (Oksanen et al. 2020) and data visualization with ggplot2 package (Wickham 2016).
Remote Sensing of Vegetation Recovery
The proportion of vegetated marsh surface before and after runnel construction was measured using publicly available aerial imagery of the National Agriculture Imagery Program (NAIP) through the United States Department of Agriculture of 2012—2018. Aerial imagery (RBG – Infrared) was acquired in the summer and fall (July – October) with resolutions of 1 m (2012, 2014) improving to 0.6 m (2016, 2018, 2021). Vegetated and non-vegetated areas (i.e., water, wrack, bare mud, floating algae, and roads) were classified from the normalized difference vegetation index (NDVI) across the Pine Island Marsh of Plum Island Estuary (see Fig. 1) through pixel classification with the Classification Wizard Tool in ArcGIS Pro (Esri, Redlands, CA). The outline of each pool was manually drawn based on 2012 imagery, and a 5 m buffer was created around the pool outline to capture potential pool expansion. The vegetated and non-vegetated surface areas were tabulated from classified pixels within each pool outline. Spatial accuracy assessments were conducted for each year across the Pine Island Marsh to verify the vegetation classification methods. Stratified random points (n = 50) were visually ground-truthed based on NAIP, Google Earth, and Leaf-off imagery. The User Accuracy, Producer Accuracy, and Kappa Index of Agreement were calculated to understand the accuracy of the vegetated – unvegetated classification (Congalton 1991).
Results
Hydrology
The efficacy of the runnels to drain both pools after high tides improved during the first three years post-restoration and diverged after 2018 (Table 1). Although pre-restoration monitoring was not completed for hydrology, it was assumed that the pools remained flooded permanently based on field observation and lack of outlet. The percent flood duration time, a proxy for the ability of the pool to drain after high tides, declined by 25% and 76% for the north and south pools, respectively, by 2018. The timing and high tide elevation of the tides between the channels and both pools converged between 2015 – 2018. While pools still took longer to drain than channels, lags in drainage diminished over time (Fig. 4). The runnel for the north pool became clogged with vigorous S. alterniflora growth and sediment sometime after 2018. Drainage through the north runnel had decreased and lagged substantially after high tides in 2021, creating permanent flooded conditions in the pool. The surrounding marsh platform continued to benefit, however, as the flooding duration decreased from 10% of the monitoring period in 2015 to 5% in 2021. In the south pool, flooding duration continued to decline to 13% by 2021 and saturation of the marsh platform remained relatively low and unchanged by the end of the monitoring period.
Water level elevations for hydrologic channel (blue) and pool (orange) water level recorders in Summer/Fall 2015 (after runnel construction), Summer 2016, Fall 2018, and Fall 2021. The north pool is shown on left and south pool on the right. The average salt marsh platform elevation (black dashed line) and pool elevation (green dashed line) are shown for each site
Vegetation
The vegetation communities of the pools and platforms were set on a trajectory of recovery and improved health and required 3 – 4 years to differentiate from pre-restoration conditions (Fig. 5, Online Resource 1). In the pool habitat, percent cover of S. alterniflora cover increased by 31% north and 37% south and high marsh graminoid cover, the aggregate of S. patens, D. spicata, and J. gerardii, increased 7% north and 27% south while the drained bare areas declined by 21% north and 63% south from pre-restoration conditions to 2021. S. alterniflora cover peaked in Fall 2018 for the north pool and subsequently was reduced by 39% in 2021 as non-live cover increased by 21%. In the south pool habitat, S. alterniflora cover declined by half between 2018 – 2021 and was predominantly replaced by high marsh graminoid cover. The non-live cover continued to decline in the same timeframe, resulting in a total loss of 64% since restoration.
Two-dimensional NMDS ordinations found divergent recoveries of the vegetation community between the north (stress = 7.2) and south pools (stress = 12.8; Fig. 6). The low stress values (< 15) suggest the NMDS ordinations are useful at interpreting and understanding the vegetation community (McCune and Grace 2002). At the north pool site, the pool plots remained relatively stagnant over time, associated with non-live (r2 = 0.90; correlation regression with NMDS) and S. alterniflora cover (r2 = 0.92). Dissimilarity between pre-restoration conditions increased through 2018 (R = 0.501, p < 0.001), although the vegetation community partially reverted in 2021 (R = 0.199; Table 2). At the south pool site (Fig. 6), the pool plots progressed towards a vegetation community with greater S. patens cover (r2 = 0.71) and less non-live cover (r2 = 0.87), resembling the vegetation of the platform plots in 2016. The vegetation community only required two full growing seasons to differentiate from Spring 2015 conditions (R = 0.147, p = 0.003) and progressed towards high marsh habitat through 2021 (R = 0.939). SIMPER analyses indicated that S. alterniflora and non-live cover drove dissimilarity in the pools over time (Table 3), indicating the vegetation community mainly improved through the colonization of the low marsh grass.
Non-metric multidimensional scaling ordination (NMDS) north pool (Top) and south pool (Bottom) for the mean locations of each habitat and season. Vegetation species that were highly and significantly correlated (r2 > 20%, p < 0.05) are overlain in main NMDSs. Species abbreviations are DISPI = Distichlis spicata, SPALT = Spartina alterniflora, SPPAT = S. patens, NONLIVE – VAUCH = Non-live Cover
The adjacent platforms also trended toward a less saturated environment with the recovery of high marsh graminoids and declines of S. alterniflora. The percent cover of low marsh grass initially increased and peaked in 2016 to 24% north and 22% south and then subsequently declined to 8% north, and 7% south in 2021. In contrast, high marsh graminoid cover continually increased throughout monitoring (+ 35% north, + 49% south). The HMH ratio reflected the similar trajectories with large increases at both sites over time (+ 0.27 north, + 0.21 south), illustrating the simultaneous gains in high marsh graminoid cover and losses of S. alterniflora. At both sites, non-live cover declined through 2018 and increased slightly as S. alterniflora declined and high marsh graminoids were yet unable to fully colonize all of the bare patches. Plot-level richness of the vegetation community increased in both habitats over time with noticeable increases of high marsh forbs on the salt marsh platform including Agalinis maritima, Limonium nashii, Plantago maritima, and Solidago sempervirens.
Multivariate analyses of the platform plots confirmed convergent recoveries of high marsh habitat. In the ordinations, both sites progressed towards a vegetation community with higher S. patens cover (r2 = 0.95 north) and less S. alterniflora (r2 = 0.84 south). The platforms of both sites required at least three years to differentiate from pre-restoration conditions (R = 0.289, p < 0.001 north; R = 0.406, p < 0.001 south). The contribution of S. alterniflora declined after 2016 and was replaced by S. patens as the largest contributor of dissimilarity, demonstrating the continued increase in high marsh grasses of S. patens and D. spicata as the driver of the recoveries.
Elevation
Elevations of each habitat zone across the pool sites remained relatively unchanged and within the accuracy of the RTK-GPS (± 3.0 cm) after runnel construction (Table 4). The difference in elevation between the pool and immediate platform increased by 3 cm at the north site and decreased by 1.5 cm at the south site. At the north site, the divergence of elevations was driven by elevation gains in the platform and subsidence in the pool plots. The small convergence of elevations at the south site was the result of elevation loss for both the platform and pool.
Remote Sensing of Vegetation Recovery
The remote sensing analysis provided a site-level understanding of the recovery of the vegetation community as the vegetation expanded beyond the scope of the pool edge monitoring. Spatial accuracy assessments suggested accurate vegetated – unvegetated classification of the marsh surface with Kappa Coefficient values ranging 0.81 – 0.88 (Online Resource 2). Pool expansion and waterlogging of surrounding marsh platform was ongoing before runnel construction with decreases of 2% (0.003 ha) and 12% (0.12 ha) of vegetated marsh area in the north and south pools, respectively (Fig. 7, Table 5). In the south pool, vegetation gains were restricted along the perimeter of the pool between 2015 – 2018 resulting in gains of 42% (0.37 ha) of vegetated area since 2014. Between 2018 – 2021, however, vegetation greatly expanded into the interior of the pool, resulting in total gains of 124% (1.10 ha) since 2014. In the north pool, vegetation expansion was minor since restoration with an increase of 13% (0.02 ha). Revegetation was consistent along the entire pool perimeter including the emergence of several patches within the southern pool interior by 2021. It should be noted that two non-hydrologically connected pools just north of the north pool continued to expand and these unvegetated areas were measured within the 5 m buffer, interfering with the estimate of vegetation recovery.
Remote sensing analysis of south (Top) and north pools (Bottom). Runnel construction took place in Spring 2015. National Agriculture Imagery Program aerial imagery of 2018 is background for all panels. Vegetated areas are shown in green and unvegetated areas in black. The 5 m buffer of the pool outline is shown in yellow
Discussion
Connection of Pools to Hydrologic Pathways
Despite the limited hydrologic data (1 month), which can yield highly variable results year-to-year, the runnels altered existing pool hydrology through reconnection with tidal channels and enhanced drainage of the surrounding platform marsh. Between 2015 – 2018, runnels at both sites met restoration goals of improving drainage especially considering the pools were permanently flooded before construction. Each system required at least 2 – 3 growing seasons to reflect similar timing and elevation of high tides as well as drainage based on lagging water level elevations following each high tide in 2016. An immediate improvement of hydrology is well-documented for landscape-scale salt marsh restoration projects that manipulate tidal hydrology (Buchsbaum et al. 2006; Smith et al. 2009; Raposa et al. 2019), however, long-term monitoring (> 5 years) of water elevations provides additional insight into the gradual recovery of the tidal cycle unique to drained pool systems (see Smith et al. 2009; Karberg et al. 2018). Continued declines in flood duration at the south pool through six years post-restoration, continue to support gradual recovery of tidal cycles. It should be noted that the peak of the 18.6-year metonic cycle coincided with the beginning of this study, but the slightly lower higher tides (5 – 10 cm) expected following the peak through 2024 are unlikely to be responsible for the increased drainage observed following runnel installation (Chambers et al. 2003).
Hydrologic gains were more evident in the south pool than in the north pool, especially during the 2018 – 2021 monitoring period. Contrasting runnel and hydrology trajectories between the south and north pools after 2018 impacted the recoveries of the vegetation community. Enhanced drainage and tidal flushing, especially 5 – 10 cm belowground, can ameliorate biogeochemical stress on the vegetation community including reducing salinity, sulfides, and anoxia (Flynn et al. 1999). The inability of the system to fully drain each tidal cycle restricts the colonization of S. alterniflora to the outer edges of the pool and may prevent high marsh graminoids from becoming dominant on the adjacent marsh platform. The salt marsh platform of the north site continued to see improvements in drainage in 2021 (Table 1), despite less drainage in the pool, suggesting the runnels, though partially clogged, still improved biogeochemical conditions of the adjacent marsh platform to facilitate high marsh graminoid increases.
Recovery of the Vegetation Community
Runnels facilitated significant, yet divergent, recoveries of the vegetation community of the pool and adjacent high marsh platform. Restoration goals of improving growing conditions of salt marsh grasses were achieved on the marsh platform and at pool edges of the south pool and partially achieved in the north. Besterman et al. (2022) outlined the recovery of the vegetation community from runnels in three phases: drainage of standing water and increase in bare peat, establishment of early colonizers like S. alterniflora and Salicornia spp., and incremental increase in high marsh graminoids species. The south pool followed the recovery model well as it underwent loss of standing water, increase of S. alterniflora in 2015 – 2018 followed by replacement of high marsh graminoids in 2021. In the north pool, S. alterniflora had variable increases in 2015 – 2018 followed by a decline in 2021, attributed to the runnel becoming clogged.
The vegetation community of marsh platforms had an initial minor peak in S. alterniflora within two growing seasons followed by the replacement of high marsh graminoids as observed by Perry et al. (2021). Despite limited drainage in the north pool by 2021, the marsh platform had similar high marsh graminoids cover and HMH ratio to the south site. Drainage may have beneficially impacted the marsh platform as much as 5 m away from the pool perimeter (Raposa et al. 2019) without increased rates of decomposition and subsidence (Perry et al. 2021). Remote sensing analysis and field visits confirmed this observation with the revegetation of pannes around the edges of the south pool and expansion of vegetation along the entire perimeters of both pools (Fig. 5). Vegetation classification from aerial imagery or field plots extending further into the marsh platform should be considered in future monitoring plans for evaluating pool drainage as monitoring pool edges cannot capture the full extent of the recovery (see Ganju et al. 2017; Wasson et al. 2019).
Trajectory of Pool Recovery
The main restoration goal typically associated with the use of runnels is to “push” the expanding pool or oversaturated area back into the negative feedback loop where vegetation on the marsh platform maintains itself in a dynamic balance (Burdick and Roman 2012). Ideally, connecting the pools to tidal exchange will set the system on a long-term trajectory to recover elevation loss and sustain a high marsh vegetation community. Previous drainage studies (Raposa 2008; Raposa et al. 2019; Perry et al. 2021; Besterman et al. 2022) have documented improvement of tidal exchange in oversaturated areas within years and gradual recolonization by halophyte vegetation over 4 – 7 growing seasons. A lag of 1 – 2 growing seasons after restoration activities was noted for substantial S. alterniflora growth in the pools, which was likely driven by incremental amelioration of soil and porewater chemistry (Raposa 2008; Smith et al. 2009). Recovery timeframes observed in runnel projects have also been documented in other tidal restoration efforts (i.e., culvert replacements, impoundment removals, and living shorelines) with immediate recoveries of hydrology and porewater chemistry and gradual recovery of vegetation within 5 – 10 years (Konisky et al. 2006; Smith and Warren 2012; Bilkovic and Mitchell 2017).
A crucial question that remains about runnel projects is the ability of the pool to develop a dendritic hydrological pattern and build in elevation fast enough to close the gap and become similar to the marsh platform. Continued gains of HMGs throughout the drained pool depends on elevations gains in the long-term. In this study, the difference in pool edge and platform elevation at the south site would require at least 70 years based on the observed elevation gain rate of 2.1 mm yr−1 from 2015 to 2021. Through surface elevation table (SET) monitoring and modeling, revegetating pools in New England had increased accretion rates of 2 – 8 mm yr−1 greater than the adjacent marsh interior (Wilson et al. 2014; Mariotti et al. 2020). Historic aerial photography has shown that complete pool recovery requires multi-decadal timeframes (Wilson et al. 2009, 2014; Smith and Pellew 2021). Long-term field monitoring of surface elevation (via surveying and/or SETs) at future drainage projects should be conducted to better understand pool recovery trajectories (Mariotti 2016).
Management Considerations of Pool Drainage
Runnelling appears to be an effective tool for drainage of large, ponded areas resulting from historic marsh manipulations but should not be widely applied to all pools on the landscape. Pools are natural features of stable interior marshes and provide nesting, foraging, and refugia habitat for nekton and avian species (Adamowicz and Roman 2005; Raposa and Roman 2006; Smith and Niles 2016). Through interpretation of stratigraphic coring and historic aerial photography, coastal ecologists better understand that pools undergo natural decadal cycles of birth, expansion, merging, and recovery (Wilson et al. 2009, 2010, Smith and Pewell 2021). Isolated pool features on the marsh can be relatively stable and surrounding platform marsh can maintain elevation with current rates of SLR and adequate sediment supply (Mariotti 2016).
However, the very large pools that developed in the twentieth century following abandonment of embankments and ditches are unnatural legacies of past agricultural practices (Adamowicz et al. 2020). Rather than allow these pools to expand and degrade the underlying peat for decades (with losses in marsh elevation and stored carbon), runnels can be used to drain shallower portions and revegetate, rehabilitating the marsh platform. Pools with unstable, non-vertical shorelines (e.g., evidence of soil slumping and calve-off events), depths of less than 20 cm (Adamowicz and Roman 2005), shifts of the surrounding vegetation community from high marsh meadows to pannes, and buildup of algal mats on the shoreline (Pethick 1974; Wasson et al. 2017) could be candidates for runnel application. Additionally, future projects should include a long-term monitoring program that reflects project goals to gauge performance. Hydrology, vegetation (field and remote sensing), surface elevation, and avian monitoring are common metrics that may be used to evaluate the trajectory of the pool recovery (see Neckles et al. 2002; Ganju et al. 2017; Adamowicz et al. 2020). During post-restoration monitoring, maintenance of runnels or site-specific adaptive management should be considered to ensure hydrology and vegetation gains are not lost as in the case of north pool (see Knight et al. 2021).
Conclusions
As of six years post-restoration, project goals of tidal exchange reconnection, vegetation establishment in the pool interior, and improvement of surrounding marsh habitat were achieved in the south and partially met in the north site. Runnelling holds promise to be a valuable restoration tool for coastal managers and ecologists to prevent runaway marsh collapse and pool expansion in the marsh interior and improve the resilience of the surrounding high marsh. Although tidal exchange and drainage are achieved relatively quickly, the vegetation community will require at least several years for substantial colonization of S. alterniflora and high marsh graminoids into the pool edge. Complete pool recovery to high marsh with small, interspersed pools may take several decades as the elevation of the basins reaches parity with the marsh platform (Wilson et al. 2014), though increasing rates of SLR (> 5 mm yr−1) may prevent complete recovery. Although not an immediate or permanent fix to stem marsh degradation from the interaction of SLR with relict agricultural features, runnelling has the potential to stimulate the recovery of valuable high marsh habitats from large and expanding unvegetated pools. Such efforts may increase resiliency for decades or at least until new high marsh is formed from upland migration (Besterman et al. 2022).
Data Availability
The datasets generated during the current study are available in the Figshare Repository ‘Pine Island Runnel, Great Marsh, Massachusetts 2014 – 2021 Dataset’, https://doi.org/10.6084/m9.figshare.19666242. The dataset includes vegetation community, hydrology, surface elevation, plot locations, and geospatial analysis results as well as R-script for water level elevation and multi-variate analyses. Gespatial datasets including shapefiles, spatial accuracy assessments, and vegetation classification rasters are available upon request.
References
Adamowicz SC, Roman CT (2005) New England salt marsh pools: A quantitative analysis of geomorphic and geographic features. Wetlands 25:279–288
Adamowicz SC, Wilson G, Burdick DM, Ferguson W, Hopping R (2020) Farmers in the marsh: Lessons from history and case studies for the future. Wetland Sci Practice 2020:183–195
Anderson CM, Treshow M (1980) A review of environmental and genetic factors that affect height in Spartina alterniflora Loisel. (Salt marsh cord grass). Estuaries 3:168–176
Berkowitz JF, VanZomeren CM, Piercy CD, White JR (2018) Evaluation of coastal wetland soil properties in a degrading marsh. Estuarine Coast Shelf Sci 212:311–317
Besterman AF, Jakuba RW, Ferguson W, Brennan D, Costa JE, Deegan LA (2022) Buying time with runnels: A climate adaption tool for salt marshes. Estuaries and Coasts. https://doi.org/10.1007/s12237-021-01028-8
Bilkovic DM, Mitchell MM (2017) Designing Living Shoreline Salt Marsh Ecosystems to Promote Coastal Resilience. In: Bilkovic DM, Mitchell MM, La Peyre MK, Toft JD (eds) Living Shorelines: The Science and Management of Nature-Based Coastal Protection. CRC Press, Boca Raton, LA, pp 293–316
Bozek CM, Burdick DM (2005) Impacts of seawalls on saltmarsh plant communities in the Great Bay Estuary, New Hampshire USA. Wetlands Ecol Manage 12:553–568
Broomberg KD, Bertness MD (2005) Reconstructing New England salt marsh losses using historical maps. Estuaries and Coasts 28:823–832
Buchsbaum RN, Catena J, Hutchins E, James-Pirri MJ (2006) Changes in salt marsh vegetation, Phragmites australis, and nekton in response to increased tidal flushing in a New England salt marsh. Wetlands 26:544–557
Burdick DM, Moore GE, Adamowicz SC, Wilson GM, Peter CR (2020a) Mitigating the legacy effects of ditching in a New England salt marsh. Estuaries and Coasts 43:1672–1679
Burdick DM, Peter CR, Fischella B, Tyrrell M, Allen J, Mora J, Raposa K, Goldstein J, Feurt C, Crane L (2020b) Synthesizing NERR sentinel site data to improve coastal wetland management across New England data report. NEERS Science Collaborative. https://nerrssciencecollaborative.org/media/resources/Burdick_DataReport.pdf. Accessed 6 Jan 2023
Burdick DM, Roman CT (2012) Salt Marsh Response to Tidal Restriction and Restoration: A Summary of Experiences. In: Roman CT, Burdick DM (eds) Tidal Marsh Restoration: A Synthesis of Science and Management. Island Press, Washington DC, pp 373–383
Burns CJ, Alber M, Alexander CR (2021) Historical changes in the vegetated area of salt marshes. Estuaries and Coasts 44:162–177
Cavatorta JR, Johnston M, Hopkinson C, Valentine V (2003) Patterns of sedimentation in a salt marsh-dominated estuary. Biol Bull 205:239–241
Chambers LG, Osgood DT, Bart DJ, Montalto F (2003) Phragmites australis Invasion and Expansion in Tidal Wetlands: Interactions among Salinity, Sulfide, and Hydrology. Estuaries and Coasts 26:398–406
Chambers LG, Steinmuller HE, Breithaupt JL (2019) Toward a mechanistic understanding of “peat collapse” and its potential contribution to coastal wetland loss. Ecology. https://doi.org/10.1002/ecy.2720
Congalton RG (1991) A review of assessing the accuracy of classifications of remotely sensed data. Remote Sens Environ 37:35–46
Crosby SC, Sax DF, Palmer ME, Booth HS, Deegan LA, Bertness MD, Leslie HM (2016) Salt marsh persistence is threatened by predicted sea-level rise. Estuarine Coast Shelf Sci 181:93–99
Day JW, Kemp GP, Reed DJ, Cahoon DR, Boumans RM, Suhayda JM, Gambrell R (2011) Vegetation death and rapid loss of surface elevation in two contrasting Mississippi delta salt marshes: the role of sedimentation, autocompaction and sea-level rise. Ecol Eng 37:229–240
DeLaune RD, Nyman JA, Patrick WH Jr (1994) Peat collapse, ponding and wetland loss in a rapidly submerging coastal marsh. J Coast Res 10:1021–1030
Ewanhuck PJ, Bertness MD (2004) Structure and organization of a northern New England salt marsh plant community. Ecology 92:72–85
Fagherazzi S, Wiberg PL, Temmerman S, Struyf E, Zhao Y, Raymond PA (2013) Fluxes of water, sediments, and biogeochemical compounds in salt marshes. Ecol Process. https://doi.org/10.1186/2192-1709-2-3
Flynn KM, Mendelssohn IA, Wilsey BJ (1999) The effect of water level management on the soils and vegetation of two coastal Louisiana marshes. Wetlands Ecol Manage 7:193–218
Ganju NK, Defne Z, Fagherazzi S (2020) Are elevation and open-water conversion of salt marshes connected? Geophys Res Lett. https://doi.org/10.1029/2019GL086703
Ganju NK, Defne Z, Kirwan ML, Fagherazzi S, D’Alpaos A, Carniello L (2017) Spatially integrative metrics reveal hidden vulnerability of microtidal salt marshes. Nature Communications. https://doi.org/10.1038/ncomms14156
Gedan KB, Silliman BR, Bertness MD (2009) Centuries of human-driven change in salt marsh ecosystems. Ann Rev Marine Sci 1:117–141
Gehman AM, McLenaghan NA, Byers JE, Alexander CR, Pennings SC, Alber M (2018) Effects of small-scale armoring and residential development on the salt marsh-upland ecotone. Estuaries and Coasts 41:54–67
Gjerdrum C, Elphick DS, Rubega M (2005) Nest site selection and nesting success in saltmarsh breeding sparrows: The importance of nest habitat, timing, and study site differences. The Condor 107:849–862
Hill TD, Anisfield SC (2021) VulnToolkit: Analysis of tidal datasets. https://CRAN.R-project.org/package=VulnToolkit. Acceessed 6 Jan 2023
Himmelstein J, Vinent OD, Temmerman S, Kirwan ML (2021) Mechanisms of pond expansion in a rapidly submerging marsh. Front Marine Sci. https://doi.org/10.3389/fmars.2021.704768
Hopkinson CS, Morris JT, Wollheim WM, Raymond PA (2018) lateral marsh edge erosion as a source of sediments for vertical marsh accretion. J Geophys Res: Biogeosciences 123:2444–2465
Karberg JM, Beattie KC, O’Dell DI, Omand KA (2018) Tidal hydrology and salinity drives salt marsh vegetation restoration and Phragmites australis control in New England. Wetlands 38:993–1003
Kirwan ML, Megonigal JP (2013) Tidal wetland stability in the face of human impacts and sea-level rise. Nature 504:53–60
Knight JM, Marx SK, Dale PER (2021) Assessment of runnelling as a form of mosquito control in saltmarsh: Efficacy environmental impacts and management. Wetlands Ecol Manage 30:1109–1127
Konisky RA, Burdick DM, Dionne M, Neckles HA (2006) a regional assessment of salt marsh restoration and monitoring in the Gulf of Maine. Restor Ecol 14:516–525
Langston AK, Vinent OD, Herbert ER, Kirwan ML (2020) Modeling long-term salt marsh response to sea level rise in the sediment-deficient Plum Island Estuary, MA. Limnol Oceanogr 65:2142–2157
Mariotti G (2016) Revisiting salt marsh resilience to sea level rise: Are ponds responsible for permanent land loss? J Geophys Res: Earth Surface 121:1391–1407
Mariotti G, Spivak AC, Luk SY, Ceccherini G, Tyrrell M, Eagle Gonneea M (2020) Modeling the spatial dynamics of marsh ponds in New England salt marshes. Geomorphology. https://doi.org/10.1016/j.geomorph.2020.107262
McCune B, Grace JB (2002) Analysis of Ecological Communities. MjM Software Design, Gleneden Beach, Oregon
Mendelssohn IA, Morris JT (2001) Eco-physiological controls on the productivity of Spartina alterniflora Loisel. In: Winstein MP, Kreeger DA (eds) Concepts and Controversies in Tidal Marsh Ecology. Kluwer Academic Publishers, Boston, MA, pp 59–80
Meredith WH, Saveikis DE, Stachecki CJ (1985) Guidelines for “open marsh water management” in Delaware’s salt marshes – objectives, system designs, and installation procedures. Wetlands 5:119–133
Millette TL, Argow BA, Marcano E, Hayward C, Hopkinson CS, Valentine V (2010) Salt marsh geomorphological analyses via integration of multitemporal multispectral remote sensing with LIDAR and GIS. J Coast Res 26:809–816
Mitsch WJ, Gosselink JG (2015) Wetlands, 5th edn. John Wiley and Sons, Inc., Hoboken, New Jersey
Mora JW, Burdick DM (2013) The impact of man-made earthen barriers on the physical structure of New England tidal marshes (USA). Wetlands Ecol Manage 21:387–398
Neckles HA, Dionne M, Burdick DM, Roman CT, Buchsbaum R, Hutchins E (2002) A monitoring protocol to assess tidal restoration of salt marshes on local and regional scales. Restor Ecol 10:556–563
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O'Hara RB, Simpson GL, Solymos P, Henry M, Stevens H, Szoecs E, Wagner H (2020) vegan: Community Ecology Package. https://CRAN.R-project.org/package=vegan. Accessed 6 Jan 2023
Payne AR, Burdick DM, Moore GE (2019) Potential effects of sea-level rise on salt marsh elevation dynamics in a New Hampshire estuary. Estuaries and Coasts 42:1405–1418
Perry DC, Ferguson W, Thornber CS (2021) Salt marsh climate change adaptation: Using runnels to adapt to accelerating New England salt marsh. Restor Ecol. https://doi.org/10.1111/rec.13466
Pethick JS (1974) The distribution of salt pans on tidal salt marshes. J Biogeog 1:57–62
Pontee N (2013) Defining coastal squeeze: A discussion. Ocean Coast Manage 84:204–207
Qi M, MacGregor J, Gedan K (2021) Biogeomorphic patterns emerge with pond expansion in deteriorating marshes affected by relative sea level rise. Liminol Oceanog 66:1036–1049
R Core Team (2022) R: A language and environment for statistical computing. Vienna, Austria. http://www.r-project.org/. Accessed 6 Jan 2023
Raposa K (2008) Early Ecological Responses to Hydrologic Restoration of a Tidal Pond and Salt Marsh Complex in Narragansett Bay, Rhode Island. J Coast Res 55:180–192
Raposa K, Roman CT (2006) Seasonal habitat-use patterns of nekton in a tide-restricted and unrestricted New England salt marsh. Wetlands 21:451–461
Raposa KB, Weber RL, Ekberg MC, Ferguson W (2017) Vegetation dynamics in Rhode Island salt marshes during a period of accelerating sea level rise and extreme sea level events. Estuaries and Coasts 40:640–650
Raposa KB, Weber RL, Ferguson W, Hollister J, Rozsa R, Maher N, Gettman A (2019) Drainage enhancement effects on a waterlogged Rhode Island (USA) salt marsh. Estuarine Coast Shelf Sci. https://doi.org/10.1016/j.ecss.2019.106435
Redfield AC (1972) Development of a New England salt marsh. Ecol Monogr 42:201–237
Roman CT, James-Pirri MJ, Heltshe JG (2001) Monitoring salt marsh vegetation: A protocol for the long-term coastal ecosystem monitoring program at Cape Cod National Seashore. National Park Service Inventory and Monitoring, Department of the Interior. https://irma.nps.gov/DataStore/DownloadFile/147528. Accessed 6 Jan 2023
Schepers L, Brennand P, Kirwan ML, Guntenspergen GR, Temmerman S (2020) Coastal marsh degradation into ponds induces irreversible elevation loss to sea level in a microtidal system. Geophys Res Lett. https://doi.org/10.1029/2020GL089121
Schepers L, Kirwan ML, Guntenspergen GR, Temmerman S (2017) Spatio-temporal development of vegetation die-off in a submerged coastal marsh. Limnol Oceanogr 62:137–150
Shriver GW, Vickery PD, Hodgman TP, Gibbs JP (2007) Flood tides affect breeding ecology of two sympatric sharp-tailed sparrows. The Auk 124:552–560
Smith DC, Konrad V, Koulouris H, Hawes E, Borns HW Jr (1989) Salt marshes as a factor in the agriculture of northeastern North America. Agricultural History 63:270–294
Smith J, Niles L (2016) Are salt marsh pools suitable sites for restoration? Wetland Sci Practice 2016:101–109
Smith JAM, Pellew M (2021) Pond dynamics yield minimal net loss of vegetation cover across an unditched salt marsh landscape. Estuaries and Coasts 44:1534–1546
Smith JAM, Adamowicz SC, Wilson GM, Rochlin I (2021) “Waffle” pools in ditched salt marshes: assessment, potential causes, and management. Wetlands Ecol Manage 30:1081–1097
Smith SM, Roman CT, James-Pirri MJ, Chapman K, Portnoy J, Gwilliam E (2009) Response of plant communities to incremental hydrologic restoration of a tide-restricted salt marsh in southern New England (Massachusetts, U.S.A.). Restor Ecol 17:606–618
Smith SM, Warren RS (2012) Vegetation Responses to Tidal Restoration. In: Roman CT, Burdick DM (ed) Tidal Marsh Restoration: A Synthesis of Science and Management. Island Press, Washington DC, pp 59 – 80
Sweet, WV, Hamlington BD, Kopp RE, et al (2022) Global and regional sea level rise scenarios for the United States: Updated mean projections and extreme water level probabilities along U.S. coastlines. NOAA Technical Report NOS 01. National Oceanic and Atmospheric Administration, National Ocean Service. https://tidesandcurrents.noaa.gov/publications/techrpt83_Global_and_Regional_SLR_Scenarios_for_the_US_final.pdf. Accessed 6 Jan 2023
Temmerman S, Bouma TJ, Govers G, Lauwaet D (2005) Flow paths of water and sediment in a tidal marsh: relations with marsh developmental stage and tidal inundation height. Estuaries 28:338–352
Temmerman S, Govers G, Wartel S, Meire P (2003) Spatial and temporal factors controlling short-term sedimentation in a salt marsh and freshwater tidal marsh, Scheldt Estuary, Belgium, SW Netherlands. Earth Surf Process Landf 8:739–755
Tiner RW (2009) Field Guide to Tidal Wetland Plants of the Northeastern United states and Neighboring Canada. University of Massachusetts Press, Amherst, MA
Van Huissteden J, Van De Plassche O (1998) Sulphate reduction as a geomorphological agent in tidal marshes (‘Great Marshes’ at Barnstable, Cape Cod, USA). Earth Surf Process Landf 23:223–236
Vincent RE, Burdick CM, Dionne M (2013) Ditching and ditch-plugging in New England salt marshes: Effects on hydrology, elevation, and soil characteristics. Estuaries and Coasts 36:610–625
Vincent RE, Burdick DM, Dionne M (2014) Ditching and ditch-plugging in New England salt marshes: Effects on plant communities and self-maintenance. Estuaries and Coasts 37:354–368
Vinent OD, Herbert ER, Coleman DJ, Himmelstein JD, Kirwan ML (2021) Onset of runaway fragmentation of salt marshes. One Earth 4:506–516
Warren RS, Niering WA (1993) Vegetation change of a northeast tidal marsh: Interaction of sea-level rise and marsh accretion. Ecology 74:96–103
Wasson K, Ganju NK, Defne Z, Endris C, Elsey-Quirk T, Thorne KM, Freeman CM, Guntenspergen G, Nowacki DJ, Raposa KB (2019) Understanding tidal marsh trajectories: Evaluation of multiple indicators of marsh persistence. Environ Res Lett. https://doi.org/10.1088/1748-9326/ab5a94
Wasson K, Jeppesen R, Endris C, Perry DC, Woolfolk A, Beheshti K, Rodriguez M, Eby R, Watson EB, Rahman F, Haskins J, Hughes BB (2017) Eutrophication decreases salt marsh resilience through proliferation of algal mats. Biol Conserv 212:1–11
Watson EB, Wigan C, Davey EW, Andrews HM, Bishop J, Raposa KB (2017) Wetland loss patterns and inundation-productivity relationships prognosticate widespread salt marsh loss for southern New England. Estuaries and Coasts 40:662–681
Watson EB, Szura K, Wigand C, Raposa KB, Blount K, Cencer M (2016) Sea level rise, drought, and the decline of Spartina patens in New England marshes. Biol Conserv 196:173–181
Wickham H (2016) ggplot2: Elegant Graphics for Data Analysis. Springer-Verlag New York. https://ggplot2.tidyverse.org. Accessed 6 Jan 2023
Wigand C, Ardito T, Chaffee C, Ferguson W, Paton S, Raposa K, Vandemoer C, Watson E (2017) A climate change adaption strategy for management of coastal marsh systems. Estuaries and Coasts 40:682–693
Wilson CA, Hughes ZJ, FitzGerald DM, Hopkinson CS, Valentine V, Kolker AS (2014) Saltmarsh pool and tidal creek morphodynamics: Dynamic equilibrium of northern latitude saltmarshes? Geomorphology 213:99–115
Wilson KR, Kelley JT, Croitoru A, Dionne M, Belknap DF, Steneck R (2009) Stratigraphic and ecophysical characterizations of salt pools: Dynamic landforms of the Webhannet Salt Marsh, Wells, ME, USA. Estuaries and Coasts 32:855–870
Wilson KR, Kelley JT, Tanner BR, Belknap DF (2010) Probing the origins and stratigraphic signature of salt pools from north-temperate marshes in Maine, U.S.A. J Coast Res 26:1007–2016
Wolfe R (1996) Effects of Open Water Management on selected tidal marsh resources: A review. J Am Mosq Cont Assoc 12:701–712
Wolfe R, Zarebiki P, Meredith W (2021) The evolution of saltmarsh mosquito control water Management practices relative to coastal resiliency in the Mid-Atlantic and northeastern United States. Wetlands Ecol Manag. https://doi.org/10.1007/s11273-021-09817-5
Acknowledgements
We would like to thank the Rachel Carson and Parker River National Wildlife Refuges for their cooperation and support of the research. The Refuge staff including Susan Adamowicz, Nancy Pau, Frank Drauszewski, Bob Springfield, and Katie Bland were incredibly helpful and supportive of field work. Funding for this research was provided by Hurricane Sandy funds (DOI FWS/R5/CGS #F14AP00522 and DOI FWS/CGS #F15 AC01066) administered through the United States Fish and Wildlife Service and by the Atlantic Coast Joint Venture, grant number F21AP00933. Contribution number 589 of the Jackson Estuarine Laboratory, University of New Hampshire.
Funding
Funding for this research was provided by Hurricane Sandy funds (DOI FWS/R5/CGS #F14AP00522 and DOI FWS/CGS #F15 AC01066) administered through the United States Fish and Wildlife Service and by the Atlantic Coast Joint Venture, grant number F21AP00933.
Author information
Authors and Affiliations
Contributions
McKown, Burdick, Moore, Peter, Payne, and Gibson made substantial contributions to the data collection in the field and review and editing of the manuscript. Burdick and Moore were responsible for the conception, funding acquisition, experimental design, and supervision of the study. McKown, Peter, Payne, and Gibson were responsible for the data analysis, data visualization, and data interpretation. McKown was responsible for the original draft of the manuscript. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors read, reviewed, and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Ethics Committee Approval
Approval from ethics committees at the University of New Hampshire (IACUC, IRP) were not required for this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
McKown, J.G., Burdick, D.M., Moore, G.E. et al. Runnels Reverse Mega-pool Expansion and Improve Marsh Resiliency in the Great Marsh, Massachusetts (USA). Wetlands 43, 35 (2023). https://doi.org/10.1007/s13157-023-01683-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-023-01683-6