Abstract
Background
Patients with cortisol secreting adrenal adenomas present with Cushing’s syndrome (CS), while 5–15% of subjects with adrenal incidentalomas have subclinical hypercortisolism (SH) as they have biochemical abnormalities suggesting autonomous cortisol secretion without associated clinical features of CS.
Goals
Examine HPA function immediately after resection of either of these adenomas and utilize the data to decide on initiating glucocorticoid replacement.
Methods
ACTH, cortisol, and DHEA-S levels were measured frequently for 8 h after adrenalectomy in 14 patients with CS and 19 others with incidentalomas + SH. Glucocorticoids were withheld before/during surgery and administered 6–8 h postoperatively to those who had cortisol levels of <3 ug/dL (83 nmol/L).
Results
Preoperatively, incidentalomas + SH patients had larger tumors, higher ACTH, and DHEA-S but lower dexamethasone-suppressed serum cortisol levels than those with CS. Postoperatively, ACTH levels increased in both groups: (90.1 ± 31.6; 24.1 ± 14.4 ng/L, respectively; P < 0.001). Postoperative ACTH levels correlated negatively with preoperative Dexamethasone-suppressed cortisol concentrations in both groups. Patients with CS had steeper decline in cortisol concentrations than those with incidentalomas + SH. All patients with CS had hypocortisolemia requiring glucocorticoid therapy for several months, while only 5/19 with incidentalomas + SH had cortisol levels <3 ug/dL;(83 nmol/L) 6–8 h after adrenalectomy and received hydrocortisone replacement therapy for ≤4 weeks.
Conclusions
Surgical stress stimulates HPA function even in patients with hypercortisolemia. Patients with incidentalomas + SH have incomplete HPA suppression that allows more robust response to surgical stress than that observed in patients with CS. HPA assessment immediately after surgical resection of adrenal incidentalomas identified those requiring glucocorticoid replacement before discharge.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recent data suggest that 5–15% of patients with incidentally discovered adrenal tumors are shown to have abnormalities suggestive of autonomous cortisol secretion even though they present clinically with minimal, if any clinical features of Cushing’s syndrome and are commonly described as having subclinical hypercortisolism [1,2,3,4,5,6]. Although patients with adrenal incidentalomas and subclinical hypercortisolism do not have the classic clinical features of Cushing’s syndrome, many are obese and some have metabolic abnormalities such as impaired glycemia, insulin resistance or frank diabetes mellitus, and hypertension [1,2,3,4,5,6,7] while others have low bone mineral density and reduced bone quality [8,9,10], as well as higher incidence of cardiovascular events [11, 12]. Many of these clinical abnormalities improve after adrenalectomy [13,14,15,16,17]. When tested biochemically, patients with subclinical hypercortisolism have normal or minimally elevated urinary free cortisol, low plasma ACTH, an abnormal dexamethasone-suppressed serum cortisol, and a low serum DHEA-S levels [1,2,3,4,5,6,7, 18].
Laparoscopic adrenalectomy has been the main treatment option for patients with adrenal tumors. Most studies addressing the peri-operative management of patients with adrenal hypercortisolism have reported that irrespective of how mild the hypercortisolism was, such patients were given glucocorticoids before, during and after adrenalectomy [17, 19,20,21,22,23,24]. Postoperatively, most studies reported that glucocorticoids are continued for weeks to months before attempts were made to test for recovery of HPA function [19,20,21,22,23,24,25]. However, a recent study [25] followed a protocol whereby peri-operative glucocorticoids were not administered until the morning after adrenalectomy if the serum cortisol was concentration that was <5 ug/dL (138 nmol/L) and/ or when Cosyntropin-stimulated level was <18 ug/dL (497 nmol/L).
Variability also exists among institutions in the peri-operative management of another cause of hypercortisolism, namely, that of patients with ACTH-secreting pituitary adenomas. In managing the latter group of patients, we have followed a protocol over the past 30 years whereby glucocorticoid therapy is withheld until documentation of hypocortisolism is established [26,27,28,29,30]. The approach offered us the opportunity to examine peri-operative hormonal alterations and demonstrate their importance in predicting need for replacement therapy, as well as future recurrences [26,27,28,29,30].
In the current investigation, we explored the value of extending the approach followed in patients with corticotroph adenomas [26,27,28,29,30] to patients with adrenal hypercortisolism. The primary goal of the study was to examine rapid alteration in HPA function in patients with presumably suppressed axis and appreciate the modulating impact of surgical stress in that setting. We postulated that surgical stress is a powerful stimulus for HPA activation even in patients with hypercortisolism. We also postulated that patients with subclinical hypercortisolism will have a more robust activation of the HPA axis such that some may not require postoperative glucocorticoid replacement therapy.
Patients characteristics, methods, and study design
This is a prospective observational study that included two groups of consecutive patients who had unilateral laparoscopic adrenalectomy for the removal of cortisol secreting adenomas. One group had classic clinical features of Cushing’s syndrome while the other presented with adrenal incidentalomas who were demonstrated on subsequent biochemical testing to have alterations in HPA function that are detailed below. None of the patients received exogenous glucocorticoids during the year preceding their evaluation nor were they taking medications or had other illnesses that could influence HPA function or serum cortisol measurements [31]. The study was approved by the Institutional Research Board of UH Cleveland Medical Center and informed consent was obtained from participants. Since serum DHEA-S levels are gender and age-dependent, we obtained data on 94 healthy subjects with similar age and gender distribution who served as a control group.
Patients with incidentalomas and subclinical hypercortisolism
This group of patients (n = 19) represent consecutive subjects who underwent surgical adrenalectomy for a unilateral adrenal mass that was incidentally discovered on imaging studies performed for various clinical indications. The protocol followed at our institution in evaluating patients with adrenal incidentalomas included the following clinical and biochemical assessments and was applied to all enrolled subjects. Clinical evaluation of patients enrolled in this study showed that none had a history of malignant disease but some had hypertension (n = 10/19), diabetes/impaired glucose tolerance (n = 6/19), and all were overweight or clearly obese (BMI: 28-44 kg/m2) and in 8/19 the obesity was central. None of these patients had myopathy, purple striae, or supraclavicular fat fullness while some had posterior fat fullness on physical examination. All had normal 24-h urinary and/or plasma metanephrines. Screening tests for hyperaldosteronism among those who had hypertension (N = 10) and/or unprovoked hypokalemia (n = 1) were also negative. All patients had the one-mg overnight dexamethasone suppression test after having completed a 24-h urinary free cortisol and creatinine determination. Plasma ACTH, serum cortisol and DHEA-S levels were obtained before and the morning after dexamethasone administration. Adequate absorption and/or metabolism of dexamethasone was confirmed by measuring its serum level (>5.6 nmol/L) in all patients.
Subclinical hypercortisolism was defined on the basis of biochemical characteristics and the lack of prominent clinical features of hypercortisolism that included plethora, myopathy, supraclavicular fat pad, thin skin, ecchymosis and unexplained osteoporosis, or bone fractures. The biochemical characteristics defining subclinical hypercortisolism included a baseline plasma ACTH of ≤20 ng/L, a low or low-normal age-adjusted and gender-adjusted serum DHEA-S level and a normal or minimally elevated 24 h urinary free cortisol excretion (<60 ug/day; 165 nmol) in addition to a dexamethasone-suppressed serum cortisol of >1.8 ug/dL (50 nmol/L). Individual data on the 19 patients with subclinical hypercortisolism are shown in Fig. 1. Review of the CT scan on all patients showed that none had demonstrable abnormalities in the contralateral adrenal gland. The decision to proceed with adrenalectomy in this group was based on tumor size (>4 cm) and/or increased urinary free cortisol (>50 ug/gm creatinine; 138 nmol).
Individual preoperative data on 19 patients with adrenal incidentalomas and subclinical hypercortisolism. Shown here are baseline plasma ACTH in ng/L (left column), Post-dexamethasone serum cortisol (middle column) and baseline serum DHEA-S levels (right column). All patients had a plasma ACTH of <20 ng/L and a post-dexamethasone level of >1.8 ug/dL along with low age and gender adjusted serum DHEA-S levels. To convert plasma ACTH levels from ng/L to pmol/L multiply by 0.222. To convert serum cortisol levels from ug/dL to nmol/L multiply by 27.59. To convert serum DHEA-S levels from ug/dL to umol/L multiply by 0.027
Patients with classic adrenal Cushing’s syndrome
The 14 patients in this group presented with clinical features highly suggestive of hypercortisolism and their biochemical and imaging data were consistent with unilateral adrenal tumors. Their clinical manifestations included: unexplained weight gain (n = 14/14), hypertension (n = 13/14), diabetes/impaired glucose tolerance (n = 8/14), myopathy (n = 12/14), plethora (11/14), osteoporosis/fractures (n = 4/14), and impaired gonadal function (n = 12/14) and 10/14 had striae while 11/14 had prominent supraclavicular fat pad.
Biochemical evidence for hypercortisolism in this group of patients included a low baseline plasma ACTH of <10 ng/L, a low age and gender- adjusted serum DHEA-S level and an elevated 24 h urinary free cortisol excretion to >twice the upper limits of normal in addition to a dexamethasone-suppressed serum cortisol of >1.8 ug/dL(50 nmol/L).
Peri-operative management protocol
The protocol followed in the current study was modified from the one established in patients undergoing surgical resection of ACTH secreting pituitary adenomas [28,29,30] and stipulated that glucocorticoids were not administered before and during adrenalectomy. Blood samples were drawn before and at one, 2, 4, 6, and 8 h after adrenalectomy while patients were frequently evaluated clinically and biochemically for evidence for adrenal insufficiency. Blood samples were sent for the determination of plasma ACTH, serum cortisol and DHEA-S levels. Results of serum cortisol levels were usually available within 1–3 h whereas those of ACTH and DHEA-S were not immediately reported. Hydrocortisone was administered to patients with clinical symptoms (hypotension, tachycardia) and / or those who had serum cortisol levels of ≤3 ug/dL (83 nmol/L) during the first 8 post adrenalectomy hours. Patients whose serum cortisol levels remained at ≥5 ug/dL (138 nmol/L) by the 8th postoperative hour were monitored clinically and continued without any glucocorticoid therapy while another blood sample obtained the morning after surgery. Patients with a serum cortisol of >3 ug/dL (83 nmol/L) but less than 5 ug/dL (138 nmol/L) at the 8th postoperative hour were monitored for 4–12 additional hours and were started on hydrocortisone therapy if they have symptoms of adrenal insufficiency or if their serum cortisol remained below 5 ug/dL (138 nmol/L).
Pharmacokinetic data analysis
In light of the minimal rise in plasma ACTH after adrenalectomy in patients with Cushing’s syndrome, the expected increase in serum cortisol from the contralateral adrenal is presumed to be negligible or even non-existent (please see below under the results section). That offered a unique opportunity to estimate the endogenous plasma cortisol half-life. We calculated volume of distribution and plasma half-life of cortisol in the circulation using published methods [32]. We first plotted cortisol concentrations against time and then calculated the area under the curve for each subject. We also plotted the log-transformed cortisol levels against time and estimated the slope of the regression line to determine the elimination rate constant. The half-life was calculated by dividing loge2 (0.693) by the elimination constant.
Laboratory analysis
Plasma ACTH, as well as serum cortisol and DHEA-S concentrations were measured using standard established methods as described recently. Plasma ACTH and DHEA-S values below the assay detectability limits (<5 ng/L and <15 ug/dL, respectively) were included as being 4 ng/L and 14 ug/dL, respectively. Serum dexamethasone levels were measured by Lab Corporation of America, Burlington, NC.
Statistical analysis
Data are presented as mean ± standard deviation (SD), unless stated otherwise. The peri-operative data in each group were first analyzed using the Kruskal–Wallis test, as a non-parametric alternative to analysis of variance test and then comparisons between groups were done using the Wilcoxon Rank Sum test for non-parametric measurements. Categorical data were compared using Chi square and Fisher exact tests. Differences were considered significant when the two-sided P-values were less than 0.05. All data analyses were made using the SPSS program.
Results
Pre-operative data
The preoperative data on all patients are illustrated in Table 1. Individual data on the 19 patients with subclinical hypercortisolism are shown in Fig. 1. Both groups of patients had low serum DHEA-S levels (P < 0.001) as compared to similar values observed in the healthy age-matched and gender-matched subjects. When compared to those with Cushing’s syndrome, patients with subclinical hypercortisolism had larger tumor sizes, higher preoperative ACTH and DHEA-S but lower dexamethasone-suppressed serum cortisol levels (Table 1).
Peri-operative data
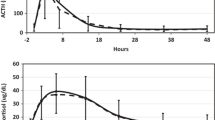
Clinically, all patients in both groups did well and none had any clinical signs or symptoms to indicate adrenal insufficiency. Plasma ACTH and serum cortisol levels on the two groups of patients are illustrated in Fig. 2
Top Panel: Plasma ACTH levels measured immediately after unilateral adrenalectomy in patients with adrenal Cushing’s syndrome (CS) and others with incidentalomas and subclinical hypercortisolism (SH). Plasma ACTH levels measured on all occasions were lower in patients with CS than in those with SH. To convert plasma ACTH values from ng/L to pmol/L multiply by 0.222. Lower Panel: Serum cortisol levels measured immediately after unilateral adrenalectomy in patients with adrenal Cushing’s syndrome (CS) and others with incidentalomas and subclinical hypercortisolism (SH). Serum cortisol levels measured at one hour and at all the times thereafter were lower (P < 0.001) in patients with CS than the comparable values in those with SH. To convert serum cortisol values from ug/dL to nmol/L multiply by 27.59
In patients with classic adrenal Cushing’s syndrome, plasma ACTH levels increased to variable degrees ranging from 8 to 43 ng/L with a mean 24.2 ± 14.1 and a median of 24 ng/L an hour after adrenalectomy and declined quickly thereafter to low or undetectable levels (Fig. 2). Serum DHEA-S levels in patients with Cushing’s syndrome declined further after adrenalectomy and were undetectable by the 8th postoperative hour.
Serum cortisol levels in all patients declined gradually over the 8 h of evaluation after adrenalectomy (Fig. 2) although the decline was steeper in those with Cushing’s syndrome. Importantly, one hour after adrenalectomy, serum cortisol levels deceased in every patient with Cushing’s syndrome with the respective mean values declining from 19.9 ± 1.9 to 15.7 ± 3.1 ug/dL; 554 ± 50 vs. 432 ± 66 nmol/L; (P < 0.001). All such patients had serum cortisol levels of <3 ug/dL (83 nmol/L) by the 6th hour and <2 ug/dL 55 nmol/L) by the 8th hour after adrenalectomy at which time hydrocortisone therapy was initiated.
Plasma ACTH levels increased after adrenalectomy in all patients with subclinical hypercortisolism and ranged from 55 to 194 with a mean of 90.1 ± 28 and a median peak level of 87 ng/L declining slowly thereafter to levels that were in the 10–20 ng/L range by the 8th hour (Fig. 2). Serum DHEA-S levels in patients with subclinical hypercortisolism did not change during the 8 postoperative hours. The decline in serum cortisol levels was slower and less steep when compared to that observed in patients with Cushing’s syndrome. (Fig. 2). At the 6th–8th postoperative hours only 5/19 patients with subclinical hypercortisolism had serum cortisol levels at ≤3 ug/dL (83 nmol/L) and these 5 were started on hydrocortisone therapy. At the 8th postoperative hour serum cortisol levels were ≥5 ug/dL (138 nmol/L) in 12 of the remaining 14 subjects and ≥4.4 (121 nmol/L) in 2 additional subjects, who had levels >5 ug/dL (138 nmol/L) on subsequent testing 4–12 h later. All of the latter 14 subjects remained without glucocorticoid supplementation.
The 5 subjects who developed adrenal insufficiency after adrenalectomy had higher preoperative urinary free cortisol excretion (53.6 ± 5.7 vs. 44.3 ± 6.1 ug/gm creatinine; 143.5 ± 18 vs. 127.1 ± 16.4 nmol/L, respectively; P = 0.023) but similar dexamethasone-suppressed serum cortisol levels (4.2 ± 1.1 vs. 3.9 ± 1.1 ug/dL; 113 ± 32 vs. 105 ± 39.5 nmol/L, respectively; p = 0.11) as compared to the 14 who did not. However, peak postoperative plasma ACTH levels were lower (P < 0.01) in the 5 subjects who developed adrenal insufficiency with levels ranging from 55 to 67 and a mean of 61.4 ± 5.6 ng/L as compared to the 14 who did not who had ACTH plasma concentrations levels ranging from 65 to 194 with a mean of 96.4 ± 29.9 ng/L.
The data depicting the relationship between the preoperative dexamethasone-suppressed serum cortisol levels and postoperative plasma ACTH in all patients are illustrated in Fig. 3. There was a strong negative correlation between the dexamethasone-suppressed serum cortisol and the postoperative rise in plasma ACTH following surgery observed within each group (Fig. 3) although the line depicting the relationship was steeper in patients with subclinical hypercortisolism.
Correlation between peak plasma ACTH observed after adrenalectomy and the preoperative serum cortisol response to dexamethasone suppression in patients with incidentalomas and subclinical hypercortisolism (solid triangles) and those with adrenal Cushing’s syndrome (solid circles). The drawn lines represent the best fit for the data in each group
Estimation of cortisol plasma half-life
The minimal rise in plasma ACTH after surgery in patients with Cushing’s syndrome suggested that there was negligible new cortisol secretion from the contralateral gland. The rapid decline in serum cortisol level one hour after adrenalectomy is supportive of our contention. The graph depicting the relationship between serum cortisol vs. time in patients with Cushing’s syndrome was a straight line even when the concentration reached low values; suggesting lack of interference of other steroids in the cortisol assay. This offered the unique opportunity to calculate the endogenous cortisol serum half-life. As illustrated in Fig. 4, plasma half-life of cortisol ranged from 1.31 to 2.51 h with a mean of 1.74 ± 0.34 and a median of 1.67 h. Figure 4 also illustrates the significant positive relation between the calculated plasma half-life and the preoperative serum cortisol.
Correlation between preoperative serum cortisol levels and the calculated plasma half-life of cortisol in the circulation in 14 patients with adrenal adenomas and classical Cushing’s syndrome. There was a significant positive correlation between the cortisol half-life and initial cortisol concentration ((P < 0.001)
Clinical data/ follow up management
The final pathology on all patients was consistent with adrenal adenoma.
Patients with Cushing’s syndrome were started on hydrocortisone therapy at the 6th–8th postoperative hour and the dose was tapered slowly over several months as described earlier in other patients with ACTH-dependent Cushing’s [26,27,28,29,30]. Hydrocortisone therapy was required for 7–14+ months with a mean of 9.3+ and a median of 9.8 months. As stated earlier, 5/19 patients were discharged home on hydrocortisone therapy while the remaining 14 did not receive postoperative glucocorticoids. Serum cortisol levels were measured weekly in the 5 subjects who were treated with hydrocortisone. Such measurements were obtained 24-h after the last oral dose of exogenous hydrocortisone. Glucocorticoid therapy was tapered quickly in the latter 5 subjects and discontinued within 4 weeks of surgery. When tested 8–13 weeks after adrenalectomy, all 19 subjects have normal AM cortisol (12.2 ± 1.1 ug/dL; 338 ± 35 nmol/L), normal plasma ACTH (27 ± 7 ng/L) but their serum DHEA-S levels were still low (33.6 ± 12 ug/dL) and similar to their respective preoperative values. Patients with a baseline serum cortisol of ≤10 ug/dL (280 nmol/L) had a normal response to Cosyntropin stimulation (n = 7). Dexamethasone suppression testing was repeated during the first 6 postoperative months in 11/19 and all had serum cortisol levels of <1.8 ug/dL (50 nmol/L).
Discussion
The study provided detailed and unique set of data after unilateral adrenalectomy in patients with adrenal Cushing’s syndrome and others with incidentalomas and subclinical hypercortisolism. To our knowledge, this would be the first thorough investigation of HPA alterations in these patients conducted according to an established protocol that avoids the confounding effects of exogenously administered glucocorticoids. The study offered an opportunity to appreciate the variations in the HPA response to a powerful stimulus in patients with different degrees of HPA suppression and the chance to estimate endogenous cortisol plasma half-life.
Multiple studies from our institution and others demonstrated that the postoperative period is a potent stimulus for HPA activation [33,34,35,36]. The current study supplement our own data on the perioperative alterations in HPA function in patients with ACTH secreting pituitary adenomas after pituitary adenomectomy [29, 30]. In the latter studies, we demonstrated that within the first few hours following resection of corticotroph adenomas plasma ACTH increased slightly but significantly before it started to decline reaching low levels by 24 h [29, 30]. We believe, the postoperative period is a powerful stimulus for HPA activation even in subjects whose axis is somewhat suppressed by endogenous hypercortisolism. It was of interest to note that the mean incremental rise in plasma ACTH that was observed after corticotroph adenomectomy (25.5 ± 9 ng/L) was very similar to that observed in patients with adrenal Cushing’s syndrome (21.4 ± 4 ng/L). These values should be contrasted with the incremental increase in plasma ACTH (74.8 ± 13 ng/L) observed in patients with subclinical hypercortisolism and that reported earlier (201.5 ± 98.8 ng/L) in subjects with normal HPA function [34, 35].
The decline in serum cortisol in patients with Cushing’s syndrome was rapid and consistent with its known plasma half-life [32]. In point of fact all 14 patients with Cushing’s syndrome had serum cortisol levels of ≤3 ug/dL (83 nmol/L) 6 h after the procedure. This should be contrasted with what was observed in patients with Cushing’s disease following corticotroph adenomectomy where it took over 30 h on average to reach such low cortisol levels [29, 30]. We believe the reason behind that was the fact that patients with Cushing’s disease had two hyperplastic adrenal gland that could respond to the endogenous ACTH surge noted after surgery [29, 30]. The higher transient surge in plasma ACTH levels observed after adrenalectomy in patients with incidentalomas and subclinical hypercortisolism would explain the slower decline in serum cortisol in these patients as compared to others with Cushing’s syndrome.
The current investigation demonstrated that patients with subclinical hypercortisolism did not have a completely suppressed HPA function as they mounted a robust response to the surgical stress. This was clinically relevant as it indicated, as was demonstrated in the study that the majority of patients with subclinical hypercortisolism did not require postoperative glucocorticoid therapy while such therapy in the minority of these patients was necessary for only a short period of time. It becomes evident that patients with subclinical hypercortisolism had variable degrees of HPA suppression that would dictate whether the need for postoperative hydrocortisone supplementation. The data indicate that among patients with subclinical hypercortisolism, those who had relatively higher preoperative urinary free cortisol excretion had less activation of their HPA axis as determined by the postoperative rise in plasma ACTH levels. The relatively modest increase in plasma ACTH after adrenalectomy in the latter group of patients predicted the need for glucocorticoid replacement after surgery. In light of the small number of subjects with subclinical hypercortisolism who developed postoperative adrenal insufficiency and the overlap in values within that group it would be hard to set a cutoff level predicting need for glucocorticoid replacement. Earlier studies examined potential preoperative data that could predict the development of adrenal insufficiency after adrenalectomy [19, 24, 37,38,39]. While one study found it hard to predict the development of adrenal insufficiency, others suggested that the duration of glucocorticoid deficiency can be predicted by a combination of preoperative data that include high urinary free cortisol excretion, lower plasma ACTH concentrations and a higher dexamethasone-suppressed serum cortisol levels [19, 27, 37, 38]. An earlier study conducted in a small number of patients suggested that preoperative CRH testing could potentially predict postoperative adrenal insufficiency [39]. It is not known whether preoperative Cosyntropin testing could achieve that objective.
Published perioperative data on patients with CS and those incidentalomas and hypercortisolism are limited as most reports have included patients who received glucocorticoids. However, in one study, glucocorticoid therapy was withheld until the morning after adrenalectomy [25] at which time treatment with hydrocortisone was initiated in those who had a baseline serum cortisol level of <5 ug/dL (138 nmol/L) and/or a peak response to Cosyntropin of <18 ug/dL (497 nmol/L). In reviewing the data included in that study one can appreciate significant heterogeneity in enrolled subjects whereby some clearly had Cushing’s syndrome (low plasma ACTH and high urinary free cortisol) and others had low urinary free cortisol excretion and/or dexamethasone-suppressed serum cortisol of <1.8 ug/dL;(50 nmol/L). The extreme heterogeneity in that study makes it hard to compare to our own investigation.
The decline in serum cortisol levels in patients with Cushing’s syndrome an hour after adrenalectomy indicated that the modest rise in plasma ACTH levels did not stimulate significant cortisol secretion from the contralateral adrenal gland. This provided the opportunity to calculate plasma half-life of endogenously secreted cortisol in subjects without renal or hepatic dysfunction. The calculated cortisol plasma half-life in our study was very similar to that reported in the literature [32]. Importantly, we noted that the plasma cortisol half-life strongly correlated with the initial serum concentration at the time of adrenalectomy (Fig. 4). We believe this was an important observation as it could have major implications on the interaction between the dose and frequency of hydrocortisone administration to patients with deficiency.
Although the data on patients with subclinical hypercortisolism are limited to 19 subjects, we believe our protocol of avoiding administering glucocorticoid before, during or immediately after adrenalectomy is safe and practical as was previously documented in patients undergoing corticotroph adenomectomy [29, 30]. None of the patients in the current study or in any of the studies on corticotroph adenomas had any clinical adverse events related to withholding glucocorticoid therapy in the peri-operative period. Now that cortisol assays are commonly utilized in most medical centers, the results should be available to physicians within a few hours. A blood sample drawn at 8 h after adrenalectomy in such patients can provide a relatively quick answer to whether glucocorticoid therapy is needed or not. A repeat serum cortisol measurement the morning after adrenalectomy would be helpful in those where the 8 h sample is not over 5 ug/dL 138 nmol/L). Patients with clear Cushing’s syndrome can be started on replacement therapy immediately after surgery while those with subclinical hypercortisolism can await until documentation of hypocortisolemia is obtained. One advantage for following this approach even in patients with adrenal Cushing’s syndrome is that the absence of hypocortisolemia at the 8th post adrenalectomy hour should raise concern for persistent disease as would expect in patients with metastatic adrenal carcinoma.
The results of serum cortisol levels might not as immediately available in some medical centers. One option to follow in such situations would be to avoid glucocorticoid replacement until the 8th postoperative hour at which time a blood sample is obtained before instituting therapy. A data-driven decision can be made at a later stage when the result becomes available. Although we believe that obtaining plasma ACTH levels at 1–2 h after adrenalectomy would be extremely helpful, we recognize that such measurement would not as practical as that of cortisol since many centers may not have the facilities to perform the assay on the former hormone in a timely manner.
In summary, the current study demonstrate that the stress of the peri-operative period is powerful enough to activate the HPA axis even in patients with endogenous hypercortisolism. Activation of the HPA axis in this setting is dependent on the degree of excessive cortisol secretion and thus, surgical resection of an incidental adrenal mass associated with subclinical hypercortisolism would not require glucocorticoid therapy in most patients. Monitoring cortisol and ACTH levels in the peri-operative 8 h after adrenalectomy in the latter group of patients can identify those who might need short term glucocorticoid therapy.
References
M. Reincke, Subclinical Cushing’s syndrome. Endocrinol. Metab. Clin. North Am. 29(1), 43–56 (2000)
M. Terzolo, G. Reimondo, S. Bovio, A. Angeli, Subclinical Cushing’s syndrome. Pituitary 7(4), 217–223 (2004)
V. Morelli, B. Masserini, A.S. Salcuni, C. Eller-Vainicher, C. Savoca, R. Viti, F. Coletti, G. Guglielmi, C. Battista, L. Lorio, P. Beck-Peccoz, B. Ambrosi, M. Arosio, A. Scillitani, I. Chiodini, Subclinical hypercortisolism: correlation between biochemical diagnostic criteria and clinical aspects. Clin. Endocrinol. 73(2), 161–166 (2010)
I. Chiodini, Clinical review: diagnosis and treatment of subclinical hypercortisolism. J. Clin. Endocrinol. Metab. 96(5), 1223–1236 (2011)
M. Terzolo, A. Pia, G. Reimondo, Subclinical Cushing’s syndrome: definition and management. Clin. Endocrinol. 76(1), 12–18 (2012)
L.K. Neiman, Update on subclinical Cushing’s syndrome. Curr. Opin. Endocrinol. Diabetes Obese 22(3), 180–184 (2015)
M. Terzolo, A. Pia, A. Alì, G. Osella, G. Reimondo, S. Bovio, F. Daffara, M. Procopio, P. Paccotti, G. Borretta, A. Angeli, Adrenal incidentaloma: a new cause of the metabolic syndrome? J. Clin. Endocrinol. Metab. 87(3), 998–1003 (2002)
I. Chiodini, V. Morelli, B. Masserini, A.S. Salcuni, C. Eller-Vainicher, R. Viti, F. Coletti, G. Guglielmi, C. Battista, V. Carnevale, L. Iorio, P. Beck-Peccoz, M. Arosio, B. Ambrosi, A. Scillitani, Bone mineral density, prevalence of vertebral fractures and bone quality in patients with adrenal incidentalomas with and without subclinical hypercortisolism: an Italian Multicenter Study. J. Clin. Endocrinol. Metab. 94(9), 3207–3214 (2009)
C. Eller-Vainicher, V. Morelli, F.M. Uliveri, S. Palmieri, V.V. Zhukouskaya, E. Cairoli, R. Pino, A. Naccarato, A. Scillitani, P. Beck-Peccoz, I. Chiodini, Bone quality, as measured by trabecular bone score in patients with adrenal incidentalomas with and without subclinical hypercortisolism. J. Bone Miner. Res. 27(10), 2223–2230 (2012)
V. Morelli, C. Eller-Vainicher, A.S. Salcuni, F. Coletti, L. Iorio, G. Muscogiuri, S. Della Casa, M. Arosio, B. Ambrosi, P. Beck-peccoz, I. Chiodini, Risk of new vertebral fractures in patients with adrenal incidentaloma with and without subclinical hypercortisolism: a multicenter longitudinal study. J. Bone Miner. Res. 26(8), 1816–1821 (2011)
L. Tauchmanovà, R. Rossi, B. Biondi, M. Pulcrano, V. Nuzzo, E.A. Palmieri, S. Fazio, G. Lombardi, Patients with subclinical Cushing’s syndrome due to adrenal adenoma have increased cardiovascular risk. J. Clin. Endocrinol. Metab. 87(11), 4872–4878 (2002)
V. Morelli, G. Reimondo, Giordano, S. Della Casa, C. Policola, S. Palmieri, A.S. Salcuni, A. Dolci, M. Mendola, M. Arosio, B. Ambrosi, A. Scillitani, E. Ghigo, P. Beck-Peccoz, M. Terzolo, I. Chiodini, Long-term follow-up in adrenal incidentalomas: an Italian multicenter study. J. Clin. Endocrinol. Metab. 99(3), 827–834 (2014)
I. Chiodini, V. Morelli, A.S. Salcuni, C. Eller-Vainicher, M. Torlontano, F. Coletti, L. Iorio, A. Cuttitta, A. Ambrosio, L. Vicentini, F. Pellegrini, M. Copetti, P. Beck-Peccoz, M. Arosio, B. Ambrosi, V. Trischitta, A. Scillitani, Beneficial metabolic effects of prompt surgical treatment in patients with an adrenal incidentaloma causing biochemical hypercortisolism. J. Clin. Endocrinol. Metab. 95(6), 2736–2745 (2010)
M. Iacobone, M. Citton, M. Scarpa, G. Viel, M. Boscaro, D. Nitti, Systematic review of surgical treatment of subclinical Cushing’s syndrome. Br. J. Surg. 102(4), 318–330 (2015)
I. Akaza, T. Yoshimoto, F. Iwashima, C. Nakayama, M. Doi, H. Izumiyama, Y. Hirata, Clinical outcome of subclinical Cushing’s syndrome after surgical and conservative treatment. Hypertens. Res. 34(1), 1111–1115 (2011)
I. Bancos, F. Alahdab, R.K. Crowley, V. Chortis, D.A. Delivanis, D. Erickson, N. Natt, M. Terzolo, W. Arlt, W.F.J.R. Young, M.H. Murad, Improvement of cardiovascular risk factors after adrenalectomy in patients with adrenal tumors and subclinical Cushing’s syndrome: A systematic review and meta-Analysis. Eur. J. Endocrinol. 175(6), R283–R295 (2016)
M. Iacobone, M. Citton, G. Viel, R. Boetto, I. Bonadio, I. Mondi, S. Tropea, D. Nitti, G. Favia, Adrenalectomy may improve cardiovascular and metabolic impairment and ameliorate quality of life in patients with adrenal incidentalomas and subclinical Cushing’s syndrome. Surgery 152(6), 991–997 (2012)
M.C. Dennedy, A.K. Annmalai, O. Prankerd-Smith, N. Freeman, K. Vengopal, J. Graggaber, O. Koulouri, A.S. Powlson, A. Shaw, D.J. Halsall, M. Gunnell, Low DHEA-S: A sensitive and specific test for the detection of subclinical hypercortisolism in adrenal incidentalomas. J. Clin. Endocrinol. Metab. 102, 786–792 (2017)
C. Eller-Vainicher, V. Morelli, A.S. Salcuni, M. Torlontano, F. Coletti, L. Iorio, A. Cuttitta, A. Ambrosio, L. Vicentini, V. Carnevale, P. Beck-Peccoz, M. Arosio, B. Ambrosi, A. Scillitani, I. Chiodini, Post-surgical hypocortisolism after removal of an adrenal incidentaloma: is it predictable by an accurate endocrinological work-up before surgery? Eur. J. Endocrinol. 162(1), 91–99 (2010)
G. Di Dalmazi, C.M. Berr, M. Fassnacht, F. Beuschlein, M. Reincke, Adrenal function after adrenalectomy for subclinical hypercortisolism and Cushing’s syndrome: A systematic review of the literature. J. Clin. Endocrinol. Metab. 99, 2637–2645 (2014)
C.M. Berr, G. Di Dalmazi, A. Osswald, K. Ritzel, M. Bidlingmaier, L.L. Geyer, M. Treitl, K. Hallfeldt, W. Rachinger, N. Reisch, R. Blaser, J. Schopohl, F. Beuschlein, M. Reincke, Time to recovery of adrenal function after curative surgery for Cushing’s syndrome depends on etiology. J. Clin. Endocrinol. Metab. 100(4), 1300–1308 (2015)
M. Fassnacht, W. Arlt, I. Bancos, H. Dralle, J. Newell-Price, A. Sahdev, A. Tabarin, M. Terzolo, S. Tsagarakis, O.M. Dekkers, Management of adrenal incidentalomas: European Society of Endocrinology Clinical Practice Guideline in collaboration with the European Network for the Study of Adrenal Tumors. Eur. J. Endocrinol. 175(2), G1–G34 (2016)
S.H. Lee, K.H. Song, J. Kim, S. Park, S.H. Ahn, H. Kim, Y.Y. Cho, S. Suh, B.J. Kin, J.H. Kim, J.M. Koh, New diagnostic criteria for subclinical hypercortisolism using postsurgical hypocortisolism: the co-work of adrenal research study. Clin. Endocrinol. 86(1), 10–18 (2017)
V. Morelli, L. Minelli, C. Eller-Vainicher, S. Palmieri, E. Cairoli, A. Spada, M. Arosio, I. Chiodini, Predictability of hypoadrenalism occurrence and duration after adrenalectomy for ACTH independent hypercortisolism. J. Endocrinol. Invest. 41(4), 485–493 (2018)
D. Ortiz, J.W. Finding, T.B. Carroll, B.R. Javorsky, A.A. Carr, D.B. Evans, T.W. Yen, T.S. Wang, Cosyntropin stimulation testing on postoperative day 1 allows for selective glucocorticoid replacement therapy after adrenalectomy for hypercortisolism: Results of a novel, multidisciplinary institutional protocol. Surgery 159(1), 259–265 (2016)
B.M. Arafah, O.H. Pearson. in Cushing’s syndrome. ed. by R.E. Rakel. Conn’s Current Therapy, 36th edn., (WB Saunders Co, Philadelphia, 1984), p. 472–477
A. Krikorian, D. Abdelmannan, W.R. Selman, B.M. Arafah, Cushing disease: use of perioperative serum cortisol measurements in early determination of success following pituitary surgery. Neurosurg. Focus 23(3), E6–E11 (2007)
D. Abdelmannan, W.R. Selman, B.M. Arafah, Perioperative management of Cushing’s disease. Rev. Endocr. Metab. Disord. 11, 127–134 (2010)
D. Abdelmannan, J. Chaiban, W.R. Selman, B.M. Arafah, Recurrences of ACTH-secreting adenomas after pituitary adenomectomy can be accurately predicted by perioperative measurements of plasma ACTH levels. J. Clin. Endocrinol. Metab. 98(4), 1458–1465 (2013)
N. El Asmar, A. Rajpal, W.R. Selman, B.M. Arafah, The value of perioperative levels of ACTH, DHEA, and DHEAS and tumor size in predicting recurrence of Cushing disease. J. Clin. Endocrinol. Metab. 103(2), 477–485 (2018)
V. Bansal, N. El Asmar, W.R. Selman, B.M. Arafah, Pitfalls in the diagnosis and management of Cushing’s syndrome. Neurological. Focus 38, 1–11 (2015)
H. Derendorf, H. Mollmann, J. Barth, C. Mollmann, S. Tunn, M. Kreig, Pharmacokinetics and oral bioavailability of hydrocortisone. J. Clin. Pharmacol. 31, 473–476 (1991)
B. Chernow, R. Alexander, R.C. Smallridge, E.R. Thompson, D. Cook, D. Beardsley, M.P. Fink, C.R. Lake, J.R. Fletcher, Hormonal responses to graded surgical stress. Arch. Intern. Med. 147, 1273–1278 (1987)
R. Udelsman, J.A. Norton, S.E. Jelenich, D.S. Goldstein, W.M. Linehan, D.L. Loriaux, G.P. Chrousos, Responses of the hypothalamic-pituitary-adrenal and renin-angiotensin axes and the sympathetic system during controlled surgical and anesthetic stress. J. Clin. Endocrinol. Metab. 64, 986–994 (1987)
K. El-Sibai, A. Rajpal, W.R. Selman, B.M. Arafah, The impact peri-operative dexamethasone administration on the normal hypothalamic pituitary adrenal response to major surgical procedures. Endocrine 58, 134–142 (2017)
A.F. Cheres, N. ElAsmar, A. Rajpal, W.R. Selman, B.M. Arafah, Perioperative hypothalamic pituitary adrenal function in patients with silent corticotroph adenomas. Pituitary 20(4), 471–476 (2017)
V. Morelli, L. Minelli, C. Eller-Vainicher, S. Palmieri, E. Cairoli, A. Spada, M. Arosio, I. Chiodini, Predictability of hypoadrenalism occurrence and duration after adrenalectomy for ACTH-independent hypercortisolism. J. Endocrinol. Investig. 41, 485–493 (2018)
A. Prete, R.M. Paragliola, F. Bottiglieri, C.A. Rota, A. Pontecorvi, R. Salvatori, S.M. Corsello, Factors predicting the duration of adrenal insufficiency in patients successfully treated for Cushing’s disease an nonmalignant primary adrenal Cushing’s syndrome. Endocrine 55, 969–980 (2017)
M. Reincke, J. Nieke, G.P. Krestin, W. Saeger, B. Allolio, W. Winkelmann, Precrinical Cushing’s syndrome in adrenal incidentalomas: comparison with adrenal Cushing’s syndrome. J. Clin. Endocrinol. Metab. 75, 826–832 (1992)
Funding
Local/departmental
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional Review Board and with the 1964 Helsinki declaration and later amendments or comparable ethical standards.
Informed consent
An informed consent was obtained from all participants in the study.
Rights and permissions
About this article
Cite this article
Khawandanah, D., ElAsmar, N. & Arafah, B.M. Alterations in hypothalamic-pituitary-adrenal function immediately after resection of adrenal adenomas in patients with Cushing’s syndrome and others with incidentalomas and subclinical hypercortisolism. Endocrine 63, 140–148 (2019). https://doi.org/10.1007/s12020-018-1769-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-018-1769-z