Abstract
Orthodontic archwires, especially nickel-titanium (NiTi) ones, are a major source of nickel release. Despite their importance, no in vivo studies have assessed the ion release from conventional or new generations of NiTi archwires (copper-included and epoxy-coated). This double-blind parallel randomized clinical trial was done on 84 saliva specimens sampled at two time points from 42 orthodontic patients. The patients were randomly divided into three groups of NiTi, copper NiTi (NiTiCu), and epoxy NiTi archwires (n = 14 each). The effects of 2-month treatment, wire types, gender, and age on pretreatment, posttreatment, and time-dependent differences were analyzed statistically (α = 0.05). Salivary nickel concentrations were 10.4571 ± 0.77168 and 11.0779 ± 0.81232 μg/L, respectively, in the baseline and second month. This increase was significant (P = 0.0000, paired t test). The extents of nickel increase in different groups were 0.8279 ± 0.14 (NiTi), 0.6493 ± 0.10 (NiTiCu), and 0.385 ± 0.11 (epoxy-coated NiTi) μg/L (P = 0.0000, one-way analyses of variance (ANOVA)). Differences between each two archwire types were significant (P = 0.000, Tukey). Age and gender did not play a role. It can be concluded that NiTi archwires might increase nickel salivary levels. Epoxy-coated NiTi followed by copper NiTi archwires might release less nickel compared to conventional NiTi ones.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alloys used in orthodontics contain nickel which is the most common etiology of type-IV cell-mediated sensitivity worldwide, affecting about one out of every four people [1–8]. By their corrosion, orthodontic appliances release nickel into the saliva and body [2, 4, 9–14].
The nickel content of nickel-titanium (NiTi) archwires is about seven times greater than that of conventional stainless steel (SS) archwires [15]; hence, they might pose a higher risk at nickel release. Various methods have been proposed to reduce the nickel release. Mainly TiO2 and to a lesser degree Cr2O3 form a passivation anti-oxide layers over the NiTi alloy surface [10, 11, 16, 17]. Nevertheless, this layer can be easily removed under various thermal, mechanical, and chemical stresses existing in the mouth such as the friction between brackets and orthodontic archwires [2–5, 7, 9, 11, 18–22]. Another method is to use metal injection molding (MIM) brackets that are single-piece (no soldering) and hence free of intra-bracket galvanic corrosion [23, 24]. Yet, their ion release rate is assessed in merely one study [24].
Brackets are not the sole source of nickel discharge. Archwires—which are never studied in vivo—matter as well [24]. According to the three available in vitro studies, NiTi archwires can be modified to release less nickel: either by the inclusion of copper (Cu) in their alloy or by coating them with a protective layer (especially epoxy) [23, 25, 26]. However, relevant in vitro studies are a few (and also lacking many factors affecting ion release), and in vivo studies are absent. Therefore, this study was carried out to measure salivary nickel in fixed orthodontic patients before treatment, and 2 months after beginning of fixed treatment using MIM brackets and three different NiTi archwires.
Subjects and Methods
This double-blind parallel randomized clinical trial was done on 42 orthodontic patients, who were divided into three groups of NiTi, copper NiTi, and epoxy NiTi (n = 14 × 3). There were two time points of 42 observations each: baseline (pretreatment) and 60 days after the treatment initiation. Subjects or their parents signed written consent forms, being informed that their participation was completely voluntary, and that they could leave the study without any penalties, and without any outcome on the delivery of their routine treatment procedures. The study ethics were approved by the institutional review board of the university according to the Helsinki declaration (RCT code: IRCT2015120725408N1). All the materials studied are already used in common practice; therefore, we did not find any damage caused to patients by randomizing them to different archwire groups.
Sample
Screened for eligibility were 48 patients visiting the Department of Orthodontics, Jundishapur University (Ahvaz, Iran), during 2015 to 2016. The inclusion criteria were the clinical need for fixed orthodontic therapy and the presence of all permanent teeth (ignoring the third molars). The exclusion criteria were histories of earlier orthodontic treatments or allergic reactions, as well as the presence of systemic diseases, medicine consumption, alcohol drinking, smoking, dental decays, or metallic tooth restorations [7, 24, 27].
Randomization and Blinding
A computer-generated random table was used to randomize the included subjects into three different groups. The involved parties (patients, laboratory technician, and the author responsible for the statistical analyses) were unaware of the archwire allocations. Only the orthodontist who had randomized the allocations and had concealed them within a sealed envelope knew them. Saliva had been sent to the laboratory within coded bottles (hence, the technician was blinded). The data groups were anonymized until the end of statistical analysis procedures.
Orthodontic Treatments
Orthodontic protocols were all similar between three groups, except for the archwires. They were free of dental extraction and involved both jaws. No soldered orthodontic bands, headgears, or other metal appliances which could release nickel (and disrupt the findings) were used. The appliances were four MIM orthodontic tubes (Fairfield Orthodontic, Fairfield, CT, USA) on the first molars and bonded 0.022-in.-slot stainless steel MIM brackets (AISI 316L, Gem Petit, Fairfield Orthodontic) on all teeth except the molars [24, 27]. The only differing item was the archwires: in three groups, NiTi, copper NiTi, and epoxy NiTi (all produced by American Orthodontic, Sheboygan, WI, USA) were utilized. All three archwires were 0.014-in. round, during the study period.
Saliva Sampling and Nickel Measuring
The sampling was performed once immediately before starting the orthodontic treatment, and once 2 months later. Before each sampling session, a list of nickel-rich foods/drinks was handed to the patients, who were requested not to consume any of them and also not to irrigate with fluoridated mouthwashes during the 24 h. before the next visit, until after saliva sampling. A same type of toothpaste (Crest Regular, Procter & Gamble, Cincinnati, OH, USA) was handed to all patients. They were taught proper methods of oral hygiene control, both verbally and in written form. They were asked to avoid brushing their teeth in the day scheduled for saliva sampling.
Saliva sampling was carried out in the morning. Patients irrigated their mouth with distilled water for 30 s. After a 2-min wait, they ejected the unstimulated saliva into bottles of polyethylene free of nickel and cleansed beforehand with acetone and distilled water. Saliva bottles were sent to the Chemistry University. After centrifuging, protein/debris removal, and dilution with nitric acid, each specimen was subjected to atomic absorption spectrophotometry at 0.01 μg/L accuracy limit (AA280Z GTA120, Varian, Mulgrave, Australia), thrice. The average of the three analyses per specimen was recorded as the main value [7, 24].
Statistical Analysis
The before-after differences between the two time points (i.e., the delta value) were calculated in each patient. Descriptive statistics and 95 % confidence intervals were computed for nickel values and delta values. The analyses (correlation coefficients, t tests, as well as one-way analyses of variance (ANOVA) and three-way repeated-measures analyses of covariance (ANCOVA)) were conducted using the SPSS program (v 20.0, IBM, Armonk, NY, USA). The alpha was determined as 0.05.
Results
Of the 48 evaluated patients, 6 were excluded before the beginning of the study. The patients aged 19.79 ± 4.09 years (range 13 to 27). Average ages in the NiTi, copper NiTi, and epoxy-coated NiTi groups were respectively 20.07 ± 4.51, 19.29 ± 3.65, and 20.00 ± 4.31 years (P = 0.860, one-way ANOVA). The numbers of males and females were uniformly six and eight, respectively, in each group.
Inter-group Differences
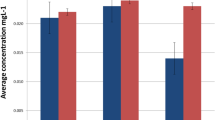
No significant difference was observed between nickel values measured at baseline (one-way ANOVA’s P = 0.162) or in the 60th day (ANOVA’s P = 0.118, Fig. 1, Table 1).
Changes During the Study Course
Salivary nickel of the whole sample (regardless of the archwire types (n = 42 patients)) increased from a baseline concentration of 10.4571 ± 0.77168 μg/L (95 % CI 10.2167 to 10.6976) to an average of 11.0779 ± 0.81232 μg/L in the 60th day (95 % CI 10.8247 to 11.3310, Fig. 1, Table 1). This increase was significant (0.6207 ± 0.21825 μg/L (95 % CI 0.5527 to 0.6887), P = 0.00000, paired t test). There was a perfect and positive correlation between the nickel values recorded at both time points (rho = 0.959, P = 0.00000).
Inter-group Differences Between Time-Dependent Nickel Changes
The one-way ANOVA’s comparison of delta nickel values in three groups indicated a significant difference between the archwires (P = 0.0000, Fig. 1, Table 1). According to the Tukey post hoc test, each of the pairwise comparisons between different pairs of archwires was significant as well (all three P values ≤0.001, Fig. 1, Table 1).
Assessment of all Variables
According to the three-way repeated-measures ANCOVA, the effects of time (F = 27.04, P = 0.00000) and the interaction of time with archwire types (F = 47.42, P = 0.00000) were significant. The latter indicated that the rates of increase in nickel release differed between the archwires. Other interactions were all non-significant (all P values >0.120). The effects of age (F = 0.002, P = 0.963), and sex (F = 0.859, P = 0.360) were not significant.
No significant role was observed for age (all P values >0.520, Spearman coefficient), in terms of pretreatment nickel levels, 60th-day nickel levels, and the extents of increases in time. The same was observed for sex (all P values >0.300, point-biserial coefficient).
Discussion
The findings of this study indicated that salivary nickel release increases in patients under treatment with MIM brackets and NiTi wires. Several articles have reported comparable increases either immediately following the beginning of treatment or about 30 days after the beginning [5, 7, 24, 28]. However, the largest part of the literature has not observed such increases [6, 7, 11, 16, 19, 28]. There are also other researches with different outcomes such as a reduction in nickel amounts [5]. In any case, such alterations in salivary nickel are not comparable to the up-to-800-μg amount of nickel introduced daily to the body [2, 6, 11, 19, 24]. On the other hand, it should be taken into consideration that the cytotoxic/genotoxic/mutagenic effects of this metal are not limited to dose-dependent ones. There are dose-independent effects, as well as effects intensified because of chronic exposures, plus local effects caused by the affinity of the metal to some organs (kidneys, in the case of nickel) which might increase the dose considerably within a local tissue [1, 7–11, 16, 18, 24].
In this study, the lowest rate of time-dependent increase was observed in the epoxy-coated NiTi group, followed by copper NiTi archwires, and then conventional NiTi wires with the highest amount of time-dependent increase. This confirmed the success of manufactures in adopting strategies to reduce the ion release, such as creating new NiTi alloys (containing copper) or coating the archwires with epoxy resin. In vitro studies have shown that epoxy-coated-NiTi and NiTiCu archwires might have a smaller corrosion rate compared to conventional NiTi, probably due to the lower oxidation-reduction potential of NiTiCu in the proximity of MIM brackets, as well as the layer of epoxy which might delay or eliminate the removal of the TiO2 film in the mouth [23, 25, 26]. Our findings also showed that age or sex might not influence either the baseline salivary nickel amounts (the normal population), or the salivary nickel levels in orthodontic patients undergoing treatment for 2 months, or the pattern of nickel increase. This was similar to the findings of the only available study in this regard [24]. Although the patterns of increase differed in various archwire groups, the final nickel concentrations were not statistically different. A reason might be that the source of ion release was not exclusively the archwires, but was a combination of brackets and archwires. The similarity of brackets in all three groups could eclipse the real contrast existing between the archwires.
This study was limited by some factors. Although in vivo designs allow obtaining more generalizable results, there are too many variables involved [1, 6, 9, 11, 18]. We hope that some level of control was present in this study, at least because of the exclusion criteria and the longitudinal nature of the study. The significant findings observed might confirm this. Furthermore, due to the lack of sample size predetermination based on power calculations, the insignificant results (but not the significant ones) should be approached cautiously.
The use of the same bracket in different archwire groups of this clinical trial might confound the findings. Finally, MIM brackets might have a less galvanic corrosion tendency than do conventional brackets [23, 26]. Hence, future studies should compare the effects of different combinations of MIM/conventional brackets with various NiTi/SS archwires.
Conclusions
Two months of orthodontic therapy using MIM brackets and NiTi archwires can increase salivary nickel, regardless of the archwire type in use. However, the pattern of increase depends on the archwire type: its increase will be lighter if epoxy-coated archwires (or to a lesser degree, copper NiTi archwires) are utilized. Overall, the nickel increase was subtle in comparison with daily nickel intake, and as long as only the dose is of concern, orthodontic treatment is less likely a threat to health. The baseline nickel or its increases are least likely influenced by sex or age.
References
Menezes LM, Quintao CA, Bolognese AM (2007) Urinary excretion levels of nickel in orthodontic patients. Am J Orthod Dentofac Orthop 131:635–638
House K, Sernetz F, Dymock D, Sandy JR, Ireland AJ (2008) Corrosion of orthodontic appliances–should we care? Am J Orthod Dentofac Orthop 133:584–592
Eliades T, Athanasiou AE (2002) In vivo aging of orthodontic alloys: implications for corrosion potential, nickel release, and biocompatibility. Angle Orthod 72:222–237
Hwang CJ, Shin JS, Cha JY (2001) Metal release from simulated fixed orthodontic appliances. Am J Orthod Dentofac Orthop 120:383–391
Matos de Souza R, Macedo de Menezes L (2008) Nickel, chromium and iron levels in the saliva of patients with simulated fixed orthodontic appliances. Angle Orthod 78:345–350
Kocadereli L, Atac PA, Kale PS, Ozer D (2000) Salivary nickel and chromium in patients with fixed orthodontic appliances. Angle Orthod 70:431–434
Agaoglu G, Arun T, Izgi B, Yarat A (2001) Nickel and chromium levels in the saliva and serum of patients with fixed orthodontic appliances. Angle Orthod 71:375–379
Natarajan M, Padmanabhan S, Chitharanjan A, Narasimhan M (2011) Evaluation of the genotoxic effects of fixed appliances on oral mucosal cells and the relationship to nickel and chromium concentrations: an in-vivo study. Am J Orthod Dentofac Orthop 140:383–388
Amini F, Jafari A, Amini P, Sepasi S (2012) Metal ion release from fixed orthodontic appliances—an in vivo study. Eur J Orthod 34:126–130
Amini F, Borzabadi Farahani A, Jafari A, Rabbani M (2008) In vivo study of metal content of oral mucosa cells in patients with and without fixed orthodontic appliances. Orthod Craniofac Res 11:51–56
Amini F, Rakhshan V, Mesgarzadeh N (2012) Effects of long-term fixed orthodontic treatment on salivary nickel and chromium levels: a 1-year prospective cohort study. Biol Trace Elem Res 150:15–20
Amini F, Rakhshan V, Sadeghi P (2012) Effect of fixed orthodontic therapy on urinary nickel levels: a long-term retrospective cohort study. Biol Trace Elem Res 150:31–36
Amini F, Shariati M, Sobouti F, Rakhshan V (2016) Effects of fixed orthodontic treatment on nickel and chromium levels in gingival crevicular fluid as a novel systemic biomarker of trace elements: a longitudinal study. Am J Orthod Dentofac Orthop. doi:10.1016/j.ajodo.2015.10.023
Khaneh Masjedi M, Haghighat Jahromi N, Niknam O, Hormozi E, Rakhshan V (2016) Effects of fixed orthodontic treatment using conventional (two-piece) versus metal injection moulding brackets on hair nickel and chromium levels: a double-blind randomized clinical trial. Eur J Orthod. doi:10.1093/ejo/cjw017
Amini F, Rakhshan V, Pousti M, Rahimi H, Shariati M, Aghamohamadi B (2012) Variations in surface roughness of seven orthodontic archwires: an SEM-profilometry study. Korean J Orthod 42:129–137
Eliades T, Trapalis C, Eliades G, Katsavrias E (2003) Salivary metal levels of orthodontic patients: a novel methodological and analytical approach. Eur J Orthod 25:103–106
Heravi F, Moayed MH, Mokhber N (2015) Effect of fluoride on nickel-titanium and stainless steel orthodontic archwires: an in-vitro study. J Dent (Tehran, Iran) 12:49–59
Hafez HS, Selim EM, Kamel Eid FH, Tawfik WA, Al-Ashkar EA, Mostafa YA (2011) Cytotoxicity, genotoxicity, and metal release in patients with fixed orthodontic appliances: a longitudinal in-vivo study. Am J Orthod Dentofac Orthop 140:298–308
Fors R, Persson M (2006) Nickel in dental plaque and saliva in patients with and without orthodontic appliances. Eur J Orthod 28:292–297
Mikulewicz M, Chojnacka K (2011) Cytocompatibility of medical biomaterials containing nickel by osteoblasts: a systematic literature review. Biol Trace Elem Res 142:865–889
Freitas MP, Oshima HM, Menezes LM (2011) Release of toxic ions from silver solder used in orthodontics: an in-situ evaluation. Am J Orthod Dentofac Orthop 140:177–181
Macedo de Menezes L, Cardoso Abdo Quintão C (2010) The release of ions from metallic orthodontic appliances. Semin Orthod 16:282–292
Siargos B, Bradley TG, Darabara M, Papadimitriou G, Zinelis S (2007) Galvanic corrosion of metal injection molded (MIM) and conventional brackets with nickel-titanium and copper-nickel-titanium archwires. Angle Orthod 77:355–360
Amini F, Harandi S, Mollaei M, Rakhshan V (2015) Effects of fixed orthodontic treatment using conventional versus metal-injection molding brackets on salivary nickel and chromium levels: a double-blind randomized clinical trial. Eur J Orthod 37:522–530
Kim H, Johnson JW (1999) Corrosion of stainless steel, nickel-titanium, coated nickel-titanium, and titanium orthodontic wires. Angle Orthod 69:39–44
Varma DP, Chidambaram S, Reddy KB, Vijay M, Ravindranath D, Prasad MR (2013) Comparison of galvanic corrosion potential of metal injection molded brackets to that of conventional metal brackets with nickel-titanium and copper nickel-titanium archwire combinations. J Contemp Dent Pract 14:488–495
Amini F, Mollaei M, Harandi S, Rakhshan V (2015) Effects of fixed orthodontic treatment on hair nickel and chromium levels: a 6-month prospective preliminary study. Biol Trace Elem Res 164:12–17
Petoumenou E, Arndt M, Keilig L, Reimann S, Hoederath H, Eliades T, Jager A, Bourauel C (2009) Nickel concentration in the saliva of patients with nickel-titanium orthodontic appliances. Am J Orthod Dentofac Orthop 135:59–65
Author’s Contribution
Mashallah Khaneh Masjedi searched the literature, conceived the assessment of effects of orthodontic treatment with different archwires on nickel release, supervised the experiments, mentored the thesis, and interpreted the findings. Ozra Niknam searched the literature, conceived the assessment of effects of orthodontic treatment with different archwires on nickel release, designed and performed the experiments, wrote the thesis, interpreted the findings, and participated in drafting the article. Nima Haghighat Jahromi and Pedram Javidi participated in the literature search and experiments. Vahid Rakhshan searched the literature, conceived the assessment of the role of age and gender, specified and implemented the statistical analyses, interpreted and discussed the findings, and drafted/revised the article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Source of Funding
The study was funded by the authors.
Rights and permissions
About this article
Cite this article
Khaneh Masjedi, M., Niknam, O., Haghighat Jahromi, N. et al. Effects of Fixed Orthodontic Treatment Using Conventional, Copper-Included, and Epoxy-Coated Nickel-Titanium Archwires on Salivary Nickel Levels: A Double-Blind Randomized Clinical Trial. Biol Trace Elem Res 174, 27–31 (2016). https://doi.org/10.1007/s12011-016-0690-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-016-0690-7