Abstract
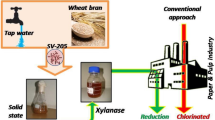
Microwave irradiation (MWI) was used as pretreatment of wheat bran and eucalyptus kraft pulp to examine its effect on xylanase production by Bacillus halodurans FNP 135 using solid state fermentation and biobleaching with xylanase, respectively. Irradiation of wheat bran under optimized conditions (600 W, 6 min, and 20 % consistency) resulted in 56.8, and 31.7 % increase in xylanase yield and water absorbance of wheat bran and 17.3 % reduction in reducing sugars content. Optimized MWI of kraft pulp at 850 W, 2 min, and 20 % consistency led to 0.9 % increase in brightness, 10 % decrease in kappa number, 7.7 % increase in water absorbance, 4.6 % decrease in tear factor, 0.9 % increase in burst factor, and 7.5 % increase in viscosity. Also, MWI enhanced xylanase-mediated biobleaching by increasing brightness (1.1 %) and decreasing kappa number (14.3 %) and leading to a total of about 20 % reduction in chlorine consumption. MWI is an economical, efficient, and environment-friendly pretreatment of wheat bran and pulp for enhanced enzyme yield and rapid heating, respectively.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The cost of the substrate alone amounts to 30–40 % of the total cost of industrial enzymes [1]. Thus, abundantly available low-cost agricultural residues (lignocelluloses) have a great potential as substrates for industrial enzyme production. Food waste, especially agricultural residues like rice bran, rice husk, and wheat bran contain lignocellulose as the major component. Lignocelluloses are preferred inducers and substrates especially in solid state fermentation (SSF) which has many advantages over submerged fermentation in terms of its less energy requirement, cheap medium composition, high enzyme concentration and simple equipment. However, lignocellulose consists of cellulose microfibrils embedded in a matrix of hemicelluloses and pectin with lignin deposited in the cell walls to give rigidity and strength [2]. Lignin, which occupies around 30 % of the lignocellulosic residues [3], represents a major obstacle for the attack by microorganisms and penetration of enzymes into the substrate. Pretreatment of the substrate is an important step before SSF to reduce lignin content, decrease crystallinity of the substrate, and increase porosity and specific surface area thereby making the substrates more susceptible to the growth of microorganisms and hydrolytic action of enzymes [4–6]. Pretreatments by pulverization, steam explosion, acid hydrolysis, alkali soaking, and organosolve treatment have been used to promote the growth of microorganisms on lignocelluloses and their enzymatic degradation [7–10]. However, most of these pretreatment methods require high-temperature or high-pressure reactions and the application of high dose of chemicals which may be toxic to the enzymes or the fermentative microorganisms, and their removal is costly and complicated [11].
As compared to conduction/convection heating, microwaves allow direct interaction between a heated object and an applied electromagnetic field to create heat. Therefore, heating is volumetric and rapid when microwave is used to treat lignocelluloses. It is hypothesized that this unique heating feature results in an explosion effect among particles and improves disruption of recalcitrant structures of lignocelluloses. In addition, electromagnetic field generated during microwaving might create nonthermal effects that also accelerate destruction of crystal structures [12].
There are reports on the effect of microwave-assisted chemical pretreatment of lignocellulosic materials on their enzymatic hydrolysis [10, 13–16] and on enzyme yield in solid state fermentation [6]. However, there are few reports on microwave irradiation (MWI) of lignocelluloses without the addition of other chemicals such as alkali, acid, or H2O2 for pretreatment before enzymatic hydrolysis or microbial growth [11].
MWI has been used in combination with H2O2 for bleaching of different pulps and has led to increased brightness in short time and reduced chemical consumption at high pulp consistency, as compared to conventional heating [17–19]. However, there are no reports on the effect of MWI of kraft pulp on xylanase performance and the resulting paper and fiber properties. This study reports the effect of MWI of (1) wheat bran on xylanase production by solid state fermentation and (2) kraft pulp on xylanase-mediated bleaching, chlorine consumption and physico-mechanical properties of the resulting paper.
Materials and Methods
Raw Material and Microwave Treatment
Wheat bran was purchased from local grain market and stored in a sealed plastic bag at room temperature. Unbleached eucalyptus kraft pulp (kappa number, 17.2 and brightness, 29.75) was provided by Ballarpur paper mills, Yamunanagar, Haryana, India.
Microwave pretreatments were carried out at a frequency of 2.45 GHz. Wheat bran and kraft pulp (20 % consistency) were placed in a sealed glass vessel and irradiated at 600 W and 850 W, respectively, for different durations.
Microorganism
Bacillus halodurans FNP 135 was isolated in our laboratory from landfill site in Chandigarh city (Gene Bank accession no. HM 484173, deposited MTCC# 10957) and was maintained on Horikoshi (HK) medium [20]. It produced a thermo-alkali-stable, cellulase-free xylanase that worked optimally at 75 °C, pH 9–9.5.
Xylanase Production
Xylanase production was carried out by solid state fermentation according to previously optimized conditions. Separately sterilized dry wheat bran (5 g), 1.16 ml of 10 % Na2CO3, and 2.34 ml tap water were mixed and inoculated with 0.5 ml of 16-h-old inoculum grown in HK medium. The SSF was carried out at 37 °C for 68 h under 95 % relative humidity with intermittent shaking. Control experiments were simultaneously conducted using unirradiated wheat bran. The fermented bran was soaked in 0.02 % Tween 80 and shaken at 160 rpm for 30 min, and the suspension was filtered and centrifuged at 12,000 × g for 10 min. The supernatant was used as crude xylanase enzyme.
Enzyme and Reducing Sugar Content Assays
Xylanase activity was assayed with 0.5 % Birchwood xylan as substrate in 100 mM Tris–Cl buffer, pH 9 at 75 °C for 5 min [21]. One unit of xylanase was defined as the amount of enzyme required to release 1 μmol of xylose from birchwood xylan per minute under standard assay conditions. It was expressed as IU/gram dry substrate (gds). For reducing sugar content measurement, 1 g of oven-dried untreated or microwave-irradiated wheat bran was soaked in sufficient amount of distilled water, vigorously stirred, and filtered. The filtrate was centrifuged at 10,000 × g for 10 min, and the reducing sugar content of the supernatant was determined according to Miller [22].
Xylanase-Mediated Biobleaching
Xylanase treatment of the kraft pulp was carried out under optimized conditions (15 IU/g oven-dried pulp in 100 mM Tris–Cl buffer, pH 9, at 65 °C, 5 % consistency, for 90 min) after the MWI pulp was cooled to 65 °C. All pulp bleaching experiments were carried out in triplicates. Results presented are the average of these values and standard deviation was within 5 %.
Water Absorbance
The sorbability to deionized water was measured by means of the Tea Bag method [23]. Briefly, 0.1 g of dry sample was added to a small nylon bag and sealed. The bag was dipped into deionized water with vigorous stirring and the increase in weight was examined every 2 min until equilibrium. The absorption was expressed as:
where Q is the sorbability at equilibrium, W 0 is weight of dry sample, and W 1 is total weight of water-sorbing wheat bran or kraft pulp.
Scanning Electron Microscopy (SEM)
Samples of pulp fibers were processed for scanning electron microscopy. The fibers were washed thrice with deionized water and fixed with 2.5 % glutaraldehyde solution prepared in phosphate buffer, pH 7.2, for 1 h. Fibers were separated from glutaraldehyde and washed thrice with same buffer and were gradually dehydrated with acetone gradient between 30 and 90 % and finally suspended in 100 % acetone; small pieces of fibers were air dried and placed on the stubs, mounted with silver tape, and sputter coated with gold using fine coat (JEOL ion sputter, Model JFC-1100) and examined at 10 KV.
CEPH1H2 Bleaching
Enzyme pretreated pulp was subjected to Chlorination (C), alkaline extraction and peroxide bleaching (EP), hypochlorite1 (H1), and hypochlorite2 (H2) stages. The chlorination was done at room temperature for 45 min, alkaline extraction at 70 °C for 120 min, and H1 and H2 stages at 45 °C for 120 min.
Analysis of Pulp Properties
The biobleached kraft pulp was thoroughly washed and hand sheets were prepared under standardized conditions. Different pulp properties, viz., kappa number (T 236 om-99), brightness (T 452 om-98), whiteness (T 1216), viscosity (T 230 om-99), burst factor (T 403 om-97), breaking length (T 404 om-92), tear factor (T 414 om-98), and residual chlorine (T 611) were determined according to the standard protocols of TAPPI [24].
Results and Discussion
The important factors in the MWI treatment of lignocelluloses would be microwave intensity, irradiation time, and substrate concentration. High microwave power with shorter pretreatment time or low microwave power with longer pretreatment time had similar effect on the weight loss and composition at the same energy consumption [16]. Also, samples with low-solid content (and accordingly a high water content) received more energy in microwave pretreatment, which eventually facilitated the disruption of the lignocellulosic complex [11]. In this study, microwave intensity of 600 W (850 W for kraft pulp) and 20 % consistency were optimized after both energy efficiency and minimum water usage were taken into account.
MWI of Wheat Bran
Xylanase production was carried out with wheat bran irradiated for 0–10 min (Fig. 1). Both xylanase production and water absorbance increased up to 6 min and declined thereafter. At 6 min MWI, 56.8 % increase in xylanase production (from 3363 IU/gds without MWI to 5,271 IU/gds) was obtained. MWI initially decreased the reducing sugar content of wheat bran (2 min), but increased it thereafter (Fig. 1). Radical increase in reducing sugars was observed after the wheat bran started charring. It seems that MWI results in simultaneous production and degradation of reducing sugars. MWI causes autolysis of polysaccharides with unavoidable secondary degradation of monosaccharides as the reaction takes place at elevated temperature [25]. Lignocellulosic materials appear to be better substrates than xylan for producing xylanolytic enzymes [26]. Wheat bran consists mainly of non-starch polysaccharides (41–60 %), starch (10–20 %), and protein (15–20 %) [27]. The increased susceptibility of wheat bran to MWI may be primarily attributed to the physical changes in the substrate. The MWI treatment probably ruptured the rigid structure of the biomass, opened the polysaccharides for microbial attack, increased the air space for more aeration, and consequently enhanced the xylanase production. However, after 6 min treatment, water absorbance and xylanase production declined which may be due to the reduction of polysaccharide content (carbonization) and nutrients in the substrate [6]. The decreased xylanase yield may also be due to excessive breakdown of the substrate into very small particles that reduce the air flow (porosity) for proper bacterial growth. It has been reported that pretreatment is one of the most important steps in the process of converting renewable lignocellulosic biomass into useful products. However, if the pretreatment is not efficient, the resultant residue is not easily hydrolyzed by enzymes. And if it is more severe, toxic compounds, which inhibit the microbial metabolism, are produced [3].
Zhao et al [6] showed increased susceptibility of microwave-irradiated wheat bran to microbial attack for cellulase production. Higher MWI power or time and lower water content (high consistency) led to substrate charring. To aviod the charring problem, we optimized all the conditions.
MWI of Kraft Pulp
MWI enhanced the physico-mechanical properties of the bleached pulp. MWI is a source of efficient heating of pulp with less energy and short time and can be applied to pulp at high consistencies. Wood or other fibrous materials are poor thermal conductors and reduce significantly the efficiency of heat transfer by conventional heating methods [17]. High-consistency pulp bleaching gives better brightness and reduces effluent. However, at consistencies higher than 30 %, the pulp has a tendency to aggregate, creating voids in the mass which interfere with heat transfer by conduction. This heating problem can be solved by using MWI, as it does not depend on a temperature gradient and is independent of the thermal properties of wood, which is almost transparent to microwaves [17]. Very high-consistency (up to 93 %) thermomechanical pulp has been treated with H2O2 and pulsed microwave (90 s) simultaneously, resulting in an increase of 20–25 points in brightness [18]. As simultaneous treatment of pulp with MWI and xylanase was not possible, treatments were given sequentially in this work.
The effect of MWI and MWI–xylanase treatment on kappa number and water absorbance of eucalyptus pulp was studied (Fig. 2). MWI alone did not change the kappa number but enhanced the xylanase-mediated reduction in kappa number by 5 % at 2 min, indicating that MWI may increase the access of the enzyme to the pulp. Percentage reduction of Kappa number and water absorbance by pulp followed the same pattern with their maxima at 2 min irradiation, and as the treatment time was increased, both decreased which may be due to loss of water from pulp and adverse effect of MWI on the pulp fibers, affecting the physical and mechanical properties. The pulp started charring after 6 min exposure to the MWI. As maximum absorbance of water and kappa number reduction were obtained at 850 W, 2 min, and 20 % consistency, these conditions were selected for further work.
Scanning Electron Micrograph of Pulp Fibers
Fibers in raw kraft pulp were uniform, straight, and intact with smooth surface (Fig. 3a). The MWI treatment resulted in fiber explosion which caused pores all over its surface (Fig. 3b) and which may have made fibers more susceptible to enzyme attack. This observation was also supported by the increased water absorption of MWI pulp (Fig. 2). However, as the MWI time increased, water absorbance and kappa number reduction decreased which may be the result of the formation of wider pores that affected water retention of fiber and the physical and mechanical properties after much of the water was lost. MWI renders the fibers more susceptible to both the enzymatic and chemical processes. When MWI was followed by xylanase treatment, it generated more perforations, suggesting their additive effect (Fig. 3) that rendered the fibers more accessible to chemical bleaching agents, thereby, assisting in lowering down the concentration of active chemical ingredients to be used in the subsequent bleaching process [7].
MWI–Xylanase–CEPH1H2 Treatment of Pulp
Xylanase pretreatment alone improved all physico-mechanical properties of the pulp (Table 1). Increase in viscosity might be due to the selective removal of lower-degree polymerized xylan and consequently enrichment of high molecular weight polysaccharides [28]. Increase in tear factor indicates that the fibers were not weakened despite xylan removal [29]. High-power microwave cooking of commercial black spruce plywood logs, as a pretreatment for mechanical pulping, resulted in energy savings up to 15 % and increase in hand sheet properties, viz. burst index (35 %), tear index (20 %), tensile index (13 %), and a modest reduction in brightness (10 %) [30].
MWI alone led to increase in brightness (0.9 %), whiteness (1.3 %), burst factor (0.9 %), breaking length (5.1 %), and viscosity (7.5 %) and decrease in kappa number (10 %) and tear factor (4.6 %) at the end of CEPH1H2 treatment, indicating that MWI did not damage the fiber. No information is available about the effect of MWI on physico-mechanical properties of CEPH1H2-bleached pulp.
Effect of MWI Pretreatment on Xylanase-Mediated Bleaching
The combined treatment with MWI and xylanase increased the brightness (4.5 %), whiteness, (5.9 %), breaking length (27.6 %), viscosity (9.8 %), burst factor (6.9 %), and tear factor (3.8 %) and reduced kappa number (40 %) as compared to the control (CEPH1H2-treated) pulp. MWI contributed 1.1, 0.4, 3, 0.9 and 16.7 % increase in brightness, whiteness, breaking length, viscosity, and kappa number reduction, respectively. When 20 % less concentration of bleaching chlorinated compound was used at different stages, brightness (79.04 %) of MWI–xylanase–CEPH1H2-treated pulp was equivalent (79.4 %) to that of control pulp with improved physico-mechanical properties, except for a slight reduction in tear factor (1.6 %) and whiteness (6.1 %) (Table 1). Reduction in chlorine consumption has both economic and environmental benefits.
Microwave pretreatment has several potential benefits in Kraft pulp mills. For bleachable hardwood or softwood pulps, pretreatment could be used to decrease both pulping and bleaching chemicals, process cycle time, and process energy requirements. Microwave pretreatment of hardwood coppice permitted simultaneous decreases of pulping chemicals and H-factor, a measure of process energy and time, by 30 % with comparable kappa numbers and pulp strengths [30]. Microwave treatment simultaneous with hydrogen peroxide bleaching of soda-AQ wheat straw pulp could increase brightness of pulp with similar pulp crystallinity and lower fiber length [19]. Hydrogen peroxide bleaching of softwood kraft pulp along with microwave heating resulted in brightness gain of seven points and 16 % increase in sheet density [31]. In microwave-peroxide bleaching of chemithermomechanical and high-yield sulfite pulps, maximum brightness was obtained in less time; as at high temperatures, the active peroxide bleaching component reacts rapidly with the pulp before its effectiveness is reduced by decomposition [32]. The brightness, breaking length, and burst index of bleached thermomechanical pulps have been improved with reduced peroxide concentration, higher consistency, and short bleaching time [17]. However, a corresponding decrease in tear index was also reported. Tensile strength, tearing property and crystallinity of cellulose fabric treated with microwave irradiation were reported to be higher than untreated cellulose fabric [33]. Similarly, MWI pretreatment of pulp enhanced the xylanase-mediated biobleaching, thereby improving the quality of the bleached pulp.
Conclusions
MWI of wheat bran is an economical, efficient, and environment-friendly alternative to enhance substrate accessibility for microbial growth and xylanase production by SSF. MWI of raw pulp reduced kappa number and improved physico-mechanical characteristics of pulp. Morphological changes such as cracks and holes due to the MWI treatment were observed by scanning electron microscopy of the fiber. These changes may improve the diffusion of the larger lignin macromolecules. Pretreatment with MWI enhanced the bleachability of eucalyptus kraft pulp and reduced the quantities of bleaching agents in subsequent stages.
References
Laxman, R. S., Sonawane, A. P., More, S. V., Rao, B. S., Rele, M. V., Jogdand, V. V., et al. (2005). Process Biochemistry, 40, 3152–3158.
Raven, P. H., Evert, R. F., & Eandichhorn, S. E. (1999). Biology of plants. New York: WH Freeman and Company.
Kodali, B., & Pogaku, R. (2006). Electronic Journal of Environmental, Agricultural and Food Chemistry, 5, 1253–1264.
Morrison, D., Dyk, J. S., & Pletschke, B. I. (2011). Bioresources, 6, 3132–3141.
Singh, A., Singh, N., & Bishnoi, N. R. (2010). Journal of Scientific and Industrial Research, 69, 232–237.
Zhao, X., Zhou, Y., Zheng, G., & Liu, D. (2010). Applied Biochemistry and Biotechnology, 160, 1557–1571.
Cen, P. L., & Xia, L. M. (1999). Advances in Biochemical Engineering/Biotechnology, 65, 69–92.
Liu, C. G., & Wyman, C. E. (2005). Bioresource Technology, 96, 1978–1985.
Shin, C. S., Lee, J. P., Lee, J. S., & Park, S. C. (2000). Applied Biochemistry and Biotechnology, 84–86, 237–245.
Zheng, G., Zhou, Y., Zhang, J., Cheng, K., Zhao, X., Zhang, T., et al. (2007). Journal wood chemistry and Technology, 27, 65–71.
Ma, H., Liu, W. W., Chen, X., Wua, Y. J., & Yu, Z. L. (2009). Bioresource Technology, 100, 1279–1284.
De La Hoz, A., Diaz-Ortiz, A., & Moreno, A. (2005). Chemical Society Reviews, 34, 164–178.
Azuma, J. I., Tanaka, F., & Koshijima, T. (1984). Journal of Fermentation Technology, 62, 377–384.
Ooshima, H., Aso, K., Harano, Y., & Yamamoto, T. (1984). Biotechnology Letters, 6, 289–294.
Xiong, J., Ye, J., Liang, W. Z., & Fan, P. M. (2000). Journal of South China University of Technology, 28, 84–89.
Zhu, S., Wu, Y., Yu, Z., Chen, Q., Wu, G., Yu, F., et al. (2006). Biosystems Engineering, 94, 437–442.
Law, K. N., Luo, S. G., & Valade, J. L. (1993). Journal of Pulp Paper Science, 19, 5181–5185.
Wan, J. K. S., Radoiu, M., Kalatchev, I., & Depew, M. C. (2000). Research on Chemical Intermediates, 26, 931–939.
Wu, C., Zhao, C., Li, J., & Chen, K. (2011). Advanced Materials Research, 236–238, 1307–1312.
Ikura, Y., & Horikoshi, K. (1987). Agricultural and Biological Chemistry, 51, 3143–3145.
Bailey, M. J., Biely, P., & Poutanen, K. (1992). Journal of Biotechnology, 23, 257–270.
Miller, G. L. (1959). Analytical Chemistry, 31, 426–428.
Hua, F., & Qian, M. (2001). Journal of Materials Science, 36, 731–738.
TAPPI Test Method (2000). TAPPI Press, Atlanta, GA.
Grundas, S. (2011). In Tech pp 752.
Damaso, M. C., Andrade, C. M., & Pereira, N. J. (2000). Applied Biochemistry and Biotechnology, 84–86, 1–9.
Liu, Z., Ying, Y., Li, F., Ma, C., & Xu, P. (2010). Journal of Industrial Microbiology and Biotechnology, 37, 495–501.
Kantelinen, A., Hortling, B., Sundquist, J., Linko, M., & Viikari, L. (1993). Holzforschung, 47, 318–324.
Roberts, J. C., Mc Carthy, A. J., Flynn, N. J., & Broda, P. (1990). Enzyme and Microbial Technology, 12, 210–213.
Scott, C.T., Klungness, J., Lentz, M., Horn, E., & Akhtar, M. (2003). Proceedings from the 2003 TAPPI Pulping Conference, San Diego, California.
Hageman, J., Plumet, L., & Robberechts, M .(1986). United States Patent 4,576,609 assigned to INTEROX (Belgium) (Mar. 18, 1986).
Liebergott, N., Van Lierop, B., & Heitner, C. (1987). Proceedings International Mechanical Pulping Conference, pp. 75-80.
Hou, A., Wang, X., & Wu, L. (2008). Carbohydrate Polymers, 74, 934–937.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Woldesenbet, F., Virk, A.P., Gupta, N. et al. Effect of Microwave Irradiation on Xylanase Production from Wheat Bran and Biobleaching of Eucalyptus Kraft Pulp. Appl Biochem Biotechnol 167, 100–108 (2012). https://doi.org/10.1007/s12010-012-9663-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9663-2