Abstract
Purpose of Review
Coronary computed tomography angiography (CCTA) is the diagnostic modality of choice for patients with stable chest pain. In this review, we scrutinize the evidence on the use of CCTA for the screening of asymptomatic patients.
Recent Findings
Clinical evidence suggests that CCTA imaging enhances cardiovascular risk stratification and prompts the timely initiation of preventive treatment leading to reduced risk of major adverse coronary events. Visualization of coronary plaques by CCTA also helps patients to comply with preventive medications. The presence of non-obstructive plaques and total plaque burden are prognostic for cardiovascular events. High-risk plaque features and pericoronary fat attenuation index, enrich the prognostic output of CCTA on top of anatomical information by capturing information on plaque vulnerability and coronary inflammatory burden.
Summary
Timely detection of atherosclerotic disease or coronary inflammation by CCTA can assist in the deployment of targeted preventive strategies and novel therapeutics to prevent cardiovascular disease.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Coronary computed tomography angiography (CCTA) is a non-invasive imaging method to visualize the anatomy of coronary arteries in three dimensions with a high sensitivity for the detection of coronary atherosclerosis [1]. CCTA has recently gained ground as a first step diagnostic approach for stable patients with chest pain and low/intermediate pretest probability for obstructive coronary artery disease (CAD) [2]. While functional ischemia tests inform on the presence of obstructive luminal stenoses, CCTA identifies both obstructive and non-obstructive plaques.
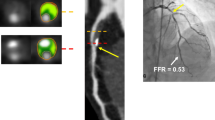
CCTA is currently indicated only for symptomatic patients, but accumulating evidence advocates for its use in asymptomatic individuals too. Besides identifying calcium and estimating total plaque burden, CCTA provides information on plaque composition and the presence of high-risk plaque (HRP) features [3]. Post-processing of CCTA images can also provide the fat attenuation index (FAI), a CCTA-derived metric that detects coronary inflammation based on gradients in pericoronary adipose tissue CT attenuation [4, 5] (Fig. 1). Therefore, CCTA is a valuable tool that beyond diagnosis also reduces the incidence of coronary events, as shown by the large randomized clinical trials SCOT-HEART (Scottish Computed Tomography of the Heart) and PROMISE (Prospective Multicenter Imaging Study for Evaluation of Chest Pain) [6, 7••].
Overview of the diagnostic tools provided by CCTA and implications for risk stratification and CVD prevention. CAD coronary artery disease, CAD-RADS Coronary Artery Disease Reporting and Data System, CCTA coronary computed tomography angiography, FAI fat attenuation index, HRP high-risk plaque, HU Hounsfield units, LAD left anterior descending, LAP low attenuation plaque, LCX left circumflex, NRS napkin ring sign, PR positive remodeling, RCA right coronary artery, SC spotty calcification, SIS segment involvement score, SSS segment stenosis score
In this review, we discuss the evolution of CCTA from a diagnostic tool of CAD into a companion diagnostic for the selection of vulnerable patients and the targeted deployment of preventative treatment. We provide an overview of novel CCTA-derived imaging biomarkers that complement the traditional anatomical information provided by CCTA and discuss the current evidence and challenges for the introduction of CCTA in everyday clinical practice as a screening tool in asymptomatic individuals.
CCTA for the Anatomical Assessment of the Coronary Arteries
Coronary Artery Calcium Score (CCS)
Coronary calcium depositions on the coronary wall, mostly suggestive of coronary atherosclerosis, can be identified and quantified in non-contrast enhanced CT images [8]. Image acquisition for Agatston CCS measurement is performed with the use of low radiation dose (< 1 mSv), and calculation of CCS is easy and inexpensive [9]. Since the seminal study of Agatston et al. the prognostic value of CCS has been confirmed in several studies [10,11,12,13] and is recommended for enhanced risk stratification for low- and intermediate-risk asymptomatic individuals (class IIb) and the deployment of statins [14, 15]. Although the “power of zero” CCS as a negative risk modifier is advocated [16], young individuals may present with “soft,” i.e., non-calcified plaques. Moreover, statins may increase plaque calcification, and therefore, the value of calcium scoring in patients under statin treatment is limited [17]. Despite its limitations, CCS is currently the most widely used imaging marker to risk stratify asymptomatic individuals.
Epicardial Coronary Artery Disease and the Significance of Non-Obstructive Plaques
CCTA is highly sensitive and has an excellent negative predictive value for obstructive CAD [18,19,20]. Patients without any luminal stenoses on CCTA have excellent prognosis with almost no cardiac events (either fatal or non-fatal) in large clinical studies with a long-term follow-up period, up to ten years [21,22,23]. Conversely, obstructive CAD is associated with increased mortality risk as shown by the CONFIRM registry [21]. Importantly, patients with non-obstructive plaques (< 50% luminal stenosis) also carry higher mortality risk compared to patients with no coronary atheromatosis [21, 23]. The importance of non-obstructive plaques has been also highlighted by the findings of a Danish registry in asymptomatic individuals, which indicated similar risk for CVD events for patients with either non-obstructive or obstructive CAD [24]. This finding is hardly unexpected, given that most culprit plaques for acute coronary syndromes are small non-obstructive atheromas [25]. This is an important clinical observation, since CCTA is currently the only imaging modality capable of detecting non-obstructive plaques in contrary to ischemia tests that rely on the presence of significant luminal stenoses. The importance of non-obstructive plaques favors the clinical use of CCTA even in asymptomatic individuals before the development of angina symptoms to deploy preventive treatment and reduce the risk of plaque rupture.
Extent of Coronary Atherosclerosis and Total Plaque Burden
Beyond the assessment of luminal stenosis, CCTA can also assess the extent of coronary atherosclerosis. Several scores have been developed to incorporate CAD extent in CCTA reporting such as the Duke index [22], segment stenosis score [26], and segment involvement score [27]. Coronary artery disease-reporting and data system (CAD-RADS) is now the recommended scoring system to describe both CAD severity and extent in CCTA images [28]. In patients with non-obstructive CAD from the CONFIRM registry, an added 6% mortality risk was observed for each additional segment with non-obstructive plaque [29]. Similarly, in patients with obstructive CAD the mortality risk increased in a dose–response relationship depending on the number of coronary vessels with obstructive CAD [21]. CAD extent is closely associated with cardiovascular risk independently of CAD severity (i.e., traditionally defined as the number of vessels with obstructive disease); extensive CAD is associated with an increased risk of MACEs equally for both patients with obstructive and non-obstructive CAD [30•]. Importantly, coronary plaque extent offers incremental prognostic value on top of clinical risk profile, the presence, and number of epicardial arteries with obstructive CAD [31]. The association of CAD extent with CVD outcomes could justify the initiation or intensification of preventative measures such as statins, especially in patients with high plaque burden (e.g., in ≥ 5 coronary segments) even in the presence of non-obstructive CAD [32].
Plaque Phenotyping and High-Risk Plaque Features
Beyond the severity and extent of coronary atherosclerosis, thanks to its high spatial resolution, CCTA can be used for plaque characterization or phenotyping. Certain anatomical plaque characteristics detected by CCTA, known as HRP features, flag plaque vulnerability and increased risk for plaque rupture [33]. These features include the presence of low-attenuation plaque (LAP), positive remodeling (PR), spotty calcification (SC), and the napkin-ring sign (NRS) [33].
LAPs, commonly defined by a mean attenuation of < 30 Hounsfield units (HU) in 3 regions of interest of a non-calcified plaque, correspond to soft plaques with a large necrotic core [34]. These plaques are well characterized as vulnerable plaques in histological studies [35] and are more frequently observed in culprit lesions for acute coronary syndromes (ACS) than in stable lesions [33]. In a cohort of 1769 stable patients with chest pain, LAP burden was a strong predictor of myocardial infarction (MI), independent of traditional CVD risk factors, CCS, and area stenosis [36]. PR is defined as an outward compensatory remodeling of the external elastic membrane leading to a diameter at the plaque site at least 10% larger than this in the proximal reference luminal area [3, 37]. Presence of PR and LAP features was associated with thin-cap fibroatheroma (TCFA) with macrophage infiltration by optical coherence tomography [38]. PR prevalence was significantly higher in culprit lesions for ACS compared to stables plaques [33]. SC is characterized as a plaque calcification with diameter less than 3 mm surrounded by non-calcified components [39]. SC detection by CCTA indicated a higher risk for the composite endpoint of cardiac mortality and non-fatal MI, irrespective of clinical risk factors and CAD severity in a cohort of 1615 patients with a 10.5-year follow-up [40]. NRS is defined by the presence of a plaque core surrounded by a ring with CT attenuation higher than the core but less than 130 HU [41]. NRS has been closely associated with vulnerable plaque characteristics such as TCFA in imaging studies [42, 43] and was highly specific for TCFA identification in pathology specimens [44]. Prospective clinical data suggest that NRS is the HRP feature most strongly associated with the risk of a future cardiac event [41, 45].
While HRP features are predictive for future MACEs, the presence of HRP features is a common finding (about 15–30%), and their positive predictive value is only modest [46, 47]. In a study of 1059 patients, coronary plaques with two HPR features (PR and LAP) were associated with significantly higher risk of coronary events compared to plaques with only one feature (22.2% vs 3.7%, respectively) [3]. The clinical significance of plaques with ≥ 2 HRP features is stressed in the latest CAD-RADS 2.0 expert consensus document which characterizes these plaques as HRPs and recommends their inclusion in the CCTA clinical reporting [48]. HRP feature detection enhances risk stratification over and above the extent of coronary atherosclerosis [49••]; however, clinical evidence for the management of such patients is limited. Based on available evidence, the detection of HRP could justify the initiation or intensification of preventive treatment such as statins, which reduce plaque progression and necrotic core volume in HRPs [50].
Coronary Inflammatory Burden by Pericoronary Fat Imaging
Imaging of pericoronary adipose tissue (PCAT) has been recently added to the available imaging markers that can be extracted from routine CCTA. PCAT attenuation is a validated method for the quantification of the inflammatory burden of the coronary arteries. The rationale of using PCAT imaging as a metric of coronary inflammation is based on experimental evidence showing phenotypic changes in adjacent PCAT in response to vascular wall inflammation (i.e., inhibition of adipocyte differentiation and intracellular lipids’ accumulation) [4, 51]. Such changes can be detected on standard CCTA images as an increase in PCAT CT attenuation. Fat attenuation index (FAI) has been developed to detect inflammation-driven changes in PCAT and is a good surrogate marker of vascular inflammation as shown by pathology, gene expression, and correlated positron emission tomography (PET) data [4, 52].
Evidence for the prognostic value of pericoronary FAI was first provided by the large prospective Cardiovascular RISk Prediction using Computed Tomography (CRISP-CT) study, which included two independent cohorts of 3912 patients undergoing clinically indicated diagnostic CCTA [53]. Increased FAI around the right coronary artery (RCA) was associated with a ~7-fold cardiac mortality risk and a ~3-fold all-cause mortality risk. These findings were independent of traditional CVD risk factors, CAD extent, and HRP feature presence. Importantly, FAI also enhanced cardiac mortality risk prognostication, beyond CVD risk factors, CAD extent, and presence of HRP [53]. FAI retained its incremental prognostic value even after the inclusion of CCS in the prognostic model for both endpoints. These findings on the prognostic value of FAI have been confirmed by two recent meta-analyses [54, 55].
Importantly, in contrary to markers of plaque burden or vulnerability (e.g., CCS or HRP) that flag irreversible anatomical changes in the vascular wall, FAI is a biomarker responsive to changes in the levels of coronary inflammation, e.g., after a plaque rupture event or the initiation of treatment. A decrease in FAI was observed in patients who started statin or anti-inflammatory treatment with anti-TNF-α, anti-IL-12/23, or anti-IL-17 for psoriasis [56,57,58]. These findings suggest that FAI could be used to select patients with evidence of coronary inflammation eligible for guiding the deployment of anti-inflammatory treatments, e.g., colchicine or novel high-cost therapeutics and monitor disease activity.
The measurement of FAI is currently clinically available as part of a regulatory-cleared medical device that can be used for the estimation of coronary inflammatory burden (FAΙ-Score) [59]. Age- and sex-specific nomograms and percentile curves are generated for FAI-Score around each of the major coronary arteries as reference diagrams to individually interpret FAI-Score in clinical practice, which can be used together with the risk factor profile and CCTA-derived information on anatomical disease to estimate the 8-year individualized risk for a fatal cardiac event [59].
Preventative CCTA Imaging: Ready for Prime Time?
Clinical Benefit of CCTA Use: Moving from Symptomatic to Asymptomatic Populations
The clinical use of CCTA for patients with chest pain is cost-effective, reducing invasive coronary angiogram referrals and cardiovascular morbidity and mortality, as shown by real-world data from a large UK population, including about 2,000,000 individuals [60]. Evidence from large randomized, multi-center clinical trials indicates that CCTA imaging refines CVD risk stratification in stable patients with chest pain and is associated with a reduction of CVD risk, which is most possibly attributed to the early deployment of preventive measures. In the SCOT-HEART study, including 4146 patients with stable chest pain followed-up for ~ 5 years, CCTA on top of standard care decreased the risk for fatal or non-fatal coronary events compared to standard care alone [7••]. The PROMISE study showed better MACE prognostication with CCTA than with functional testing in ~ 9000 individuals [6]. Also, a subanalysis of the ISCHEMIA trial showed that CCTA performed better than functional testing in MACE prediction [61].
On the other hand, evidence on the use of CCTA imaging in asymptomatic individuals is limited. In the large, multicenter, prospective CONFIRM study, CCTA did not offer incremental prognostic value when added to a model including risk factors and CCS [62, 63]. However, significant changes in CCTA technology and clinical practice have been introduced since the conduction of these studies. Ongoing studies, such as the SCOT-HEART 2 (NCT03920176), are expected to provide a definitive answer to the clinical value of CCTA imaging in asymptomatic participants with at least one risk factor. Currently, only the use of CCS is indicated in asymptomatic individuals for enhanced risk stratification, while the use of CCTA remains a class III indication in asymptomatic patients based on the latest clinical guidelines [14, 64, 65].
The main argument against the use of CCTA for population screening is the exposure of patients to ionizing radiation and the risks associated with the use of intravenous iodinated contrast agents. Nonetheless, the overall radiation dose for CCTA has been considerably reduced over the last decade through better patient preparation, the implementation of low-dose tube voltage, and improved data acquisition protocols with the latest technology CT scanners [66,67,68], which can bring CCTA dose down to ~ 1 mSv levels similar to that used for calcium scoring.
The Rationale of Using CCTA as a Screening Tool
CCTA is an appropriate imaging method for screening, as it is inexpensive and highly sensitive for coronary atherosclerosis detection. Clinical risk score such as the SCORE-2 [14] or ASCVD [69] has only modest prognostic performance and cannot reliably detect patients with subclinical atherosclerosis at risk for coronary events. Coronary plaques may remain latent for years and untreated before they develop any symptoms either due to obstructive luminal stenosis or acute rupture. Also, given that most plaques that rupture are non-obstructive, waiting for patients to become symptomatic and develop angina before referring them for diagnostic testing unnecessarily delays treatment. In the recent Danish study that included 9533 asymptomatic individuals age 40 years or older, almost half of them (46%) had evidence of coronary atherosclerosis [30•]. Screening by CCTA could permit the early detection of coronary plaques and lead to the timely administration of preventive treatment (e.g., low-dose aspirin or statins) to prevent plaque progression or rupture. CCTA screening for the detection of anatomical disease or vulnerable plaques could be also justified in individuals with risk factors not adequately captured by clinical risk scores, such as autoimmune conditions, chronic infections, or high lipoprotein(a) to exclude significant disease [70,71,72].
Moreover, and despite clinical guidelines [14], current clinical practice is already changing. At least 28% of patients in the large CONFIRM registry undergoing CCTA were asymptomatic [29], in which referral for CCTA to exclude significant CAD was based on the grounds of history of peripheral arterial disease, cerebrovascular disease, or multiple CAD risk factors. A multinational survey also showed 34% of CCTA referrals were ordered “for the exclusion of CAD in clinically healthy patients” [73].
CCTA as a Companion Diagnostic for Lipid Lowering and Anti-Inflammatory Treatments
Initiation of statins is currently favored for patients at high or very high CVD risk based on clinical risk scores [14]. CCS can be used to decide upon earlier (for patients with CCS > 100th or > 75th percentile) or delayed (zero CCS) deployment of statins [15]. Beyond CCS, high total plaque burden, presence of HRP, or high FAI-Score could be used to identify high risk patients likely to benefit from earlier or more aggressive intervention with lipid lowering treatments (LLT), although robust evidence on the effectiveness of this approach is currently lacking. A more refined approach for the tailored up-titration of LLT could consider atherosclerosis regression or progression in response to LLT and the administration of newer therapeutics such as PCSK9 inhibitors or inclisiran.
Residual cardiovascular risk, beyond lipid lowering, includes the tackling of vascular inflammation which is a major cause of atherosclerosis development, plaque progression, and rupture. Methods for the detection of vascular inflammation are useful to detect vulnerable individuals at risk for events. Next to LLT, CCTA could help in the precise deployment of anti-inflammatory agents to those patients that need them the most. Colchicine administration is currently recommended for high CVD-risk patients, especially in the secondary prevention setting [14], although so far, we have been currently lacking a companion diagnostic to guide the administration of novel high-cost anti-inflammatory therapeutics with evidence of coronary inflammation. Currently, the only method for the detection of residual inflammatory risk is the use of plasma biomarkers of inflammation such as C-reactive protein which however are non-specific to arterial inflammation. In a large meta-analysis of available data from prospective clinical studies, we have shown that the use of plasma biomarkers of inflammation provided only minimal increments in risk prognostication over the clinical risk profile [49••]. Conversely, the use of imaging, particularly the combination of FAI with HRP in CCTA may be a more efficient approach to capture residual inflammatory risk (Fig. 2), with significant additive prognostic information offered on top of clinical risk profile or CAD extent [49••]. Pericoronary FAI mapping and FAI-Score estimation could be used to monitor disease activity and residual risk despite control of traditional risk factors and help in the deployment of novel therapeutics such as canakinumab [74], rilonacept, or ziltivekimab [75]. The role of CCTA as a screening tool in asymptomatic individuals is presented in Table 1.
According to a meta-analysis of available published evidence from clinical studies (n = 351,628 individuals), plasma biomarkers offer only minimal incremental prognostic information on top of clinical risk factors for major adverse cardiovascular events. Conversely, the added prognostic value of imaging biomarkers on top of patient risk profile and coronary atherosclerosis extent is significantly greater than that of plasma biomarkers. The combination of high-risk plaque features with the perivascular fat attenuation index (FAI) by CT was associated with the maximum added prognostic information among the studied vascular inflammation biomarkers. CRP C-reactive protein, HRP high-risk plaque, IL-6 interleukin-6, PET/CT positron emission tomography/computed tomography, PVAT perivascular adipose tissue, TNF-α tumor necrosis factor α (Reprinted from: Antonopoulos AS et al. J Am Coll Cardiol Img. 2022 Mar, 15 (3) 460–471, with permission from Elsevier) [49••]. (Parts of the figures were drawn by using pictures from Servier Medical Art (http://smart.servier.com/), licensed under a Creative Commons Attribution 3.0 Unported License (https://creativecommons.org/licenses/by/3.0/))
Current Tools and Future Perspectives
CCTA can reliably detect the presence and extent of coronary atherosclerosis and plaque characteristics related to adverse outcomes (HRP features). Post-processing of CCTA images can also contribute to the identification of coronary inflammatory burden by pericoronary fat imaging. All these modalities maximize the prognostic output of CCTA, identifying CVD risk beyond clinical risk factors. The combination of risk factor profile, CAD extent, plaque phenotype, and coronary inflammatory burden into a single metric [59] paves the way for the deployment of precision medicine in cardiovascular prevention. For example, patients with high inflammatory burden could benefit from statin and/or anti-inflammatory treatment such as colchicine [14]. Data from the Oxford Risk Factors And Non Invasive Imaging Study (ORFAN Study), comprising a large dataset of 250,000 individuals with available CCTA datasets and a clinical follow-up of up to 15 years, is expected to provide further insights into the clinical use of CCTA in low-risk populations and provide a benchmark for developing and testing novel Artificial Intelligence (AI)-derived risk markers [76] with appropriate validation before regulatory approvals for clinical use [77]. Algorithms for the automated segmentation of coronary plaques and quantification of total plaque burden are already under development and could be ready soon for clinical use, refining risk prediction. Beyond coronary atherosclerosis, automated algorithms have been developed to automate the quantification measure epicardial adipose tissue volume in CCTA images from the ORFAN study [78]. Application of AI on CCTA images has been successfully applied to generate novel risk metrics and radiomic features with prognostic value, such as the radiomic texture of PCAT associated with tissue fibrosis and vascularity which is prognostic of MACEs [79] or the inflammatory footprint of COVID-19 [80] to assess COVID-19–related CVD risk and guide treatment. Other studies based on CT radiomics reported culprit lesion discrimination [81], discrimination of acute MI from unstable angina [82], and enhanced risk prognostication for MACEs [83]. The integration of AI tools in cardiovascular prevention and risk stratification is only around the corner, and large real-world CCTA datasets such as ORFAN are invaluable resources towards this end.
Conclusions
The detection of non-obstructive disease, high-risk plaques, and coronary inflammation by pericoronary fat imaging, as well as the steadily decreasing doses of ionizing radiation used, provides solid rationale on the use of CCTA for detecting asymptomatic individuals at risk for events. In the real-world practice, CCTA usage is already gaining ground as a screening tool, and ongoing studies are expected to provide an answer on the efficacy of this approach to reduce CVD risk and hard clinical endpoints. Next to refined risk stratification, CCTA has the potential to be used as a companion diagnostic for the tailored deployment of lipid lowering or anti-inflammatory treatments to those patients likely to benefit the most. Therefore, the conceptual framework and technological prerequisites on the preventative use of CCTA are mature, but stronger clinical evidence is required on the cost-effectiveness of this approach and introduction into clinical care pathways for the screening asymptomatic individuals or guiding treatments.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Nicol ED, Norgaard BL, Blanke P, Ahmadi A, Weir-McCall J, Horvat PM, Han K, Bax JJ, Leipsic J. The future of cardiovascular computed tomography: Advanced analytics and clinical insights. JACC Cardiovasc Imaging. 2019;12(6):1058–72.
Knuuti J, Wijns W, Saraste A, Capodanno D, Barbato E, Funck-Brentano C, Prescott E, Storey RF, Deaton C, Cuisset T, et al. 2019 ESC guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J. 2020;41(3):407–77.
Motoyama S, Sarai M, Harigaya H, Anno H, Inoue K, Hara T, Naruse H, Ishii J, Hishida H, Wong ND, et al. Computed tomographic angiography characteristics of atherosclerotic plaques subsequently resulting in acute coronary syndrome. J Am Coll Cardiol. 2009;54(1):49–57.
Antonopoulos AS, Sanna F, Sabharwal N, Thomas S, Oikonomou EK, Herdman L, Margaritis M, Shirodaria C, Kampoli AM, Akoumianakis I, et al. Detecting human coronary inflammation by imaging perivascular fat. Sci Transl Med. 2017;9(398).
Antonopoulos AS, Angelopoulos A, Tsioufis K, Antoniades C, Tousoulis D. Cardiovascular risk stratification by coronary computed tomography angiography imaging: Current state-of-the-art. Eur J Prev Cardiol. 2022;29(4):608–24.
Hoffmann U, Ferencik M, Udelson JE, Picard MH, Truong QA, Patel MR, Huang M, Pencina M, Mark DB, Heitner JF, et al. Prognostic value of noninvasive cardiovascular testing in patients with stable chest pain: Insights from the PROMISE trial (Prospective Multicenter Imaging Study for Evaluation of Chest Pain). Circulation. 2017;135(24):2320–32.
•• Investigators S-H, Newby DE, Adamson PD, Berry C, Boon NA, Dweck MR, Flather M, Forbes J, Hunter A, Lewis S, et al. Coronary CT angiography and 5-year risk of myocardial infarction. N Engl J Med. 2018;379(10):924–33. Findings from this study suggest that CCTA imaging in patients with stable chest pain is associated with lower fatal or non-fatal cardiac events compared to standard care.
Agatston AS, Janowitz WR, Hildner FJ, Zusmer NR, Viamonte M Jr, Detrano R. Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol. 1990;15(4):827–32.
Hong JC, Blankstein R, Shaw LJ, Padula WV, Arrieta A, Fialkow JA, Blumenthal RS, Blaha MJ, Krumholz HM, Nasir K. Implications of coronary artery calcium testing for treatment decisions among statin candidates according to the ACC/AHA cholesterol management guidelines: A cost-effectiveness analysis. JACC Cardiovasc Imaging. 2017;10(8):938–52.
Geisel MH, Bauer M, Hennig F, Hoffmann B, Lehmann N, Mohlenkamp S, Kroger K, Kara K, Muller T, Moebus S, et al. Comparison of coronary artery calcification, carotid intima-media thickness and ankle-brachial index for predicting 10-year incident cardiovascular events in the general population. Eur Heart J. 2017;38(23):1815–22.
Yeboah J, Young R, McClelland RL, Delaney JC, Polonsky TS, Dawood FZ, Blaha MJ, Miedema MD, Sibley CT, Carr JJ, et al. Utility of nontraditional risk markers in atherosclerotic cardiovascular disease risk assessment. J Am Coll Cardiol. 2016;67(2):139–47.
Blaha MJ, Whelton SP, Al Rifai M, Dardari Z, Shaw LJ, Al-Mallah MH, Matsushita K, Rozanski A, Rumberger JA, Berman DS, et al. Comparing risk scores in the prediction of coronary and cardiovascular deaths: Coronary artery calcium consortium. JACC Cardiovasc Imaging. 2021;14(2):411–21.
Bell KJL, White S, Hassan O, Zhu L, Scott AM, Clark J, Glasziou P. Evaluation of the incremental value of a coronary artery calcium score beyond traditional cardiovascular risk assessment: A systematic review and meta-analysis. JAMA Intern Med. 2022;182(6):634–42.
Visseren FLJ, Mach F, Smulders YM, Carballo D, Koskinas KC, Back M, Benetos A, Biffi A, Boavida JM, Capodanno D, et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur Heart J. 2021;42(34):3227–337.
Mach F, Baigent C, Catapano AL, Koskinas KC, Casula M, Badimon L, Chapman MJ, De Backer GG, Delgado V, Ference BA, et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur Heart J. 2020;41(1):111–88.
Mortensen MB, Falk E, Li D, Nasir K, Blaha MJ, Sandfort V, Rodriguez CJ, Ouyang P, Budoff M. Statin trials, cardiovascular events, and coronary artery calcification: Implications for a trial-based approach to statin therapy in MESA. JACC Cardiovasc Imaging. 2018;11(2 Pt 1):221–30.
Puri R, Nicholls SJ, Shao M, Kataoka Y, Uno K, Kapadia SR, Tuzcu EM, Nissen SE. Impact of statins on serial coronary calcification during atheroma progression and regression. J Am Coll Cardiol. 2015;65(13):1273–82.
Miller JM, Rochitte CE, Dewey M, Arbab-Zadeh A, Niinuma H, Gottlieb I, Paul N, Clouse ME, Shapiro EP, Hoe J, et al. Diagnostic performance of coronary angiography by 64-row CT. N Engl J Med. 2008;359(22):2324–36.
Budoff MJ, Dowe D, Jollis JG, Gitter M, Sutherland J, Halamert E, Scherer M, Bellinger R, Martin A, Benton R, et al. Diagnostic performance of 64-multidetector row coronary computed tomographic angiography for evaluation of coronary artery stenosis in individuals without known coronary artery disease: Results from the prospective multicenter ACCURACY (Assessment by Coronary Computed Tomographic Angiography of Individuals Undergoing Invasive Coronary Angiography) trial. J Am Coll Cardiol. 2008;52(21):1724–32.
Chao SP, Law WY, Kuo CJ, Hung HF, Cheng JJ, Lo HM, Shyu KG. The diagnostic accuracy of 256-row computed tomographic angiography compared with invasive coronary angiography in patients with suspected coronary artery disease. Eur Heart J. 2010;31(15):1916–23.
Min JK, Dunning A, Lin FY, Achenbach S, Al-Mallah M, Budoff MJ, Cademartiri F, Callister TQ, Chang HJ, Cheng V, et al. Age- and sex-related differences in all-cause mortality risk based on coronary computed tomography angiography findings results from the International Multicenter CONFIRM (Coronary CT Angiography Evaluation for Clinical Outcomes: An International Multicenter Registry) of 23,854 patients without known coronary artery disease. J Am Coll Cardiol. 2011;58(8):849–60.
Min JK, Shaw LJ, Devereux RB, Okin PM, Weinsaft JW, Russo DJ, Lippolis NJ, Berman DS, Callister TQ. Prognostic value of multidetector coronary computed tomographic angiography for prediction of all-cause mortality. J Am Coll Cardiol. 2007;50(12):1161–70.
Finck T, Hardenberg J, Will A, Hendrich E, Haller B, Martinoff S, Hausleiter J, Hadamitzky M. 10-year follow-up after coronary computed tomography angiography in patients with suspected coronary artery disease. JACC Cardiovasc Imaging. 2019;12(7 Pt 2):1330–8.
Mortensen MB, Dzaye O, Steffensen FH, Botker HE, Jensen JM, Ronnow Sand NP, Kragholm KH, Sorensen HT, Leipsic J, Maeng M, et al. Impact of plaque burden versus stenosis on ischemic events in patients with coronary atherosclerosis. J Am Coll Cardiol. 2020;76(24):2803–13.
Stone GW, Maehara A, Lansky AJ, de Bruyne B, Cristea E, Mintz GS, Mehran R, McPherson J, Farhat N, Marso SP, et al. A prospective natural-history study of coronary atherosclerosis. N Engl J Med. 2011;364(3):226–35.
Al-Mallah MH, Qureshi W, Lin FY, Achenbach S, Berman DS, Budoff MJ, Callister TQ, Chang HJ, Cademartiri F, Chinnaiyan K, et al. Does coronary CT angiography improve risk stratification over coronary calcium scoring in symptomatic patients with suspected coronary artery disease? Results from the prospective multicenter international CONFIRM registry. Eur Heart J Cardiovasc Imaging. 2014;15(3):267–74.
Ahmed AI, Han Y, Al Rifai M, Alnabelsi T, Nabi F, Chang SM, Chamsi-Pasha MA, Nasir K, Mahmarian JJ, Cainzos-Achirica M, et al. Added prognostic value of plaque burden to computed tomography angiography and myocardial perfusion imaging. Atherosclerosis. 2021;334:9–16.
Cury RC, Leipsic J, Abbara S, Achenbach S, Berman D, Bittencourt M, Budoff M, Chinnaiyan K, Choi AD, Ghoshhajra B, et al. CAD-RADS 2.0–2022 coronary artery disease-reporting and data system: An expert consensus document of the society of cardiovascular computed tomography (SCCT), the American College of Cardiology (ACC), the American College of Radiology (ACR), and the North America Society of Cardiovascular Imaging (NASCI). JACC Cardiovasc Imaging. 2022;15(11):1974–2001.
Chow BJ, Small G, Yam Y, Chen L, McPherson R, Achenbach S, Al-Mallah M, Berman DS, Budoff MJ, Cademartiri F, et al. Prognostic and therapeutic implications of statin and aspirin therapy in individuals with nonobstructive coronary artery disease: Results from the CONFIRM (COronary CT Angiography EvaluatioN For Clinical Outcomes: An InteRnational Multicenter registry) registry. Arterioscler Thromb Vasc Biol. 2015;35(4):981–9.
• Fuchs A, Kuhl JT, Sigvardsen PE, Afzal S, Knudsen AD, Moller MB, de Knegt MC, Sorgaard MH, Nordestgaard BG, Kober LV, et al. Subclinical coronary atherosclerosis and risk for myocardial infarction in a Danish cohort: A prospective observational cohort study. Ann Intern Med. 2023;176(4):433–42. This study indicates that 46% of asymptomatic persons have coronary atheromatosis detected by CCTA.
Bittencourt MS, Hulten E, Ghoshhajra B, O’Leary D, Christman MP, Montana P, Truong QA, Steigner M, Murthy VL, Rybicki FJ, et al. Prognostic value of nonobstructive and obstructive coronary artery disease detected by coronary computed tomography angiography to identify cardiovascular events. Circ Cardiovasc Imaging. 2014;7(2):282–91.
Chow BJW, Yam Y, Small G, Wells GA, Crean AM, Ruddy TD, Hossain A. Prognostic durability of coronary computed tomography angiography. Eur Heart J Cardiovasc Imaging. 2021;22(3):331–8.
Motoyama S, Kondo T, Sarai M, Sugiura A, Harigaya H, Sato T, Inoue K, Okumura M, Ishii J, Anno H, et al. Multislice computed tomographic characteristics of coronary lesions in acute coronary syndromes. J Am Coll Cardiol. 2007;50(4):319–26.
Motoyama S, Kondo T, Anno H, Sugiura A, Ito Y, Mori K, Ishii J, Sato T, Inoue K, Sarai M, et al. Atherosclerotic plaque characterization by 0.5-mm-slice multislice computed tomographic imaging. Circ J. 2007;71(3):363–6.
Virmani R, Burke AP, Farb A, Kolodgie FD. Pathology of the unstable plaque. Prog Cardiovasc Dis. 2002;44(5):349–56.
Williams MC, Kwiecinski J, Doris M, McElhinney P, D’Souza MS, Cadet S, Adamson PD, Moss AJ, Alam S, Hunter A, et al. Low-attenuation noncalcified plaque on coronary computed tomography angiography predicts myocardial infarction: Results from the multicenter SCOT-HEART trial (Scottish Computed Tomography of the HEART). Circulation. 2020;141(18):1452–62.
Glagov S, Weisenberg E, Zarins CK, Stankunavicius R, Kolettis GJ. Compensatory enlargement of human atherosclerotic coronary arteries. N Engl J Med. 1987;316(22):1371–5.
Nakazato R, Otake H, Konishi A, Iwasaki M, Koo BK, Fukuya H, Shinke T, Hirata K, Leipsic J, Berman DS, et al. Atherosclerotic plaque characterization by CT angiography for identification of high-risk coronary artery lesions: A comparison to optical coherence tomography. Eur Heart J Cardiovasc Imaging. 2015;16(4):373–9.
Lu G, Ye W, Ou J, Li X, Tan Z, Li T, Liu H. Coronary computed tomography angiography assessment of high-risk plaques in predicting acute coronary syndrome. Front Cardiovasc Med. 2021;8:743538.
Finck T, Stojanovic A, Will A, Hendrich E, Martinoff S, Hausleiter J, Hadamitzky M. Long-term prognostic value of morphological plaque features on coronary computed tomography angiography. Eur Heart J Cardiovasc Imaging. 2020;21(3):237–48.
Otsuka K, Fukuda S, Tanaka A, Nakanishi K, Taguchi H, Yoshikawa J, Shimada K, Yoshiyama M. Napkin-ring sign on coronary CT angiography for the prediction of acute coronary syndrome. JACC Cardiovasc Imaging. 2013;6(4):448–57.
Kashiwagi M, Tanaka A, Kitabata H, Tsujioka H, Kataiwa H, Komukai K, Tanimoto T, Takemoto K, Takarada S, Kubo T, et al. Feasibility of noninvasive assessment of thin-cap fibroatheroma by multidetector computed tomography. JACC Cardiovasc Imaging. 2009;2(12):1412–9.
Yang DH, Kang SJ, Koo HJ, Chang M, Kang JW, Lim TH, Baek S, Han S, Lee PH, Roh JH, et al. Coronary CT angiography characteristics of OCT-defined thin-cap fibroatheroma: A section-to-section comparison study. Eur Radiol. 2018;28(2):833–43.
Maurovich-Horvat P, Schlett CL, Alkadhi H, Nakano M, Otsuka F, Stolzmann P, Scheffel H, Ferencik M, Kriegel MF, Seifarth H, et al. The napkin-ring sign indicates advanced atherosclerotic lesions in coronary CT angiography. JACC Cardiovasc Imaging. 2012;5(12):1243–52.
Nerlekar N, Ha FJ, Cheshire C, Rashid H, Cameron JD, Wong DT, Seneviratne S, Brown AJ. Computed tomographic coronary angiography-derived plaque characteristics predict major adverse cardiovascular events: A systematic review and meta-analysis. Circ Cardiovasc Imaging. 2018;11(1):e006973.
Williams MC, Moss AJ, Dweck M, Adamson PD, Alam S, Hunter A, Shah ASV, Pawade T, Weir-McCall JR, Roditi G, et al. Coronary artery plaque characteristics associated with adverse outcomes in the SCOT-HEART study. J Am Coll Cardiol. 2019;73(3):291–301.
Ferencik M, Mayrhofer T, Bittner DO, Emami H, Puchner SB, Lu MT, Meyersohn NM, Ivanov AV, Adami EC, Patel MR, et al. Use of high-risk coronary atherosclerotic plaque detection for risk stratification of patients with stable chest pain: A secondary analysis of the PROMISE randomized clinical trial. JAMA Cardiol. 2018;3(2):144–52.
Cury RC, Leipsic J, Abbara S, Achenbach S, Berman D, Bittencourt M, Budoff M, Chinnaiyan K, Choi AD, Ghoshhajra B, et al. CAD-RADS 2.0–2022 coronary artery disease-reporting and data system: An expert consensus document of the society of cardiovascular computed tomography (SCCT), the American College of Cardiology (ACC), the American College of Radiology (ACR), and the North America Society of Cardiovascular Imaging (NASCI). J Cardiovasc Comput Tomogr. 2022;16(6):536–57.
•• Antonopoulos AS, Angelopoulos A, Papanikolaou P, Simantiris S, Oikonomou EK, Vamvakaris K, Koumpoura A, Farmaki M, Trivella M, Vlachopoulos C, et al. Biomarkers of vascular inflammation for cardiovascular risk prognostication: A meta-analysis. JACC Cardiovasc Imaging. 2022;15(3):460–71. This study consists the largest meta-analysis of the prognostic value of vascular inflammation biomarkers, suggesting that CCTA-derived biomarkers enhance CVD risk prognostication.
Park HB, Arsanjani R, Sung JM, Heo R, Lee BK, Lin FY, Hadamitzky M, Kim YJ, Conte E, Andreini D, et al. Impact of statins based on high-risk plaque features on coronary plaque progression in mild stenosis lesions: Results from the PARADIGM study. Eur Heart J Cardiovasc Imaging. 2023.
Antoniades C, Tousoulis D, Vavlukis M, Fleming I, Duncker DJ, Eringa E, Manfrini O, Antonopoulos AS, Oikonomou E, Padro T, et al. Perivascular adipose tissue as a source of therapeutic targets and clinical biomarkers. Eur Heart J. 2023.
Kwiecinski J, Dey D, Cadet S, Lee SE, Otaki Y, Huynh PT, Doris MK, Eisenberg E, Yun M, Jansen MA, et al. Peri-coronary adipose tissue density is associated with (18)F-sodium fluoride coronary uptake in stable patients with high-risk plaques. JACC Cardiovasc Imaging. 2019;12(10):2000–10.
Oikonomou EK, Marwan M, Desai MY, Mancio J, Alashi A, Hutt Centeno E, Thomas S, Herdman L, Kotanidis CP, Thomas KE, et al. Non-invasive detection of coronary inflammation using computed tomography and prediction of residual cardiovascular risk (the CRISP CT study): A post-hoc analysis of prospective outcome data. Lancet. 2018;392(10151):929–39.
Sagris M, Antonopoulos AS, Simantiris S, Oikonomou E, Siasos G, Tsioufis K, Tousoulis D. Pericoronary fat attenuation index-a new imaging biomarker and its diagnostic and prognostic utility: A systematic review and meta-analysis. Eur Heart J Cardiovasc Imaging. 2022;23(12):e526–36.
Kato S, Utsunomiya D, Horita N, Hoshino M, Kakuta T. Prognostic significance of the perivascular fat attenuation index derived by coronary computed tomography: A meta-analysis. Hellenic J Cardiol. 2022;67:73–5.
Goeller M, Tamarappoo BK, Kwan AC, Cadet S, Commandeur F, Razipour A, Slomka PJ, Gransar H, Chen X, Otaki Y, et al. Relationship between changes in pericoronary adipose tissue attenuation and coronary plaque burden quantified from coronary computed tomography angiography. Eur Heart J Cardiovasc Imaging. 2019;20(6):636–43.
Dai X, Yu L, Lu Z, Shen C, Tao X, Zhang J. Serial change of perivascular fat attenuation index after statin treatment: Insights from a coronary CT angiography follow-up study. Int J Cardiol. 2020;319:144–9.
Elnabawi YA, Oikonomou EK, Dey AK, Mancio J, Rodante JA, Aksentijevich M, Choi H, Keel A, Erb-Alvarez J, Teague HL, et al. Association of biologic therapy with coronary inflammation in patients with psoriasis as assessed by perivascular fat attenuation index. JAMA Cardiol. 2019;4(9):885–91.
Oikonomou EK, Antonopoulos AS, Schottlander D, Marwan M, Mathers C, Tomlins P, Siddique M, Kluner LV, Shirodaria C, Mavrogiannis MC, et al. Standardized measurement of coronary inflammation using cardiovascular computed tomography: Integration in clinical care as a prognostic medical device. Cardiovasc Res. 2021;117(13):2677–90.
Weir-McCall JR, Williams MC, Shah ASV, Roditi G, Rudd JHF, Newby DE, Nicol ED. National trends in coronary artery disease imaging: Associations with health care outcomes and costs. JACC Cardiovasc Imaging. 2023;16(5):659–71.
Maron DJ, Hochman JS, Reynolds HR, Bangalore S, O’Brien SM, Boden WE, Chaitman BR, Senior R, Lopez-Sendon J, Alexander KP, et al. Initial invasive or conservative strategy for stable coronary disease. N Engl J Med. 2020;382(15):1395–407.
Cho I, Chang HJ, Sung JM, Pencina MJ, Lin FY, Dunning AM, Achenbach S, Al-Mallah M, Berman DS, Budoff MJ, et al. Coronary computed tomographic angiography and risk of all-cause mortality and nonfatal myocardial infarction in subjects without chest pain syndrome from the CONFIRM Registry (coronary CT angiography evaluation for clinical outcomes: An international multicenter registry). Circulation. 2012;126(3):304–13.
Cho I, Al’Aref SJ, Berger A, Ó Hartaigh B, Gransar H, Valenti V, Lin FY, Achenbach S, Berman DS, Budoff MJ, et al. Prognostic value of coronary computed tomographic angiography findings in asymptomatic individuals: A 6-year follow-up from the prospective multicentre international CONFIRM study. Eur Heart J. 2018;39(11):934–41.
Narula J, Chandrashekhar Y, Ahmadi A, Abbara S, Berman DS, Blankstein R, Leipsic J, Newby D, Nicol ED, Nieman K, et al. SCCT 2021 Expert consensus document on coronary computed tomographic angiography: A report of the society of cardiovascular computed tomography. J Cardiovasc Comput Tomogr. 2021;15(3):192–217.
Arnett DK, Blumenthal RS, Albert MA, Buroker AB, Goldberger ZD, Hahn EJ, Himmelfarb CD, Khera A, Lloyd-Jones D, McEvoy JW, et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: A report of the American College of Cardiology/American Heart Association Task Force on clinical practice guidelines. Circulation. 2019;140(11):e596–646.
Schmermund A, Marwan M, Hausleiter J, Barth S, Bruder O, Kerber S, Korosoglou G, Leber A, Moshage W, Schroder S, et al. Declining radiation dose of coronary computed tomography angiography: German cardiac CT registry experience 2009–2014. Clin Res Cardiol. 2017;106(11):905–12.
Stocker TJ, Deseive S, Leipsic J, Hadamitzky M, Chen MY, Rubinshtein R, Heckner M, Bax JJ, Fang XM, Grove EL, et al. Reduction in radiation exposure in cardiovascular computed tomography imaging: Results from the PROspective multicenter registry on radiaTion dose Estimates of cardiac CT angIOgraphy iN daily practice in 2017 (PROTECTION VI). Eur Heart J. 2018;39(41):3715–23.
Si-Mohamed SA, Boccalini S, Lacombe H, Diaw A, Varasteh M, Rodesch PA, Dessouky R, Villien M, Tatard-Leitman V, Bochaton T, et al. Coronary CT angiography with photon-counting CT: First-in-human results. Radiology. 2022;303(2):303–13.
Lloyd-Jones DM, Huffman MD, Karmali KN, Sanghavi DM, Wright JS, Pelser C, Gulati M, Masoudi FA, Goff DC Jr. Estimating longitudinal risks and benefits from cardiovascular preventive therapies among medicare patients: The million hearts longitudinal ASCVD risk assessment tool: A special report from the American Heart Association and American College of Cardiology. Circulation. 2017;135(13):e793–813.
Antonopoulos AS, Simantiris S, Benetos G, Vlachopoulos C, Tsioufis K, Tousoulis D. Screening of asymptomatic patients with elevated lipoprotein(a) levels by coronary computed tomography angiography. Eur J Prev Cardiol. 2022;29(15):2049–51.
Karpouzas GA, Malpeso J, Choi TY, Li D, Munoz S, Budoff MJ. Prevalence, extent and composition of coronary plaque in patients with rheumatoid arthritis without symptoms or prior diagnosis of coronary artery disease. Ann Rheum Dis. 2014;73(10):1797–804.
Fragkou PC, Moschopoulos CD, Dimopoulou D, Triantafyllidi H, Birmpa D, Benas D, Tsiodras S, Kavatha D, Antoniadou A, Papadopoulos A. Cardiovascular disease and risk assessment in people living with HIV: Current practices and novel perspectives. Hellenic J Cardiol. 2023;71:42–54.
Maurer MH, Zimmermann E, Schlattmann P, Germershausen C, Hamm B, Dewey M. Indications, imaging technique, and reading of cardiac computed tomography: Survey of clinical practice. Eur Radiol. 2012;22(1):59–72.
Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377(12):1119–31.
Ridker PM, Devalaraja M, Baeres FMM, Engelmann MDM, Hovingh GK, Ivkovic M, Lo L, Kling D, Pergola P, Raj D, et al. IL-6 inhibition with ziltivekimab in patients at high atherosclerotic risk (RESCUE): A double-blind, randomised, placebo-controlled, phase 2 trial. Lancet. 2021;397(10289):2060–9.
Antoniades C, Patel P, Antonopoulos AS. Using artificial intelligence to study atherosclerosis, predict risk and guide treatments in clinical practice. Eur Heart J. 2023;44(6):437–9.
Antonopoulos AS, Tsioufis K. Cardiometabolic risk assessment by imaging: current status and future perspectives. Eur J Prev Cardiol. 2022;28(18):2056–8.
West HW, Siddique M, Williams MC, Volpe L, Desai R, Lyasheva M, Thomas S, Dangas K, Kotanidis CP, Tomlins P, et al. Deep-learning for epicardial adipose tissue assessment with computed tomography: Implications for cardiovascular risk prediction. JACC Cardiovasc Imaging. 2023;16(6):800–16.
Oikonomou EK, Williams MC, Kotanidis CP, Desai MY, Marwan M, Antonopoulos AS, Thomas KE, Thomas S, Akoumianakis I, Fan LM, et al. A novel machine learning-derived radiotranscriptomic signature of perivascular fat improves cardiac risk prediction using coronary CT angiography. Eur Heart J. 2019;40(43):3529–43.
Kotanidis CP, Xie C, Alexander D, Rodrigues JCL, Burnham K, Mentzer A, O’Connor D, Knight J, Siddique M, Lockstone H, et al. Constructing custom-made radiotranscriptomic signatures of vascular inflammation from routine CT angiograms: A prospective outcomes validation study in COVID-19. Lancet Digit Health. 2022;4(10):e705–16.
Lin A, Kolossvary M, Cadet S, McElhinney P, Goeller M, Han D, Yuvaraj J, Nerlekar N, Slomka PJ, Marwan M, et al. Radiomics-based precision phenotyping identifies unstable coronary plaques from computed tomography angiography. JACC Cardiovasc Imaging. 2022;15(5):859–71.
Si N, Shi K, Li N, Dong X, Zhu C, Guo Y, Hu J, Cui J, Yang F, Zhang T. Identification of patients with acute myocardial infarction based on coronary CT angiography: The value of pericoronary adipose tissue radiomics. Eur Radiol. 2022;32(10):6868–77.
Shang J, Ma S, Guo Y, Yang L, Zhang Q, Xie F, Ma Y, Ma Q, Dang Y, Zhou K, et al. Prediction of acute coronary syndrome within 3 years using radiomics signature of pericoronary adipose tissue based on coronary computed tomography angiography. Eur Radiol. 2022;32(2):1256–66.
Funding
Dr. Antonopoulos is supported by the Hellenic Foundation for Research and Innovation (HFRI, Grant Number 00468) and from the European Union’s Horizon 2020 Research and Innovation Programme (Grant agreement number 965286—MAESTRIA).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Antonopoulos is the inventor of patents US10,695,023B2 and GR20180100510 licensed through exclusive license to Caristo Diagnostics Ltd., an Oxford University spinout company. Dr. Simantiris has nothing to disclose.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Antonopoulos, A.S., Simantiris, S. Preventative Imaging with Coronary Computed Tomography Angiography. Curr Cardiol Rep 25, 1623–1632 (2023). https://doi.org/10.1007/s11886-023-01982-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11886-023-01982-8