Abstract
An efficient and atom economic modification of a previously reported synthetic pathway to tetrasubstituted thiophenes is described. The previously published synthetic methodology involved a one pot procedure starting with ketene dithioacetal and an appropriate secondary amine, and subsequent reaction with Na2S and phenacyl bromide. However, the liberated methanethiolate by-product was competed with enethiolate intermediate for phenacyl bromide, which reduced the yield and imposed the necessity to use two molar equivalents of α-haloketone reagent to increase the yield of the target thiophene products. In the present work, the proposed modification consisted in isolation of the intermediate enethiolate derivative, thereby reducing quantity of the α-haloketone to one molar equivalent. Moreover, the reaction conditions were optimized to attain optimum base/solvent combination to improve the yield of the target derivatives. Following our modified procedure, three series of new 3-amino-4-cyanothiophene derivatives were synthesized and isolated in high yields and high purity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Substituted and heteroannulated thiophene nucleus constitute crucial structural motifs found in a number of FDA-approved drugs of various pharmacological activities (Gramec et al. 2014) such as the antidepressant drug Duloxetine, the antihypertensive Eprosartan, and the antipsychotic Olanzapine. In addition, a number of thiophene-containing scaffolds are currently in clinical trials (Court et al. 2016; Sarker et al. 2015). The 3-amino-4-carbonitrile thiophene derivative I was documented as a kinase inhibitor (Cheeseright et al. 2009; Workman & Collins, 2013). The 3-aminothiophene-2-acylhydrazone derivative II was reported as analgesic and anti-inflammatory agent (Alves, Barreiro, Suzana, & Moreira, 2014). Whilst, the 3-amino-thiophene carbohydrazide derivative III was reported as inhibitor of angiogenesis (Papakyriakou et al. 2014) (Fig. 1).
On the other hand, piperazine moiety is considered as an important scaffold in a large number of biologically active candidates (Al-ghorbani et al. 2015; Singh et al. 2015; Walayat et al. 2019). Existence of piperazine moiety is known to increase the water solubility of the compounds, and piperazine ring is considered a privileged structural element in medicinal chemistry which can be used to build a library of compounds to be tested against certain receptors (Al-ghorbani et al. 2015). For example, 4-[(4-arylpiperazin-1-yl)methyl]-5-substituted-thiophenes IV exhibited allosteric enhancer activity at the A1 adenosine receptor (Romagnoli et al. 2012) (Fig. 1).
On the basis of the broad spectrum pharmacological profile observed by thiophene-based compounds, the development of new synthetic strategies, optimization, and greening of current synthetic routes may be beneficial to the pharmaceutical industry.
Literature has been enriched with numerous reports that describe various synthetic pathways to access thiophene derivatives with different substitution pattern (Li 2009). Particular attention has been given to ketene N,S- and S,S-acetals that have been used in the synthetic process of thiophene-containing compounds including 3-amino-4-cyanothiophenes bearing a substituted amino group at position 5. (Ahmed 2008; Farhat et al. 2016; Gompper and Töpfl 1962; Gruner et al. 2008; Thomae et al. 2009; Zhang et al. 2016).
In 2009, Thomae et al. (2009) reported a one-pot four step procedure for the synthesis of tetrasubstituted thiophenes in high yields (Scheme 1). The versatility of this method allowed for the access to variously substituted thiophenes, especially 3-amino-4-cyanothiophene derivatives, the synthesis of which by the common synthetic routes was found to be unsatisfactory. Motivated by these verdicts, herein we report the synthesis of novel 3-amino-4-cyanothiophenes 4a–g, 6a–k and 7. Furthermore, the optimization of the reaction conditions was performed aiming to improve the yield, purity and economy of the reaction (Schemes 2, 3).
Experimental
All the chemicals were purchased from various commercial suppliers and were used without further purification. The melting points were determined using Stuart SMP20 apparatus and are uncorrected. The IR spectra were acquired using Shimadzu IR-435 spectrophotometer and the values are represented in cm−1. Bruker AVANCETM III 400 MHz spectrophotometer was used for the acquisition of 1H NMR (400 MHz) and 13C NMR (100 MHz) spectra of the new compounds using DMSO-d6 a solvent. Nuclear magnetic resonance spectra were processed on MestreNova 11.0.4 program using residual solvent peak as the internal standard. Chemical shifts and coupling constants are presented in ppm and in Hz, respectively. Both IR and NMR spectra were carried out at Faculty of Pharmacy, Cairo University, Cairo, Egypt. Elemental analyses were carried out at The Regional Center for Mycology and Biotechnology, Al-Azhar University, Cairo, Egypt. The preparation of 3,3-dimethylthio-2-cyanoacrylonitrile (Thomae et al. 2009; Yu and Cai 2003) (1) and 2-chloro-N-arylacetamides 5a–i (Al-Nadaf et al. 2010; Kumar et al. 2014; Ma et al. 2011; Monforte et al. 2014; Rajak et al. 2012) was performed as previously reported.
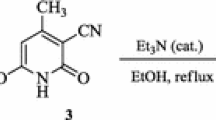
2-(4-Phenylpiperazine-1-carbonothioyl)malononitrile (3)
A mixture of compound 1 (11.92 g, 70 mmol) and 1-phenylpiperazine (11.36 g, 11.0 mL, 70 mmol) in DMF (70 mL) was stirred at 70 °C for 2 h. Thereafter, Na2S ⋅ 9H2O (16.81 g, 70 mmol) was added, and the reaction mixture was stirred at 70 °C for further 2 h. The mixture was diluted with water (40 mL) and filtered. The filtrate was cooled to 0 °C, then added to a 500 mL of a stirred saline acetic acid-sodium acetate solution (NaCl: 11.69 g, 200 mmol; CH3COONa: 12 g, 150 mmol; CH3COOH: 75 mL, 1250 mmol). The precipitate formed was filtered, washed with water (100 mL), air-dried until a constant weight, then crystallized from water-acetic acid (1:1) mixture.
Yield: 85%. mp 130–131 °C. IR (cm−1); 2194, 2164 (C≡N). 1H NMR (ppm): 3.21 (t, 4H, J = 4.9 Hz, CH2), 4.27 (t, 4H, J = 4.9 Hz, CH2), 6.86–7.30 (m, 6H, ArH, CH(CN)2. 13C NMR (ppm): 49.0, 50.5, 51.0 (piperazine Cs and CH(CN)2), 115.9, 119.5, 123.0, 129.3, 151.3 (Ar. Cs and CN), 195.4 (C=S). Anal. Calcd for C14H14N4S: C, 62.20; H, 5.22; N, 20.72. Found: C, 62.47; H, 5.36; N, 21.04.
4-Amino-5-aroyl-2-(4-phenylpiperazin-1-yl)-3-thiophenecarbonitriles 4a–g
Method A
A mixture of 1 (0.51 g, 3 mmol) and 1-phenylpiperazine (0.49 g, 0.47 mL, 3 mmol) in DMF (5 mL) was stirred at 70 °C for 75 min. Then, Na2S ⋅ 9H2O (0.72 g, 3 mmol) was added, and the stirring was continued at 70 °C for additional 2 h. The corresponding phenacyl bromide (6 mmol) was added portionwise to the reaction mixture, and the mixture was stirred at 70 °C for 2 h. Finally, the reaction was basified with K2CO3 (0.41 g, 3 mmol) and stirring was continued at 70 °C for 90 min. After cooling, the reaction mixture was poured onto ice water (50 mL) and stirred for 1 h. The precipitate formed was filtered, washed with ethanol (2 × 20 mL), then with diethyl ether (10 mL), dried and crystallized from a suitable solvent.
Method B
A solution of compound 3 (0.70 g, 2.6 mmol), and potassium carbonate (0.72 g, 5.2 mmol) in ethanol–water mixture (1:1, 25 mL) was added to a heated solution of the corresponding phenacyl bromide (2.6 mmol) in ethanol (25 mL). The reaction mixture was heated under reflux for 1 h. After cooling, the solid product was filtered, washed with ethanol (2 × 20 mL), oven-dried and crystallized from the suitable solvent.
4-Amino-5-benzoyl-2-(4-phenylpiperazin-1-yl)-3-thiophenecarbonitrile (4a)
Yield: 74% (method B), crystallized from ethanol-ethyl acetate (1:1) mixture; mp 205–207 °C; IR (cm−1): 3371, 3271 (NH2), 2198 (C≡N), 1612 (C=O).1H NMR (ppm): 3.31 (t, 4H, J = 5.2 Hz, CH2), 3.76 (t, 4H, J = 5.2 Hz, CH2), 6.80–7.63 (m, 10H, ArH), 7.96 (s, 2H, NH2, D2O exchangeable); 13C NMR (ppm): 47.2, 49.6 (piperazine Cs); 77.2, 93.6, 115.1, 115.6, 119.4, 126.8, 128.4, 129.0, 130.5, 140.8, 150.0, 158.9, 167.1 (Ar. Cs and C≡N); 184.0 (C=O). Anal. Calcd for C22H20N4OS: C, 68.02; H, 5.19; N, 14.42. Found: C, 68.31; H, 5.27; N, 14.35.
4-Amino-5-(3-bromobenzoyl)-2-(4-phenylpiperazin-1-yl)-3-thiophenecarbonitrile (4b)
Yield: 23% (method A); 71% (method B), crystallized from ethanol-ethyl acetate (1:1) mixture; mp 233–235 °C; IR (cm−1): 3394, 3259 (NH2); 2198 (C≡N); 1597 (C=O); 1H NMR (ppm): 3.31–3.33 (m, 4H, CH2), 3.78–3.80 (m, 4H, CH2), 6.80–7.73 (m, 9H, ArH), 8.00 (s, 2H, NH2, D2O exchangeable); 13C NMR (ppm): 47.2, 49.6 (piperazine Cs); 77.0, 93.4, 115.0, 115.6, 119.4, 121.8, 125.7, 129.0, 129.4, 130.7, 133.2, 142.9, 150.0, 159.4, 167.2 (Ar. Cs and C≡N); 181.8 (C=O). Anal. Calcd for C22H19BrN4OS: C, 56.54; H, 4.10; N, 11.99. Found: C, 56.76; H, 3.98; N, 12.17.
4-Amino-5-(4-bromobenzoyl)-2-(4-phenylpiperazin-1-yl)-3-thiophenecarbonitrile (4c)
Yield: 47% (method A); 74% (method B), crystallized from ethanol-ethyl acetate (1:1) mixture; mp 225–227 °C. IR (cm−1): 3383, 3282 (NH2); 2198 (C≡N); 1600 (C=O); 1H NMR (ppm): 3.31 (t, 4H, J = 5.0 Hz, CH2), 3.77 (t, 4H, J = 5.0 Hz, CH2), 6.80–7.26 (m, 5H, ArH), 7.56 (d, 2H, J = 8.4 Hz, ArH), 7.68 (d, 2H, J = 8.4 Hz, ArH), 7.99 (s, 2H, NH2, D2O exchangeable); 13C NMR (ppm): 47.3, 49.6 (piperazine Cs); 77.1, 93.5, 115.1, 115.7, 119.5, 124.1, 129.0, 129.0, 131.5, 139.8, 150.0, 159.3, 167.1 (Ar. Cs and C≡N); 182.6 (C=O). Anal. Calcd for C22H19BrN4OS: C, 56.54; H, 4.10; N, 11.99. Found: C, 56.70; H, 4.17; N, 12.23.
4-Amino-5-(4-chlorobenzoyl)-2-(4-phenylpiperazin-1-yl)-3-thiophenecarbonitrile (4d)
Yield: 27% (method A); 77% (method B), crystallized from ethanol-ethyl acetate (1:1) mixture; mp 218–220 °C. IR (cm−1): 3383, 3286 (NH2), 2198 (C≡N), 1600 (C=O); 1H NMR (ppm): 3.31–3.33 (m, 4H, CH2), 3.78 (t, 4H, J = 5.0 Hz, CH2), 6.80–7.26 (m, 5H, ArH), 7.55 (d, 2H, J = 8.4 Hz, ArH), 7.64 (d, 2H, J = 8.4 Hz, ArH), 7.99 (s, 2H, NH2, D2O exchangeable); 13C NMR (ppm): 47.3, 49.7 (piperazine Cs); 77.1, 93.5, 115.1, 115.7, 119.5, 128.6, 128.8, 129.1, 135.3, 139.5, 150.1, 159.3, 167.1 (Ar. Cs and C≡N); 182.6 (C=O). Anal. Calcd for C22H19ClN4OS: C, 62.48; H, 4.53; N, 13.25. Found: C, 62.61; H, 4.70; N, 13.57.
4-Amino-5-(3-methoxybenzoyl)-2-(4-phenylpiperazin-1-yl)-3-thiophenecarbonitrile (4e)
Yield: 68% (method B), crystallized from ethanol-ethyl acetate (1:1) mixture; mp 209–210 °C. IR (cm−1): 3375, 3275 (NH2), 2954, 2927, 2831 (aliphatic C–H), 2198 (C≡N), 1612 (C=O); 1H NMR (ppm): 3.31 (t, 4H, J = 5.0 Hz, CH2), 3.76 (t, 4H, J = 5.0 Hz, CH2), 3.80 (s, 3H, OCH3), 6.80–7.42 (m, 9H, ArH), 7.96 (s, 2H, NH2, D2O exchangeable); 13C NMR (ppm): 47.3, 49.6 (piperazine Cs); 55.2 (OCH3); 77.1, 93.7, 112.0, 115.1, 115.7, 116.3, 118.9, 119.4, 128.3, 129.0, 129.7, 142.2, 150.0, 159.1, 167.1 (Ar. Cs and C≡N); 183.7 (C=O). Anal. Calcd for C23H22N4O2S: C, 66.01; H, 5.30; N, 13.39. Found: C, 66.23; H, 5.39; N, 13.58.
4-Amino-5-(4-methoxybenzoyl)-2-(4-phenylpiperazin-1-yl)-3-thiophenecarbonitrile (4f)
Yield: 88% (method B), crystallized from ethanol-ethyl acetate (1:1) mixture; mp 220–222 °C. IR (cm−1): 3379, 3278 (NH2), 2935, 2835 (aliphatic C–H), 2202 (C≡N), 1604 (C=O); 1H NMR (ppm): 3.30–3.32 (m, 4H, CH2), 3.75–3.77 (m, 4H, CH2), 3.81 (s, 3H, OCH3), 6.80–7.65 (m, 9H, ArH), 7.88 (s, 2H, NH2, D2O exchangeable); 13C NMR (ppm): 47.3, 49.6 (piperazine Cs); 55.3 (OCH3); 77.3, 93.5, 113.7, 115.2, 115.7, 119.4, 128.9, 129.0, 133.2, 150.0, 158.6, 161.1, 166.8 (Ar. Cs and C≡N); 183.4 (C=O). Anal. Calcd for C23H22N4O2S: C, 66.01; H, 5.30; N, 13.39. Found: C, 66.18; H, 5.28; N, 13.61.
4-Amino-5-(3-nitrobenzoyl)-2-(4-phenylpiperazin-1-yl)-3-thiophenecarbonitrile (4g)
Yield: 77% (method B), crystallized from toluene; mp 244–246 °C. IR (cm−1): 3394, 3263 (NH2), 2198 (C≡N), 1600 (C=O), 1527, 1342 (NO2); 1H NMR (ppm): 3.31–3.34 (m, 4H, CH2), 3.79–3.81 (m, 4H, CH2), 6.80–8.36 (m, 11H, 9 ArH and NH2); 13C NMR (ppm): 47.2, 49.7 (piperazine Cs), 76.9, 93.2, 115.0, 115.6, 119.4, 121.5, 125.1, 129.0, 130.4, 133.1, 141.9, 147.7, 150.0, 159.8, 167.2 (Ar. Cs and C≡N), 180.7 (C=O). Anal. Calcd for C22H19N5O3S: C, 60.96; H, 4.42; N, 16.16. Found: C, 61.23; H, 4.37; N, 16.40.
2-Chloro-N-(substituted) phenylacetamides 5j,k
Chloroacetyl chloride (5.65 g, 4 mL, 50 mmol) was added dropwise to a stirred solution of the corresponding 3-aminobenzamide derivatives (50 mmol), and TEA (5.06 g, 7 mL, 50 mmol) in DCM (50 mL) at 0 °C. The reaction mixture was stirred at room temperature for 8 h, then the solvent was evaporated. The residue was washed with water, air-dried until a constant weight, and recrystallized from aqueous ethanol.
N-(3-(2-Chloroacetamido)-4-methylphenyl)benzamide (5j)
Yield: 73%, mp 186–187 °C. IR (cm−1): 3232, 3201 (NH), 1678, 1643 (C=O); 1H NMR (ppm): 2.18 (s, 3H, CH3), 4.32 (s, 2H, CH2), 7.19–7.97 (m, 8H, ArH), 9.70 (s, 1H, CONH, D2O exchangeable), 10.25 (s, 1H, CONH, D2O exchangeable). 13C NMR (ppm): 17.2 (CH3); 43.1 (CH2); 117.2, 117.9, 127.2, 127.6, 128.3, 130.2, 131.5, 134.8, 135.4, 137.2 (Ar. Cs); 164.8, 165.3 (2 C=O). Anal. Calcd for C16H15ClN2O2 (302.76): C, 63.48; H, 4.99; N, 9.25. Found: C, 63.39; H, 4.52; N, 8.96.
N-(3-(2-Chloroacetamido)-4-methoxyphenyl)benzamide (5k)
Yield: 87%, mp 178–180 °C. IR (cm−1): 3379, 3271 (NH), 1697, 1643 (C=O); 1H NMR (ppm): 3.85 (s, 3H, CH3), 4.40 (s, 2H, CH2), 7.05–8.38 (m, 8H, ArH), 9.52 (s, 1H, CONH, D2O exchangeable), 10.20 (s, 1H, CONH, D2O exchangeable). 13C NMR (ppm): 43.4 (CH2); 55.9 (CH3); 111.0, 114.9, 117.3, 126.2, 127.5, 128.3, 131.4, 132.0, 134.9, 146.0 (Ar. Cs); 164.6, 165.1 (2 C=O). Anal. Calcd for C16H15ClN2O3 (318.76): C, 60.29; H, 4.74; N, 8.79. Found: C, 60.54; H, 5.01; N, 8.27.
3-Amino-4-cyano-5-(4-phenylpiperazin-1-yl)-N-(aryl)-2-thiophene-carboxamides 6a–k
A solution of compound 3 (0.81 g, 3 mmol) and K2CO3 (0.83 g, 6 mmol) in ethanol–water (1:1) mixture (25 mL) was added portionwise to a heated solution of an appropriate 2-chloro-N-phenylacetamide 5a–k (3 mmol) in ethanol (25 mL). The reaction mixture was heated under reflux for 1 h with continuous stirring, then cooled to room temperature. The precipitated solid product was filtered, washed with ethanol (2 × 20 mL), oven-dried and crystallized from a suitable solvent.
3-Amino-4-cyano-5-(4-phenylpiperazin-1-yl)-N-phenyl-2-thiophenecarboxamide (6a)
Yield: 70%, crystallized from ethyl acetate, mp 188–189 °C. IR (cm−1): 3441, 3387, 3325 (NH2/NH), 2194 (C≡N), 1635 (C=O); 1H NMR (ppm): 3.34–3.36 (m, 4H, CH2), 3.73–3.75 (m, 4H, CH2), 6.82–7.62 (m, 10H, ArH), 6.90 (s, 2H, NH2, D2O exchangeable), 9.10 (s, 1H, NH, D2O exchangeable); 13C NMR (ppm): 47.3, 49.7 (piperazine Cs); 79.1, 85.4, 115.4, 115.7, 119.5, 120.5, 122.8, 128.3, 129.0, 139.2, 150.2, 154.8, 162.4, 164.6 (Ar. Cs, C≡N, and C=O). Anal Calcd for C22H21N5OS: C, 65.49; H, 5.25; N, 17.36. Found: C, 65.73; H, 5.34; N, 17.61.
3-Amino-N-(2-bromophenyl)-4-cyano-5-(4-phenylpiperazin-1-yl)-2-thiophenecarboxamide (6b)
Yield: 68%, crystallized from ethyl acetate, mp 210–212 °C. IR (cm−1): 3444, 3367, 3329 (NH2/NH), 2198 (C≡N), 1635 (C=O); 1H NMR (ppm): 3.35 (t, 4H, J = 5.0 Hz, CH2), 3.73 (t, 4H, J = 5 Hz, CH2), 6.82–7.67 (m, 11H, 9 ArH and NH2), 8.69 (s, 1H, NH, D2O exchangeable); 13C NMR (ppm): 47.4, 49.7 (piperazine Cs); 79.0, 85.2, 115.4, 115.7, 119.4, 119.5, 126.8, 127.5, 127.9, 129.0, 132.4, 136.6, 150.2, 154.7, 162.0, 164.7 (Ar. Cs, C≡N, and C=O). Anal Calcd for C22H20BrN5OS: C, 54.78; H, 4.18; N, 14.52. Found: C, 54.97; H, 4.22; N, 14.67.
3-Amino-N-(2-chlorophenyl)-4-cyano-5-(4-phenylpiperazin-1-yl)-2-thiophenecarboxamide (6c)
Yield: 70%, crystallized from ethyl acetate, mp 214–216 °C. IR (cm−1): 3448, 3390, 3336 (NH2/NH), 2198 (C≡N), 1635 (C=O); 1H NMR (ppm): 3.34–3.36 (m, 4H, CH2), 3.72–3.75 (m, 4H, CH2), 6.82–7.62 (m, 11H, 9ArH and NH2), 8.75 (s, 1H, NH, D2O exchangeable). 13C NMR (ppm): 47.4, 49.7 (piperazine Cs); 79.0, 85.3, 115.4, 115.7, 115.8, 119.5, 126.4, 127.3, 128.4, 129.0, 129.3, 135.2, 150.2, 154.7, 162.1, 164.7 (Ar. Cs, C≡N, and C=O). Anal. Calcd for C22H20ClN5OS: C, 60.34; H, 4.60; N, 15.99. Found: C, 60.62; H, 4.73; N, 16.26.
3-Amino-N-(4-chlorophenyl)-4-cyano-5-(4-phenylpiperazin-1-yl)-2-thiophenecarboxamide (6d)
Yield: 77%, crystallized from ethyl acetate, mp 215–216 °C. IR (cm−1): 3456, 3320, 3309 (NH2/ NH), 2194 (C≡N), 1627 (C=O); 1H NMR (ppm): 3.35–3.37 (m, 4H, CH2), 3.73–3.75 (m, 4H, CH2), 6.81–7.66 (m, 9H, ArH), 6.94 (s, 2H, NH2, D2O exchangeable), 9.22 (s, 1H, NH, D2O exchangeable); 13C NMR (ppm): 47.4, 49.7 (piperazine Cs); 79.0, 85.1, 115.4, 115.8, 119.5, 121.9, 126.4, 128.2, 129.0, 138.3, 150.2, 155.1, 162.3, 164.7 (Ar. Cs, C≡N, and C=O). Anal. Calcd for C22H20ClN5OS: C, 60.34; H, 4.60; N, 15.99. Found: C, 60.13; H, 4.75; N, 16.23.
3-Amino-4-cyano-N-(2,4-dichlorophenyl)-5-(4-phenylpiperazin-1-yl)-2-thiophene carboxamide (6e)
Yield: 68%, crystallized from ethyl acetate, mp 235–237 °C. IR (cm−1): 3429, 3398, 3325 (NH2/ NH), 2206 (C≡N), 1635 (C=O); 1H NMR (ppm): 3.34–3.36 (m, 4H, CH2), 3.74 (t, 4H, CH2), 6.81–7.65 (m, 10H, 8 ArH and NH2), 8.83 (s, 1H, NH, D2O exchangeable); 13C NMR (ppm): 47.3, 49.7 (piperazine Cs); 78.9, 85.1, 115.3, 115.7, 119.5, 127.4, 128.4, 128.7, 129.0, 129.5, 129.6, 134.5, 150.2, 154.9, 162.0, 164.8 (Ar. Cs, C≡N, and C=O). Anal. Calcd for C22H19Cl2N5OS: C, 55.94; H, 4.05; N, 14.83. Found: C, 56.23; H, 4.01; N, 15.06.
3-Amino-4-cyano-N-(4-ethylphenyl)-5-(4-phenylpiperazin-1-yl)-2-thiophenecarboxamide (6f)
Yield: 80%, crystallized from ethyl acetate, mp 180–182 °C. IR (cm−1): 3475, 3437, 3336 (NH2/ NH), 2962, 2931, 2873, 2804 (aliphatic CH), 2198 (C≡N), 1635 (C=O); 1H NMR (ppm): 1.15 (t, 3H, J = 7.6 Hz, CH3), 2.54 (q, 2H, J = 7.6 Hz, CH2), 3.34 (t, 4H, J = 5.0 Hz, CH2), 3.72 (t, 4H, J = 5.0 Hz, CH2), 6.81–7.53 (m, 9H, ArH), 6.88 (s, 2H, NH2, D2O exchangeable), 9.03 (s, 1H, NH, D2O exchangeable); 13C NMR (ppm): 15.7 (CH3); 27.6 (CH2); 47.4, 49.7 (piperazine Cs); 79.2, 85.6, 115.4, 115.7, 119.5, 120.7, 127.5, 129.0, 136.8, 138.2, 150.2, 154.5, 162.3, 164.5 (Ar. Cs, C≡N, and C=O). Anal. Calcd for C24H25N5OS: C, 66.80; H, 5.84; N, 16.23. Found: C, 67.12; H, 5.99; N, 16.59.
3-Amino-4-cyano-N-(4-methylphenyl)-5-(4-phenylpiperazin-1-yl)-2-thiophenecarboxamide (6g)
Yield: 71%, crystallized from ethyl acetate, mp 194–195 °C. IR (cm−1): 3441, 3421, 3313 (NH2/ NH), 2904, 2835 (aliphatic C–H), 2194 (C≡N), 1627 (C=O); 1H NMR (ppm): 2.24 (s, 3H, CH3), 3.36 (t, 4H, J = 5.0 Hz, CH2), 3.73 (t, 4H, J = 5.0 Hz, CH2), 6.81–7.50 (m, 9H, ArH), 6.87 (s, 2H, NH2, D2O exchangeable), 9.02 (s, 1H, NH, D2O exchangeable); 13C NMR (ppm): 20.4 (CH3); 47.4, 49.7 (piperazine Cs); 79.2, 85.6, 115.4, 115.8, 119.5, 120.7, 128.8, 129.0, 131.8, 136.7, 150.2, 154.5, 162.3, 164.5 (Ar. Cs, C≡N, and C=O). Anal. Calcd for C23H23N5OS: C, 66.16; H, 5.55; N, 16.77. Found: C, 66.47; H, 5.67; N, 16.89.
3-Amino-4-cyano-N-(3-nitrophenyl)-5-(4-phenylpiperazin-1-yl)-2-thiophenecarboxamide (6h)
Yield: 77%, crystallized from acetic acid, mp 238–240 °C. IR (cm−1): 3429, 3352, 3321 (NH2/NH), 2198 (C≡N), 1627 (C=O), 1530, 1346 (NO2); 1H NMR (ppm): 3.35–3.37 (m, 4H, CH2), 3.74–3.76 (m, 4H, CH2), 6.81–8.66 (m, 11H, 9 ArH and NH2), 9.52 (s, 1H, NH, D2O exchangeable); 13C NMR (ppm): 47.4, 49.7 (piperazine Cs); 78.8, 84.8, 114.2, 115.4, 115.8, 116.9, 119.6, 126.0, 129.0, 129.5, 140.8, 147.7, 150.2, 155.8, 162.5, 164.9 (Ar. Cs, C≡N, and C=O). Anal. Calcd for C22H20N6O3S: C, 58.92; H, 4.49; N, 18.74. Found: C, 59.34; H, 4.37; N, 19.01.
3-Amino-N-(3-benzamidophenyl)-4-cyano-5-(4-phenylpiperazin-1-yl)thiophene-2-carboxamide (6i)
Yield: 84%, mp 230–232 °C. IR (cm−1): 3464, 3410, 3367, 3336 (NH2/2 NH); 2191 (C≡N); 1662, 1639 (2 C=O). 1H NMR (ppm): 3.35–3.37 (m, 4H, CH2), 3.73–3.75 (m, 4H, CH2), 6.81–8.20 (m, 16H, 14 ArH and NH2), 9.20 (s, 1H, NH, D2O exchangeable), 10.25 (s, 1H, NH, D2O exchangeable). 13C NMR (ppm): 47.4, 49.7 (piperazine Cs); 79.1, 85.5, 113.2, 115.3, 115.4, 115.7, 116.3, 119.5, 127.6, 128.3, 128.3, 129.0, 131.4, 135.0, 139.1, 139.4, 150.2, 154.8, 162.4, 164.7, 165.4 (Ar. Cs, C≡N, and C=O). Anal. Calcd for C29H26N6O2S (522.63): C, 66.65; H, 5.01; N, 16.08. Found: C, 66.23; H, 5.15; N, 15.82.
3-Amino-N-(5-benzamido-2-methylphenyl)-4-cyano-5-(4-phenylpiperazin-1-yl)thiophene-2-carboxamide (6j)
Yield: 73%, mp 260–262 °C. IR (cm−1): 3468, 3406, 3340, 3305 (NH2/2 NH); 2198 (C≡N); 1662, 1635 (2 C=O). 1H NMR (ppm): 2.16 (s, 3H, CH3), 3.34–3.36 (m, 4H, CH2), 3.71–3.73 (m, 4H, CH2), 6.81–7.96 (m, 15H, 13 ArH and NH2), 8.81 (s, 1H, NH, D2O exchangeable), 10.21 (s, 1H, NH, D2O exchangeable). 13C NMR (ppm): 17.4 (CH3), 47.4, 49.7 (piperazine Cs), 79.2, 85.7, 115.4, 115.7, 117.7, 117.7, 118.9, 119.5, 127.6, 128.3, 129.0, 129.9, 131.4, 134.9, 136.4, 137.0, 150.2, 154.2, 162.4, 164.6, 165.3 (Ar. Cs, C≡N, and C=O). Anal. Calcd for C30H28N6O2S (536.65): C, 67.14; H, 5.26; N, 15.66. Found: C, 67.30; H, 5.12; N, 15.87.
3-Amino-N-(5-benzamido-2-methoxyphenyl)-4-cyano-5-(4-phenylpiperazin-1-yl)thiophene-2-carboxamide (6 k)
Yield: 87%, mp 264–266 °C. IR (cm−1): 3452, 3390, 3321 (NH2/2 NH); 2210 (C≡N); 1666, 1635 (2 C=O). 1H NMR (ppm): 3.35–3.37 (m, 4H, CH2), 3.73–3.75 (m, 4H, CH2), 3.84 (s, 3H, OCH3), 6.82–8.08 (m, 15H, 13 ArH and NH2), 8.25 (s, 1H, NH, D2O exchangeable), 10.16 (s, 1H, NH, D2O exchangeable). 13C NMR (ppm): 47.3, 49.7 (piperazine Cs), 56.0 (OCH3), 79.1, 85.7, 110.8, 115.3, 115.7, 115.7, 116.6, 119.5, 126.9, 127.5, 128.3, 129.0, 131.3, 131.9, 134.9, 146.5, 150.2, 154.2, 161.6, 164.4, 165.0 (Ar. Cs, C≡N, and C=O). Anal. Calcd for C30H28N6O3S (552.65): C, 65.20; H, 5.11; N, 15.21. Found: C, 64.83, H, 5.35, N, 14.92.
Ethyl 3-amino-4-cyano-5-(4-phenylpiperazin-1-yl)thiophene-2-carboxylate (7)
A solution of ethyl chloroacetate (0.37 g, 0.32 mL, 3 mmol) in absolute ethanol (25 mL) was added to a solution of compound 3 (0.81 g, 3 mmol), and potassium carbonate (0.83 g, 6 mmol) in water–ethanol (1:1) mixture (25 mL). The reaction mixture was heated under reflux with continuous stirring for 1 h. After cooling, the mixture was poured onto water (50 mL). The precipitated solid was filtered, oven-dried, and crystallized from ethanol.
Yield: 62%, mp 137–139 °C. IR (cm−1): 3448, 3336 (NH2), 2974, 2893, 2831 (aliphatic C–H), 2202 (C≡N), 1647 (C=O); 1H NMR (ppm): 1.22 (t, 3H, J = 7.08, CH3), 3.32 (t, 4H, J = 5.10, CH2), 3.73 (t, 4H, J = 5.06, CH2), 4.15 (q, 2H, J = 7.08, CH2), 6.66 (s, 2H, NH2, D2O exchangeable), 6.81–7.26 (m, 5H, ArH); 13C NMR (ppm): 14.4 (CH3); 47.3, 49.4 (piperazine Cs); 59.3 (CH2); 77.9, 82.2, 115.3, 115.7, 119.4, 129.0, 150.1, 155.4, 162.7, 166.1 (Ar. Cs, C≡N, and C=O). Anal. Calcd for C18H20N4O2S (356.44): C, 60.65; H, 5.66; N, 15.72. Found: C, 60.38; H, 5.78; N, 15.98.
Results and discussion
The previously reported sequential one-pot procedure developed by Thomae et al.(2009) consisted of reacting ketene dithioacetal 1 with an appropriate secondary amine to give the intermediate ketene N,S-acetal A, which upon treatment with sodium sulfide afforded the enethiolate derivative B. Further reaction of B with a number of activated halides, and subsequent base-catalyzed cyclization of the resulting intermediate C gave rise to the thiophene derivatives D (Scheme 1).
In the aforementioned method, the conversion of the ketene N,S-acetal A to the corresponding enethiolate intermediate B liberated a molar equivalent of sodium methanethiolate which competed for the activated halide in the subsequent step, thereby reducing the yield of the intended thiophene derivative D (Scheme 1). Although this problem has been brought under control using two molar equivalents of the activated halides (Thomae et al. 2009), this procedure suffered from a major economic drawback because of the use of excess of the expensive halides.
In an effort to optimize the reaction conditions, elimination of methanethiolate side-product from the reaction medium was supposed to be an appropriate manoeuvre. This had been achieved by isolating the enethiolate derivative B in its enethiol form. It was hypothesized that this extra step would improve both the reaction economy and the purity of the isolated products in the downstream reactions.
Herein, the synthetic pathway to the target thiophene derivatives 4–7 is shown in Schemes 2, 3. As a model example, the enethiolate derivative 2 was prepared via the reaction of compound 1 with 1-phenylpiperazine following previously procedure (Thomae et al. 2009). Initial attempts to isolate this intermediate in its enethiol form (compound 3) consisted of acidification of the cooled reaction mixture with aqueous acetic acid or dil. HCl. Although this trial furnished a solid in a satisfactory yield, it suffered from lack of reproducibility from batch to batch with a sticky brown mass frequently forming during the isolation process. A product of higher purity and of higher yield (85%) has been achieved via replacement of the aqueous acidic solution by a saline acetic acid-sodium acetate buffer, Scheme 2.
The IR spectrum of compound 3 revealed the presence of two bands at 2194 and 2164 cm−1 assignable to the two C≡N groups. Its 1H NMR spectrum showed two triplet signals at δ 3.22 and 4.28 ppm of the piperazine ring protons; in addition to multiplet signals between δ 6.86–7.30 ppm corresponding to the five aromatic hydrogens of the phenyl ring. The presence of phenylpiperazine moiety was also confirmed by 13C NMR analysis, which showed the signals of the aliphatic carbons of piperazine ring in addition to a signal at δ 195.4 ppm attributable to the C=S carbon.
With the aim to explore the most suitable reaction conditions for the synthesis of the target thiophene derivatives 4a–g, we tried to synthesize the derivative 4a by reacting compound 3 with only 1 molar equivalent of phenacyl bromide in DMF using potassium carbonate as a base. However, this trial was unsuccessful, imposing further optimization of the reaction conditions using different solvent/base combinations (Table 1). The results revealed that when the reaction was carried out in DMF as a solvent in presence of a strong base such as NaOH, 4a was obtained in about 43% yield, however, the use of TEA as a base was found to be more reproducing (67% yield). Switching the solvent to ethanol gave a comparable yield (64%). Surprisingly, ethanol/K2CO3 combination yielded compound 4a in 59% yield. The optimum solvent/base combination for the preparation of 4a was found to be ethanol–water (3:1) mixture and K2CO3 as a base (74% yield).

In this work, the new compounds 4a–d were prepared following the procedure reported by Thomae et al. (2009) (method A) as well as by our optimized procedure (method B). The yields obtained by the two methods were compared and presented in Table 2. The results showed that compounds 4a–d prepared following method B were obtained in higher yields ranging from 71 to 77%. Accordingly, compounds 4e–g were prepared following method B and were isolated in high purity and high yields.

The structure of compounds 4a–g was confirmed by IR, 1H NMR and 13C NMR spectroscopic analyses. Their IR spectra showed two bands in the range of 3394–3259 cm−1 indicating NH2 function. In addition, bands indicating C≡N group were observed in the range of 2202–2198 cm−1. It is worth to mention that, the bands corresponding to C=O group were observed at a relatively low wavenumber (1612–1581 cm−1), which might be attributed to the high conjugation of C=O with the neighbouring aromatic ring systems as well as the possibility of hydrogen bonding formation with the adjacent NH2 protons (Naguib and El-Nassan 2016). The 1H NMR spectra of compounds 4a–g showed a broad exchangeable singlet signal at δ 7.88–8.00 ppm corresponding to NH2 protons. In the 13C NMR spectra of these compounds, the C=O carbons appeared at δ 180.7–184 ppm.
Similarly, the thiophene-2-carboxamide derivatives 6a–k were synthesized according to the modified procedure (method B) from compound 3 and 2-chloro-(N-aroyl)-acetamides 5a–k (Al-Nadaf et al. 2010; Kumar et al. 2014; Ma et al. 2011; Monforte et al. 2014; Rajak et al. 2012; Yu and Cai 2003) and were isolated in moderate to high yields (68–87%). It is noteworthy that compounds 6a–k were not reported earlier and were not accessible via the reported procedures (Scheme 3).
The IR spectra of compounds 6a–k showed NH2/NH bands in the range 3475–3305 cm−1. Moreover, the C≡N and amidic C=O functions appeared at 2210–2191 cm−1 and 1639–1627 cm−1, respectively. The 1H NMR spectra of these compounds showed two exchangeable singlet signals at δ 6.81–7.02 ppm and 8.25–9.52 ppm corresponding to NH2 and NH protons, respectively. The 13C NMR spectra of the compounds showed the expected number of signals for all compounds. Aliphatic CH2 carbons of the piperazine ring gave two signals between δC 47.3–49.7 ppm.
Likewise, the ester derivative 7 was prepared in 62% yield from compound 3 and ethyl chloroacetate following method B. The IR spectrum of compound 7 revealed two bands of NH2 at 3448 and 3336 cm−1, in addition to C≡N and C=O bands at 2202 and 1647 cm−1, respectively. Its 1H NMR spectrum showed triplet and quartet signals at δ 1.22 and 4.15 ppm corresponding to CH3CH2 protons, along with an exchangeable NH2 singlet signal at δ 6.66 ppm. Besides, its 13C NMR spectrum showed two signals at δ 14.4 and 59.3 ppm assignable to CH3CH2 carbons and a signal corresponding to C=O carbon at δ 166.1 ppm.
Conclusion
In summary, a modification of the procedure reported by Thomae et al. for the synthesis of tetrasubstituted thiophenes 4a–g, 6a–k and 7 was reported. Optimization of the reaction conditions comprises the isolation of the enethiol derivative 3, the use of K2CO3 or TEA as a base, and the use of ethanol, water, or a mixture of them as a solvent. The described modification eliminated the need for the use of two molar equivalents of the active halide, making it an atom economic procedure. Besides, the desired products were obtained in higher yields and in purer form.
References
Ahmed GA (2008) Heterocyclic synthesis with thiophene-2-carboxamide. Phosphorus Sulfur Silic Relat Elem 183:74–81. https://doi.org/10.1080/10426500701557005
Al-ghorbani MABB, Mamatha SV, Khanum SA, Al-ghorbani M (2015) Piperazine and morpholine: synthetic preview and pharmaceutical applications. Res J Pharm Technol 8(5):611–628. https://doi.org/10.5958/0974-360X.2015.00100.6
Al-Nadaf A, Sheikha GA, Taha MO (2010) Elaborate ligand-based pharmacophore exploration and QSAR analysis guide the synthesis of novel pyridinium-based potent β-secretase inhibitory leads. Bioorg Med Chem 18(9):3088–3115. https://doi.org/10.1016/j.bmc.2010.03.043
Alves MA, Barreiro EJ, Suzana M, Moreira A (2014) 3-Aminothiophene-2-acylhydrazones: non-toxic, analgesic and anti-inflammatory lead-candidates. Molecules 19:8456–8471. https://doi.org/10.3390/molecules19068456
Cheeseright TJ, Holm M, Lehmann F, Luik S, Gottert M, Melville JL, Laufer S (2009) Novel lead structures for p38 MAP kinase via FieldScreen virtual screening. J Med Chem 52:4200–4209. https://doi.org/10.1021/jm801399r
Court JJ, Poisson C, Ardzinski A, Bilimoria D, Chan L, Chandupatla K et al (2016) Discovery of novel thiophene-based, thumb pocket 2 allosteric inhibitors of the Hepatitis C NS5B polymerase with improved potency and physicochemical profiles. J Med Chem 59:6293–6302. https://doi.org/10.1021/acs.jmedchem.6b00541
Farhat MF, Mezoughi A, El-saghier A (2016) Utilization of 2-ylidene-4-thiazolidinones in synthesis of heterocyclic compounds part (II): transformation of (4-oxo-3-phenyl-1,3-thiazolidin-2-ylidene)malononitrile to 3-aminothiophene derivatives. Asian J Chem 28:1823–1827. https://doi.org/10.14233/ajchem.2016.19850
Gompper R, Töpfl W (1962) Carbonsäurederivate, V Substituierte Dithiocarbonsäuren und Ketenmercaptale. Chem Ber 95(12):2861–2870. https://doi.org/10.1002/cber.19620951206
Gramec D, Peterlin Mašič L, Sollner Dolenc M (2014) Bioactivation potential of thiophene-containing drugs. Chem Res Toxicol 27(8):1344–1358. https://doi.org/10.1021/tx500134g
Gruner M, Böttcher G, Gewald K (2008) Heterocondensed thiophenes and thiazoles by Thorpe-Ziegler cyclization. J Heterocycl Chem 45(4):1071–1076. https://doi.org/10.1002/jhet.5570450419
Kumar D, Khare G, Kidwai S, Tyagi AK, Singh R, Rawat DS (2014) Novel isoniazid–amidoether derivatives: synthesis, characterization and antimycobacterial activity evaluation. Med Chem Commun 6:131–137. https://doi.org/10.1039/C4MD00288A
Li JJ (2009) Name reactions, 5th edn. https://doi.org/10.1007/s13398-014-0173-7.2
Ma L, Li S, Zheng H, Chen J, Lin L, Ye X et al (2011) Synthesis and biological activity of novel barbituric and thiobarbituric acid derivatives against non-alcoholic fatty liver disease. Eur J Med Chem 46(6):2003–2010. https://doi.org/10.1016/j.ejmech.2011.02.033
Monforte A-M, Ferro S, De Luca L, Lo Surdo G, Morreale F, Pannecouque C et al (2014) Design and synthesis of N1-aryl-benzimidazoles 2-substituted as novel HIV-1 non-nucleoside reverse transcriptase inhibitors. Bioorg Med Chem 22(4):1459–1467. https://doi.org/10.1016/j.bmc.2013.12.045
Naguib BH, El-Nassan HB (2016) Synthesis of new thieno[2,3-b]pyridine derivatives as pim-1 inhibitors. J Enzyme Inhib Med Chem. https://doi.org/10.3109/14756366.2016.1158711
Papakyriakou A, Kefalos P, Sarantis P, Tsiamantas C, Xanthopoulos KP, Vourloumis D, Beis D (2014) A Zebrafish in vivo phenotypic assay to identify 3-aminothiophene-2-carboxylic acid-based angiogenesis inhibitors. Assay Drug Dev Technol 12(9):527–535. https://doi.org/10.1089/adt.2014.606
Rajak H, Kumar P, Parmar P, Thakur BS, Veerasamy R, Sharma PC et al (2012) Appraisal of GABA and PABA as linker: design and synthesis of novel benzamide based histone deacetylase inhibitors. Eur J Med Chem 53:390–397. https://doi.org/10.1016/j.ejmech.2012.03.058
Romagnoli R, Baraldi PG, Carrion MD, Cara CL, Cruz-lopez O, Salvador MK et al (2012) Synthesis and biological evaluation of 2-amino-3-(4-chlorobenzoyl)- 4-[(4-arylpiperazin-1-yl)methyl]-5-substituted-thiophenes. Effect of the 5-modification on allosteric enhancer activity at the A1 adenosine receptor. J Med Chem 55:7719–7735. https://doi.org/10.1021/jm3007504
Sarker D, Ang JE, Baird R, Kristeleit R, Shah K, Moreno V et al (2015) First-in-human Phase I study of Pictilisib (GDC-0941), a potent pan-class I phosphatidylinositol-3-kinase (PI3K) inhibitor, in patients with advanced solid tumors. Clin Cancer Res 21:77–86. https://doi.org/10.1158/1078-0432.CCR-14-0947.First-in-human
Singh K, Siddiqui HH, Shakya P, Bagga P (2015) Piperazine—a biologically active scaffold. https://doi.org/10.13040/IJPSR.0975-8232.6(10).4145-58
Thomae D, Perspicace E, Henryon D, Xu Z, Schneider S, Hesse S et al (2009) One-pot synthesis of new tetrasubstituted thiophenes and selenophenes. Tetrahedron 65(50):10453–10458. https://doi.org/10.1016/j.tet.2009.10.021
Walayat K, Mohsin N, Aslam S, Ahmad M (2019) An insight into the therapeutic potential of piperazine-based anticancer agents. https://doi.org/10.3906/kim-1806-7
Workman P, Collins I (2013) Modern cancer drug discovery: Integrating targets, technologies, and treatments for personalized medicine. In: Neidle S, Denny W, Rewcastle W (eds) Cancer drug design and discovery, 2nd edn. https://doi.org/10.1016/B978-0-12-396521-9.00001-2
Yu S-Y, Cai Y-X (2003) Synthesis of polysubstituted pyrimidines from ketene dithioacetals using KF/Al2O3 catalyst. Synth Commun 33:3989–3995. https://doi.org/10.1081/SCC-120026325
Zhang L, Dong J, Xu X, Liu Q (2016) Chemistry of ketene N, S-acetals: an overview. Chem Rev 116:287–322. https://doi.org/10.1021/acs.chemrev.5b00360
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
El-Meligie, S.E.M., Khalil, N.A., El-Nassan, H.B. et al. Efficient synthesis of new 3-amino-4-cyanothiophene derivatives. Chem. Pap. 74, 2491–2500 (2020). https://doi.org/10.1007/s11696-020-01070-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01070-z