Abstract
The sleeve gastrectomy (SG) is the most popular weight loss procedure in the USA. Despite its popularity, little is definitively known about the variables that correlate with weight loss. We performed a literature search to find studies that reported variables that correlated with weight loss following SG. Forty-eight articles were identified and included. These articles covered 36 different factors predictive of weight loss while only including five predictive models. Only 12.5% of multivariate analyses evaluated sufficiently reported their results. The factors that predict weight loss following SG cannot be concluded due to the inconsistency in reporting and the methodological flaws in analysis. Reporting factors that predict weight loss should be standardized, and methods should be changed to allow physicians to use the data presented.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bariatric surgery is rapidly gaining popularity across the USA and across the world. Despite this, there is a relative paucity of data showing what correlates with success and failure after surgery. This work has been previously done for Roux-en-Y gastric bypass but not for SG [1]. Personalized weight loss predictions are of utmost importance for bariatric surgeons due to the large unexplained variances observed in published studies. Weight loss outcomes, the primary outcome for most patients undergoing bariatric surgery, and weight loss predictions have the power to help surgeons educate the patients about procedures best suited for them or make better care plans for the specific patient. This can increase patient satisfaction following bariatric surgery and decrease revisional patients and non-responders.
Methods
Articles were identified by searching PubMed, MEDLINE, and the Cochrane Library using the search query “Sleeve gastrectomy” AND (model OR influence OR mapping OR prediction OR predictive OR predicts OR predictor OR predict OR predictors) for PubMed and similar queries for MEDLINE and the Cochrane Library. Additionally, the references of each article were reviewed for inclusion.
Papers were identified for review by title and abstract. Articles underwent full-text review if they were done on human subjects and referenced, in the abstract, the effect of any variable on weight loss. After full-text review, studies were included if they presented factors that affected weight loss at or greater than 6 months. Studies with more than one bariatric surgery included in the analysis were included if independent statistical analysis was performed for each surgery or if at least 50% of subjects underwent SG.
Each study was independently reviewed twice to collect important information like authors, number of subjects, statistical test performed, significant variables, non-significant variables, and if presented a model to predict weight loss.
Each study was also classified as univariate or multivariate depending on which statistical tests were performed and adequately reported. If both univariate and multivariate methods are reported, the significant factors are reported from the multivariate analysis.
Results
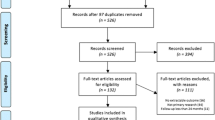
Overall, 1487 studies were identified in our initial search. We eliminated 1373 articles because they did not meet our inclusion criteria, leaving 114 for full-text review. Additionally, from the references of these 114 studies, 6 studies were identified as meeting the criteria for full-text review.
Of the 120 papers that met criteria for full-text review, 72 were excluded because they did not meet inclusion criteria leaving 48 studies for this review. In these studies, eight different statistical methods of identifying variables that affected or were correlated with weight loss were used. Of all studies reviewed, only five presented a model (two multivariate and three univariate) with two other studies presenting only a graphical explanation of their model without any numerical representation and six others presenting an incomplete model for predicting weight loss. Of 14 studies that reported results of a multilinear regression, only two reported the entire mathematical model with coefficients and variables.
There were 16 studies that used multivariate models to find variables that affect weight loss and 32 that used univariate methods to find variables that affect weight loss. Details of all 48 studies can be found in Table 1. From the 48 studies included in this review, 36 different variables were found to correlate with weight loss. All 36 variables and the studies that found them to be significant and non-significant can be found in Table 2. The majority of these variables [45] were analyzed in five or fewer analysis. There were also numerous other variables that were not found to affect weight loss as seen in Table 1. Of the 16 multivariate analyses, only 2 fully reported their finding in a way that they could be verified by other researchers.
Of the variables that affect weight loss age, BMI, bougie size, DM, gender, HTN, short-term postoperative weight loss, preop weight loss, and sleep apnea (SA) were the only variables studied in more than five studies. Of these nine variables, only six were studied over five times in a multivariate analysis. All variables found to be significant in multivariate studies, and the studies that found them to be significant and non-significant can be found in Table 3.
BMI was found to be a negative predictor of weight loss. Seventeen studies investigated BMI as a predictor of weight loss. Twelve of these studies used multivariate analysis to determine the effect of BMI on weight loss. Many methods were reported including the β coefficient from the regression analysis, the odds ratio of failing for every point BMI gained, and viewing BMI as a categorical variable in a regression analysis.
Of the 12 multivariate studies that found BMI to be a significant predictor, 9 found it to be a negative predictor and 2 found it to be a positive predictor. In one of the two studies that BMI was a positive predictor, the model predicted BMI reduction instead of EWL% [23]. When predicting EWL%, the higher the BMI of the patient, the more weight they have to lose to achieve the same EWL%, and often even though larger patients are predicted to lose less EWL%, they often lose more weight.
Cottam et al. reported two models in their paper where they modeled both EWL% and BMI reduction on the same population and BMI was a strong negative predictor for EWL% and a strong positive predictor for BMI reduction [49]. The only other study where BMI was positively associated with weight had a sample size of seven sleeve patients and so is likely due to random error [3]. There was also one study that found that baseline BMI was not predictive of weight loss [27]. However, in this study, the authors included postoperative weight loss which was far more predictive of weight loss than baseline BMI, and so, BMI, while important, did not achieve statistical significance.
Bougie size was studied in 8 of the 49 papers with 3 papers finding that it did affect weight loss and 5 finding that it did not affect weight loss. The range of bougie size was fairly large (32–60) across the studies, and each study compared different sizes. In the three studies that found bougie size to be significant, the bougie size comparisons were 32 to 40, < 36 to > 44, and 32–36 to 38–60, and in the five that found it to not affect weight loss, the bougie comparisons were 32 to 36, 33 to 42, 32 to 42, 40 to 60, and 32 to 44 [7, 11, 18, 24,25,26, 40, 47].
However, these studies that found bougie size to be significant were somewhat misleading. Hady et al. compared bougie sizes 32 and 40 [7]. The initial BMI of the two groups was significantly different at the 0.001 level. Patients in the bougie size of 40 groups started with the mean BMI 5.46 points higher, and at 6 months, the patients with a bougie size of 40 have a mean BMI that is 5.05 higher. This shows that those in the larger bougie size group had a greater BMI reduction; however, since only %EWL, proportional measure, is reported, they appear to lose less weight even though they had a greater BMI reduction.
Discounting this study due to fault in their conclusion, there are two studies that found bougie size to be significant. However, Ellatif et al. only found bougie size to be significant 4 years after surgery. At every follow-up point before 4 years, it was non-significant. At 1 year, the p value was 0.3 [26]. So, during the typical postoperative weight loss period, only one study found bougie size to be significant and the difference was from 32 to 36, and 38–60. They did not report on how many patients received each specific bougie size [40].
Age was studied in 18 different papers. Of these 18 papers, 14 used multivariate analysis to look at the impact of age on weight loss. Of these 14, there was an even split with 7 finding age to affect weight loss and 7 finding that it does not affect weight loss. The studies that did find that age affects weight loss were consistent in reporting age as a negative predictor.
Diabetes was studied in 12 different papers, 10 of which used a form of multivariate analysis. It was found to be a negative predictor in the three multivariate analysis where it was significant and it was not found to be significant in 7 multivariate analysis.
Gender was studied in 16 different papers, 12 of which used a form of multivariate analysis. It was found to be predictive twice out of the 12 studies, and in both of the studies, female gender was found to be predictive of lower weight loss.
Hypertension was studied in eight different papers, all of which were multivariate analysis. It was found to be predictive three times out of the eight studies and in the two studies which reported whether it was a positive or negative predictor; it was a negative predictor. The other study never specified the effect (positive or negative) of HTN on weight loss.
Preoperative weight loss was studied in seven different papers, three of which were multivariate analysis. It was found to be predictive only once out of the three multivariate studies. It was found to be a positive predictor of weight loss in that study. However, in the other two univariate analysis that found it to significantly affect weight loss, it was found to be a positive predictor in one and a negative predictor in the other.
Short-term postoperative weight loss was studied in seven different papers, three of which were multivariate analysis. In all seven studies, it was found to be predictive of weight loss following surgery. The correlation was found as early as 1 month, but the exact best time following surgery varied from paper to paper.
SA was studied in six different papers, all of which were multivariate analysis. It was found to be predictive only once and was a negative predictor. However, all other times, SA was not found to be a predictor of weight loss. A more detailed explanation of why SA is unlikely to be truly predictive of weight loss can be found in Supplementary Materials File 2.
Discussion
While bariatric surgery is not a new field and the sleeve gastrectomy is a well-established procedure within the specialty, there is little known about why patients fail to achieve adequate weight loss after the surgery. Predictive modeling can help to mediate this problem by finding correlation between variables and future weight loss. Alarmingly, only two papers give us enough information to replicate their studies.
This foreknowledge gained through predictive modeling can help doctors immensely as once these problem patients are identified, doctors can start a more targeted approach and warn the patient of the risks of inadequate weight loss. There are two general times to make predictions: the first is presurgery based on the patient’s baseline demographics, and the other is in the short term following the procedure based on the patient’s weight loss curve and the patient’s demographics.
Preoperative predictions regarding the SG are particularly useful since the SG has been shown on average to be a less effective procedure in terms of weight loss when compared to the Roux-en-Y gastric bypass or the duodenal switch. This information is also of use if the surgeon or patient still feels that the SG is the right procedure for them. If they know that the patient has a high likelihood of not completing their goals, they can make a more targeted care plan for the patient. This could include many things such as more follow-up visits, more recommended exercise, phone calls to check up on them, and their nutrition. If a patient and surgeon know before the surgery how much weight the patient is expected to lose, they can consider other surgeries or make a more targeted approach to follow up care so that the patient can succeed.
Postoperative predictions are somewhat less useful as a patient has already undergone the procedure and so cannot choose to start with a more invasive procedure, or with a more targeted care plan. However, they allow surgeons to identify non-responders early in the postoperative treatment and change the individual treatment to better suit the patient. This information can help the surgeon make a targeted care program for the patient still within the typical weight loss window of about the first year.
There are many different ways to predict weight loss following the SG. In simple terms, these tests can be broken into two categories: univariate and multivariate analysis. Univariate analysis like t tests are easily understood and easily misinterpreted. These types of tests very often look promising but are subject to underlying causes and differences in group demographics. Multivariate analysis on the other hand can be somewhat harder to interpret; however, they consider all variables at once which allows the effect of group demographic differences to be mitigated as long as the confounding variable is measured and entered into the model. Multivariate analyses also allow for interacting variables to be more fully studied.
While neither univariate or multivariate analysis will prove causation in SG weight loss prediction, multivariate analysis offers the tools to make prediction models which account for many measurable variables.
These prediction models while not based upon a causational relationship can be very useful in a bariatric center. Patients should be informed before surgery of a personalized weight loss expectation for each procedure that they consider. This will help them choose a procedure appropriate to them when balanced with complication rates or choose a more targeted care plan to help them succeed with the SG. Also, in the early postoperative period, these models can help surgeons identify patients who will not meet their weight loss goals and try preventative measures to help their patients succeed.
Overall, these studies add to the understanding about what variables may correlate with weight loss following the sleeve gastrectomy. There are many variables that are hard to judge if they truly correlate with weight loss because of reporting practice and statistical methods. To rectify this, we recommend a change in how variables are reported so that in future studies, this effect can be measured more accurately.
In some multivariate studies, such as the one reported by Goldenshluger et al., the results are reported as the variables that affect weight loss and an associated p value [30]. This, while useful, fails to tell the reader the true impact of the results. The real question that needs to be answered is to what extent does that variable affect weight loss and in which direction, positive or negative. Also, in six studies, a multilinear regression is reported with all of the variables and beta coefficients; however, they do not report the constant. Without this, the model cannot be validated by other researchers and the entirety of their results is ineffective in a clinical setting.
We recommend reporting the entire model as did Cottam et al. and Goulart et al. so that surgeons can apply these findings in their own practices to improve bariatric care [8, 49]. These studies reported their entire models in the form %EWL = c + B1V1 + B2V2 + … + BiVi where c is a constant from linear regression output, i is the number of significant variables, each V is a different variable labeled 1-I, and each B is the beta coefficient associated with that variable. Another way which would also be equally acceptable is to present the model in a table. An example of an acceptable table can be found in Supplementary Materials File 1. That value c in the equation above is not multiplied by any variable but is necessary for the model to be validated by others.
We also recommend a decrease in the amount of studies which make their final conclusion based upon univariate comparisons. While there are numerous reasons to make univariate comparisons, which are valid and sometimes necessary in bariatric surgery, chopping up the population into subsets over and over again reduces the ability to make accurate conclusions due to the increased likelihood of a type 1 error [50]. If five tests are performed, then there is a 22% chance that incorrectly reports a difference. If you run eight tests, there is a 33% chance that at least one is reported incorrectly reported as significant. A more detailed mathematical explanation can be found in the Supplementary Materials File 2. This high type 1 error rate is why there is need for repeated testing by independent studies to determine the likelihood of a variable affecting weight loss. While this error is present in both univariate and multivariate analysis, univariate comparisons often show differences that are better ascribed to other measured variables where multivariate analysis mitigates this effect.
We also recommend more studies to use interacting variables in their models. It often is true that one variable’s effect on the outcome is mediated by another variable. An example of this would be temperature. The temperature outside does affect how hot people feel, but the degree of the response is affected by the humidity. The same thing can happen in weight loss predictions, for example, Cottam et al. found that DM and HTN were interacting variables in predicting weight loss. HTN and DM individually predicted some decrease in weight loss, but together, the effect was greater than the added effects of the individual comorbidities.
Conclusion
Early postoperative weight loss and baseline BMI are highly correlated with long-term weight loss and should be considered in future weight loss prediction models. All predictive models should be completely reported so that they can be easily validated in other research settings. Current reporting practices are inadequate for continued collaborative research and must change to allow other researcher to use and validate their results.
References
Livhits M, Mercado C, Yermilov I, et al. Preoperative predictors of weight loss following bariatric surgery: systematic review. Obes Surg. 2012;22(1):70–89. https://doi.org/10.1007/s11695-011-0472-4.
Nagao Y, Diana M, Vix M, et al. Age impact on weight loss and glycolipid profile after laparoscopic sleeve gastrectomy: experience with 308 consecutive patients. Surg Endosc. 2014;28(3):803–10. https://doi.org/10.1007/s00464-013-3261-4.
Gras-Miralles B, Haya JR, Moros JM, et al. Caloric intake capacity as measured by a standard nutrient drink test helps to predict weight loss after bariatric surgery. Obes Surg. 2014;24(12):2138–44. https://doi.org/10.1007/s11695-014-1306-y.
Contreras JE, Santander C, Court I, et al. Correlation between age and weight loss after bariatric surgery. Obes Surg. 2013;23(8):1286–9. https://doi.org/10.1007/s11695-013-0905-3.
Jambhekar A, Maselli A, Robinson S, et al. Demographics and socioeconomic status as predictors of weight loss after laparoscopic sleeve gastrectomy: a prospective cohort study. Int J Surg. 2018;54(Pt A):163–9. https://doi.org/10.1016/j.ijsu.2018.04.025.
Figura A, Ahnis A, Stengel A, et al. Determinants of weight loss following laparoscopic sleeve gastrectomy: the role of psychological burden, coping style. and Motivation to Undergo Surgery J Obes. 2015;2015:626010. https://doi.org/10.1155/2015/626010.
Hady HR, Olszewska M, Czerniawski M, et al. Different surgical approaches in laparoscopic sleeve gastrectomy and their influence on metabolic syndrome: a retrospective study. Medicine (Baltimore). 2018 Jan;97(4):e9699. https://doi.org/10.1097/MD.0000000000009699.
Goulart A, Leão P, Costa P, et al. Doctor, how much weight will I lose?—a new individualized predictive model for weight loss. Obes Surg. 2016;26(6):1357–9. https://doi.org/10.1007/s11695-016-2134-z.
Sherman WE, Lane AE, Mangieri CW, et al. Does preoperative weight change predict postoperative weight loss after laparoscopic sleeve gastrectomy? Bariatr Surg Pract Patient Care. 2015;10(3):126–9. https://doi.org/10.1089/bari.2015.0023.
Major P, Droś J, Kacprzyk A, et al. Does previous abdominal surgery affect the course and outcomes of laparoscopic bariatric surgery? Surg Obes Relat Dis. 2018;14(7):997–1004. https://doi.org/10.1016/j.soard.2018.03.025.
Hawasli A, Jacquish B, Almahmeed T, et al. Early effects of bougie size on sleeve gastrectomy outcome. Am J Surg. 2015;209(3):473–7. https://doi.org/10.1016/j.amjsurg.2014.10.011.
Manning S, Pucci A, Carter NC, et al. Early postoperative weight loss predicts maximal weight loss after sleeve gastrectomy and roux-en-Y gastric bypass. Surg Endosc. 2015;29(6):1484–91. https://doi.org/10.1007/s00464-014-3829-7.
Chew CAZ, Tan IJ, Ng HJH, et al. Early weight loss after laparoscopic sleeve gastrectomy predicts midterm weight loss in morbidly obese Asians. Surg Obes Relat Dis. 2017;13(12):1966–72. https://doi.org/10.1016/j.soard.2017.05.016.
Obeidat F, Shanti H. Early weight loss as a predictor of 2-year weight loss and resolution of comorbidities after sleeve gastrectomy. Obes Surg. 2016 Jun;26(6):1173–7. https://doi.org/10.1007/s11695-015-1903-4.
Janse Van Vuuren MA, Strodl E, White KM, et al. Emotional food cravings predicts poor short-term weight loss following laparoscopic sleeve gastrectomy. Br J Health Psychol. 2018;23:532–43. https://doi.org/10.1111/bjhp.12302.
Philouze G, Voitellier E, Lacaze L, et al. Excess body mass index loss at 3 months: a predictive factor of long-term result after sleeve gastrectomy. J Obes. 2017;2017:2107157. https://doi.org/10.1155/2017/2107157.
Perrone F, Bianciardi E, Benavoli D, et al. Gender influence on long-term weight loss and comorbidities after laparoscopic sleeve gastrectomy and roux-en-Y gastric bypass: a prospective study with a 5-year follow-up. Obes Surg. 2016;26(2):276–81. https://doi.org/10.1007/s11695-015-1746-z.
Garay M, Balagué C, Rodríguez-Otero C, et al. Influence of antrum size on gastric emptying and weight-loss outcomes after laparoscopic sleeve gastrectomy (preliminary analysis of a randomized trial). Surg Endosc. 2018;32(6):2739–45. https://doi.org/10.1007/s00464-017-5972-4.
Sioka E, Tzovaras G, Oikonomou K, et al. Influence of eating profile on the outcome of laparoscopic sleeve gastrectomy. Obes Surg. 2013;23(4):501–8. https://doi.org/10.1007/s11695-012-0831-9.
Bekheit M, Abdel-Baki TN, Gamal M, et al. Influence of the resected gastric volume on the weight loss after laparoscopic sleeve gastrectomy. Obes Surg. 2016;26(7):1505–10. https://doi.org/10.1007/s11695-015-1981-3.
Salamat A, Afrasiabi MR, Lutfi RE. Is a “retained fundus” seen on postoperative upper gastrointestinal series after laparoscopic sleeve gastrectomy predictive of inferior weight loss? Surg Obes Relat Dis. 2017;13(7):1145–51. https://doi.org/10.1016/j.soard.2017.01.032.
Angrisani L, Cutolo PP, Buchwald JN, et al. Laparoscopic reinforced sleeve gastrectomy: early results and complications. Obes Surg. 2011;21(6):783–93. https://doi.org/10.1007/s11695-011-0400-7.
Steinbeisser M, McCracken J, Kharbutli B. Laparoscopic sleeve gastrectomy: preoperative weight loss and other factors as predictors of postoperative success. Obes Surg. 2017;27(6):1508–13. https://doi.org/10.1007/s11695-016-2520-6.
Spivak H, Rubin M, Sadot E, et al. Laparoscopic sleeve gastrectomy using 42-French versus 32-French bougie: the first-year outcome. Obes Surg. 2014;24(7):1090–3. https://doi.org/10.1007/s11695-014-1199-9.
Weiner RA, Weiner S, Pomhoff I, et al. Laparoscopic sleeve gastrectomy—influence of sleeve size and resected gastric volume. Obes Surg. 2007;17(10):1297–305.
Abd Ellatif ME, Abdallah E, Askar W, et al. Long term predictors of success after laparoscopic sleeve gastrectomy. Int J Surg. 2014;12(5):504–8. https://doi.org/10.1016/j.ijsu.2014.02.008.
Cottam A, Billing J, Cottam D, et al. Long-term success and failure with SG is predictable by 3 months: a multivariate model using simple office markers. Surg Obes Relat Dis. 2017;13(8):1266–70. https://doi.org/10.1016/j.soard.2017.03.016.
Dilektasli E, Erol MF, Cayci HM, et al. Low educational status and childhood obesity associated with insufficient mid-term weight loss after sleeve gastrectomy: a retrospective observational cohort study. Obes Surg. 2017;27(1):162–8. https://doi.org/10.1007/s11695-016-2273-2.
Holsen LM1, Davidson P, Cerit H, Hye T, Moondra P, Haimovici F, Sogg S, Shikora S, Goldstein JM, Evins AE, Stoeckel LE. Neural predictors of 12-month weight loss outcomes following bariatric surgery. Int J Obes 2018;42(4):785–793. doi: https://doi.org/10.1038/ijo.2017.190.
Goldenshluger M, Goldenshluger A, Keinan-Boker L, et al. Postoperative outcomes, weight loss predictors, and late gastrointestinal symptoms following laparoscopic sleeve gastrectomy. J Gastrointest Surg. 2017;21(12):2009–15. https://doi.org/10.1007/s11605-017-3585-9.
Goitein D, Zendel A, Westrich G, et al. Postoperative swallow study as a predictor of intermediate weight loss after sleeve gastrectomy. Obes Surg. 2013 Feb;23(2):222–5. https://doi.org/10.1007/s11695-012-0836-4.
McNickle AG. Bonomo SR predictability of first-year weight loss in laparoscopic sleeve gastrectomy. Surg Endosc. 2017;31(10):4145–9. https://doi.org/10.1007/s00464-017-5467-3.
Martin DJ, Lee CM, Rigas G, et al. Predictors of weight loss 2 years after laparoscopic sleeve gastrectomy. Asian J Endosc Surg. 2015 Aug;8(3):328–32. https://doi.org/10.1111/ases.12193.
Andersen JR, Aadland E, Nilsen RM, et al. Predictors of weight loss are different in men and women after sleeve gastrectomy. Obes Surg. 2014;24(4):594–8. https://doi.org/10.1007/s11695-013-1124-7.
Hansen N, Hardin E, Bates C, et al. Preoperative change in 6-minute walk distance correlates with early weight loss after sleeve gastrectomy. JSLS. 2014;18(3) https://doi.org/10.4293/JSLS.2014.00383.
Watanabe A, Seki Y, Haruta H, et al. Preoperative weight loss and operative outcome after laparoscopic sleeve gastrectomy. Obes Surg. 2017;27(10):2515–21. https://doi.org/10.1007/s11695-017-2697-3.
Miller-Matero LR, Bryce K, Saulino CK, et al. Problematic eating behaviors predict outcomes after bariatric surgery. Obes Surg. 2018;28(7):1910–5. https://doi.org/10.1007/s11695-018-3124-0.
Du X, Luo R, Chen YY, et al. Resected gastric volume has no influence on early weight loss after laparoscopic sleeve gastrectomy. Surg Obes Relat Dis. 2018;14(2):129–35. https://doi.org/10.1016/j.soard.2017.11.005.
Vidal P, Ramón JM, Busto M, et al. Residual gastric volume estimated with a new radiological volumetric model: relationship with weight loss after laparoscopic sleeve gastrectomy. Obes Surg. 2014;24(3):359–63. https://doi.org/10.1007/s11695-013-1113-x.
Sánchez-Santos R, Masdevall C, Baltasar A, et al. Short- and mid-term outcomes of sleeve gastrectomy for morbid obesity: the experience of the Spanish national registry. Obes Surg. 2009;19(9):1203–10. https://doi.org/10.1007/s11695-009-9892-9.
Moser F, Marconetto M, Gorodner V, et al. Sweet eating habit: does this affect the results after sleeve gastrectomy? J Laparoendosc Adv Surg Tech A. 2016;26(4):270–5. https://doi.org/10.1089/lap.2016.0084.
Shanti H, Obeidat F. The impact of family members on weight loss after sleeve gastrectomy. Surg Obes Relat Dis. 2016;12(8):1499–503. https://doi.org/10.1016/j.soard.2016.04.002.
Schag K, Mack I, Giel KE, Ölschläger S, Skoda EM, von Feilitzsch M, Zipfel S, Teufel M. The impact of impulsivity on weight loss four years after bariatric surgery. Nutrients. 2016, 8(11). pii: E721.
Rosas U, Hines H, Rogan D, et al. The influence of resected gastric weight upon weight loss after sleeve gastrectomy. Am Surg. 2015;81(12):1240–3.
Obeidat FW, Shanti HA, Mismar AA, et al. Volume of resected stomach as a predictor of excess weight loss after sleeve gastrectomy. Obes Surg. 2014;24(11):1904–8. https://doi.org/10.1007/s11695-014-1330-y.
Sevinçer GM, Konuk N, Bozkurt S, et al. Food addiction and the outcome of bariatric surgery at 1-year: prospective observational study. Psychiatry Res. 2016;244:159–64. https://doi.org/10.1016/j.psychres.2016.07.022.
Parikh M, Gagner M, Heacock L, et al. Laparoscopic sleeve gastrectomy: does bougie size affect mean %EWL? Short-term outcomes. Surg Obes Relat Dis. 2008;4(4):528–33. https://doi.org/10.1016/j.soard.2008.03.245.
Obeidat F, Shanti H, Mismar A, et al. The magnitude of antral resection in laparoscopic sleeve gastrectomy and its relationship to excess weight loss. Obes Surg. 2015;25(10):1928–32. https://doi.org/10.1007/s11695-015-1642-6.
Cottam S, Cottam D, Cottam A, et al. The use of predictive markers for the development of a model to predict weight loss following vertical sleeve gastrectomy. Obes Surg. 2018;28:3769–74. https://doi.org/10.1007/s11695-018-3417-3.
Wang R, Lagakos SW, Ware JH, et al. Statistics in medicine—reporting of subgroup analyses in clinical trials. N Engl J Med. 2007;357(21):2189–94.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Samuel Cottam has no conflict of interest to declare. Daniel Cottam, the corresponding author, reports personal fees from Medtronic, outside the submitted work. Austin Cottam has no conflict of interest to declare.
Statement of Human and Animal Rights
This study did not involve performing any procedures. Thus, it was done in accordance with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Since this is a retrospective study, the formal consent is not required for this type of study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cottam, S., Cottam, D. & Cottam, A. Sleeve Gastrectomy Weight Loss and the Preoperative and Postoperative Predictors: a Systematic Review. OBES SURG 29, 1388–1396 (2019). https://doi.org/10.1007/s11695-018-03666-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-018-03666-7