Abstract
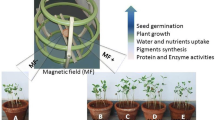
This mini-review aims to assess how magnetic fields (MFs) have been shown to affect in vitro plant growth and development and the practical uses of this technology. Magnetic or electromagnetic fields have shown effects on morphogenesis from different initial explants; on growth-related parameters of in vitro shoots, roots, somatic embryos, and callus; and on the photosynthetic pigment profile, level of stress-induced alanine production, activity of stress-related enzymes, and endogenous levels of cytokinins and auxins. These effects have depended in part on the intensity and duration of exposure of the applied field and in part on the species and in vitro conditions, such as explant type or medium consistency. In vitro growth and development has been manipulated in a series of species, including field crops (soybean, alfalfa, wheat), herbs and medicinal plants (mojito mint, peppermint, spearmint, Calendula officinalis), horticultural crops (potato, sugar beet, wild Solanum spp.), fruits (beach plum), ornamentals (hybrid Cymbidium, hybrid Phalaenopsis, duckweed, Krainzia longiflora, Spathiphyllum), a weed (Haplopappus gracilis), and trees (cork oak, Paulownia sp.). MFs thus have the potential of being used to manipulate the growth and development of plants in vitro and serve as a novel system to open up novel avenues of research in plant science.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Magnetic Fields and Plant Life
All life on Earth is under the influence of the Earth’s geomagnetic field (mean GMF = 5 × 10−5 Tesla [T]), which is generated from the combination of several magnetic fields (MFs) and a dipole on the surface of the Earth, while being influenced by atmospheric flow, crust flow, and other local electric anomalies (Olsen et al. 2010). The impact of external MFs or electromagnetic fields (EMFs) on plant life has been studied since the early 20th century (Savostin 1930). An MF is a vector field in the neighborhood of constant magnets or electric currents that is specified by both direction and strength and is characterized by magnetic flux density (measurement in T) and MF strength (measurement in amperes [A]/meter [m]). An EMF is generated from the acceleration of charged particles and has two components: an electric field surrounding all charged particles and an MF produced by the motion of charged particles (Macintyre 2000).
Experiments on the influence of MFs or EMFs have shown that they are able to have positive effects on the growth, development, and metabolic processes of plants, mainly in early stages of germination or seedling growth (Hirota et al. 1999; reviewed in Vasilevski 2003; Belyavskaya 2004; Galland and Pazur 2005). These effects depended on the type of magnet; the intensity, polarity, and orientation of the MF; and the duration of exposure. Despite knowledge of these concepts, studies on plant morphogenesis and growth under in vitro conditions were only conducted and published after the turn of the millennium, except for two pioneering experiments (Dijak et al. 1986; Celestino et al. 1998).
In this review, we present information from all studies that have applied MFs to manipulate plant growth and development in vitro. Water can be magnetized then used to alter plant growth and development, but magnetized water has not been used in in vitro systems (Teixeira da Silva and Dobránszki 2014). As a nonchemical stressor, the effective use of MFs would provide a nontoxic means to increase productivity, change growth form, or provide a sterile study platform for the mechanisms underlying MF-related stress responses in plants. The review is divided into two sections: the first is a summary of the literature that covers all studies in vitro that have employed MFs, and the second presents hypotheses and possible mechanisms by which MFs affect plant growth and development in vitro.
Effects of Magnetic Fields on Plant Micropropagation and In Vitro Development
Several studies have shown that MFs can affect the development of cells and tissues cultured in vitro. Shoot and root formation rates of Paulownia tissue culture increased when nodes were exposed to external MFs (2.9–4.8 microTesla [mT] for 2.2, 6.6, and 19.8 s or 0.1–0.3 T during the culture period) compared to the control (Ham et al. 2004; Yaycili and Alikamanoglu 2005; Çelik et al. 2008). Paulownia tomentosa and Paulownia fortunei, in response to an MF of 2.9–4.8 mT applied for 19.8 s, increased the shoot regeneration percentage, fresh weight, length, leaf number, and chlorophyll (chl) content after 28 d of culture from nodal sections of 3-mo-old seedlings (Yaycili and Alikamanoglu 2005). Shoot formation of P. tomentosa exposed to a magnetic flux density of 2.9–4.8 mT for 2.2 s increased from 61.9 to 82.5%; the contents of total chl, chl a, and chl b increased; and the total RNA content of the treated tissues doubled compared to the control (Çelik et al. 2008). However, in contrast to the earlier results of Yaycili and Alikamanoglu (2005), increasing the exposure time to 19.8 s decreased the regeneration percentage to 45% and was accompanied by a decrease in chl and RNA contents. Similarly, Atak et al. (2003) found that both regeneration and growth of soybean (Glycine max (L.) Merr.) shoot-tip cultures exposed to MFs (2.9–4.6 mT) for 2.2 and 6.6 s increased 87 and 74%, respectively, relative to the control (62%), while rooting percentage increased 26 and 36%, respectively, relative to the control (14%). Chl content of leaves was the highest after exposure for 2.2 s. However, exposure for 19.8 s decreased all parameters relative to the control.
Belyavskaya (2004) indicated that a weak magnetic field (WMF; 100 nT–0.5 mT) affected the development of cells and tissues of sugar beet (Beta vulgaris L. var. saccharifera) and two cultivars derived from Haplopappus gracilis (Nutt.) A. Gray cultivated in vitro. The cell index of sugar beet culture started to decrease 11 d after exposure to WMF and was most decreased (47% decrease) compared to GMF (control) on the 13th day of culture. However, by the 24th day of culture, there were no significant differences in the cell number of cultures exposed or not exposed to WMF. Callus production of both strains of H. gracilis decreased by 15% compared to GMF after a 5-d-long culture under WMF and by 14 or 21% after 10-d-long cultivation under WMF, depending on the strain. No differences between WMF and GMF were detected in callus cell number after 5 d, but it was lower after 10-d-long cultivation in WMF than in GMF. The regeneration of plants from the callus of peppermint (Mentha × piperita L.), spearmint (Mentha spicata), mojito mint (Mentha villosa), and Calendula officinalis L. was studied after exposing callus to MF with different intensities, including 0.4 × 10−4 T (GMF), 3 × 10−4 T high-intensity static MF, and 0 T for 22 to 96 h (Criveanu and Taralunga 2006). Mentha species had the highest developmental response (regeneration and growth dynamics) in response to 3 × 10−4 T while C. officinalis responded best to 0 T MF. Both stimulating and inhibiting effects at near 0 T MF (i.e., “super weak” MF) conditions were detected in in vitro shoot-tip and nodal segment cultures of ‘Desirée’ potato (Solanum tuberosum L.) on root, stem, and leaf growth, depending on the exposure period (Rakosy-Tican et al. 2005). If cultures were kept for 14 d at near 0 T MF, no significant effect was detected, but exposure for 28 d stimulated the growth of roots (by about 50%) and leaf surfaces (about a 40% increase in leaf length and a 37% increase in leaf width), increased the leaf number/shoot (from 9 to 12), and doubled the photosynthetic pigment content. Even after two to three subcultures under normal GMF conditions, plants continued to form large leaf surfaces. However, when experiments were repeated, vegetative growth was inhibited, suggesting the role of the initial explants in response to MF. When examining the responses of other wild Solanum species (S. chacoense [Chaco potato], S. microdontum, S. verrucosum), vegetative in vitro growth was inconsistent: it could be inhibited, stimulated, or unchanged, regardless of the species. The stimulation of in vitro vegetative growth was connected to variation in the variable component of MFs at the beginning of growth, presumably during cell expansion. The values of GMF and geoelectrical fields affected the in vitro callus development of Krainzia longiflora (Corneanu et al. 2004) by affecting the mitotic division spindle. Exposure to twofold GMF reduced callus formation only slightly but a screened (i.e., dampened) geoelectrical field significantly affected the development of calli. When GMF was natural, a screened geoelectrical field decreased callus diameter from 38.4 to 34.8 mm in clone α and from 15.3 to 9 mm in clone β. However, twofold GMF increased callus diameter from 42.7 to 45.3 mm in clone α but decreased it from 19.4 to 11.2 mm in clone β. Shoot number/explant was decreased by the geoelectrical field at each GMF level (i.e., at natural GMF and at twofold GMF) and the percentage of necrosis doubled in clone β when twofold GMF and screened geoelectrical field were applied. Photosynthetic pigment contents (chl a, chl b, and carotenoids) were reduced when callus was exposed to twofold GMF, but the level of reduction depended on the type of callus. Under normal GMF, isoperoxidase activity was higher (by as much as 123%) in anthocyanin-type callus but under twofold GMF, its level was significantly higher (by as much as 369%) in normal, green callus.
A low EMF (0.02, 0.05, or 0.15 V, applied for 20 h in an initial range of 1–44 h) applied directly after protoplast isolation stimulated somatic embryogenesis from mesophyll protoplasts of alfalfa (Medicago sativa L.; Dijak et al. 1986). The highest number of somatic embryos developed when an EMF of 0.02 V was applied for 20 h. An alfalfa clone (‘Regen S’) that was not embryogenic under control conditions could produce somatic embryos in the same developmental pattern as the embryogenic clone ‘Rangelander’. In this case, EMF appeared to have the effect of overcoming the recalcitrance of ‘Regen S’ to embryogenesis. EMF stimulated development by improving the aggregation of protoplasts caused by the establishment of protoplast polarity. Exposure of three different embryogenic lines of cork oak (Quercus suber L.) somatic embryos to extremely low-frequency MF (50 Hz, 15 μT) for 8 wk affected the morphogenic response. The number of detachable somatic embryos decreased, but the decrease was significant only in one of the three genotypes (‘G3.27’) examined (Celestino et al. 1998). Neither germination nor the percentage of plant formation from somatic embryos was influenced by extremely low sinusoidal EMF. However, under suppressed GMF (0 T), germination percentage and plant formation increased significantly (from 44.6 to 58.3% and from 5.7 to 15.3%, respectively), although how GMF was suppressed was not explained.
When duckweed (Spirodela oligorrhiza (Kurz) Hegelm.) cell culture was exposed to sinusoidally varying MF (60 and 100 Hz/0.7 mT), there were specific metabolic stress effects (Parola et al. 2005). Alanine was produced only under EMF conditions, suggesting that EMF treatment altered the biochemical processes in which free radicals were involved and that alanine production was a stress-induced response. The addition of vitamin C, a free-radical scavenger, reduced the accumulation of alanine. A similar result, i.e., enhanced alanine production, was found by Monseline et al. (2003) when duckweed plants (S. oligorrhiza) were exposed to sinusoidally varying MF (SVMF) (60 and 100 Hz/0.7 mT) for 24 h. Monseline et al. (2003) demonstrated that a variety of different stresses (anoxia, air exposure, salinity, hyperosmosis, hypertonic stress, heat shock, freezing, cold shock, heavy-metal stress, starvation, water-stress deficiencies in nitrogen and phosphorous, EMF, microgravity, red/far-red pulses) caused the production and accumulation of alanine in response to free-radical production; conversely, the production and accumulation of alanine was reduced by the suppression of free radicals. They proposed, therefore, that alanine production and accumulation is a universal stress signal in living organisms. The average fresh weight of 28-d-old embryos increased when mature zygotic embryos of wheat (Triticum aestivum L.) ‘Flamura-85’ were exposed to (more specifically, run under) MFs with different flux intensities between 2.9 and 4.8 mT, at 1 m/s for 2.2 and 19.8 s (Alikamanoglu and Sen 2011). The percentage increase depended on the exposure period: it was 25% after 2.2 s exposure and 30% after 19.8 s exposure. Similarly, some biochemical parameters were also affected by MF treatment: the amount of total protein increased by 12 and 14% and chl content increased by 32 and 35% after exposure to MF treatment for 2.2 and 19.8 s, respectively. The activities of stress-related enzymes such as superoxide dismutase (SOD), catalase (CAT), peroxidase (POX), and ascorbate peroxidase (APX) also increased. The activity of total SOD increased by 62 and 88% after exposure to MF treatment for 2.2 s and 19.8 s, respectively. After 19.8 s, POX activity increased by about 80%, CAT activity by 73%, and APX activity by 62%. The endogenous production of reactive oxygen species (ROS) is a general response to environmental stresses. Induction of the antioxidant enzyme system in plants, as a defense against ROS, is the part of the plant’s defenses and acclimatization process under unfavorable or suboptimal conditions (i.e., stress conditions) to maintain plant growth (Boguszewska and Zagdańska 2012).
Decreases were observed in callus growth rate (from 0.181 to 0.175), relative growth rate (from 1.441 to 0.655), and relative callus growth rate (from 0.052 to 0.022) when mature bread wheat (Triticum aestivum L.) embryos were exposed to MF (8.8 and 17.6 T); this negative effect became more pronounced as the MF increased (Kahrizi et al. 2013).
EMF in the range of 48–115 kA/m was applied to in vitro nodal segment culture of beach plum (Prunus maritima Marshall; Yan et al. 2009). Increasing field strength stimulated regeneration and growth from explants with 2.3-folds more sprouts (shoot buds) being induced when an EMF of 97 kA/m was applied for 10 min. The multiplication rate was 5.2- or 3.1-folds higher than the control, depending on the auxin content of the culture medium. The multiplication rate was higher if medium contained 0.2 mg/l indole-3-butyric acid (IBA) instead of 0.2 mg/l α-naphthaleneacetic acid (NAA) in addition to 2.0 mg/l zeatin, and longer in vitro roots were produced from plants exposed to EMF of 97 kA/m, increasing the survival during acclimatization, although survival was not quantified.
However, studies on the effects of MFs on micropropagated plants remain scanty, especially with permanent magnets that have moderate intensities (1 mT to 1 T). This range of MFs can hypothetically be used widely in practical tissue culture chambers, as shown by results for ornamental orchids (hybrid Phalaenopsis and Cymbidium) and Spathiphyllum (Tanaka et al. 2010; Van et al. 2011a, 2011b, 2012; Tables 1 and 2). However, the effects of MFs on plant growth and development are species-specific, and the exact properties of MFs (i.e., intensity, polarity, duration of exposure, and type of magnet) are variables that still need to be individually tested for different genotypes. In these studies, increasing the MF intensity from GMF (5 × 10−6 T) to 0.1–0.2 T, north or south pole resulted in positive (highest chl content) and negative (inhibition of leaf emergence) effects on Phalaenopsis in vitro plantlet growth (Van et al. 2011b; Table 1), but had no clear influence on Cymbidium plantlet development other than negatively affecting plant height. Testing the same level of MF (0.1–0.2 T, north or south pole), external application of MF increased chl content of the third leaf (reported as SPAD value) and the total number of leaves but slightly decreased total dry weight of in vitro Spathiphyllum plantlet shoots (Table 1; Van et al. 2012).
The proliferation of Phalaenopsis protocorm-like bodies (PLBs) was affected not only by the application of external MFs (0.1, 0.15, 0.2 T; north or south pole) but also by the substrates used (Tanaka et al. 2010; Van et al. 2011a). An MF of 0.1 T north or south pole significantly increased the fresh and dry weights of PLB clusters after culture in liquid medium for 2 mo. When a similar experimental design was used to test the same cultivar on solid medium, the fresh and dry weights of PLB clusters increased when 0.2 T north pole MF was applied, although an MF of 0.1 T north or south pole had no effect (Table 1; Tanaka et al. 2010; Van et al. 2011a). The difference may be because liquid medium by itself is more sensitive and active under exposure to MF than solid medium (Pang and Deng 2008; Teixeira da Silva and Dobránszki 2014).
The In Vitro Milieu for Understanding the Possible Mechanistic Response to Magnetic Fields
How do intensity, polarity, and duration of exposure of MFs affect plantlet development in vitro? The effects of MFs on plants have been studied widely over at least 20 years. However, the results have usually been inconsistent and at times even contradictory, and there is still no clear mechanistic explanation for how MFs affect plant growth and development. Some reports have shown negative effects while others have shown positive effects or no effects. MF intensity and duration of exposure affect plant growth to different degrees (Alikamanoğlu et al. 2007; Atak et al. 2007). Most research on this topic has concentrated on the effects of a WMF (100 nT–0.5 mT) or “super weak” MF (magnetic vacuum)—as in space-like conditions—where MF intensity is nearly 0 nT and has usually focused on the effects of seed germination and seedling development under the action of MFs.
Changes in gene expression levels and stress-related responses.
There are also some studies on the effects of very high MF (>10 T) on gene expression in Arabidopsis thaliana (L.) Heynh. (Paul et al. 2006). An MF of 15 T induced the expression of an Adh/GUS transgene in both the roots and the leaves of an A. thaliana transgenic line (Paul et al. 2006). Comparative analysis of gene expression patterns in a subsequent experiment (Paul et al. 2006) showed that MF induced changes in gene expression when 3-wk-old plants were exposed to MF (MFs of 14 T for 2.5 h or of 21 T for 2.5 and 6.5 h). Out of 8000 genes examined, 114 were differentially expressed (2.5-folds more than the control after exposure to 21 T MF), and many of them were related to stress responses (stress-related genes or transcription factors) and ion transport functions. However, in other cases when there were stress-related changes, such as the accumulation of alanine, there was also a simultaneous increase in the activity of stress-related enzymes (ATX, CAT, POX) (Monseline et al. 2003; Parola et al. 2005; Alikamanoglu and Sen 2011) after exposing tissues to MFs with an increasing order of magnitude of mT (0.7 mT to axenic culture of duckweed and 2.9 or 4.8 mT to mature wheat embryos), although no further experimental proof exists about whether and how these enzymatic changes are connected with changes in gene expression.
Changes in endogenous levels of plant growth regulators.
MFs (2.9–4.6 mT for 2.2, 6.6, or 19.8 s) increased cytokinin and auxin synthesis in Paulownia tissue culture, increasing shoot regeneration and root induction (Atak et al. 2003; Çelik et al. 2008). An analogous process may have caused the observed effect of MFs on the development of Phalaenopsis plantlets (Van et al. 2011b), but future studies would need to assess endogenous levels of plant growth regulators over the entire developmental period.
Changes in growth, development, and biomass production.
A positive influence of MFs was observed during micropropagation shoot-tip culture of soybean (Atak et al. 2007) and nodal segment culture of Paulownia (P. tomentosa and P. fortunei; Yaycili and Alikamanoglu 2005; Alikamanoğlu et al. 2007) and beach plum (Yan et al. 2009). P. tomentosa plants exposed to 2.9–4.8 mT for 19.8 s had higher plant fresh weight, leaf number, and chlorophyll (chl) content after 4 wk of culture than control plants not exposed to MFs (Yaycili and Alikamanoglu 2005; Alikamanoğlu et al. 2007). The fresh weight of plantlets regenerated from soybean shoot tips exposed to MF of 2.9–4.6 mT for 2.2 and 19.8 s was higher than that of controls (Atak et al. 2007). Similarly, beach plum sprouts resulting from nodal segment culture showed higher fresh and dry weights and height when treated with an MF of 97 kA/m for 10 min (Yan et al. 2009). In the Van et al. (2011b) study, within the range of MFs tested (i.e., 0.1–0.2 T), the greatest influence on Phalaenopsis was when plantlets were exposed to MFs of 0.1 T south. In a previous, closely related study (Van et al. 2011a) in which Phalaenopsis PLBs were exposed to different MFs, 0.1 T south also had the greatest influence on the fresh and dry weights of regenerated PLBs (Table 1). However, a plausible explanation or mechanism for these effects was not (and could not be) provided, and it continues to be a difficult experimental parameter to standardize, measure, or test.
Belyavskaya (2004) mentioned that prolonged exposure of pale flax (Linum bienne L.) plants and lentil (Lens culinaris L.) roots to WMF may have different biological effects at the cell, tissue, and organ levels than shorter exposures. In the Van et al. (2011a) study, exposure to different durations of an MF of 0.15 T had some effects on the growth of Phalaenopsis plants. In particular, plantlets exposed to an MF for 3 vs. 1 or 2 mo had significantly higher total fresh and dry weights of shoots and roots and had more leaves, but the same treatment inhibited shoot elongation of Cymbidium plantlets (Van et al. 2012; Tables 1 and 2). As for Spathiphyllum, exposure to 0.15 T had no significant influence on plantlet development other than increasing the chl content (SPAD value), with longer exposure giving higher chl levels (i.e., 8 wk > 4 wk > 2 wk) (Van et al. 2012; Tables 1 and 2). Another closely related study showed that Phalaenopsis PLBs exposed to 0.15 T at either the north or south pole for a longer period of time, i.e., 7 vs. 2 wk, resulted in greater biomass of newly formed PLBs (Van et al. 2011a). MFs can thus be applied to increase biomass in Phalaenopsis PLBs and plantlets. Were MFs to be applied to other crops, greater biomass accumulation, productivity, or yield might be possible, making tissue culture laboratories more productive per unit area. At a wider scale, in a system where space restrictions apply (such as in a space station), the induction of greater yield of harvestable plant material would have obvious advantages. In contrast to Phalaenopsis, Paulownia tissue cultures were inhibited when exposed to an MF of 2.9 to 4.8 mT for longer than 6.6 s (Yaycili and Alikamanoglu 2005). Atak et al. (2003) reported similar inhibition of soybean tissue culture when exposed to 2.9–4.6 mT for 2.2, 6.6, or 19.8 s.
MFs in the range of intensities tested (i.e., from 0.1 to 0.2 T), independent of polarity and exposure duration, increased the chl content of the third leaf of Spathiphyllum shoots; conversely, the chl content decreased in the third leaf of Cymbidium shoots exposed to 0.15 T MF for 3 mo, regardless of polarity (Van et al. 2012; Tables 1 and 2). The same effect on plant growth was confirmed by several studies on different plants. Studies by Atak et al. (2003, 2007) and Çelik et al. (2008) on the effects of MFs of 2.9–4.6 mT on Paulownia node cultures and soybean tissue culture at different exposure times also confirmed that the total chl content was increased by MFs.
The Van et al. (2012) study found that an increase in intensity from 0.1 to 0.2 T or short- or long-term exposure to MFs of 0.15 T did not have a significant effect on Spathiphyllum (except for reducing the dry weight of Spathiphyllum shoots) and Cymbidium plant growth compared to the control, except for plant height (i.e., stem elongation) of Cymbidium shoots (Tables 1 and 2). However, Ham et al. (2004) found that an increase in MF from 0 to 0.15 T increased multiplication rate and rooting of in vitro Paulownia plantlets. The Van et al. (2011a) study on the effects of duration of exposure of 0.15 T (north or south) on Phalaenopsis PLB proliferation showed that longer exposure (7 vs. 2 wk) resulted in greater biomass of newly formed PLBs and smaller average number of newly formed PLBs. An MF of 0.1 T south (vs. 0.15 T or 0.2 T, north or south) resulted in the greatest fresh and dry weights of regenerated PLBs (Table 1). Regarding Phalaenopsis plantlet growth in vitro, exposure to MFs of 0.15 T for 3 mo (vs. 1 or 2 mo) of either polarity resulted in the highest total fresh and dry weights of shoots and roots (Van et al. 2011b).
Conclusion
The purpose of this review is to synthesize how MFs or EMFs can influence plant growth and development in vitro. Based on advances achieved to date, both MFs and EMFs have been shown to influence the in vitro development and growth of plants. Regardless of the actual mechanism at work, the following positive aspects of MFs can be noted: (a) MFs, when applied to small explants as a constant force, have the ability to stimulate growth, and (b) the in vitro environment can serve as an ideal test condition since it requires minimal space and limited material and can be reproduced with confidence (see Tanaka et al. 2010, for a model system). Even though there are positive aspects, some points of caution are advised: (a) cytogenetic and molecular analyses are urgently required to assess whether the changes caused to plant growth and development also irreversibly alter the cellular and genetic mechanisms of plants, and (b) researchers are advised about extrapolating in vitro results and scaling-up to greenhouse or field experiments since in vitro explants are often in close proximity to the applied MF. The response of plants after in vitro growth may differ and if the force of the MF is removed at the time of acclimatization then a plant may respond by reverting to a wild-type state, or the in vitro effect of the MFs may be lost or become diluted. Therefore, extension of systematic studies conducted under an in vitro milieu might facilitate and promote better understanding of a plant’s behavior in response to MFs, particularly from a theoretical viewpoint. It would help to identify and understand how MF-related techniques can improve growth and yield and how the direction of plant development can be regulated by using such a biophysical technology.
References
Alikamanoglu S, Sen A (2011) Stimulation of growth and some biochemical parameters by magnetic field in wheat (Triticum aestivum L.) tissue culture. Afr J Biotechnol 10:10957–10963
Alikamanoğlu S, Yaycılı O, Atak Ç, Rzakoulieva A (2007) Effect of magnetic field and γ-gamma radiation on Paulownia tomentosa tissue culture. Biotechnol Biotechnol Equip 21:49–53
Atak Ç, Çelik Ö, Olgun A, Alikamanoğlu S, Rzakoulieva A (2007) Effect of magnetic field on peroxidase activities of soybean tissue culture. Biotechnol Biotechnol Equip 21:166–171
Atak Ç, Emiroğlu Ö, Alikamanoğlu S, Rzakoulieva A (2003) Stimulation of regeneration by magnetic field in soybean (Glycine max L. Merrill) tissue cultures. J Cell Mol Biol 2:113–119
Belyavskaya NA (2004) Biological effects due to weak magnetic field on plants. Adv Space Res 34:1566–1574
Boguszewska D, Zagdańska B (2012) ROS as signaling molecules and enzymes of plant response to unfavorable environmental conditions, oxidative stress. In: Lushchak V (ed) Molecular mechanisms and biological effects. InTech, Croatia, pp 341–362
Celestino C, Picazo ML, Toribio M, Alvarez-Ude JA, Bardasano JL (1998) Influence of 50 Hz electromagnetic fields on recurrent embryogenesis and germination of cork oak somatic embryos. Plant Cell Tissue Organ Cult 54:65–69
Çelik Ö, Atak Ç, Rzakulieva A (2008) Stimulation of rapid regeneration by a magnetic field in Paulownia node cultures. J Cent Eur Agric 9:297–304
Corneanu M, Corneanu GC, Marinescu G, Băbeanu C, Morariu VV (2004) In vitro explant development in the presence of some extraterrestrial factors. http://www.gbm.bio.uaic.ro/index.php/gbm/article/download/251/242 (last accessed 21 January 2015)
Criveanu HR, Taralunga G (2006) Influence of magnetic fields of variable intensity on behaviour of some medicinal plants. J Cent Eur Agric 7:643–648
Dijak M, Smith DL, Wilson TJ, Brown DCW (1986) Stimulation of direct embryogenesis from mesophyll protoplasts of Medicago sativa. Plant Cell Rep 5:468–470
Galland P, Pazur A (2005) Magnetoreception in plants. J Plant Res 118:371–389
Ham LH, Van NTK, Vinh DN (2004) The impact of magnetic fields on in vitro culture system. Appl Biotechnol Newsl 1+2:42–49, in Vietnamese
Hirota N, Nakagawa J, Kitazawa K (1999) Effects of a magnetic field on the germination of plants. J Appl Phys 85:5717–5719
Kahrizi D, Cheghamirza K, Akbari L, Rostami-Ahmadvandi H (2013) Effects of magnetic field on cell dedifferentiation and callus induction derived from embryo culture in bread wheat (Triticum aestivum L.) genotypes. Mol Biol Rep 40:1651–1654
Macintyre SA (2000) Magnetic field measurement. http://engineering.dartmouth.edu/dartmag/docs/macintyre.pdf (last accessed 21 January 2015)
Monseline EB, Parola AH, Kost D (2003) Low-frequency electromagnetic fields induce a stress effect upon higher plants, as evident by the universal stress signal, alanine. Biochem Biophys Res Comm 302:427–434
Olsen N, Hulot G, Sabaka TJ (2010) Sources of the geomagnetic field and the modern data that enable their investigation. In: Freeden W, Nashed MZ, Sonar T (eds) Handbook of Geomathematics. Springer, Berlin, Heidelberg, pp 106–124
Pang XF, Deng B (2008) Investigation of changes in properties of water under the action of a magnetic field. Sci China Ser G: Phys, Mech Astro 51:1621–1632
Parola AH, Kost D, Katsir G, Monselise BE, Cohen-Luria R (2005) Radical scavengers suppress low frequency EMF enhanced proliferation in cultured cells and stress effects in higher plants. Environmentalist 25:103–111
Paul AL, Ferl RJ, Meisel MW (2006) High magnetic field induced changes of gene expression in Arabidopsis. Bio Magnetic Res Technol 4:1–10
Rakosy-Tican L, Aurori CM, Morariu VV (2005) Influence of near null magnetic field on in vitro growth of potato and wild Solanum species. Bioelectromagnetics 26:548–557
Savostin PW (1930) Magnetic growth relations in plants. Planta 12:327–330
Tanaka M, Van PT, Teixeira da Silva JA, Ham LH (2010) Novel magnetic field system: application to micropropagation of horticultural plants. Biotechnol Biotechnol Equip 24:2160–2163
Teixeira da Silva JA, Dobránszki J (2014) Impact of magnetic water on plant growth. Env Exp Biol 12(4):137–142
Van PT, Teixeira da Silva JA, Ham LH, Tanaka M (2011a) Effects of permanent magnetic fields on the proliferation of Phalaenopsis protocorm-like bodies using liquid medium. Sci Hortic 128:479–484
Van PT, Teixeira da Silva JA, Ham LH, Tanaka M (2011b) The effects of permanent magnetic fields on Phalaenopsis plantlet development. J Hortic Sci Biotechnol 86:473–478
Van PT, Teixeira da Silva JA, Ham LH, Tanaka M (2012) Effects of permanent magnetic fields on growth of Cymbidium and Spathiphyllum. In Vitro Cell Dev Biol Plant 48:225–232
Vasilevski G (2003) Perspectives of the application of biophysical methods in sustainable agriculture. Bulg J Plant Physiol Special Issue:179–186
Yan D, Guo Y, Zai X, Qin P (2009) Effects of electromagnetic fields exposure on rapid micropropagation of beach plum (Prunus maritima). Ecol Engin 35:597–601
Yaycili O, Alikamanoglu S (2005) The effect of magnetic field on Paulownia tissue cultures. Plant Cell Tissue Organ Cult 83:109–114
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: John Finer
Rights and permissions
About this article
Cite this article
A. Teixeira da Silva, J., Dobránszki, J. How do magnetic fields affect plants in vitro?. In Vitro Cell.Dev.Biol.-Plant 51, 233–240 (2015). https://doi.org/10.1007/s11627-015-9675-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-015-9675-z