Abstract
Soil pollution with toxic elements is a recurrent issue due to environmental disasters, fossil fuel burning, urbanization, and industrialization, which have contributed to soil contamination over the years. Therefore, the remediation of toxic metals in soil is always an important topic since contaminated soil can affect the environment, agricultural safety, and human health. Many remediation methods have been developed; however, it is essential to ensure that they are safe, and also take into account the limitation of each methodology (including high energy input and generation of residues). This scenario has motivated this review, where we explore soil contamination with arsenic, lead, mercury, and chromium and summarize information about the methods employed to remediate each of these toxic elements such as phytoremediation, soil washing, electrokinetic remediation, and nanoparticles besides elucidating some mechanisms involved in the remediation. Considering all the discussed techniques, nowadays, different techniques can be combined together in order to improve the efficiency of remediation besides the new approach of the techniques and the use of one technique for remediating more than one contaminant.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
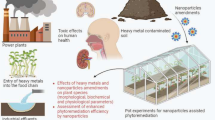
The environment is constantly being exposed to different chemical elements from natural and anthropogenic sources. Due to industrialization and urbanization in many regions of the world, the release of toxic elements from anthropogenic sources continues to rise (Wu et al. 2016). When toxic elements such as arsenic (As), lead (Pb), chromium (Cr), and mercury (Hg) accumulate in sediments, they can enter the food chain as bottom sediments in aquatic ecosystems and subsequently threaten the aquatic flora and fauna directly or indirectly (Hsu et al. 2016).
Environment contamination with toxic elements is not restricted to the aquatic ecosystem: soil can also be contaminated and has, therefore, become an important issue of environmental protection worldwide. The most significant anthropogenic sources of toxic metals accumulated in soil include effluents from industrial activities, sewage from domestic and industrial sources, and vehicular emissions, as illustrated in Fig. 1 (Soodan et al. 2014; Su et al. 2014).
Water contamination can also culminate in soil contamination, especially when contaminated water is used in irrigation. Recently, a case of contamination with arsenic became a serious problem when contaminated water was employed to irrigate rice, which could cause hazardous effects on human health (Sohn 2014).
Although this issue has been explored by various authors (Adegbeye et al. 2020; Allevato et al. 2019; Asgari Lajayer et al. 2017; Chen et al. 2015, 2018; Motuzova et al. 2014; Nriagu and Pacyna 1988), and despite the development of numerous remediation methodologies (Bolade et al. 2020; Bolan et al. 2014; Fernández et al. 2005; Gavrilescu 2004; Liang et al. 2016; Mao et al. 2015; Qin et al. 2019; Salt et al. 1995), environmental contamination by toxic metals remains a recurrent problem.
Toxic metals are considered soil contaminants when (I) they persist in soil for a long time after their introduction, (II) they have high concentration in discarded products compared with their concentration in the receiving environment, (III) they can be easily transferred from an environment to another where direct or indirect exposure can occur, (IV) they can be interconverted to different species and become more bioavailable or toxic, adversely affecting the ecosystem and the human health. Furthermore, the presence of toxic metals can inhibit organic compound biodegradation in soil (Wuana and Okieimen 2011).
Recently, Rio Doce, a river in Brazil, made the international headlines because of the mining tailing dam disaster that happened in November 2015: toxic metals including arsenic, copper, and mercury contaminated the river and resulted in the largest Brazilian environmental disaster (dos Reis et al. 2017; Escobar 2015). In 2014, a catastrophic branch of the Mount Polley mine, in Canada, released 25 million cubic meters of tailings into the West Basin of the Quesnel Lake, where metals and metalloids such as As, copper (Cu), francium (Fr), and manganese (Mn) were detected at concentrations above British Columbia’s freshwater sediment quality guidelines (Petticrew et al. 2015). These disasters can have long-lasting harmful effects on the environment. A classic example is the disaster in the Aznalcollar pyrite mine in Sevilla, Spain. In 1998, the waste dumps broke, releasing 9 × 105 m3 of toxic tailings into the Agrio and Guadiamar rivers. Despite the investments made to remediate the affected area, residual soil pollution can still be detected nowadays (García-Carmona et al. 2017).
In view of these facts, new studies about the toxic effects of metals and new strategies for their remediation must be implemented, so that satisfactory results can be achieved by means of simple methodologies that use non-toxic reagents at low cost. This review was designed using mainly studies from the last 5 years, using three different sources of research (ScienceDirect, Scopus, and Google Scholar), and keywords related to “soil remediation” were employed in the research.
Remediation techniques
Several remediation methodologies have been developed to solve or to minimize the impact of contamination. These technologies include physical, chemical, and biological methods and are summarized in Fig. 2 (Song et al. 2017).
Physical remediation uses physical technologies. Capping consists of placing a suitable isolating material over contaminated soil to isolate the contaminants. Soil replacement refers to replacing contaminated soil with non-contaminated soil (Khalid et al. 2016; Song et al. 2017).
Chemical remediation employs chemical reactions to remove contaminants and includes immobilization, soil washing, and encapsulation. Immobilization reduces contaminant mobility, bioaccessibility, leachability, and bioavailability through complexation, adsorption, and precipitation. Soil washing removes soil contaminants by extraction, whereas encapsulation involves mixing the contaminant with other materials, such as concrete, to immobilize the contaminant (Khalid et al. 2016).
Biological remediation uses microorganisms or plants to remove contaminants from soil. The most common bioremediation method is phytoremediation, which employs plants to remediate contaminated soil (Khalid et al. 2016).
In addition to their classification into physical, chemical, and biological methods, remediation methods can be divided into two main strategies: in situ remediation and ex situ remediation. In situ remediation is carried out at the originally contaminated site without the need to move the soil or the sediment itself. Ex situ remediation is conducted by excavating and treating the contaminated soil in a place away from the original site (Song et al. 2017). Ex situ and in situ techniques have specific benefits and costs. In situ remediation techniques are favored over ex situ techniques because excavation is expensive and excavators are exposed to adverse health risks posed by the contaminants (Kuppusamy et al. 2016).
Lead
Sources of lead and soil contamination with lead
Lead (Pb) is a toxic metal that has been included in the United States Environmental Protection Agency (U.S. EPA) “probable carcinogen” list. The major sources of contamination with lead include solid waste incineration, mining operations, and the chemical, metallurgical, and petrochemical industries (Du et al. 2014; Pinto and Al-Abed 2017).
The earliest record of lead mining dates back to as far as 6500 BC in Turkey. During the Roman period, lead was employed in the building of aqueducts and as a pigment in paint. It was also used to sweeten wine and to preserve fruit by addition of lead(II) acetate (Dapul and Laraque 2014).
Compared with other toxic metals, lead is highly persistent in soil due to its low solubility. It has harmful effects on human health, which is the reason why it has been banned from gasoline formulation (Cristaldi et al. 2017). The accepted safe lead concentration in soil is 400 μg g−1 in residential areas (Dapul and Laraque 2014).
The contamination of Pb in Brazil is largely from urban waste; approximately 215 thousand tons of municipal solid waste are generated daily. About 40.9% of this waste goes directly to the inadequate landfill and open dumps, causing contamination of the soil (Costa et al. 2019).
Toxic effects of lead
Lead accounts for one of the oldest occupational intoxication cases, dating back to the Christian era. The toxic effects of inorganic lead include neurological disorders, hypertension, cognitive impairment, and IQ loss. For all these reasons, it has been established that there is no safe level for lead exposure in children (Gidlow n.d.; Wu et al. 2016). Furthermore, Pb, which is initially present in the blood, is stored in bone for years, so bone becomes a long-term chronic source of lead back into the bloodstream (Laidlaw et al. 2017). Lead also binds to calcium-activated proteins with higher affinity than calcium, hence interfering in various calcium-dependent cellular functions (Dapul and Laraque 2014).
Poisoning with inorganic lead can be treated by chelation therapy, which helps to eliminate the metal. Although chelators such as CaNa2EDTA and meso-2,3-dimercaptosuccinic acid (DMSA) exert protective effects against lead, CaNa2EDTA can cause renal toxicity especially during treatment with high doses, whereas DMSA may cause appetite loss and nausea (Zhai et al. 2015).
The use of lead in the petrochemical industries is decreasing; in fact, the only compound that is currently produced in this industry is tetra-ethyl lead. Nevertheless, lead naphthenates and stereates are still employed as stabilizers for plastics (Gidlow n.d.).
Tetra-ethyl lead and inorganic lead have distinct toxicological profiles. Tetra-ethyl lead produces acute toxic psychosis, but the early symptoms can be easily missed. However, continuing exposure reveals further symptoms of toxic psychosis, and even coma may occur. Unfortunately, there is no specific antidote for organic lead poisoning other than sedation and supportive treatment (Gidlow n.d.).
Remediation of lead in soil
Intense use of lead in fossil fuels, paints, and plumbing can pollute the soil. In view of this problem, numerous methodologies have been developed to remediate lead in soil, including soil washing, phytoremediation, use of phosphates to immobilize lead, and, more recently, the use of nanoparticles.
Soil immobilization
Soil immobilization consists of adding reagents or materials into the contaminated soil to reduce the mobility of toxic substances and their solubility, thereby preventing toxic metals from migrating to other environmental media (Fig. 3) (Derakhshan et al. 2018). This method is relatively inexpensive. Nonetheless, if soil properties change, pollutants can become available again, so constant monitoring is crucial (Derakhshan et al. 2018).
The immobilization of metals can be evaluated by sequential extraction, which is a procedure that evaluates the leachability of metal in different extractor solutions. In soil, the exchangeable metal form is usually the most mobile and toxic form. A decrease in the exchangeable phase reduces mobility (Tao et al. 2017).
In soil, many materials can immobilize lead. The sludge from drinking water treatment (DWTS) can be used to this end because it contains iron and aluminum oxides that can bind lead and decrease its mobility. Recently, Souza and colleagues reported that this DWTS can efficiently reduce lead mobility and bioaccessibility (Souza et al. 2018). After a period of 4 months, samples containing a contaminated soil/sludge ratio of 1:1 present less than 3% of lead concentration in exchangeable fractions, and lead bioaccessibility decreased by 28.8% (Souza et al. 2018).
Okkenhaug and co-workers employed a mixture of oxyhydroxide powder (CFH-12) (2%) and limestone (1%) to remediate lead in soil. They also tested zerovalent iron (2%). According to the authors, all the sorbents decrease lead in water and extractable phase significantly (89% and 99% respectively). In addition, remediation remains stable over a 4-year experimental period, which indicates that the method is highly efficient (Okkenhaug et al. 2016).
Natural polymers like lignin, carboxymethyl cellulose, and sodium alginate can effectively immobilize lead. According to Tao et al. (Tao et al. 2017), all these materials diminish lead leachability and bioaccessibility, but lignin provides the best results: it lowers lead concentration in the exchangeable fraction to less than 10%. Furthermore, lignin forms a three-dimensional molecular structure bearing oxygen-containing functional groups that efficiently bind lead (Tao et al. 2017).
Remediation with phosphate
The addition of phosphate is another in situ technique for lead remediation. Lead phosphates are 44 orders of magnitude less soluble than other lead minerals such as PbSO4, PbS, PbCO3, and PbO, which is important for effective immobilization (Zeng et al. 2017). The suggested mechanisms for lead immobilization include lead retention by hydroxyapatite in ionic exchange with calcium and reaction of lead with hydroxyapatite in solution, to form stable pyromorphite-type minerals like [Pb5(PO4)3X; X = F, Cl, Br, and OH] with Kps 10–71.6, 10–84.4, 10–78.1, 10–76.8, respectively, as demonstrated by the following equations (Melamed et al. 2003; Zeng et al. 2017):
The method can be more efficient in acid soil because lead solubilization allows it to react with phosphorus. Therefore, remediation can be divided into two stages: soil acidification and addition of phosphorus, followed by the addition of limestone to make the soil alkaline. However, these procedures can be expensive and problematic (Laidlaw et al. 2017).
According to Seshadri and colleagues, countless phosphorus compounds, soluble and insoluble, can stabilize lead. These authors measured lead bioaccessibility in soil amended with diammonium phosphate (DAP) and with reactive (Sechura, SPR) and unreactive natural phosphate rock (Christmas Island; CPR), so that they could study how effective lead immobilization was. They showed that reactive SPR and DAP reduce lead bioaccessibility by 67.1% and 78.9%, respectively (Seshadri et al. 2017).
Yuan and co-workers verified that the addition of iron hydroxyl phosphate (FeHP) in soil effectively immobilizes lead. The DTPA-extractable phase decreases with increasing FeHP concentration in soil: FeHP at 4 wt% provides 55% lead immobilization (Yuan et al. 2017).
Although effective immobilization results have been achieved, some aspects must be considered before using phosphates to immobilize lead, including the risk of eutrophication in water environment and the increase in arsenic, selenium, and antimony leachability due to competition of these oxoanions for the adsorption site in soil as also reported by Hafsteinsdóttir et al. (Hafsteinsdóttir et al. 2014; Zeng et al. 2017). These authors treated the soil of East Antarctica with triple superphosphate and phosphate rock and a buffer of magnesium carbonate and magnesium oxide and observed higher arsenic leachability. They also noted that the time required for lead leachability to reduce to values below the value of the untreated landfill was 1 month because the presence of chlorides at high concentration of (from the ocean) and organic matter slowed the process of lead fixation on phosphorus through formation of pyromorphite (Hafsteinsdóttir et al. 2014).
Phytoremediation
Phytoremediation has been employed to remediate lead in soil. Phytoextraction and phytostabilization are particularly relevant to lead-contaminated soil. Phytoextraction entails using plants to absorb and to translocate lead to tissues, which is accumulated in the biomass, and can be removed by harvesting, while phytostabilization uses plants to stabilize lead in situ, by complexation or precipitation, in order to reduce the risks posed by this contaminant to the environment and the human health (Laidlaw et al. 2017) as illustrated in Fig. 4.
For phytoextraction to be effective, some characteristics are necessary including fast plant growth and a combination of high lead accumulation and large biomass to ensure efficient lead removal. Even though some plants can hyperaccumulate lead, they have small biomass and grow slowly, while high biomass-yielding non-hyperaccumulating plants generally lack the ability to accumulate high lead levels (Gupta et al. 2013; Laidlaw et al. 2017).
The uptake mechanism of metals by plants depends on the metal bioavailability and involves the root interception of the metal ions, their entry in the roots and their translocation through mass flow and diffusion. The phytostabilization mechanism indicates that plants sequester the metals in the rhizosphere through metal complexes with organic compounds, adsorption on roots surfaces, and metal accumulation in root tissues (Jabeen et al. 2009; Sabir et al. 2014). The metallothioneins (MTs) are metal-binding proteins with low molecular weight which provide thiol groups for metal chelation, participating in the phytostabilization mechanism. Their production by the plants enhances the metal translocation and sequestration, since they may be transported into vacuoles for long-time sequestration (Jabeen et al. 2009). It is important to take into account that each plant has its own phytostabilization and phytoextraction mechanism: Sedum alfredii H stabilizes Pb by induction of glutathione biosynthesis that binds Pb in the roots and the phytoextraction of Pb occurs by induction and accumulation of phytochelatin that binds metals in aboveground parts (Sabir et al. 2014).
According to Yildirim and Sasmaz, Phlomis sp. is a good phytoremediator of lead. These authors described lead concentrations of 4180, 1424, and 1050 mg kg−1 in contaminated soil and in plant roots and shoots, showing that this plant has a high root and shoot enrichment coefficients and can be applied as bioaccumulator (Yildirim and Sasmaz 2017).
The use of more than one plant for soil decontamination has also been explored (as shown in Table 1). Recently, Desjardins and colleagues studied the use of three different plants, grown in mono- and polyculture, for phytoremediation of trace metals. They reported that combinations of two (F. arundinacea and M. sativa) and three (S. miyabeana, F. arundinacea, and M. sativa) plants resulted in higher lead accumulation than the use of M. sativa monoculture: 21.6 ± 3.9, 20.6 ± 4.7, and 6.3 ± 1.2 mg Pb m−2, respectively (Desjardins et al. 2018).
Although some authors have examined the combination of phytoremediation and chelating agents (Attinti et al. 2017; Babaeian et al. 2016; Mani et al. 2015; Zaier et al. 2014) in an attempt to increase metal bioavailability and plant uptake, it is important to evaluate the potential toxicity of chelating agents to plants and soil microorganisms, as well as the degradation of such agents (Evangelou et al. 2007).
Remediation using nanoparticles
Metallic nanoparticles have been used to remediate lead. However, caution must be taken to avoid using a nanoparticle that might pollute the environment. In this context, iron nanoparticles are a suitable option (Laidlaw et al. 2017).
Remediation depends on the standard redox potential (E0) of the metal contaminant in relation to the standard redox potential of iron (E0 = − 0.44 V). If E0 of the toxic metal is significantly lower than iron E0, the pollutant will be adsorbed in iron. On the other hand, if E0 of the toxic metal is significantly higher than iron E0, the toxic metal will undergo reduction and precipitate (Laidlaw et al. 2017). Lead (E0 = − 0.13 V) is a metal that can be either adsorbed or reduced, and the iron nanoparticles can also be composed of oxides/hydroxides that can co-precipitate with lead, diminishing lead solubility in soil (Laidlaw et al. 2017).
Lead can be distributed in a more stable fraction in the soil such as the organic, residual, and Fe-Mn fractions by using ZVIN-Starch (starch stabilized zerovalent iron nanoparticles) measuring 9.28 nm as described by Okuo and colleagues (Okuo et al. 2018). Treatment of contaminated soil with 1000 mg kg−1 ZVIN-Starch decreases the lead mobility factor by 18.87%.
Gil-Díaz and co-workers studied the efficiency of zerovalent iron nanoparticles (ZVINs) for lead immobilization in acidic soil and observed that these nanoparticles reduce leachability by 98% (Gil-Díaz et al. 2014). Recently, Moazallahi et al. also reported on the efficiency of ZVINs for lead immobilization. Among the three different synthesized ZVINs, the most effective is ZVINEDTA (ZVINs stabilized by EDTA), which diminishes lead release by 86.4% (Moazallahi et al. 2017).
Various types of soil can be treated with iron nanoparticles. Emadi and colleagues reported that ZVINs measuring 50 nm decrease extractable lead with increasing ZVIN dose at contamination levels of 50 and 150 mg kg−1. Some kinds of soil (sandy, acidic, and calcareous soils) have been evaluated, to show that acidic soil experiences the greatest reduction in the extractable lead (Emadi et al. 2016).
Hydroxyapatite (HAP) is another nanomaterial that is effective for remediation. Li et al. studied lead immobilization in soil by hydroxyapatite at the nano and micrometer scale (nHAP and mHAP, respectively). Although both these phosphate-based materials effectively immobilize lead, mHAP is even better than nHAP (Li et al. 2014).
Nanoadsorbents of TiO2 have also been evaluated for remediation of lead in soil. Recently, Khalaf et al. synthesized three different mesoporous TiO2 by using CTAB, SDS, and PEG as cationic, anionic, and non-ionic surfactant template, respectively, and obtained the materials designated Ti C, Ti S, and Ti P. The authors reported spontaneous lead adsorption on TiO2 surfaces due to negative ΔG0ads values; lead adsorption follows the order Ti S > Ti C > Ti P. The total adsorptive capacity of these materials in soil containing 100 mg L−1 lead is 97.6, 99.1, and 95.3% for Ti C, Ti S, and Ti P, respectively (Khalaf et al. 2018).
Arsenic
Sources of arsenic and soil contamination with arsenic
Arsenic (As) is a crystalline metalloid, a natural element with intermediate features between metals and non-metals. It is the 20th most abundant element in the geosphere and is considered extremely toxic to the environment and human health. Arsenic minerals and compounds are usually soluble; however, arsenic mobility is limited because it has a strong tendency to adsorb onto clays, organic matter, and hydroxides (Singh et al. 2015; Tarvainen et al. 2013; Vithanage et al. 2017).
The primary source of arsenic in the environment is its release from arsenic-enriched minerals (including volcanic rocks, coal, and geothermal waters) due to geochemical factors. Anthropogenic actions that release arsenic include industrial processes, mining, coal combustion, wood preservation, fossil fuel processing, application of phosphate fertilizers and herbicides, and incineration of municipal and industrial wastes (Singh et al. 2015; Wang and Mulligan 2006).
According to the U.S. Environmental Protection Agency, the permissible limit of arsenic in soil is 24 mg kg−1. Table 2 summarizes soil contamination with arsenic in different countries (Singh et al. 2015).
Toxic effects of arsenic and its different species
Arsenic speciation is an important topic to investigate why many species have different mobility in soil and distinct toxicity. Arsenic speciation depends on its source and on environmental conditions like moisture content, redox potential, and microorganisms (Fayiga and Saha 2016). Different arsenic species can be found in organic or inorganic compounds, and arsenic exists mainly in four oxidation states: (i) As(V), arsenate; (ii) As(III), arsenite; (iii) As0, arsenic; and (iv) As−3, arsine. As(III) compounds are more mobile than As(V) compounds, suggesting that As(III) migration in the soil is more favorable (Guemiza et al. 2017).
In general, inorganic arsenic species are more toxic than organic ones. As(III) is the most toxic arsenic species, and organic forms of As(III) have been reported to be more toxic than inorganic ones (Fayiga and Saha 2016; Singh et al. 2015).
Methanogenic bacteria convert inorganic arsenic to organic forms by reducing As(V) to As(III) and methylating both these species to give monomethylarsonic acid (CH3AsO(OH)2 or MMA) and dimethylarsinic acid ((CH3)2AsOOH or DMA). This conversion can be represented by the following equation (Jaishankar et al. 2014):
Other organic arsenic species exist, including trimethylarsine oxide ((CH3)3AsO or TMAO), arsenobetaine ((CH3)3As+CH2COOH or AsB), arsenocholine (AsC), arsenosugars (AsS), and arsenolipids (Fayiga and Saha 2016; Singh et al. 2015).
The organic forms MMA and DMA have been widely used as pesticides and herbicides; DMA is also employed as a cotton defoliant. Therefore, in terms of toxicity, these arsenic forms can be ordered as follows: MMA(III) > As(III) > As(V) > DMA(V) > MMA(V) (Singh et al. 2015).
The toxic effects of arsenic are well documented: cancer, pigmentation changes, hyperkeratosis, neurological disorders, muscular weakness, and cardiovascular diseases such as hypertension and stroke. Moreover, long-term exposure to low arsenic doses can induce varying chronic health effects (McGrory et al. 2017; Mohan and Pittman 2007). Women are especially affected by arsenic during pregnancy—this element underlies spontaneous abortion, stillbirth, and preterm birth rates (Ahmad et al. 2001).
Remediation of arsenic in soil
In view of the environmental and health problems caused by soil contamination with arsenic, there has been increasing concern about soil remediation, and various strategies and methods have been developed to remediate arsenic (Song et al. 2017). Table 3 shows numerous methodologies for the remediation of arsenic in the soil.
Phytoremediation of arsenic in soil
Phytoremediation, which is also known as phytoaccumulation, phytoabsorption, or phytosequestration, uses plants to extract pollutants like toxic metal(oid)s through the plant roots, causing the pollutants to translocate and to accumulate in aboveground biomass. This method is viewed as an ecologically responsible alternative for environmental remediation (Alkorta et al. 2004; Mahar et al. 2016).
Phytoremediation offers the following advantages: it can effectively extract toxic metal(oid)s non-intrusively, it is easy to implement and to maintain, it reduces the amount of waste going to landfills, it is inexpensive, and it can be employed with other techniques to improve the remediation efficiency. Nevertheless, some disadvantages can be cited: it is limited by contaminant depth (roots), solubility, and availability; it requires a long time of exposure; it depends on the climate season, and contaminants can be transferred to the food chain (Alkorta et al. 2004).
To be applied in phytoremediation, a plant should be tolerant of high levels of the contaminant, have a rapid growth rate, and accumulate reasonable levels of the contaminant (Alkorta et al. 2004). Table 4 depicts the plants that are most frequently employed to remediate arsenic in the soil.
Recently, Kumar et al. reported that arsenic successfully accumulates in Pongamia pinnata: about 1129–3322 mg kg−1 of dry weight in roots, with bioaccumulation and translocation factors > 1. Therefore, this plant can potentially remediate arsenic in soil (Kumar et al. 2017).
Genetic engineering has attracted attention because it can help to improve the levels of plant tolerance to contaminants and their accumulation in plants. Nahar and colleagues modified the tobacco (Nicotiana tabacum) genome with the Arabidopsis thaliana AtACR2 gene. They found that transgenic tobacco is more tolerant of arsenic than wild-type tobacco, which helps to promote the field of genetic engineering applied to soil remediation (Nahar et al. 2017).
Coupling phytoremediation with the addition of bacteria enhances remediation. Franchi and co-workers observed that phytoextraction assisted by indigenous PGP bacteria (PGPB) makes the phytoremediation of arsenic 85% more effective (Franchi et al. 2017).
Recently, Lei and colleagues used the plant Pteris vittata to remediate arsenic for 2 years. They reported 16.09% remediation efficiency calculated from arsenic uptake by P. vittata (Lei et al. 2018). However, this plant did not provide a level of remediation with efficiency as high as the efficiency reported for other plants discussed in this topic.
Soil washing
Soil washing uses washing solutions to extract pollutants from soil. The extraction process is controlled by dissolution of the metal(oid)-mineral bond, which is followed by dispersion of the contaminant in the washing solution (Fig. 5) (Ko et al. 2006).
In this methodology, selecting the washing solution(s) is the most important step: the extraction efficiency of a certain contaminant depends on the washing solution features, but the soil characteristics and the bond strength must also be considered (Jang et al. 2007). Particle size distribution in the soil can also be determinant in this method: in most cases, the contaminants will concentrate on the fine particle fraction. Thus, information about soil characteristics including soil particle fractions can significantly improve the efficiency of the method (Ko et al. 2006).
Several washing solutions for remediation of arsenic in soil have been studied, e.g., NaOH, H3PO4, EDTA, oxalate, and HCl. Arsenic solubility increases with increasing pH, so an alkaline washing solution is best to treat arsenic (Cao et al. 2016). Cao et al. reported that oxalate is able to remove 52% of arsenic from contaminated soil. Moreover, soil washing combined with zerovalent iron treatment gives arsenic removal as high as 94% after 120 min (Cao et al. 2016). Different washing solutions can be combined to increase the remediation efficiency. The sequence of washing solutions phosphoric acid-oxalic acid-Na2EDTA (POE) removes 41.9% of arsenic as reported by Wei and co-workers (Wei et al. 2016a).
Soil washing extracts toxic metal(oid)s adsorbed onto soil and reduces the volume of contaminated soil. Furthermore, this method can be applied to large contaminated areas because it is efficient and fast (Jang et al. 2007). Nevertheless, this method can modify the soil properties related to soil quality, including soil texture, water holding capacity, organic matter content, and total nitrogen concentration (Im et al. 2015). For this reason, Wang and colleagues recently investigated how various washing solutions impact arsenic removal from soil. According to these authors, NaOH decreases the total phosphorus, organic carbon, and nitrogen content; H3PO4 lowers Ca, Mg, Al, Fe, and Mn concentrations, but it increases total nitrogen and phosphorous contents; and EDTA decreases the total organic carbon, phosphorous, Ca, Mg, Al, Fe, and Mn contents. However, the soil treated with NaOH provides the best conditions for wheat to grow compared with the other washing solutions (Wang et al. 2017b).
Simultaneous application of oxalic acid and dithionite to extract arsenic from amorphous and crystalline iron oxides present in the soil is less effective. Lee et al. observed that the use of each solution alone is not efficient, either. During the application of oxalic acid after dithionite is added to soil, the amount of arsenic bound to amorphous iron oxides and dithionite produces reduced forms of sulfur, which reacts with the extracted arsenic to form an arsenic sulfide precipitate as residual fraction. In turn, simultaneous application of oxalic acid and dithionite removes 74% of arsenic bound to amorphous iron oxides and 65% of arsenic bound to crystalline iron oxides (Lee et al. 2018).
Electrokinetic remediation
Electrokinetic remediation (EKR) consists in applying an electrical field to the contaminated soil to remove toxic metal(oid)s. The main goal is to get the contaminants to migrate under the applied electric field via electroosmosis, electromigration, or electrophoresis (Shin et al. 2017; Virkutyte et al. 2002).
During electroosmosis, the soil moisture moves from the anode to the cathode of an electrolytic cell. During electromigration, ions are transported to the electrode of opposite charge. During electrophoresis, contaminants bound to mobile matter can be transported and eliminated under an electric field (Fig. 6) (Virkutyte et al. 2002).
The advantages of EKR include its potential applicability to several pollutants and its high efficiency and time-effectiveness in medium with low permeability such as soil (Kim et al. 2005). Nonetheless, this method has drawbacks: (i) it consumes a high amount of electricity, which raises remediation costs; (ii) the resulting OH− reacts with cations, to form sediments that decrease the diffusive flow because they clog the spacing between soil particles; (iii) the gas produced during electrolytic dissociation is attracted to the electrode surface, which slows remediation down because bubbles cover the cathode surface and increase resistance (Suzuki et al. 2013; Virkutyte et al. 2002).
Ryu and colleagues obtained positive results when they used the EKR method to remediate arsenic in soil. They observed that NaOH addition increased the removal of both As(III) and As(V), which was 25% in one of the samples (Ryu et al. 2017).
To boost EKR efficiency, different methods can be combined with EKR. Soil washing can be accomplished before EKR, as reported by Mao and co-workers. The latter authors employed a KH2PO4 solution for the washing and, after this process, they conducted EKR and achieved arsenic removal efficiency of 52% in the cathode area (Mao et al. 2017). Phytoremediation can also be used with EKR to enhance remediation efficiency. Mao and colleagues observed that both these methodologies could be coupled to improve remediation. The latter authors reported that arsenic bioaccumulation in plants (Indian mustard, spinach, and cabbage) goes up, indicating that EKR is a good method to be coupled with phytoremediation (Mao et al. 2016).
Remediation using nanoparticles
Nowadays, there has been increasing interest in the use of nanoparticles (NPs) in environmental remediation. NPs have small size, high reactivity, and large surface area, which help to improve the adsorption of contaminants. For example, iron oxide NPs offer 10 times greater sorption capacity than microscale particles (Liang and Zhao 2014). The advantages of this method are its low cost, low energy demand, and applicability to in situ treatment. However, it is important to consider the effects of NPs on the environment (Wang et al. 2012b).
Many NPs have been studied for the adsorption (and consequently remediation) of arsenic (Goswami et al. 2012; Nabi et al. 2009; Zhang et al. 2010). Iron oxide NPs such as hematite and magnetite and zerovalent iron NPs are the most promising for this purpose (Liang and Zhao 2014; Zhang et al. 2010). The mechanism of interaction of As and iron nanoparticle compounds, such as FeS, is different depending on the oxidation of As. The As(V) interacts with FeS particles by forming an outer sphere complexation and chemical complexation, and the As(III) at pH > 6 interacts by surface sorption, on the other hand, in pH 5, the interaction occurs by the following equation (Gong et al. 2016):
Some studies have employed zerovalent iron NPs (ZVI NPs) to remediate arsenic in soil [61,62]. Gil-Díaz and co-workers reported that arsenic in the exchangeable fraction decreases by 70% in the presence of 5% ZVI NPs (Gil-Díaz et al. 2017).
Magnetite NPs (magNPs) can also be used to decontaminate soil polluted with arsenic. Yang and colleagues observed that magNPs reduce arsenic concentration in the easily extractable phase due to adsorption of the contaminant onto the NPs (Yang et al. 2017). Liang et al. studied As(V) immobilization by magNPs and reported a 93% decrease in As(V) in the water-leachable phase, indicating that these NPs can stabilize arsenic in soil and reduce its mobility (Liang and Zhao 2014).
Recently, Li et al. synthesized ZVI NPs supported on zeolite (Z-NZV) to remediate arsenic in a real sample. They observed that arsenic adsorption depended on the soil pH: alkaline soil promoted co-precipitation of As(V) and iron hydroxide, thus enhancing arsenic immobilization. Nevertheless, in both types of soil, acid, and alkaline, the available arsenic concentration decreased: 30 mg kg−1 Z-NZV in the soil samples lowered arsenic to 30% and 13% of the initial arsenic concentration in acid soil and in alkaline soil, respectively (Li et al. 2018).
Babaee and Mulligan employed Fe/Cu NPs to remediate arsenic in a chromated copper arsenate (CCA)-contaminated soil. The NP solution effectively reduced arsenic leachability: the water-soluble arsenic was transferred to the nanoparticle phase, and the water-leachable arsenic concentration decreased by 92% (Babaee and Mulligan 2018).
NPs of binary oxides can also be prepared to immobilize arsenic in soil, thereby working as an in situ technique. Fe-Mn binary oxide NPs efficiently immobilize arsenic: water-leachable arsenic decreases by 91–96%, and the toxicity characteristic of the leaching procedure (TCLP) reduces by 94–98% (An and Zhao 2012).
Mercury
Sources of mercury and soil contamination with mercury
Mercury (Hg) and its compounds are potentially hazardous to all organisms. Trace levels of this element occur naturally in the environment through volcanic activities and geological weathering. However, anthropogenic action has increased mercury release into the environment by a factor of three to ten. The largest sources of mercury emission are fossil fuel burning, which releases elemental mercury, and artisanal and small-scale gold mining: these activities release more than 1000 tons of mercury each year (Esdaile and Chalker 2018; Odumo et al. 2014; Pogrzeba et al. 2016; Provencher et al. 2014). Mercury is emitted into the atmosphere primarily as Hg0 and is removed from the atmosphere after its oxidation to Hg2+, which is then deposited in either aquatic or terrestrial environment, where it is more persistent than in any ecosystem (Lamborg et al. 2014; Xu et al. 2015).
Mercury emission from anthropogenic sources is approximately 2500 tons yearly. This emission is distributed into the air, water, and land at a 45:7:48 ratio (Odumo et al. 2014). In this scenario, regulatory agencies have estimated guideline mercury levels in soil to protect both the environment and human health. In the UK, the inorganic mercury guideline level depends on land use; the lowest value is 8 μg g−1. In Canada, this value is 6.6 μg g−1. In the Netherlands, the maximum permissible added content of mercury is 1.9 μg g−1 (Tipping et al. 2010).
Toxic effects of mercury and its different species
Mercury is recognized as a highly toxic metal that causes severe harmful effects to human health and the environment. Brain damage, central nervous system (CNS) malfunctioning, memory loss, cardiac disease, liver damage, blindness, and loss of sensation are among its toxic effects on human health. Additionally, mercury can affect pregnant women and their babies because this metal can pass through the placenta and lead to low birth weight, as well as delayed neurodevelopment, growth, and development (Ha et al. 2017; Mahbub et al. 2017).
The mercury species can exist both in the inorganic and organic form and their toxicity and bioavailability depend on the form. Inorganic mercury exists both as elemental mercury (Hg0) and divalent mercury (Hg2+). Exposure to Hg0 occurs mainly through dental amalgam, and workers at small-scale gold mining can also be exposed to this mercury species. The most soluble form of mercury is mercury chloride: it is more easily transported than other inorganic forms; moreover, it serves as a substrate for methylation. Hg0 and its amalgams are less toxic than inorganic soluble forms. In the event of contamination with inorganic mercury, the target organs include the brain and kidneys. High mercury ingestion results not only in brain and kidney damage but also in digestive tract disorder, which can culminate in death (Ha et al. 2017; Rice et al. 2014; Sysalová et al. 2017).
The most common form of organic mercury is methylmercury (MeHg), which is the major source of organic mercury in the environment. The main source of MeHg is seafood and fish (Ha et al. 2017; Rice et al. 2014). Compared with other mercury forms, MeHg has the highest bioavailability and can cause permanent injury to the CNS because it is easily absorbed by the digestive tract (approximately 95% absorption) (Ha et al. 2017; Rice et al. 2014).
Remediation of mercury in soil
During soil remediation, Hg cannot be degraded in the same way as organic contaminants. Therefore, different methodologies to remediate mercury in soil had to be developed, including soil washing, thermal treatment, phytoremediation, and use of nanoparticles (Xu et al. 2015).
Soil washing
The soil washing method can be applied to remediate mercury in soil. This method uses an extractant solution that decreases the mercury concentration in soil. This method depends on soil characteristics like particle size and organic matter (OM) content. In soil, OM at high levels tends to bind mercury, thereby interfering in mercury distribution in particle size fractions and in mercury mobilization, which limits the effectiveness of this method (Xu et al. 2015).
Although soil washing is inexpensive compared with thermal treatment, and even though a smaller volume of soil has to be further treated or disposed after soil washing, issues may arise when (i) mercury is strongly bound to soil particles owing to high levels of insoluble humic substances, (ii) mercury is present in all particle size fractions, (iii) differences in density or surface properties between mercury-bearing and clean particles are not significant (Xu et al. 2015).
Different extractants can be employed to remediate mercury, including EDTA, thiosulfate, and iodide (I−) solutions. Iodide increases the solubility of mercury in soil by forming a soluble complex, HgI42−, as shown by the following equation (Wang et al. 2012b):
EDTA and thiosulfate solutions can remove mercury with 30% efficiency. Nevertheless, better results can be achieved by using a mixture of HCl and KI, which can provide mercury removal efficiency as high as 77%. The only problem is that this mixture changes the soil properties (Wang et al. 2012b).
Sodium sulfite is also an extractant solution that promotes mercury desorption in soil (Lu et al. 2017; Qi et al. 2017). Recently, Qi and colleagues obtained 92.05% mercury desorption by using 0.7 mol L−1 sodium sulfite after 24 h (Qi et al. 2017).
The soil washing method can also employ supercritical fluids like CO2. To this end, mercury must be strongly chelated with a ligand that is soluble in the supercritical fluid, and the resulting complex must be soluble in the medium (He et al. 2015).
Thermal treatment
Thermal treatment employs high temperatures to remove mercury from the soil through volatilization followed by condensation of mercury vapors into liquid elemental mercury (Fig. 7) (Xu et al. 2015).
Whereas this method provides acceptable levels of decontamination, it consumes high amounts of energy for temperatures above 600 °C to be reached, and the treated soil is not suitable for agricultural reuse (Ma et al. 2015).
Inorganic mercury is usually present as Hg0 or as Hg(II) compounds like HgS, HgO, and HgCO3 in soil. When the thermal treatment temperature reaches 600–800 °C, these mercury compounds are converted into gaseous elemental mercury, which can be easily recovered (Wang et al. 2012b).
The temperature employed during thermal treatment depends on the phase to which mercury is linked and on mercury speciation. Temperatures ranging from 460 to 700 °C afford greater efficiency (Sierra et al. 2016; Xu et al. 2015).
Hung et al. reported high mercury removal when they used this method. Despite the high temperature of the thermal treatment, 700 °C, these authors obtained 97.8% of mercury removal from a soil simultaneously contaminated with mercury and pentachlorophenol (Hung et al. 2016).
Recent studies have been geared toward reducing energy costs by decreasing the treatment temperature and by using solar energy. Zhao and co-workers removed 70% of mercury by treating soil at 350 °C for 30 min in engineering-scale experiments (Zhao et al. 2018). Citric acid can also be used to reduce the treatment temperature. Ma et al. showed that citric acid reduces the energy input by 35% compared with the traditional thermal treatment method. More specifically, mercury concentration decreases from 134 to 1.1 mg/kg when a temperature of 400 °C is used for 60 min (Ma et al. 2015). Sierra and colleagues controlled thermal desorption by using a solar furnace, which is a sustainable alternative for this treatment. They reported a total mercury release of more than 80% at 280 °C, indicating that the technique is efficient (Sierra et al. 2016).
Phytoremediation
Phytoremediation can also help to remediate mercury in soil and involves phytostabilization, phytoextraction, and phytovolatilization. Phytostabilization immobilizes mercury in soil through mercury absorption and accumulation in roots, thereby preventing mercury migration by soil erosion. During phytoextraction, mercury is absorbed and translocated by roots into plant parts lying above the ground, which can then be harvested. Phytovolatilization relies on the high volatility of mercury, which is taken up by roots, transported through the xylem, and released from cellular tissues into the atmosphere. Efficient mercury absorption by plants depends on factors that affect the availability of the contaminants, e.g., soil pH, organic matter, microbial biomass, and competitive cations (Sarwar et al. 2017b; Xu et al. 2015).
Some plants listed in Table 5 can be applied in phytoremediation. Marrugo-Negrete’s research group studied the plant Jatropha curcas and observed that mercury concentration behaves as follows: roots > leaves > stems. The highest cumulative mercury absorption occurs between the second and the third month of exposure. Although the mercury concentration absorbed by aerial parts is low, the researchers concluded that this plant can be used in phytoremediation because it can adapt to eroded soil and is easily found near mining areas affected by pollution (Marrugo-Negrete et al. 2015).
As recently reported by Franchi and colleagues, addition of bacteria to the phytoremediation process can increase the remediation efficiency. The authors verified that indigenous PGP bacteria (PGPB) boost the phytoaccumulation efficacy for both B. juncea and L. albus. In the former plant, mercury phytoaccumulation reaches 45% (Franchi et al. 2017).
Mobilizing agents such as EDTA and HCl can improve this methodology. Rodríguez et al. tested EDTA and HCl solutions and observed that EDTA reduces plant (Lupinus albus) growth. Although mercury absorption takes place, phytoextraction is not significantly higher compared with the control sample. In turn, HCl improves root growth, and mercury uptake is 3.7 times higher compared with the control sample (Rodríguez et al. 2016).
Remediation using nanoparticles
In recent years, immobilization of toxic metal(oid)s by NPs has become a promising alternative. Mercury tends to form strong bonds with sulfur because it behaves as a soft Lewis acid. Interestingly, HgS formation is considered one of the primary mercury sinks in the environment (Gong et al. 2012). Possible mercury reactions in sulfidic soil include (Gong et al. 2012):
The affinity between mercury and sulfur makes mercury immobilization in soil with FeS NPs a promising remediation methodology. For example, Gong et al. reported that FeS NPs decrease leachable mercury in soil by 90–93% (Gong et al. 2012).
Selenium (Se) belongs to the same group as sulfur in the Periodic Table and also has affinity for mercury. Therefore, SeNPs can be a promising material to remediate mercury in soil. SeNPs can efficiently immobilize mercury through formation of HgSe, which is a less toxic compound. About 45.8–57.1% and 39.1–48.6% of mercury can be converted into HgSe under anaerobic and aerobic conditions, respectively (Wang et al. 2017a).
Zerovalent iron NPs can also be employed to remediate soil contaminated with mercury. Gil-Díaz and colleagues investigated the use of zerovalent iron NPs for mercury immobilization in soil and observed that, at 10%, these NPs decrease the mercury exchangeable phase by 63–90% (Gil-Díaz et al. 2017).
Wang and colleagues recently explored the use of SeNPs stabilized by extracellular polymeric substances (EPS) from Citrobacter freundii Y9 and observed that SeNP EPS at low doses promote high Hg0 immobilization (over 88.8%) (Wang et al. 2018).
Chromium
Sources of chromium and soil contamination with chromium
Chromium (Cr) is the 21st most abundant metal in the Earth’s crust. It is extremely toxic to the environment and the human health. It occurs naturally in weathering rocks and volcanic gases and is adsorbed by minerals present in soil particles, such as manganese (Mn), aluminum (Al), and iron (Fe, as ferrochromite (Fe2Cr2O4)). Natural sources emit an estimated 43 thousand tons of chromium per year worldwide. Anthropogenic actions, like the production of pigments in textile industries and the tanning of leather in tanneries, also contribute to the release of this element into the environment (Hsu et al. 2015; Nriagu and Pacyna 1988; Shahid et al. 2017).
According to the State Environmental Protection Administration (SEPA) in China, the permissible limit of chromium in soil is 250 mg kg−1 (Khan et al. 2008). Table 6 depicts the contamination of soil with this element in different countries.
In Brazilian agriculture, the soil is disturbed by toxic metals such as Cr due to coal mines (Rodriguez-Iruretagoiena et al. 2015) and also from agricultural supplies such as fertilizers (Turra et al. 2010). It is also important to notice that since Brazil is among the five largest producers of leather, processing about 42 million cattle hides per year, the impact of this activity may be the release of Cr in the soil due to the inappropriate disposal (Araújo et al. 2014).
Toxic effects of chromium and its different species
Chromium speciation is an important topic because its species display distinct toxicity and soil mobility. The most common and stable chromium species in the environment is Cr(III), which forms a complex with organic matter present in soil and aquatic environments. Cr(III) can also occur in the form of chromic oxide (Cr2O3), hydroxide (Cr(OH)3), or sulfate (Cr2(SO4)3.12 H2O), which have low solubility at pH < 5.5. Indeed, the soil pH governs Cr(III) adsorption and desorption. Because Cr(III) compounds are highly stable in soil, they have lower mobility in plant roots. In addition, this species is less toxic than hexavalent Cr(VI), which is considered the most harmful form. Cr(VI) comes associated with chromate (CrO42−) or dichromate (Cr2O72−) ions, which are toxic to living organisms and promote carcinogenesis, mutagenesis, and teratogenesis. Furthermore, Cr(VI) is soluble in water; that is, it is more bioavailable, and consequently has greater mobility in soils and plants (Prado et al. 2016; Shahid et al. 2017; Sinha et al. 2018).
Remediation of chromium in soil
Due to the harmful effects of Cr to the environment and health, different methods of Cr remediation were studied. The remediation methods described in this section are in situ, which minimize the soil disturbance and cost as described in the “Remediation techniques” section. However, conditions such as climate, soil permeability, depth of contamination, and deep leaching of chemicals must be carefully examined (Liu et al. 2018).
Phytoremediation
Phytoremediation has been employed to remediate chromium in soil. In a study by Shahandeh and Hossner, thirty-six plant species of plants were investigated for Cr phytoremediation, although some species showed to be tolerant to Cr, they reported the difficulty of plants to be tolerant to Cr (VI) toxicity (Shahandeh and Hossner 2006). However, other studies reported some plants that can be tolerant to Cr (VI) and employed for soil phytoremediation as shown in Table 7.
Diwan et al. investigated the phytoremediation of Cr by different species of Indian mustard. Among the studied genotype, Pusa Jai Kisan is a hyperaccumulator of Cr with a maximum amount of 1680 μg Cr g−1 (for the dry weight) besides to be tolerant to the oxidative stress caused by Cr (Diwan et al. 2008).
Panikum (Panicum antidotale), Napier grass (Pennisetum purpureum), squash (Cucurbita pepo), cotton (Gossypium hirsutum), and sunflower (Helianthus annuus) were evaluated as Cr phytoremediator by Lotfy and Mostafa. The range of Cr removal by these plants was 29.9 to 36.5% and the sunflower roots showed the highest Cr accumulation about 58% (Lotfy and Mostafa 2014).
Recently, oregano was studied for Cr phytoremediation by Levizou and co-workers (Levizou et al. 2019). They reported a bioaccumulation of Cr(VI) of 4300 mg kg−1 for the dry biomass in both roots and aerial part in a soil containing a range of 150–200 mg kg−1 of Cr (Levizou et al. 2019).
Although some plants can be used as bioaccumulator, Cr can cause stress and decrease the effectiveness of the remediation. In view of this fact, microbial-mediated remediation can improve this process by decreasing the metal stress in the plant and also by altering soil pH, release of chelators, thus improving the phytoremediation process. The inoculation of P. aeruginosa in soil enhanced the Cr accumulation in maize shoots by a factor of 4.3 by the increase production of siderophores (Pinto et al. 2018).
Bioremediation
Bioremediation remediates soil through microorganisms, which are stimulated to degrade contaminants to acceptable levels in soil and effluents (Liu et al. 2018).
The microbial mechanisms of chromium detoxification include absorption and oxidation. Absorption is known as bioaccumulation or biosorption; oxidation is called biorreduction and entails reduction of Cr(VI) to Cr(III), which is less toxic, immobile, and insoluble. Biosorption is an inexpensive, fast, and reversible physicochemical process involving the metallic species (sorbate) and the biological material (biosorbent); the surface/volume ratio is high. However, metals accumulate through various mechanisms, such as transport, formation of extracellular complexes, and precipitation (Fernández et al. 2018).
Microorganisms can remove Cr(VI) through biosorption processes because the microbial cell wall contains functional groups like carboxylate, hydroxyl, amino, and phosphate, which can bind to the ions of potentially toxic metals (Fernández et al. 2018).
Ksheminska et al. reported that Cr(VI) compounds enter the microorganism cells through the anion channel via non-selective diffusion and are sensitive to the oxidative state. Cell membranes are impermeable to Cr(III), supposedly because they form little soluble complexes (Ksheminska et al. 2006).
Electrokinetic remediation
Electrokinetic remediation is an effective and environmentally friendly technique to remove toxic metals, like Cr, from contaminated soil.
As discussed in the “Toxic effects of chromium and its different species” topic, the toxicity of the Cr(III) and Cr(VI) are different, thus the reduction of Cr(VI) to Cr(III) is also important in order to decrease it toxicity. Wei et al. added NaHSO3 to the soil before EKR and reduced 90.3% of Cr(VI) to Cr(III). The method of EKR with approaching anodes (Aas) was effective in Cr removal with 64.4% of efficiency for the total Cr concentration (Wei et al. 2016b).
The soil acidification can improve of EKR efficiency of Cr removal. Meng and co-workers reported that after addition of citric acid (0.9 mol L−1) in soil the Cr(VI) and total Cr removal rates were 77.66% and 26.97% respectively, while in soil without addition of citric acid, the removal rates were 6.23% and 19.01% for total Cr and Cr(VI), respectively, after a period of 5 days (Meng et al. 2018). Although in the study of Meng et al. the change in pH of soil after addition of acid citric was relatively small (from 8.54 before the addition to 8.43 after addition of citric acid), it is important to mention that changes in pH can affect the microbiota and nutrient uptake of the plants.
The pulsed electrokinetic remediation (PEKR) is an improvement of the EKR method by pulsing the electric field in the electrokinetic remediation with reduction of energy consumption. Mu’azu et al. reported an achievement of 89.64% in Cr remediation in bentonite soil by using PEKR (Dalhat et al. 2016).
The concern about methods that follow the principles of green chemistry is getting attention and due to this, to reduce the energy input is important in the EKR method. In view of this challenge, recently, Zhou and colleagues investigated the efficiency of solar energy with exchange electrode-EKR (EE-EKR) for Cr remediation. After the remediation, the residual concentration was 10% and 58% for Cr(VI) and total Cr, respectively (Zhou et al. 2018).
Remediation using nanoparticles
Nanoparticles are used as adsorbent material due to their high specific surface area (availability of active sites), high reactivity, and high reduction capacity. According to Cao et al. (2006), 1 g of iron nanoparticles can reduce 69.3–72.7 mg of Cr(VI) in chromium ore. Singh et al. (2012) revealed that the efficiency of the remediation of Cr(VI) in soil is 99% after 40 days of contact with 5 g L−1 iron nanoparticles (Cao et al. 2016; Singh et al. 2012).
Wang et al. synthesized iron sulfide nanoparticles (FeS NPs) stabilized with carboxymethyl cellulose (CMC) for Cr(VI) remediation. The FeS NPs decreased the Cr(VI) concentration from 4.58 mg L−1 to 46.8–80.7 μg L−1 (Wang et al. 2019).
Recently, nanomagnetic MnFe2O4 were employed for Cr immobilization in soil. Eyvazi and co-workers reported the decrease of leachability of Cr(VI) in soil from 70.95 to 4.22% using 2 g L−1 of nanomagnetic MnFe2O4 during 192-h remediation time. Besides the high efficiency of Cr(VI) immobilization, they observed a high decrease in bioaccessibility of Cr(VI) in soil with a reduction from 86.76 to 4.42% (Eyvazi et al. 2019).
Carbon nanotubes comprise layers of coiled graphene that form a cylinder. They exhibit excellent thermal, electrical, and mechanical properties and are also employed for Cr remediation (Alim et al. 2018). Upon adsorption onto the surface of multi-walled carbon nanotubes, Cr(VI) can be reduced to Cr(III), and part of the latter species can be released into solution along the adsorption process, in acidic medium (Anastopoulos et al. 2017).
Carboxylated and hydroxylated multi-walled carbon nanotubes (MWCNT-COOH and MWCNT-OH) were studied for Cr(VI) adsorption by Zhang et al. and they reported that the Cr adsorption of MWCNT-COOH and MWCNT-OH could reach 8.09 and 7.85 mg g−1. They also observed the influence of pH which its decrease enhances the Cr(VI) adsorption in both carbon nanotubes (Zhang et al. 2018).
Remediation using biochar
Biochar is another kind of material with a potential to be employed in soil remediation, which is the carbon-rich final product of the thermal degradation of organic biomass in the absence of oxygen (Lahori et al. 2017).
The mechanisms involved in the immobilization of pollutants by biochar include (i) electrostatic attraction: due to the high electronegativity of biochar, the electrostatic attraction of positively charged ions is facilitated; (ii) ion exchange: the high CEC of the biochar release cations which exchange the metal ions from soil; (iii) complexation: the surface functional groups from biochar, such as –OH, –COOH, –C=O–, and C=N, can immobilize the toxic metals by complexation; and (iv) precipitation: some mineral elements in the biochar may precipitate with metals, such as inorganic P which can precipitate Pb forming Pb10(PO4)6(OH)2 (He et al. 2019).
The efficiency of biochar-supported zerovalent iron nanoparticles (nZVI@BC) for Cr(VI) remediation was studied by Su and co-workers. The immobilization efficiency of nZVI@BC reached 100% by 8 g nZVI@BC per kg of soil for 15 days of remediation. They also reported that the exchangeable soil phase was almost completely converted to Fe-Mn oxides and organic matter, which are stable phases from the soil (Su et al. 2016).
Xu et al. reported that biochar acts as a redox process of soil active components. They evaluated the effects of Fe mineral and lactate, and observed that mineral iron decreases the Cr (VI) reduction rate by biochar due to surface oxidation and Fe-biochar complex coverage, attributed to Fe electron transfer (II) and by biochar (Xu et al. 2019).
Liu et al. demonstrated that the application of biochar in the remediation of Cr in soil is directly related to pH since the decrease of soil pH increases the availability of acid-soluble Cr. The application of biochar reduced the Cr availability rate by 55% (Liu et al. 2020).
Determination of toxic metals in soil
Toxic metal contamination in soil can be determined by a wide range of techniques and atomic absorption spectroscopy (AAS) (Souza et al. 2019), high-resolution continuum source atomic absorption spectrometry (HR CS AAS) (Souza et al. 2018), inductively coupled plasma optical emission spectroscopy (ICP-OES), and inductively coupled plasma mass spectrometer (ICP-MS) (Ran et al. 2016) are the most employed. However, different techniques can also be used for this purpose as detailed below.
Zhang and researchers synthesized a magnetic porous carbon adsorbent using a nitrogen-doped humic acid lignin coated for Cr (VI) adsorption and reduction. Analysis on AAS revealed that the adsorbent has a high adsorption capacity of 130.5 mg g−1 for Cr (VI), with partial reduction of toxic Cr (VI) to non-toxic Cr (III) (Zhang et al. 2020).
Zhao et al. quantitatively evaluated soil Pb by femtosecond-nanosecond double-pulse laser-induced rupture spectroscopy (DP-LIBS) analysis. The researchers demonstrate this technique is an efficient spectroscopic tool for analysis of Pb in soil, obtaining values of relative standard deviation (RSD), mean square cross-validation error (RMSECV), and detection limit (LOD) of 99.42%, 2.99%, 0.42% by weight and 8.13 mg/kg, respectively. They also observed that a lower density of aerosol particles from soil is produced by the filament ablation target; thus, the Pb determination was strongly dependent on the delay time between pulses (Zhao et al. 2019).
Cheng and co-workers investigated the accumulation of mercury in soil from atmospheric deposition in temperate steppe of Inner Mongolia, China. They determined the Hg by Zeeman atomic absorption spectroscopy (ZAAS) and achieved a detection limit of 0.5 mg kg−1. The average Hg content found in the topsoil and subsoil was 14.9 ± 10.4 μg kg−1 and 8.9 ± 5.8 μg kg−1, respectively (Cheng et al. 2019).
Concomitant soil decontamination of Pb, Hg, As, and Cr
In areas with high industrial/mining activity, different toxic metals can contaminate the soil, and in order to remediate these areas, it is important to take in account an efficient methodology for all the contaminants, since it is unlikely that only one of these elements will occur separately. As discussed in all previously section of this review, different methodologies have been applied for soil decontamination. Among these techniques, some can be applied for remediate multiple contaminants at the same time as showed in Table 8.
The phytoremediation is a technique that can remediate more than one toxic element, as reported by Mattina and co-workers. According to their study, tomato (Lycopersicon esculentum) showed a bioconcentration factor (BCF) of 0.18 and 0.01 for Pb and As, respectively, besides to be able to remediate organic compounds (chlordane) presenting a BCF value of 0.05 (Mattina et al. 2003). Although it is possible to remediate concomitantly different pollutants by phytoremediation, higher efficiency can be achieved using different techniques together. Mao et al. coupled the phytoremediation with electrokinetic remediation for Pb and As remediation. The acidification in the anode soil increased the water-soluble and exchangeable Pb and As, which consequently enhanced the solubility of these metals and increased their bioaccumulation in Brassica juncea, which were accumulated more in plant roots than in shoots (Mao et al. 2016).
Ferric and aluminum water treatment residues can be employed as immobilizers for remediation of multiple toxic metal(oid)s in soil according to the study by Wang and co-workers. Pb and As were transformed in more stable forms in the soil besides the decrease of As bioaccessibility by 25% (Wang et al. 2012a).
Nanoparticles also can remediate more than one metal in soil. Water treatment residual nanoparticles (nWTR) presented high efficiency in reducing the accessible forms of Hg and Cr in calcareous soil due to the formation of stable Hg and Cr species such as Hg(OH)2 amor, CrSO4. xH2O, and Cr(OH)2. Furthermore, the concentration of Hg and Pb increased in residual phase of the soil (from 69.27% and 52.62% to 93.89% and 90.05%, respectively) which is the more stable fraction of the soil (Moharem et al. 2019).
Future perspectives and challenges
A wide range of techniques can be employed to remediate soil, including the use of nanoparticles, phytoremediation, and electrokinetic remediation as mentioned in this review. However, new approaches within each of these technologies must also be explored in order to overcome the issues of each existing methodology.
Electrokinetic remediation requires considerable water and electrical energy, which can be a disadvantage. However, bioelectrokinetic remediation (Bio-EK) is a new strategy that is more environmentally friendly and which has potentially higher efficiency (Surya et al. 2018). The use of microorganisms can induce geochemical transformations of metals by oxidation or reduction (bioleaching) and facilitates precipitation for remediation (He et al. 2018). The bioleaching mechanism depends on the microorganism, but primarily, the process is driven by Fe- and S-oxidizing in aerobic conditions (Lee et al. 2011). The mechanism for Thiobacillus ferrooxidans is categorized by contact by electrostatic attachment and non-contact, with the production of extracellular substances and the formation of biofilm on the metal sulfide surface (Huang et al. 2017).
One example is a study by He et al., who reported that bio-EK enhances the efficiency of remediation of chromium in the soil to 90.67% in the presence of the bacterium Microbacterium sp. Y2 compared with the traditional EKR (80.26% of efficiency) (He et al. 2018).
Traditional phytoremediation methodologies are less economical for application on a large scale because hyperaccumulators grow slowly. Transgenic plants obtained through genetic engineering produce higher biomass, accumulate more metal, and adapt more easily to different climatic conditions, which make them effective for large-scale decontamination (Sarwar et al. 2017a). Considering that the efficiency of remediation in a plant is limited by the steps of the phytoremediation, the transgenic plants is improved in the following approaches: (i) the metal transporter proteins, which regulates the metal uptake; (ii) the translocation and storage of metal(iod)s in vacuoles; (iii) the production of detoxifying substances such as metallothioneins and phytochelatins in order to increase the plant tolerance to the pollutants; (iv) metabolic transformations of the metals in a non-toxic and volatile form. For this purpose, genes from other plant species and bacteria can also be introduced in order to improve the efficiency (Kaur et al. 2019).
Immobilization of NPs such as ZVIN onto supports is an innovative solution to overcome issues such as aggregation and formation of bulky corrosion products. Materials like carbons, resins, zeolites, and chitosan are easy to operate. Polyelectrolytes can be an alternative for soil deliverable ZVIN, which can be used for in situ remediation (Litter 2018).
Another promisor material for remediation is the biochar (BC), as discussed in the “Remediation using biochar” section. This material has a porous structure and large surface with micropores that can adsorb the pollutants present in soil (Lahori et al. 2017) (Fig. 8).
Conclusion
Undoubtedly, the environmental contamination, especially the soil contamination, is one of the great concerns nowadays and different remediation strategies have been developed to solve this issue.
Although many methods are available for soil remediation, as discussed in this review, it is important to take into account the disadvantages and specific use of them (Table 9). However, in order to overcome these disadvantages, these methodologies can be combined to increase the efficiency such as phytoremediation with electrokinetic remediation (Mao et al. 2016), soil washing and immobilization (Zhai et al. 2018), and the use of nanomaterials with phytoremediation (Zhu et al. 2019).
In conclusion, choosing the best method for soil decontamination depends on the contaminant, the treatment depth, the cost involved, and also the soil characteristics which, consequently, is not a simple choice. Furthermore, designing new methodologies and improving on the existing soil remediation methods (by reducing cost and boosting efficiency) are crucial to deal with a recurrent topic and to avoid contamination of food and the environment, as well as health hazards.
References
Adegbeye MJ, Ravi Kanth Reddy P, Obaisi AI, Elghandour MMMY, Oyebamiji KJ, Salem AZM, Morakinyo-Fasipe OT, Cipriano-Salazar M, Camacho-Díaz LM (2020) Sustainable agriculture options for production, greenhouse gasses and pollution alleviation, and nutrient recycling in emerging and transitional nations - an overview. J Clean Prod. https://doi.org/10.1016/j.jclepro.2019.118319
Ahmad SA, Sayed MH, Barua S, Khan MH, Faruquee MH, Jalil A, Hadi SA, Talukder HK (2001) Arsenic in drinking water and pregnancy outcomes. Environ Health Perspect 109:629–631. https://doi.org/10.1289/ehp.01109629
Alim S, Vejayan J, Yusoff MM, Kafi AKM (2018) Recent uses of carbon nanotubes & gold nanoparticles in electrochemistry with application in biosensing: a review. Biosens Bioelectron 121:125–136. https://doi.org/10.1016/J.BIOS.2018.08.051
Alkorta I, Hernández-Allica J, Becerril JM, Amezaga I, Albizu I, Garbisu C (2004) Recent findings on the phytoremediation of soils contaminated with environmentally toxic heavy metals and metalloids such as zinc, cadmium, lead, and arsenic. Rev Environ Sci Bio/Technology 3:71–90
Allevato E, Stazi SR, Marabottini R, D’Annibale A (2019) Mechanisms of arsenic assimilation by plants and countermeasures to attenuate its accumulation in crops other than rice. Ecotoxicol Environ Saf. https://doi.org/10.1016/j.ecoenv.2019.109701
An B, Zhao D (2012) Immobilization of As(III) in soil and groundwater using a new class of polysaccharide stabilized Fe-Mn oxide nanoparticles. J Hazard Mater 211–212:332–341. https://doi.org/10.1016/j.jhazmat.2011.10.062
Anastopoulos I, Anagnostopoulos VA, Bhatnagar A, Mitropoulos AC, Kyzas GZ (2017) A review for chromium removal by carbon nanotubes. Chem Ecol 33:572–588. https://doi.org/10.1080/02757540.2017.1328503
Antoniadis V, Polyzois T, Golia EE, Petropoulos SA (2017) Hexavalent chromium availability and phytoremediation potential of Cichorium spinosum as affect by manure , zeolite and soil ageing. Chemosphere 171:729–734. https://doi.org/10.1016/j.chemosphere.2016.11.146
Araújo SR, Demattê JAM, Vicente S (2014) Soil contaminated with chromium by tannery sludge and identified by vis-NIR-mid spectroscopy techniques. Int J Remote Sens 35:3579–3593. https://doi.org/10.1080/01431161.2014.907940
Asgari Lajayer B, Ghorbanpour M, Nikabadi S (2017) Heavy metals in contaminated environment: destiny of secondary metabolite biosynthesis, oxidative status and phytoextraction in medicinal plants. Ecotoxicol Environ Saf 145:377–390. https://doi.org/10.1016/j.ecoenv.2017.07.035
Attinti R, Barrett KR, Datta R, Sarkar D (2017) Ethylenediaminedisuccinic acid (EDDS) enhances phytoextraction of lead by vetiver grass from contaminated residential soils in a panel study in the field. Environ Pollut 225:524–533. https://doi.org/10.1016/j.envpol.2017.01.088
Babaee Y, Mulligan CN (2018) Arsenic immobilization in soil using starch-stabilized Fe/Cu nanoparticles: a case study in treatment of a chromated copper arsenate ( CCA ) -contaminated soil at lab scale. J Soils Sediments 18:1610–1619
Babaeian E, Homaee M, Rahnemaie R (2016) Chelate-enhanced phytoextraction and phytostabilization of lead-contaminated soils by carrot (Daucus carota). Arch Agron Soil Sci 62:339–358. https://doi.org/10.1080/03650340.2015.1060320
Bolade OP, Williams AB, Benson NU (2020) Green synthesis of iron-based nanomaterials for environmental remediation: a review. Environ Nanotechnol Monit Manag. https://doi.org/10.1016/j.enmm.2019.100279
Bolan N, Kunhikrishnan A, Thangarajan R, Kumpiene J, Park J, Makino T, Kirkham MB, Scheckel K (2014) Remediation of heavy metal(loid)s contaminated soils - to mobilize or to immobilize? J Hazard Mater 266:141–166. https://doi.org/10.1016/j.jhazmat.2013.12.018
Cao M, Ye Y, Chen J, Lu X (2016) Remediation of arsenic contaminated soil by coupling oxalate washing with subsequent ZVI/air treatment. Chemosphere 144:1313–1318. https://doi.org/10.1016/j.chemosphere.2015.09.105
CETESB (2016) VALORES ORIENTADORES PARA SOLO E ÁGUA SUBTERRÂNEA NO ESTADO DE SÃO PAULO. São Paulo
Chen H, Teng Y, Lu S, Wang Y, Wang J (2015) Contamination features and health risk of soil heavy metals in China. Sci Total Environ 512–513:143–153. https://doi.org/10.1016/j.scitotenv.2015.01.025
Chen L, Zhou S, Shi Y, Wang C, Li B, Li Y, Wu S (2018) Heavy metals in food crops, soil, and water in the Lihe River Watershed of the Taihu Region and their potential health risks when ingested. Sci Total Environ 615:141–149. https://doi.org/10.1016/j.scitotenv.2017.09.230
Cheng Z, Tang Y, Li E, Wu Q, Wang L, Liu K, Wang S, Huang Y, Duan L (2019) Mercury accumulation in soil from atmospheric deposition in temperate steppe of Inner Mongolia, China. Environ Pollut. https://doi.org/10.1016/j.envpol.2019.113692
Costa AM, Alfaia RG d SM, Campos JC (2019) Landfill leachate treatment in Brazil – an overview. J Environ Manag. https://doi.org/10.1016/j.jenvman.2018.11.006
Cristaldi A, Conti GO, Jho EH, Zuccarello P, Grasso A, Copat C, Ferrante M (2017) Phytoremediation of contaminated soils by heavy metals and PAHs. A brief review. Environ Technol Innov 8:309–326. https://doi.org/10.1016/j.eti.2017.08.002
Dalhat N, Usman A, Jarrah N, Alagha O (2016) Soil and sediment contamination: an international pulsed electrokinetic removal of chromium, mercury and cadmium from contaminated mixed clay soils. Soil Sediment Contam 25:757–775. https://doi.org/10.1080/15320383.2016.1213700
Danh LT, Truong P, Mammucari R, Foster N (2013) A critical review of the arsenic uptake mechanisms and phytoremediation potential of Pteris vittata. Int J Phytoremediation 16:429–453. https://doi.org/10.1080/15226514.2013.798613
Dapul H, Laraque D (2014) Lead poisoning in children. Adv Pediatr Infect Dis 61:313–333. https://doi.org/10.1016/j.yapd.2014.04.004
Derakhshan Z, Myung N, Jung C (2018) Remediation of soils contaminated with heavy metals with an emphasis on immobilization technology. Environ Geochem Health 40:927–953. https://doi.org/10.1007/s10653-017-9964-z
Desjardins D, Brereton NJB, Marchand L, Brisson J, Pitre FE, Labrecque M (2018) Complementarity of three distinctive phytoremediation crops for multiple-trace element contaminated soil. Sci Total Environ 610–611:1428–1438. https://doi.org/10.1016/j.scitotenv.2017.08.196
Ding C, Li X, Zhang T, Ma Y, Wang X (2014) Phytotoxicity and accumulation of chromium in carrot plants and the derivation of soil thresholds for Chinese soils. Ecotoxicol Environ Saf 108:179–186. https://doi.org/10.1016/J.ECOENV.2014.07.006
Diwan H, Ahmad ÆA, Iqbal ÆM (2008) Genotypic variation in the phytoremediation potential of Indian mustard for chromium. Environ Manag 41:734–741. https://doi.org/10.1007/s00267-007-9020-3
dos Reis DA, da Fonseca Santiago A, Nascimento LP, Roeser HMP (2017) Influence of environmental and anthropogenic factors at the bottom sediments in a Doce River tributary in Brazil. Environ Sci Pollut Res 24:7456–7467. https://doi.org/10.1007/s11356-017-8443-5
Du YJ, Wei ML, Reddy KR, Liu ZP, Jin F (2014) Effect of acid rain pH on leaching behavior of cement stabilized lead-contaminated soil. J Hazard Mater 271:131–140. https://doi.org/10.1016/j.jhazmat.2014.02.002
Emadi M, Savasari M, Bahmanyar MA, Biparva P (2016) Application of stabilized zero valent iron nanoparticles for immobilization of lead in three contrasting spiked soils. Res Chem Intermed 1–14. https://doi.org/10.1007/s11164-016-2494-y
Escobar H (2015) Mud tsunami wreaks ecological havoc in Brazil. Science (80-. ) 350:1138–1139
Esdaile LJ, Chalker JM (2018) The mercury problem in artisanal and small-scale gold mining. Chem Eur J 24:6905–6916. https://doi.org/10.1002/chem.201704840
Evangelou MWH, Ebel M, Schaeffer A (2007) Chelate assisted phytoextraction of heavy metals from soil. Effect, mechanism, toxicity, and fate of chelating agents. Chemosphere 68:989–1003. https://doi.org/10.1016/j.chemosphere.2007.01.062
Eyvazi B, Jamshidi-zanjani A, Khodadadi A (2019) Immobilization of hexavalent chromium in contaminated soil using nano- magnetic MnFe 2 O 4. J Hazard Mater 365:813–819. https://doi.org/10.1016/j.jhazmat.2018.11.041
Fayiga AO, Saha UK (2016) Arsenic hyperaccumulating fern: implications for remediation of arsenic contaminated soils. Geoderma 284:132–143. https://doi.org/10.1016/j.geoderma.2016.09.003
Fernández MD, Cagigal E, Vega MM, Urzelai A, Babín M, Pro J, Tarazona JV (2005) Ecological risk assessment of contaminated soils through direct toxicity assessment. Ecotoxicol Environ Saf 62:174–184. https://doi.org/10.1016/j.ecoenv.2004.11.013
Fernández PM, Viñarta SC, Bernal AR, Cruz EL, Figueroa LIC (2018) Bioremediation strategies for chromium removal: current research, scale-up approach and future perspectives. Chemosphere 208:139–148. https://doi.org/10.1016/J.CHEMOSPHERE.2018.05.166
Franchi E, Rolli E, Marasco R, Agazzi G, Borin S, Cosmina P, Pedron F, Rosellini I, Barbafieri M, Petruzzelli G (2017) Phytoremediation of a multi contaminated soil: mercury and arsenic phytoextraction assisted by mobilizing agent and plant growth promoting bacteria. J Soils Sediments 17:1224–1236. https://doi.org/10.1007/s11368-015-1346-5
Galdames A, Mendoza A, Orueta M, de Soto García IS, Sánchez M, Virto I, Vilas JL (2017) Development of new remediation technologies for contaminated soils based on the application of zero-valent iron nanoparticles and bioremediation with compost. Resour Technol 1–12. https://doi.org/10.1016/j.reffit.2017.03.008
García-Carmona M, Romero-Freire A, Sierra Aragón M, Martínez Garzón FJ, Martín Peinado FJ (2017) Evaluation of remediation techniques in soils affected by residual contamination with heavy metals and arsenic. J Environ Manag 191:228–236. https://doi.org/10.1016/j.jenvman.2016.12.041
Gavrilescu M (2004) Removal of heavy metals from the environment by biosorption. Eng Life Sci 4:219–232. https://doi.org/10.1002/elsc.200420026
Gidlow DA (n.d.) Lead toxicity. Occup Med (Chic Ill)
Gil-Díaz M, Ortiz LT, Costa G, Alonso J, Rodríguez-Membibre ML, Sánchez-Fortún S, Pérez-Sanz A, Martín M, Lobo MC (2014) Immobilization and leaching of Pb and Zn in an acidic soil treated with zerovalent iron nanoparticles (nZVI): physicochemical and toxicological analysis of leachates. Water Air Soil Pollut 225:1–13. https://doi.org/10.1007/s11270-014-1990-1
Gil-Díaz M, Alonso J, Rodríguez-Valdés E, Gallego JR, Lobo MC (2017) Comparing different commercial zero valent iron nanoparticles to immobilize As and Hg in brownfield soil. Sci Total Environ 584–585:1324–1332. https://doi.org/10.1016/j.scitotenv.2017.02.011
Gong Y, Liu Y, Xiong Z, Kaback D, Zhao D (2012) Immobilization of mercury in field soil and sediment using carboxymethyl cellulose stabilized iron sulfide nanoparticles. Nanotechnology 23:294007. https://doi.org/10.1088/0957-4484/23/29/294007
Gong Y, Tang J, Zhao D (2016) Application of iron sulfide particles for groundwater and soil remediation: a review. Water Res 89:309–320. https://doi.org/10.1016/j.watres.2015.11.063
Gong Y, Zhao D, Wang Q (2018) An overview of field-scale studies on remediation of soil contaminated with heavy metals and metalloids: technical progress over the last decade. Water Res 147:440–460. https://doi.org/10.1016/j.watres.2018.10.024
Goswami A, Raul PK, Purkait MK (2012) Arsenic adsorption using copper (II) oxide nanoparticles. Chem Eng Res Des 90:1387–1396. https://doi.org/10.1016/j.cherd.2011.12.006
Guemiza K, Coudert L, Metahni S, Mercier G, Besner S, Blais JF (2017) Treatment technologies used for the removal of As, Cr, Cu, PCP and/or PCDD/F from contaminated soil: a review. J Hazard Mater 333:194–214. https://doi.org/10.1016/j.jhazmat.2017.03.021
Gupta DK, Huang HG, Corpas FJ (2013) Lead tolerance in plants: strategies for phytoremediation. Environ Sci Pollut Res 20:2150–2161. https://doi.org/10.1007/s11356-013-1485-4
Ha E, Basu N, Bose-O’Reilly S, Dórea JG, McSorley E, Sakamoto M, Chan HM (2017) Current progress on understanding the impact of mercury on human health. Environ Res 152:419–433. https://doi.org/10.1016/j.envres.2016.06.042
Hafsteinsdóttir EG, Fryirs KA, Stark SC, Gore DB (2014) Remediation of metal-contaminated soil in polar environments: phosphate fixation at Casey Station, East Antarctica. Appl Geochem 51:33–43. https://doi.org/10.1016/j.apgeochem.2014.08.011
He F, Gao J, Pierce E, Strong PJ, Wang H, Liang L (2015) In situ remediation technologies for mercury-contaminated soil. Environ Sci Pollut Res 22:8124–8147. https://doi.org/10.1007/s11356-015-4316-y
He J, He C, Chen X, Liang X, Huang T, Yang X, Shang H (2018) Comparative study of remediation of Cr ( VI ) -contaminated soil using electrokinetics combined with bioremediation. Environ Sci Pollut Res 25:17682–17689
He L, Zhong H, Liu G, Dai Z, Brookes PC, Xu J (2019) Remediation of heavy metal contaminated soils by biochar: mechanisms, potential risks and applications in China. Environ Pollut. https://doi.org/10.1016/j.envpol.2019.05.151
Hsu L-C, Liu Y-T, Tzou Y-M (2015) Comparison of the spectroscopic speciation and chemical fractionation of chromium in contaminated paddy soils. J Hazard Mater 296:230–238. https://doi.org/10.1016/J.JHAZMAT.2015.03.044
Hsu L-C, Huang C-Y, Chuang Y-H, Chen H-W, Chan Y-T, Teah HY, Chen T-Y, Chang C-F, Liu Y-T, Tzou Y-M (2016) Accumulation of heavy metals and trace elements in fluvial sediments received effluents from traditional and semiconductor industries. Sci Rep 6:34250. https://doi.org/10.1038/srep34250
Huang T, Peng Q, Yu L, Li D (2017) The detoxification of heavy metals in the phosphate tailing-contaminated soil through sequential microbial pretreatment and electrokinetic remediation. Soil Sediment Contam An Int J 26:308–322. https://doi.org/10.1080/15320383.2017.1299685
Hung PC, Chang SH, Ou-Yang CC, Chang MB (2016) Simultaneous removal of PCDD/Fs, pentachlorophenol and mercury from contaminated soil. Chemosphere 144:50–58. https://doi.org/10.1016/j.chemosphere.2015.08.058
Im J, Yang K, Jho EH, Nam K (2015) Effect of different soil washing solutions on bioavailability of residual arsenic in soils and soil properties. Chemosphere 138:253–258. https://doi.org/10.1016/j.chemosphere.2015.06.004
Jabeen R, Ahmad A, Iqbal M (2009) Phytoremediation of heavy metals: physiological and molecular mechanisms. Bot Rev 75:339–364. https://doi.org/10.1007/s12229-009-9036-x
Jaishankar M, Tseten T, Anbalagan N, Mathew BB, Beeregowda KN (2014) Toxicity, mechanism and health effects of some heavy metals. Interdiscip Toxicol 7:60–72. https://doi.org/10.2478/intox
Jang M, Hwang JS, Choi SI (2007) Sequential soil washing techniques using hydrochloric acid and sodium hydroxide for remediating arsenic-contaminated soils in abandoned iron-ore mines. Chemosphere 66:8–17. https://doi.org/10.1016/j.chemosphere.2006.05.056
Kaur R, Yadav P, Kohli SK, Kumar V, Bakshi P, Mir BA, Thukral AK, Bhardwaj R (2019) Emerging trends and tools in transgenic plant technology for phytoremediation of toxic metals and metalloids. In: Transgenic plant technology for remediation of toxic metals and metalloids. Elsevier, pp 63–88. https://doi.org/10.1016/b978-0-12-814389-6.00004-3
Khalaf MM, Abd NYAHM, Lateef E (2018) Fine - template synthetic process of mesoporous TiO 2 using ionic / nonionic surfactants as potential remediation of Pb ( II ) from contaminated soil. Int J Environ Sci Technol 1–12. https://doi.org/10.1007/s13762-018-1790-z
Khalid S, Shahid M, Niazi NK, Murtaza B, Bibi I, Dumat C (2016) A comparison of technologies for remediation of heavy metal contaminated soils. J Geochem Explor. https://doi.org/10.1016/j.gexplo.2016.11.021
Khan S, Cao Q, Zheng YM, Huang YZ, Zhu YG (2008) Health risks of heavy metals in contaminated soils and food crops irrigated with wastewater in Beijing, China. Environ Pollut 152:686–692. https://doi.org/10.1016/J.ENVPOL.2007.06.056
Kim SO, Kim WS, Kim KW (2005) Evaluation of electrokinetic remediation of arsenic-contaminated soils. Environ Geochem Health 27:443–453. https://doi.org/10.1007/s10653-005-2673-z
Kim W, Jeon E, Jung J (2014) Field application of electrokinetic remediation for multi-metal contaminated paddy soil using two-dimensional electrode configuration. Environ Sci Pollut Res 21:4482–4491. https://doi.org/10.1007/s11356-013-2424-0
Ko I, Lee CH, Lee KP, Lee SW, Kim KW (2006) Remediation of soil contaminated with arsenic, zinc, and nickel by pilot-scale soil washing. Environ Prog 25:39–48. https://doi.org/10.1002/ep.10101
Ksheminska HP, Honchar TM, Gayda GZ, Gonchar MV (2006) Extra-cellular chromate-reducing activity of the yeast cultures. Cent Eur J Biol 1:137–149. https://doi.org/10.2478/s11535-006-0009-3
Kumar D, Tripathi DK, Liu S, Singh VK, Sharma S, Dubey NK, Prasad SM, Chauhan DK (2017) Pongamia pinnata (L.) Pierre tree seedlings offer a model species for arsenic phytoremediation. Plant Gene In Press. https://doi.org/10.1016/j.plgene.2017.06.002
Kuppusamy S, Palanisami T, Megharaj M, Venkateswarlu K, Ravi N (2016) Reviews of environmental contamination and toxicology. Springer. https://doi.org/10.1007/978-3-319-20013-2
Lahori AH, Zhanyu GUO, Zengqiang Z, Ronghua LI, Mahar A (2017) Use of biochar as an amendment for remediation of heavy metal-contaminated soils: prospects and challenges. Pedosph An Int J 27:991–1014. https://doi.org/10.1016/S1002-0160(17)60490-9
Laidlaw MAS, Filippelli GM, Brown S, Paz-Ferreiro J, Reichman SM, Netherway P, Truskewycz A, Ball AS, Mielke HW (2017) Case studies and evidence-based approaches to addressing urban soil lead contamination. Appl Geochem 83:14–30. https://doi.org/10.1016/j.apgeochem.2017.02.015
Lamborg CH, Hammerschmidt CR, Bowman KL, Swarr GJ, Munson KM, Ohnemus DC, Lam PJ, Heimbürger LE, Rijkenberg MJA, Saito MA (2014) A global ocean inventory of anthropogenic mercury based on water column measurements. Nature 512:65–68. https://doi.org/10.1038/nature13563
Lee KY, Kim HA, Lee BT, Kim SO, Kwon YH, Kim KW (2011) A feasibility study on bioelectrokinetics for the removal of heavy metals from tailing soil. Environ Geochem Health 33:3–11. https://doi.org/10.1007/s10653-010-9359-x
Lee ME, Jeon E, Tsang DCW, Baek K (2018) Simultaneous application of oxalic acid and dithionite for enhanced extraction of arsenic bound to amorphous and crystalline iron oxides. J Hazard Mater 354:91–98. https://doi.org/10.1016/j.jhazmat.2018.04.083
Lei M, Wan X, Guo G, Yang J, Chen T (2018) Phytoextraction of arsenic-contaminated soil with Pteris vittata in Henan Province, China: comprehensive evaluation of remediation efficiency correcting for atmospheric depositions. Environ Sci Pollut Res 25:124–131. https://doi.org/10.1007/s11356-016-8184-x
Levizou E, Zanni AA, Antoniadis V (2019) Varying concentrations of soil chromium ( VI ) for the exploration of tolerance thresholds and phytoremediation potential of the oregano ( Origanum vulgare ). Environ Sci Pollut Res 26:14–23
Li Z, Zhou M-M, Lin W (2014) The research of nanoparticle and microparticle hydroxyapatite amendment in multiple heavy metals contaminated soil remediation. J Nanomater 2014
Li Z, Wang L, Meng J, Liu X, Xu J, Wang F, Brookes P (2018) Zeolite-supported nanoscale zero-valent iron: new findings on simultaneous adsorption of Cd ( II ), Pb ( II ), and As ( III ) in aqueous solution and soil. J Hazard Mater 344:1–11. https://doi.org/10.1016/j.jhazmat.2017.09.036
Liang Q, Zhao D (2014) Immobilization of arsenate in a sandy loam soil using starch-stabilized magnetite nanoparticles. J Hazard Mater 271:16–23. https://doi.org/10.1016/j.jhazmat.2014.01.055
Liang L, Liu W, Sun Y, Huo X, Li S, Zhou Q (2016) Phytoremediation of heavy metal-contaminated saline soils using halophytes: current progress and future perspectives. Environ Rev er-2016-0063. https://doi.org/10.1139/er-2016-0063
Liao X, Li Y, Yan X (2016) Removal of heavy metals and arsenic from a co-contaminated soil by sieving combined with washing process. J Environ Sci (China) 41:202–210. https://doi.org/10.1016/j.jes.2015.06.017
Litter MI (2018) Future and perspectives of the use of iron nanoparticles for water and soil remediation. In: Litter MI, Quici N, Meichtry M (eds) Iron nanomaterials for water and soil treatment. Pan Stanford Publishing Pte, Singapore, pp 307–314
Liu L, Li W, Song W, Guo M (2018) Science of the total environment remediation techniques for heavy metal-contaminated soils: principles and applicability. Sci Total Environ 633:206–219. https://doi.org/10.1016/j.scitotenv.2018.03.161
Liu S, Pu S, Deng D, Huang H, Yan C, Ma H, Razavi BS (2020) Comparable effects of manure and its biochar on reducing soil Cr bioavailability and narrowing the rhizosphere extent of enzyme activities. Environ Int 134. https://doi.org/10.1016/j.envint.2019.105277
Lotfy SM, Mostafa AZ (2014) Phytoremediation of contaminated soil with cobalt and chromium. J. Geochem Explor 144:367–373. https://doi.org/10.1016/j.gexplo.2013.07.003
Lu G, Yue C, Qiu G, Guo M, Zhang M (2017) Na2S solution remediation for heavy mercury contaminated soil. J Chem Eng JAPAN 50:155–160. https://doi.org/10.1252/jcej.16we218
Ma F, Peng C, Hou D, Wu B, Zhang Q, Li F, Gu Q (2015) Citric acid facilitated thermal treatment: an innovative method for the remediation of mercury contaminated soil. J Hazard Mater 300:546–552. https://doi.org/10.1016/j.jhazmat.2015.07.055
Mahar A, Wang P, Ali A, Awasthi MK, Lahori AH, Wang Q, Li R, Zhang Z (2016) Challenges and opportunities in the phytoremediation of heavy metals contaminated soils: a review. Ecotoxicol Environ Saf 126:111–121. https://doi.org/10.1016/j.ecoenv.2015.12.023
Mahbub KR, Krishnan K, Naidu R, Andrews S, Megharaj M (2017) Mercury toxicity to terrestrial biota. Ecol Indic 74:451–462. https://doi.org/10.1016/j.ecolind.2016.12.004
Mani D, Kumar C, Patel NK (2015) Hyperaccumulator oilcake manure as an alternative for chelate-induced phytoremediation of heavy metals contaminated alluvial soils. Int J Phytoremediation 17:256–263. https://doi.org/10.1080/15226514.2014.883497
Mansouri T, Golchin A, Neyestani MR (2017) The effects of hematite nanoparticles on phytoavailability of arsenic and corn growth in contaminated soils. Int J Environ Sci Technol 14:1525–1534. https://doi.org/10.1007/s13762-017-1267-5
Mao X, Jiang R, Xiao W, Yu J (2015) Use of surfactants for the remediation of contaminated soils: a review. J Hazard Mater 285:419–435. https://doi.org/10.1016/j.jhazmat.2014.12.009
Mao X, Han FX, Shao X, Guo K, Mccomb J, Arslan Z, Zhang Z (2016) Electro-kinetic remediation coupled with phytoremediation to remove lead , arsenic and cesium from contaminated paddy soil. Ecotoxicol Environ Saf 125:16–24. https://doi.org/10.1016/j.ecoenv.2015.11.021
Mao X, Han FX, Shao X, Arslan Z, McComb J, Chang T, Guo K, Celik A (2017) Effects of operation variables and electro-kinetic field on soil washing of arsenic and cesium with potassium phosphate. Water Air Soil Pollut 228. https://doi.org/10.1007/s11270-016-3199-y
Marrugo-Negrete J, Durango-Hernández J, Pinedo-Hernández J, Olivero-Verbel J, Díez S (2015) Phytoremediation of mercury-contaminated soils by Jatropha curcas. Chemosphere 127:58–63. https://doi.org/10.1016/j.chemosphere.2014.12.073
Mattina MI, Lannucci-berger W, Musante C, White JC (2003) Concurrent plant uptake of heavy metals and persistent organic pollutants from soil. Environ Pollut 124:375–378. https://doi.org/10.1016/S0269-7491(03)00060-5
McGrory ER, Brown C, Bargary N, Williams NH, Mannix A, Zhang C, Henry T, Daly E, Nicholas S, Petrunic BM, Lee M, Morrison L (2017) Arsenic contamination of drinking water in Ireland: a spatial analysis of occurrence and potential risk. Sci Total Environ 579:1863–1875. https://doi.org/10.1016/j.scitotenv.2016.11.171
Melamed R, Cao X, Chen M, Ma LQ (2003) Field assessment of lead immobilization in a contaminated soil after phosphate application. Sci Total Environ 305:117–127. https://doi.org/10.1016/S0048-9697(02)00469-2
Meng F, Xue H, Wang Y, Zheng B, Wang J (2018) Citric-acid preacidification enhanced electrokinetic remediation for removal of chromium from chromium-residue-contaminated soil. Environ Technol 39:356–362. https://doi.org/10.1080/09593330.2017.1301565
Moazallahi M, Baghernejad M, Jafari Haghighi M, Saffari M (2017) Stabilization of lead in two artificial contaminated calcareous soils using stabilized nanoscale zero-valent iron particles with/without chelating agents. Arch Agron Soil Sci 63:565–577. https://doi.org/10.1080/03650340.2016.1227069
Mohan D, Pittman CU (2007) Arsenic removal from water/wastewater using adsorbents-a critical review. J Hazard Mater 142:1–53. https://doi.org/10.1016/j.jhazmat.2007.01.006
Moharem M, Elkhatib E, Mesalem M (2019) Remediation of chromium and mercury polluted calcareous soils using nanoparticles: sorption – desorption kinetics, speciation and fractionation. Environ Res 170:366–373. https://doi.org/10.1016/j.envres.2018.12.054
Motuzova GV, Minkina TM, Karpova EA, Barsova NU, Mandzhieva SS (2014) Soil contamination with heavy metals as a potential and real risk to the environment. J Geochem Explor 144:241–246. https://doi.org/10.1016/j.gexplo.2014.01.026
Mueller NC, Nowack B (2010) Nanoparticles for remediation: solving big problems with little particles. Elements 6:395–400. https://doi.org/10.2113/gselements.6.6.395
Nabi D, Aslam I, Qazi IA (2009) Evaluation of the adsorption potential of titanium dioxide nanoparticles for arsenic removal. J Environ Sci 21:402–408. https://doi.org/10.1016/S1001-0742(08)62283-4
Nahar N, Rahman A, Nawani NN, Ghosh S, Mandal A, Affiliations A (2017) Title: phytoremediation of arsenic from the contaminated soil using transgenic tobacco plants expressing ACR2 gene of Arabidopsis thaliana phytoremediation of arsenic from the contaminated soil using transgenic tobacco plants expressing ACR2 gene of Arabi. J Plant Physiol accepted manuscript. https://doi.org/10.1016/j.jplph.2017.08.001
Nriagu JO, Pacyna JM (1988) Quantitative assessment of worldwide contamination of air, water and soils by trace metals. Nature 333:134–139. https://doi.org/10.1038/332141a0
Odumo BO, Carbonell G, Angeyo HK, Patel JP, Torrijos M, Rodríguez Martín JA (2014) Impact of gold mining associated with mercury contamination in soil, biota sediments and tailings in Kenya. Environ Sci Pollut Res 21:12426–12435. https://doi.org/10.1007/s11356-014-3190-3
Okkenhaug G, Gebhardt KG, Amstaetter K, Lassen H, Herzel H, Mariussen E, Rossebø Å, Cornelissen G, Breedveld GD, Rasmussen G, Mulder J (2016) Antimony ( Sb ) and lead ( Pb ) in contaminated shooting range soils: Sb and Pb mobility and immobilization by iron based sorbents, a field study. J Hazard Mater 307:336–343. https://doi.org/10.1016/j.jhazmat.2016.01.005
Okuo J, Emina A, Omorogbe S, Anegbe B (2018) Synthesis, characterization and application of starch stabilized zerovalent iron nanoparticles in the remediation of Pb-acid battery soil. Environ Nanotechnol Monit Manag 9:12–17. https://doi.org/10.1016/j.enmm.2017.11.004
Petticrew EL, Albers SJ, Baldwin SA, Carmack EC, Déry SJ, Gantner N, Graves KE, Laval B, Morrison J, Owens PN, Selbie DT, Vagle S (2015) The impact of a catastrophic mine tailings impoundment spill into one of North America’s largest fjord lakes: Quesnel Lake, British Columbia, Canada. Geophys Res Lett 42:3347–3355. https://doi.org/10.1002/2015GL063345
Pinto PX, Al-Abed SR (2017) Assessing metal mobilization from industrially lead-contaminated soils located at an urban site. Appl Geochem 83:31–40. https://doi.org/10.1016/j.apgeochem.2017.01.025
Pinto AP, De Varennes A, Dias CMB, Lopes ME (2018) Microbial-assisted phytoremediation: a convenient use of plant and microbes to clean up soils. In: Ansari A, Gill S, Gill R, Lanza G, Newman L (eds) Phytoremediation. Springer, pp 21–87. https://doi.org/10.1007/978-3-319-99651-6
Pogrzeba M, Ciszek D, Galimska-Stypa R, Nowak B, Sas-Nowosielska A (2016) Ecological strategy for soil contaminated with mercury. Plant Soil 409:371–387. https://doi.org/10.1007/s11104-016-2936-8
Prado C, Chocobar Ponce S, Pagano E, Prado FE, Rosa M (2016) Differential physiological responses of two Salvinia species to hexavalent chromium at a glance. Aquat Toxicol 175:213–221. https://doi.org/10.1016/J.AQUATOX.2016.03.027
Provencher JF, Mallory ML, Braune BM, Forbes MR, Gilchrist HG (2014) Mercury and marine birds in Arctic Canada: effects, current trends, and why we should be paying closer attention. Environ Rev 22:244–255. https://doi.org/10.1139/er-2013-0072
Qi M, Yingjie Z, Peng D (2017) Desorption of mercury from contaminated soil using sodium sulfite. Water Air Soil Pollut 228. https://doi.org/10.1007/s11270-016-3213-4
Qin C, Wang H, Yuan X, Xiong T, Zhang J, Zhang J (2019) Understanding structure-performance correlation of biochar materials in environmental remediation and electrochemical devices. Chem Eng J. https://doi.org/10.1016/j.cej.2019.122977
Ran J, Wang D, Wang C, Zhang G, Zhang H (2016) Heavy metal contents, distribution, and prediction in a regional soil-wheat system. Sci Total Environ 544:422–431. https://doi.org/10.1016/j.scitotenv.2015.11.105
Rice KM, Walker EM, Wu M, Gillette C, Blough ER (2014) Environmental mercury and its toxic effects. J Prev Med Public Health 47:74–83. https://doi.org/10.3961/jpmph.2014.47.2.74
Rodríguez L, Alonso-Azcárate J, Villasenor J, Rodríguez-Castellanos L (2016) EDTA and hydrochloric acid effects on mercury accumulation by Lupinus albus. Environ Sci Pollut Res 23:24739–24748. https://doi.org/10.1007/s11356-016-7680-3
Rodriguez-Iruretagoiena A, Fdez-Ortiz de Vallejuelo S, Gredilla A, Ramos CG, Oliveira MLS, Arana G, de Diego A, Madariaga JM, Silva LFO (2015) Fate of hazardous elements in agricultural soils surrounding a coal power plant complex from Santa Catarina (Brazil). Sci Total Environ 508:374–382. https://doi.org/10.1016/j.scitotenv.2014.12.015
Ryu SR, Jeon EK, Baek K (2017) A combination of reducing and chelating agents for electrolyte conditioning in electrokinetic remediation of As-contaminated soil. J Taiwan Inst Chem Eng 70:252–259. https://doi.org/10.1016/j.jtice.2016.10.058
Sabir M, Waraich E, Hakeem KR, Ozturk M (2014) Phytoremediation: mechanisms and adaptations. In: Soil remediation and plants. Elsevier Inc, pp 85–105
Salazar MJ, Pignata ML (2014) Lead accumulation in plants grown in polluted soils. Screening of native species for phytoremediation. J Geochem Explor 137:29–36. https://doi.org/10.1016/j.gexplo.2013.11.003
Salt DE, Blaylock M, Kumar NP, Dushenkov V, Ensley BD, Chet I, Raskin I (1995) Phytoremediation: a novel strategy for the removal of toxic metals from the environment using plants. Nat Biotechnol 13:468–474. https://doi.org/10.1038/nbt0595-468
Sampanpanish P, Pongsapich W, Khaodhiar S, Khan E (2006) Chromium removal from soil by phytoremediation with weed plant species in Thailand. Water Air Soil Pollut Focus 191–206. https://doi.org/10.1007/s11267-005-9006-1
Saravanan A, Jayasree R, Hemavathy RV, Jeevanantham S, Hamsini S, Kumar PS, Yaashikaa PR, Manivasagan V, Yuvaraj D (2019) Phytoremediation of Cr (VI) ion contaminated soil using Black gram (Vigna mungo): assessment of removal capacity. J Environ Chem Eng 7:103052. https://doi.org/10.1016/j.jece.2019.103052
Sarwar N, Imran M, Rashid M, Ishaque W, Asif M, Matloob A, Rehim A, Hussain S (2017a) Chemosphere phytoremediation strategies for soils contaminated with heavy metals: modifications and future perspectives. Chemosphere 171:710–721. https://doi.org/10.1016/j.chemosphere.2016.12.116
Sarwar N, Imran M, Shaheen MR, Ishaque W, Kamran MA, Matloob A, Rehim A, Hussain S (2017b) Phytoremediation strategies for soils contaminated with heavy metals: modifications and future perspectives. Chemosphere 171:710–721. https://doi.org/10.1016/j.chemosphere.2016.12.116
Seshadri B, Bolan NS, Choppala G, Kunhikrishnan A, Sanderson P, Wang H, Currie LD, Tsang DCW, Ok YS, Kim K (2017) Potential value of phosphate compounds in enhancing immobilization and reducing bioavailability of mixed heavy metal contaminants in shooting range soil. Chemosphere 184:197–206. https://doi.org/10.1016/j.chemosphere.2017.05.172
Shahandeh H, Hossner LR (2006) Plant screening for chromium phytoremediation. Plant Screen Chromium Phytoremediation 6514:31–51. https://doi.org/10.1080/15226510008500029
Shahid M, Shamshad S, Rafiq M, Khalid S, Bibi I, Niazi NK, Dumat C, Rashid MI (2017) Chromium speciation, bioavailability, uptake, toxicity and detoxification in soil-plant system: a review. Chemosphere 178:513–533. https://doi.org/10.1016/J.CHEMOSPHERE.2017.03.074
Shin SY, Park SM, Baek K (2017) Soil moisture could enhance electrokinetic remediation of arsenic-contaminated soil. Environ Sci Pollut Res 24:9820–9825. https://doi.org/10.1007/s11356-017-8720-3
Sierra MJ, Millán R, López FA, Alguacil FJ, Cañadas I (2016) Sustainable remediation of mercury contaminated soils by thermal desorption. Environ Sci Pollut Res 23:4898–4907. https://doi.org/10.1007/s11356-015-5688-8
Singh R, Misra V, Singh RP (2012) Removal of Cr(VI) by nanoscale zero-valent iron (nZVI) from soil contaminated with tannery wastes. Bull Environ Contam Toxicol 88:210–214. https://doi.org/10.1007/s00128-011-0425-6
Singh R, Singh S, Parihar P, Singh VP, Prasad SM (2015) Arsenic contamination, consequences and remediation techniques: a review. Ecotoxicol Environ Saf 112:247–270. https://doi.org/10.1016/j.ecoenv.2014.10.009
Sinha V, Pakshirajan K, Chaturvedi R (2018) Chromium tolerance, bioaccumulation and localization in plants: an overview. J Environ Manag 206:715–730. https://doi.org/10.1016/J.JENVMAN.2017.10.033
Sohn E (2014) The toxic side of rice. Nature Outlook, 5–6. https://doi.org/10.1038/514S62a
Song B, Zeng G, Gong J, Liang J, Xu P, Liu Z, Zhang Y, Zhang C, Cheng M, Liu Y, Ye S, Yi H, Ren X (2017) Evaluation methods for assessing effectiveness of in situ remediation of soil and sediment contaminated with organic pollutants and heavy metals. Environ Int 105:43–55. https://doi.org/10.1016/j.envint.2017.05.001
Soodan RK, Pakade YB, Nagpal A, Katnoria JK (2014) Analytical techniques for estimation of heavy metals in soil ecosystem: a tabulated review. Talanta 125:405–410. https://doi.org/10.1016/j.talanta.2014.02.033
Souza LRR, Zanatta MB, da Silva IA, da Veiga MAMS (2018) Mercury determination in soil and sludge samples by HR CS GFAAS : comparison of sample preparation procedures and chemical modifiers. J Anal At Spectrom 33:1477–1485. https://doi.org/10.1039/C8JA00152A
Souza LRR, Nakadi FV, Zanatta MBT, da Veiga MAMS (2019) Reduction of bioaccessibility and leachability of Pb and Cd in soils using sludge from water treatment plant. Int J Environ Sci Technol 16:5397–5408. https://doi.org/10.1007/s13762-018-2042-y
Su C, Jiang L, Zhang W (2014) A review on heavy metal contamination in the soil worldwide: situation, impact and remediation techniques. Environ Skept Critics 3:24–38. https://doi.org/10.1037/a0036071
Su H, Fang Z, Tsang PE, Zheng L, Cheng W, Fang J, Zhao D (2016) Remediation of hexavalent chromium contaminated soil by biochar-supported zero-valent iron nanoparticles. J Hazard Mater 318:533–540. https://doi.org/10.1016/J.JHAZMAT.2016.07.039
Surya B, Lova G, Tina R, Jatnika A (2018) An overview of electrokinetic soil flushing and its effect on bioremediation of hydrocarbon contaminated soil. J Environ Manag 218:309–321. https://doi.org/10.1016/j.jenvman.2018.04.065
Suzuki T, Moribe M, Okabe Y, Niinae M (2013) A mechanistic study of arsenate removal from artificially contaminated clay soils by electrokinetic remediation. J Hazard Mater 254–255:310–317. https://doi.org/10.1016/j.jhazmat.2013.04.013
Sysalová J, Kucera J, Drtinová B, Cervenka R, Zveina O, Komárek J, Kameník J (2017) Mercury species in formerly contaminated soils and released soil gases. Sci Total Environ 584–585:1032–1039. https://doi.org/10.1016/j.scitotenv.2017.01.157
Tao X, Li A, Yang H (2017) Immobilization of metals in contaminated soils using natural. Environ Pollut 222:348–355. https://doi.org/10.1016/j.envpol.2016.12.028
Tarvainen T, Albanese S, Birke M, Ponavic M, Reimann C, Team GP (2013) Arsenic in agricultural and grazing land soils of Europe. Appl Geochem 28:2–10. https://doi.org/10.1016/j.apgeochem.2012.10.005
Tipping E, Lofts S, Hooper H, Frey B, Spurgeon D, Svendsen C (2010) Critical limits for Hg(II) in soils, derived from chronic toxicity data. Environ Pollut 158:2465–2471. https://doi.org/10.1016/j.envpol.2010.03.027
Turra C, De Nadai Fernandes EA, Antonio Stefanuto V, Arruda Bacchi M, Adrián Sarriés G, Enrique Lai Reyes A (2010) Arsenic and chromium in Brazilian agricultural supplies. J Toxicol Environ Health A 73:910–915. https://doi.org/10.1080/15287391003744971
Virkutyte J, Sillanpää M, Latostenmaa P (2002) Electrokinetic soil remediation - critical overview. Sci Total Environ 289:97–121. https://doi.org/10.1016/S0048-9697(01)01027-0
Vithanage M, Herath I, Joseph S, Bundschuh J, Bolan N, Ok YS, Kirkham MB, Rinklebe J (2017) Interaction of arsenic with biochar in soil and water: a critical review. Carbon N Y 113:219–230. https://doi.org/10.1016/j.carbon.2016.11.032
Wang S, Mulligan CN (2006) Occurrence of arsenic contamination in Canada: sources, behavior and distribution. Sci Total Environ 366:701–721. https://doi.org/10.1016/j.scitotenv.2005.09.005
Wang C, Zhao Y, Pei Y (2012a) Investigation on reusing water treatment residuals to remedy soil contaminated with multiple metals in Baiyin, China. J Hazard Mater 237–238:240–246. https://doi.org/10.1016/j.jhazmat.2012.08.034
Wang J, Feng X, Anderson CWN, Xing Y, Shang L (2012b) Remediation of mercury contaminated sites - a review. J Hazard Mater 221–222:1–18. https://doi.org/10.1016/j.jhazmat.2012.04.035
Wang X, Zhang D, Pan X, Lee DJ, Al-Misned FA, Mortuza MG, Gadd GM (2017a) Aerobic and anaerobic biosynthesis of nano-selenium for remediation of mercury contaminated soil. Chemosphere 170:266–273. https://doi.org/10.1016/j.chemosphere.2016.12.020
Wang Y, Ma F, Zhang Q, Peng C, Wu B, Li F, Gu Q (2017b) An evaluation of different soil washing solutions for remediating arsenic-contaminated soils. Chemosphere 173:368–372. https://doi.org/10.1016/j.chemosphere.2017.01.068
Wang X, Song W, Qian H, Zhang D, Pan X, Gaddd GM (2018) Stabilizing interaction of exopolymers with nano-Se and impact on mercury immobilization in soil and groundwater. Environ Sci Nano 5:456–466. https://doi.org/10.1039/C7EN00628D
Wang T, Liu Y, Wang J, Wang X, Liu B, Wang Y (2019) In-situ remediation of hexavalent chromium contaminated groundwater and saturated soil using stabilized iron sulfide nanoparticles. J Environ Manag 231:679–686. https://doi.org/10.1016/j.jenvman.2018.10.085
Wei M, Chen J, Wang X (2016a) Removal of arsenic and cadmium with sequential soil washing techniques using Na2EDTA, oxalic and phosphoric acid: optimization conditions, removal effectiveness and ecological risks. Chemosphere 156:252–261. https://doi.org/10.1016/j.chemosphere.2016.04.106
Wei X, Guo S, Wu B, Li F, Li G (2016b) Effects of reducing agent and approaching anodes on chromium removal in electrokinetic soil remediation. Front Environ Sci Eng 10:253–261. https://doi.org/10.1007/s11783-015-0791-0
Wu X, Cobbina SJ, Mao G, Xu H, Zhang Z, Yang L (2016) A review of toxicity and mechanisms of individual and mixtures of heavy metals in the environment. Environ Sci Pollut Res 23:8244–8259. https://doi.org/10.1007/s11356-016-6333-x
Wuana R a, Okieimen FE (2011) Heavy metals in contaminated soils: a review of sources, chemistry, risks and best available strategies for remediation. ISRN Ecol 2011:1–20. https://doi.org/10.5402/2011/402647
Xu J, Bravo AG, Lagerkvist A, Bertilsson S, Sjöblom R, Kumpiene J (2015) Sources and remediation techniques for mercury contaminated soil. Environ Int 74:42–53. https://doi.org/10.1016/j.envint.2014.09.007
Xu Z, Xu X, Tsang DCW, Yang F, Zhao L, Qiu H, Cao X (2019) Participation of soil active components in the reduction of Cr(VI) by biochar: differing effects of iron mineral alone and its combination with organic acid. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2019.121455
Yang K, Kim BC, Nam K, Choi Y (2017) The effect of arsenic chemical form and mixing regime on arsenic mass transfer from soil to magnetite. Environ Sci Pollut Res 24:8479–8488. https://doi.org/10.1007/s11356-017-8510-y
Yildirim D, Sasmaz A (2017) Phytoremediation of As, Ag, and Pb in contaminated soils using terrestrial plants grown on Gumuskoy mining area (Kutahya Turkey). J Geochem Explor 182:228–234. https://doi.org/10.1016/j.gexplo.2016.11.005
Yuan Y, Chai L, Yang Z, Yang W (2017) Simultaneous immobilization of lead, cadmium, and arsenic in combined contaminated soil with iron hydroxyl phosphate. J Soils Sediments 17:432–439. https://doi.org/10.1007/s11368-016-1540-0
Zaier H, Ghnaya T, Ghabriche R, Chmingui W, Lakhdar A, Lutts S, Abdelly C (2014) EDTA-enhanced phytoremediation of lead-contaminated soil by the halophyte Sesuvium portulacastrum. Environ Sci Pollut Res 21:7607–7615. https://doi.org/10.1007/s11356-014-2690-5
Zeng G, Wan J, Huang D, Hu L, Huang C, Cheng M, Xue W, Gong X, Wang R, Jiang D (2017) Precipitation, adsorption and rhizosphere effect: the mechanisms for phosphate-induced Pb immobilization in soils—a review. J Hazard Mater 339:354–367. https://doi.org/10.1016/j.jhazmat.2017.05.038
Zhai Q, Narbad A, Chen W (2015) Dietary strategies for the treatment of cadmium and lead toxicity. Nutrients 7:552–571. https://doi.org/10.3390/nu7010552
Zhai X, Li Z, Huang B, Luo N, Huang M, Zhang Q, Zeng G (2018) Remediation of multiple heavy metal-contaminated soil through the combination of soil washing and in situ immobilization. Sci Total Environ 635:92–99. https://doi.org/10.1016/j.scitotenv.2018.04.119
Zhang X, Liu J, Huang H, Chen J (2007) Chromium accumulation by the hyperaccumulator plant Leersia hexandra Swartz. Chemosphere 67:1138–1143. https://doi.org/10.1016/j.chemosphere.2006.11.014
Zhang M, Wang Y, Zhao D, Pan G (2010) Immobilization of arsenic in soils by stabilized nanoscale zero-valent iron, iron sulfide (FeS), and magnetite (Fe3O4) particles. Chin Sci Bull 55:365–372. https://doi.org/10.1007/s11434-009-0703-4
Zhang Y, Yang J, Zhong L, Liu L (2018) Effect of multi-wall carbon nanotubes on Cr ( VI ) reduction by citric acid : implications for their use in soil remediation. Environ Sci Pollut Res 25:23791–23798
Zhang T, Wei S, Waterhouse GIN, Fu L, Liu L, Shi W, Sun J, Ai S (2020) Chromium (VI) adsorption and reduction by humic acid coated nitrogen-doped magnetic porous carbon. Powder Technol 360:55–64. https://doi.org/10.1016/j.powtec.2019.09.091
Zhao T, Yu Z, Zhang J, Qu L, Li P (2018) Low-thermal remediation of mercury-contaminated soil and cultivation of treated soil. Environ Sci Pollut Res 25:24135–24142
Zhao S, Song C, Gao X, Lin J (2019) Quantitative analysis of Pb in soil by femtosecond-nanosecond double-pulse laser-induced breakdown spectroscopy. Results Phys:15. https://doi.org/10.1016/j.rinp.2019.102736
Zhou M, Xu J, Zhu S, Wang Y, Gao H (2018) Separation and purification technology exchange electrode-electrokinetic remediation of Cr-contaminated soil using solar energy. Sep Purif Technol 190:297–306. https://doi.org/10.1016/j.seppur.2017.09.006
Zhu Y, Xu F, Liu Q, Chen M, Liu X, Wang Y, Sun Y, Zhang L (2019) Nanomaterials and plants: positive effects, toxicity and the remediation of metal and metalloid pollution in soil. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2019.01.234
Zou T, Li T, Zhang X, Yu H, Luo H (2011) Lead accumulation and tolerance characteristics of Athyrium wardii (Hook.) as a potential phytostabilizer. J Hazard Mater 186:683–689. https://doi.org/10.1016/j.jhazmat.2010.11.053
Acknowledgments
The authors are grateful to Cynthia M. C. Prado Manso for reviewing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Souza, L.R.R., Pomarolli, L.C. & da Veiga, M.A.M.S. From classic methodologies to application of nanomaterials for soil remediation: an integrated view of methods for decontamination of toxic metal(oid)s. Environ Sci Pollut Res 27, 10205–10227 (2020). https://doi.org/10.1007/s11356-020-08032-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08032-8