Abstract
Using rare minnow (Gobiocypris rarus) at early-life stages as experimental models, the developmental toxicity of five widely used triazole fungicides (myclobutanil, fluconazole, flusilazole, triflumizole, and epoxiconazole) were investigated following exposure to 1–15 mg/L for 72 h. Meanwhile, morphological parameters (body length, body weight, and heart rate), enzyme activities (superoxide dismutase (SOD), glutathione S-transferase (GST), adenosine triphosphatase (ATPase), and acetyl cholinesterase (AChE)), and mRNA levels (hsp70, mstn, mt, apaf1, vezf1, and cyp1a) were also recorded following exposure to 0.2, 1.0, and 5.0 mg/L for 72 h. Results indicated that increased malformation and mortality, decreased body length, body weight, and heart rate provide a concentration-dependent pattern; values of 72 h LC50 (median lethal concentration) and EC50 (median effective concentration) ranged from 3 to 12 mg/L. Most importantly, the results of the present study suggest that even at the lowest concentration, 0.2 mg/L, five triazole fungicides also caused notable changes in enzyme activities and mRNA levels. Overall, the present study points out that those five triazole fungicides are highly toxic to the early development of G. rarus embryos. The information presented in this study will be helpful in better understanding the toxicity induced by triazole fungicides in fish embryos.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Triazole fungicides are an important class of antifungal pesticides that are widely used in agriculture to prevent fungal growth on vegetables, cereals, soybeans, seeds, and a variety of fruits (Konwick et al. 2006; Chen et al. 2008; Li and Randak 2009). Their characteristics, such as high chemical and photochemical stability and low biodegradability in the environment (Wang et al. 2011), make them persistent in soil and water (Bromillow et al. 1999). Thus, there is a concern over the wide application of the triazole fungicides and their possible detrimental effects on aquatic organism that may arise from spray drift or surface runoff after rainfall (Raudonis et al. 2004; Konwick et al. 2006).
The antifungal activity of triazole fungicides is due to their capability to interfere with steroid biosynthesis by inhibiting the CYP51 (a cytochrome P450 enzyme) that plays an important role in the conversion of lanosterol to ergosterol in fungi and yeast (de Jong et al. 2011). And, the cell membrane assembly of fungi and yeast is disturbed by blocking the synthesis of the essential membrane component ergosterol (Zarn et al. 2003). Additionally, besides the effect on CYP51, triazoles may also inhibit other P450-mediated activities resulting in mammalian toxicity. Examples of the enzymes that have been related to the developmental toxic effects of triazoles are enzymes of the CYP26 family (Menegola et al. 2006; Tiboni et al. 2009; Marotta and Tiboni 2010). Furthermore, triazole exposure can cause skeletal defects as well as craniofacial malformations and hydrocephaly by affecting the endogenous retinoic acid levels in mammalian embryo (Menegola et al. 2005; Farag and Ibrahim 2007). For aquatic organisms, an LC50 (median lethal concentration) of 20.55 mg/L for paclobutrazol has been reported for zebrafish (Danio rerio) (Ding et al. 2009). The acute toxicity of difenoconazole on zebrafish was 1.17 mg/L for larvae, 1.45 mg/L for adult fish, and 2.34 mg/L for embryos (Mu et al. 2013). The hepatic antioxidant enzymes, the RNA/DNA ratio, and hematological and plasma biochemical parameters are affected in rainbow trout (Oncorhynchus mykiss) after exposure to 0.5 mg/L propiconazole (Li et al. 2010a, b). Previous study has also detected gene expression changes in zebrafish embryos after 4-mg/L flusilazole treatment (Hermsen et al. 2011a). Therefore, further toxicological investigation of triazole fungicides for aquatic organisms is required. In particular, toxicity evaluations on aquatic organisms which include multiple parameters (conventional morphological parameters (e.g., malformation and mortality), enzyme activities, and mRNA levels) are needed.
Fish, and in particular small freshwater fish species such as rare minnow (Gobiocypris rarus), zebrafish (D. rerio), and Japanese medaka (Oryzias latipes), have been used for acute and chronic tests to investigate the toxic properties of chemicals and for ecotoxicological regulatory purposes (Zha et al. 2007; Voelker et al. 2007; Shi et al. 2008). G. rarus is a widely distributed tiny Chinese freshwater cyprinid that has a short life cycle and could become a standardized test species for China. G. rarus possesses lots of attractive features that make it a suitable model in aquatic toxicity tests (Zhong et al. 2005; Zha et al. 2007; Zhang et al. 2008). The G. rarus embryo as a useful research model also has lots of advantages, such as small in size and transparent, easy to maintain, short development cycle, rapid embryogenesis (Zha et al. 2007; Zhu et al. 2011). Earlier studies have shown that G. rarus embryos are sensitive to heavy metals, xenoestrogens, and other aquatic pollutant (Qun-Fang et al. 2002; Wang et al. 2010; Zhu et al. 2011, 2013).
In the present study, five triazole fungicides (myclobutanil, fluconazole, flusilazole, triflumizole, and epoxiconazole) were selected for a further exploration of potential developmental toxicity by G. rarus embryos. We selected different critical monitor endpoints, such as mortality rate, malformation rate, body length, body weight, and heart rate. For better understanding, the toxicity mechanisms of triazole fungicides, relative enzyme activity (superoxide dismutase (SOD), glutathione S-transferase (GST), adenosine triphosphatase (ATPase), and acetyl cholinesterase (AChE)), as well as certain mRNA levels for cell apoptosis (apaf1), metabolism (mt and cyp1a), stress response (hsp70), muscle (mstn), and blood vessel (vezf1) development were also selected as endpoints. The work presented herein aims to assess the possible influences of triazole fungicides on multilevel biomarker responses in the developing G. rarus embryos.
Materials and methods
Fish maintenance and egg production
G. rarus were purchased from the Institute of Hydrobiology, the Chinese Academy of Sciences and maintained at approximately 25 °C with a 16 :8 h light-dark cycle in a flow-through system in charcoal-filtered tap water. Water quality readings were taken weekly to monitor the following parameters: pH (7.5–8.3), hardness (6.2–6.5 °d), conductivity (423–496 μS/cm), and dissolved oxygen (7.7–8.9 mg/L). The fish cultural condition and collection of fertilized eggs were performed as described previously (Zhu et al. 2011). Briefly, fish were fed a commercial granular food (Tetra, Melle, Germany) at a daily rate of 0.1 % body weight. Females were induced to superovulate by injection of 1 IU chorionic gonadotropin (Chorulon; Intervet Co.) and then placed in chambers with mature males in spawning boxes. The ratio of females and males was 1:2. Twelve hours later, the spawning finished, and the fertilized eggs were washed twice with fish water and collected for the subsequent exposure experiments.
Chemicals and reagents
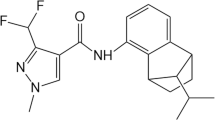
Analytical grade myclobutanil (CAS No. 88671-89-0), fluconazole (CAS No. 86386-73-4), flusilazole (CAS No. 85509-19-9), triflumizole (CAS No. 99387-89-0), and epoxiconazole (CAS No. 133855-98-8) were purchased from Sigma-Aldrich Shanghai Trading Co. Ltd (Shanghai, China), with a purity of 98.0 %. The chemical structures of five triazole fungicides are shown in Fig 1. Enzyme activity assay kits (superoxide dismutase assay kit, glutathione S-transferase assay kit, adenosine triphosphatase assay kit, and acetyl cholinesterase assay kit) were purchased from Jiancheng Bioengineering Institute (Nanjing, China). Total RNA was extracted by Trizol reagent (Invitrogen, Carlsbad, CA, USA). Reverse transcription and SYBR-green real-time quantitative PCR reagents were purchased from Takara (Dalian, China). All other reagents used in this study were of analytical grade.
Fish embryo toxicity test
The toxicity effects of five triazole fungicides on the subsequent embryonic development of G. rarus were evaluated by exposing fertilized eggs (4–32 cell stage, observed with Olympus BX41, Olympus Optical Co., Ltd., Tokyo) to a range of concentrations (1, 3, 5, 7, 9, 11, 13, and 15 mg/L). All triazoles were dissolved in dimethyl sulfoxide (DMSO, ACS reagent, ≥99.9 %) and further diluted in standard dilution water (ISO7346/3, demineralized water supplemented with 294.0 mg/L CaCl2 · 2H2O, 123.3 mg/L MgSO4 · 7H2O, 63.0 mg/L NaHCO3, and 5.5 mg/L KCl and then aerated for 24 h at 25 °C) with a final DMSO concentration of 0.2 % (v/v). The control group was exposed to 0.2 % DMSO, and control survival rate was always above 96 %. Embryos within the 4- to 32-cell stage were selected and transferred to a 24-well plate. One embryo was transferred to one well containing 2-mL freshly prepared test medium. The plates were covered with self-adhesive foil to avoid the possible effect of evaporation, and then incubated at 25 ± 1 °C with a photoperiod of 16:8 h (light/dark) for 72 h. The exposure solution was renewed every 24 h to keep the appropriate concentration of triazoles and water quality. Dead eggs were removed daily. Five replicates were set for the tests, with 30 embryos per replicate. The mortality rate and malformation rate of embryos were examined microscopically at 72 h for all treatment and control groups. Death and malformation were judged via the lethal and sublethal toxicological endpoints described by Nagel (Nagel 2002).
Developmental toxicity assays
To further explore the toxicity of five triazole fungicides on embryos, developmental parameters (body length, body weight, and heart rate), enzyme activity, and mRNA levels were also selected as observed indicators. Because of all these indicators need living embryos or larvae to measure, according to the above embryotoxicity experiments, embryos were exposed to a range of concentrations (0.2, 1.0, and 5.0 mg/L) for 72 h. Five replicates were set for the tests, with 200 embryos per replicate. The embryo cultural condition was performed according to the above established protocols.
After the incubation period, random embryos/larvae were selected for morphological and behavioral analyses. Body weight (mg) was measured by AUW220D analytical balance (Shimadzu; Tokyo, Japan). The body length (mm) and heart rate (beats/min) were counted, photographed, and videoed with a digital camera (Nikon, Japan) fixed on a dissection microscope and measured from these digital images using Image Pro Plus software (Media Cybernetics, Bethesda, MD, USA) and recorded (Zhu et al. 2013). All spontaneous movement, heart beat, and swimming speed recordings started after adaptation to 25 ± 1 °C for 5 min.
After 72-h exposure period, 30 intact embryos per concentration from treatment and control groups were selected for enzyme activity assays. The embryos were homogenized according to previous study (Ramsden et al. 2013). Homogenates were stored at −80 °C until required. Embryo homogenates were analyzed (each sample in triplicate) for total protein content and enzyme activities (SOD, GST, ATPase, and AChE). Protein content was measured by Coomasse blue protein-binding method using bovine serum albumin as standard. The enzyme activities (SOD, GST, ATPase, and AChE) were analyzed by using assay kits (Jiancheng Bioengineering Institute, Nanjing, China) following manufacturer’s instruction. Biochemistry data were normalized against homogenate protein content.
After the incubation period, 30 intact embryos per concentration from treatment and control groups were immediately frozen in liquid nitrogen and stored at −80 °C for subsequent RNA isolation. Total RNA was extracted by using Trizol reagent (Invitrogen, Carlsbad, CA, USA) following manufacturer’s instruction. The isolated RNA samples were diluted in RNase-free double-distilled water (ddH2O) and treated with DNAase I (Takara, Dalian, China) according to the manufacturer’s instructions. RNA concentration was measured on the NanoDrop spectrophotometer (ND-1000, NanoDrop Technologies Inc., Wilmington, DE), and RNA integrity was assessed on the Bioanalyzer 2100 (Agilent Technologies, Amstelveen, The Netherlands) using the RNA 6000 Nano Chip kit (Agilent technologies) by automated gel electrophoresis. RNA of 280 ng was used for cDNA synthesis with M-MLV RTase (Takara, Dalian, China) according to manufacturer’s description. Real-time (quantitative) RT-qPCR was performed using CFX96 Real-Time PCR Detection System (Bio-Rad, Hercules, CA, USA) and SYBR Premix Ex Taq II kit (Takara, Dalian, China). RT-qPCR primers for hsp70, mstn, mt, apaf1, vezf1, cyp1a, and β-actin gene were designed by previous studies (Liu et al. 2008; Yang et al. 2010; Zhu et al. 2013) and listed in Table 1. The housekeeping gene β-actin was used as an internal standard (Wang et al. 2010; Zhu et al. 2013, 2014). PCR was run in the following cycling conditions: initial denaturation at 95 °C for 10 min, 40 cycles of 95 °C for 10 s, 58 °C for 10 s, and 72 °C for 30 s. Relative expression was calculated by using a modified comparative cycle threshold (CT) method. Each individual sample was run in triplicate wells.
Statistical analysis
The LC50 and EC50 (median effective concentration) values with 95 % confidence intervals for five triazole fungicides were calculated by Trimmed Spearman-Karber statistical methods based on the percentages of abnormalities or dead organisms (Hamilton et al. 1977). mRNA relative expression was calculated using ΔΔC t method (Livak and Schmittgen 2001). To evaluate exposure data, all data were expressed as mean ± S.E.M and were analyzed by one-way ANOVA. The differences were considered significant at p < 0.05 and extremely significant at p < 0.01.
Results
Mortality rate and malformation rate
The percentage of embryos with teratogenic and lethal effects clearly increased with increasing triazole pesticide concentrations in a concentration-dependent pattern. As shown in Fig. 2a, no mortality was observed in the lowest concentration (1 mg/L). In addition, mortality rate was up to 100 % at 15 mg/L for fluconazole. Embryo malformation became apparent at 1 mg/L for fluconazole and myclobutanil, 3 mg/L for triflumizole, epoxiconazole, and flusilazole. Higher concentrations of triazole fungicides (13 mg/L for fluconazole and myclobutanil) resulted in 100 % malformation rates, and about 80 % malformation rates were observed at 15 mg/L for triflumizole, epoxiconazole, and flusilazole (Fig. 2b). Values of 72 h LC50 and EC50 with their 95 % confidence intervals are listed in Table 2. Based on the values of LC50 and EC50, the toxicity order of five triazole fungicides was fluconazole > myclobutanil > triflumizole > epoxiconazole > flusilazole.
Body length
Body length of 72-h larvae from the control group was 4.73 ± 0.05 mm (mean ± SD). Embryos exposed to triazole fungicides showed significant reduction of body length (Fig. 3a) at concentration as low as 1.0 mg/L for fluconazole, myclobutanil, and triflumizole compared with the control group. Strikingly, more than 30 % reduction for body length was observed for embryos exposed to 5.0 mg/L fluconazole and myclobutanil in comparison to control embryos. However, the highest concentration treatment (5.0 mg/L) had no significant reduction for flusilazole in comparison to control.
Body length (a), body weight (b), and heart rate (c) after embryos exposed to different concentrations of triazole fungicides at 72 h. Values that are significantly different from the control are indicated by asterisks (one-way ANOVA, *p < 0.05; **p < 0.01). Values are presented as mean ± S.E.M. Five replicates were set for the tests, with 30 embryos per replicate
Body weight
From the control group, body weight of newly hatched larval was 0.80 ± 0.06 mg. As shown in Fig. 3b, body weight was significantly decreased by 10.7 and 8.8 % in fluconazole and myclobutanil, respectively, at 0.2 mg/L. At the highest concentration, more than 10 % decrease was observed for five triazole fungicides. In addition, more than 30 % decrease were observed for fluconazole and myclobutanil at 5 mg/L.
Heart rate
From the control group, heart rate of newly hatched larval was 193.2 ± 3.7 beats/min. No significant decrease in heart rate was observed at 0.2 mg/L for five triazole fungicides compared with control group, whereas significant decreases in heart rate were observed at 1.0 mg/L for fluconazole and myclobutanil (Fig. 3c). No significant reduction of heart rate was recorded at the highest concentration (5.0 mg/L) for epoxiconazole and flusilazole.
Enzyme activity
The enzyme activities (SOD, GST, ATPase, and AChE) were detected in G. rarus embryos after exposure to different concentrations of five triazole fungicides (Fig. 4). From the control group, enzyme activities of SOD, GST, ATPase, and AChE were 27.56 ± 1.09, 24.37 ± 1.98, 2.54 ± 0.11, and 1.25 ± 0.15 U/mg protein, respectively. As shown in Fig. 4, significant increases of SOD and GST activities were observed at the lowest concentrations (0.2 mg/L) for fluconazole and myclobutanil. However, significant decrease (more than 25 %) was observed in the highest concentrations (5.0 mg/L) for all five triazole fungicides. For ATPase and AChE activities, significant decreases were observed in medium and highest concentrations for all five triazole fungicides. In addition, more than 80 % decreases of ATPase and AChE activities were observed at the highest concentrations for fluconazole and myclobutanil.
The changes in enzyme activity in embryos after exposure to various concentrations of triazole fungicides until 72 h. a SOD, b GST, c ATPase, d AChE. Values are presented as mean ± S.E.M. Values that are significantly different from the control are indicated by asterisks (one-way ANOVA, *p < 0.05; **p < 0.01). Five replicates were set for the tests, with 200 embryos per replicate
mRNA levels
The mRNA level of six candidate genes (hsp70, mstn, mt, apaf1, vezf1, and cyp1a) was detectable by RT-PCR in G. rarus embryos exposed to five triazole fungicides. In the present study, extremely significant increases were found in all concentrations for the expression of hsp70, apaf1, cyp1a, and mt genes, except the expression of vezf1 and mstn (Fig. 5). In addition, mRNA level increased with increasing triazole fungicide concentrations in a concentration-dependent pattern. For hsp70, more than 150-fold increases were observed for fluconazole at 5 mg/L. Even for flusilazole at the lowest concentration, 20.15-fold increase was found (Fig. 5a). At the highest concentration, the extremely significant increases were observed that range from 17.63- to 54.63-fold for apaf1 (Fig. 5b), 4.36- to 22.42-fold for cyp1a (Fig. 5c), and 4.93- to 9.93-fold for mt (Fig. 5d). The significant decrease was observed that ranges from 0.11- to 0.52-fold for vezf1 and mstn at the highest concentration (Fig. 5e, f).
mRNA levels after 72-h exposure to different concentrations of triazole fungicides. a hsp70, b apaf1, c cyp1a, d mt, e vezf1, f mstn. Values are presented as mean ± S.E.M. After normalization to β-actin gene, one-way ANOVA is used to analyze the differences to control. Values that are significantly different from the control are indicated by asterisks (one-way ANOVA, *p < 0.05; **p < 0.01). Five replicates were set for the tests, with 200 embryos per replicate
Discussion
The present investigation records the toxicity of five triazole fungicides on G. rarus embryo. The 72 h LC50 and EC50 range from 3 to 12 mg/L in this study. Previous studies indicated that the 48 h EC50 of triazophos for Daphnia magna was 12.92 μg/L (Liu et al. 2012), which was in some degree lower that that derived from this study. This considerable difference in acute toxicity is likely attributed to the differences about chemical structural, exposure time, and test species. Recent research on zebrafish embryos has also demonstrated that the benchmark concentration of six triazoles (flusilazole, hexaconazole, cyproconazole, triadimefon, myclobutanil, and triticonazole) ranges from 1.5 to 25 mg/L (Hermsen et al. 2011b), and which were in accordance with present study. In the present study, we also revealed a series of significant developmental changes in heart rate, body weight, and body length induced by triazole fungicides. Machera showed delayed ossification of the skull bones and cleft palate in rat embryos exposed during gestation to cyproconazole (Machera 1995). Xenopus laevis studies also showed craniofacial malformations in embryos exposed to triazoles; mainly, branchial arch malformations were found after exposure to triadimefon, which precedes craniofacial defects (Groppelli et al. 2005; Papis et al. 2006). Zebrafish embryo toxicity test also found that teratogenic effects (heart malformations, pericardial edema, head malformations, yolk sac edema, and yolk deformations) were widely observed after triazole treatment (Hermsen et al. 2011b). It can be concluded that teratogenic effects are the most frequently phenomenon caused by triazole fungicides.
Environmental toxicants can trigger biological effects at the organism level only after initiating biochemical and cellular events (Liang et al. 2007; Robinson et al. 2012). Responses range from molecular, cellular, and physiological phenomena to behavioral changes (Zhong et al. 2005; Hermsen et al. 2011b). Thus, enzyme activity and mRNA level also play a critical role in these evaluation processes (Robinson et al. 2012; Zhu et al. 2013, 2014). In this study, some relative enzyme activities and mRNA levels were chosen for better understanding of the potential effects and action mechanisms. Our result can be regarded as a type of hormesis featured by conversion from low-dose stimulation to high-dose inhibition for SOD and GST activities. Hormesis is a toxicological concept characterized by low-dose stimulation and high-dose inhibition (Calabrese and Baldwin 2000). Extensive examinations of scientific literature by Calabrese and his collaborators reported that hormetic dose-responses are common across biological systems and stressors (Calabrese and Baldwin 2000, 2001). Scientific literature provides evidence that hormesis can be caused by multiple stimuli, such as chemicals (Calabrese and Baldwin 2000; Barceló and Poschenrieder 2002), radiation (Feinendegen 2005), heat (Rea et al. 2005), and even exercise (Gomez-Cabrera et al. 2008). Similar hormetic dose-response results for SOD activity have also been reported in other species. According to Zhao et al. (2013), SOD activity was significantly increased after treated by low concentrations (from 1 to 50 mg/L) of nano-ZnO, and significantly decreased observed after treated by high concentration (100 mg/L) of nano-ZnO. SOD activity changes in Carassius auratus embryo and larvae with prolonged exposure time at different copper concentrations also showed similar hormetic dose-response patterns (Kong et al. 2013). Li and Tan (2011) found increased GST activity in rat tissues after triazole exposure. SOD is an antioxidant enzyme present in the body that provides the first line of defense against oxidative stress which converts reactive oxygen species, the superoxide anion into hydrogen peroxide, and molecular oxygen (Matés 2000). GST is important particularly in the prevention of free radical mediated oxidation of cellular macromolecules such as lipids and proteins (Seth et al. 2001) and subsequent regulation of the glutathione redox system. The decreased SOD and GST levels at high concentration indicated that the protective system of G. rarus embryos might be destroyed. In other words, triazoles could weaken the antioxidant defense system and enhance oxidative stress to damage the embryo physiology.
Vezf1 is an early development gene that encodes a zinc finger transcription factor. In the developing embryo, vezf1 is expressed in the yolk sac mesoderm and the endothelium of the developing vasculature and, in addition, in mesoderm and neuronal tissues (Kuhnert et al. 2005). Mstn, belonging to transforming growth factor (TGF) β superfamily, is a negative regulator of muscular development in mammals and fish (Grobet et al. 1997). In present study, the significantly decreased expression levels of vezf1 and mstn were observed after triazole treatment. The same trends also found after chemical (3,4-dichloroaniline and triazophos) exposure for fish embryos and larvae (Zhu et al. 2013, 2014). Decrease of vezf1 and mstn transcription mediated by triazoles could result in a block of vasculature and muscle development. Those may partly explain the high percent of observed malformations, especially for significant decrease in heart rate and body length. Stress-induced apoptosis is thought to contribute to abnormal development during embryogenesis (Yang et al. 2014). In the present investigation, several important genes that might be involved in apoptosis were investigated to elucidate the mechanisms of triazole exposure. The HSP70 family acts as a molecular chaperone that reduces stress-induced denaturation, regulates the expression of all heat shock proteins and also thought to contribute to abnormal development during embryogenesis (Yamashita 2003; Zhu et al. 2013). Cyp1a is a member of the cytochrome P450 isoenzyme family and can serve as a sensitive biomarker for certain classes of organic chemicals (Woźny et al. 2010). Apaf-1 plays an important role in p53-dependent apoptosis (Soengas et al. 1999). After triazole treatment, the expressions of hsp70, cyp1a, and apaf-1 were significantly increased in this study. The same trends were also found in other fish species (Yamashita 2003; Voelker et al. 2007; Zhu et al. 2013; Yang et al. 2014) for different hazardous substance treatment. On the basis of results from the present study, it may be hypothesized that triazole exposure induces oxidative stress; the oxidative stimulus may trigger apaf-1. Apaf-1 represses the function of the antiapoptotic gene Bcl-2 and disrupts the mitochondrial membrane (Yang et al. 2014). Once the integrity of the mitochondrial membrane is disrupted, mitochondrion releases cytochrome C and inducing caspase-independent apoptosis. However, this hypothesis requires further experiments to validate.
In conclusion, our results implied that triazole exposures can induce significant changes in the development of G. rarus embryos. We also demonstrated that five triazole fungicides caused teratogenicity in a concentration-dependent pattern. In addition, both developmental and biochemical biomarkers in the growing G. rarus embryos are very promising tools to determine the severity of toxicants. We provide further evidence that developmental and metabolism-related genes were affected at lower concentrations providing higher resolution in the attempt to define the teratogenic mechanisms of triazoles.
References
Barceló J, Poschenrieder C (2002) Fast root growth responses, root exudates, and internal detoxification as clues to the mechanisms of aluminium toxicity and resistance: a review. Environ Exp Bot 48:75–92
Bromillow RH, Evans AA, Nicholls PH (1999) Factors affecting degradation rates of five triazole fungicides in two soil types: 1. Laboratory incubations. Pestic Sci 55:1129–1134
Calabrese EJ, Baldwin LA (2000) Chemical hormesis: its historical foundations as a biological hypothesis. Hum Exp Toxicol 19:2–31
Calabrese EJ, Baldwin LA (2001) U-shaped dose-responses in biology, toxicology, and public health. Annu Rev Public Health 22:15–33
Chen PJ, Moore T, Nesnow S (2008) Cytotoxic effects of propiconazole and its metabolites in mouse and human hepatoma cells and primary mouse hepatocytes. Toxicol in Vitro 22:1476–1483
de Jong E, Barenys M, Hermsen SA, Verhoef A, Ossendorp BC, Bessems JG, Piersma AH (2011) Comparison of the mouse embryonic stem cell test, the rat whole embryo culture and the zebrafish embryotoxicity test as alternative methods for developmental toxicity testing of six 1,2,4-triazoles. Toxicol Appl Pharmacol 253:103–111
Ding F, Song WH, Guo J, Gao ML, Hu WX (2009) Oxidative stress and structure-activity relationship in the zebrafish (Danio rerio) under exposure to paclobutrazol. J Environ Sci Health B 44:44–50
Farag AT, Ibrahim HH (2007) Developmental toxic effects of antifungal flusilazole administered by gavage to mice. Birth Defects Res B Dev Reprod Toxicol 80:12–17
Feinendegen LE (2005) Evidence for beneficial low level radiation effects and radiation hormesis. Br J Radiol 78:3–7
Gomez-Cabrera MC, Domenec E, Viña J (2008) Moderate exercise is an antioxidant: upregulation of antioxidant genes by training. Free Radic Biol Med 44:126–131
Grobet L, Martin LJ, Poncelet D, Pirottin D, Brouwers B, Riquet J, Schoeberlein A, Dunner S, Ménissier F, Massabanda J, Fries R, Hanset R, Georges M (1997) A deletion in the bovine myostatin gene causes the double-muscled phenotype in cattle. Nat Genet 17:71–74
Groppelli S, Pennati R, De Bernardi F, Menegola E, Giavini E, Sotgia C (2005) Teratogenic effects of two antifungal triazoles, triadimefon and triadimenol, on Xenopus laevis development: craniofacial defects. Aquat Toxicol 73:370–381
Hamilton MA, Russo RC, Thurston RV (1977) Trimmed Spearman–Karber method for estimating median lethal concentrations in toxicity bioassays. Environ Sci Technol 12:714–720
Hermsen SA, Pronk TE, van den Brandhof EJ, van der Ven LT, Piersma AH (2011a) Chemical class-specific gene expression changes in the zebrafish embryo after exposure to glycol ether alkoxy acids and 1,2,4-triazole antifungals. Reprod Toxicol 32:245–252
Hermsen SA, van den Brandhof EJ, van der Ven LT, Piersma AH (2011b) Relative embryotoxicity of two classes of chemicals in a modified zebrafish embryotoxicity test and comparison with their in vivo potencies. Toxicol in Vitro 25:745–753
Kong X, Jiang H, Wang S, Wu X, Fei W, Li L, Nie G, Li X (2013) Effects of copper exposure on the hatching status and antioxidant defense at different developmental stages of embryos and larvae of goldfish Carassius auratus. Chemosphere 92:1458–1464
Konwick BJ, Garrison AW, Avants JK, Fisk AT (2006) Bioaccumulation and biotransformation of chiral triazole fungicides in rainbow trout (Oncorhynchus mykiss). Aquat Toxicol 80:372–381
Kuhnert F, Campagnolo L, Xiong JW, Lemons D, Fitch MJ, Zou Z, Kiosses WB, Gardner H, Stuhlmann H (2005) Dosage-dependent requirement for mouse Vezf1 in vascular system development. Dev Biol 283:140–156
Li ZH, Randak T (2009) Residual pharmaceutically active compounds (PhACs) in aquatic environment-status, toxicity and kinetics: a review. Vet Med-Czech 52:295–314
Li S, Tan Y (2011) Hormetic response of cholinesterase from Daphnia magna in chronic exposure to triazophos and chlorpyrifos. J Environ Sci (China) 23:852–859
Li ZH, Zlabek V, Grabic R, Li P, Randak T (2010a) Modulation of glutathione-related antioxidant defense system of fish chronically treated by the fungicide propiconazole. Comp Biochem Physiol C Toxicol Pharmacol 152:392–398
Li ZH, Zlabek V, Grabic R, Li P, Machova J, Velisek J, Randak T (2010b) Effects of exposure to sublethal propiconazole on theantioxidant defense system and Na+-K+-ATPase activity in brain of rainbow trout, Oncorhynchus mykiss. Aquat Toxicol 98:297–303
Liang C, Jin R, Gui W, Zhu G (2007) Enzyme-linked immunosorbent assay based on a monoclonal antibody for the detection of the insecticide triazophos: assay optimization and application to environmental samples. Environ Sci Technol 41:6783–6788
Liu Y, Wang J, Wei Y, Zhang H, Liu Y, Dai J (2008) Molecular characterization of cytochrome P450 1A and 3A and the effects of perfluorooctanoic acid on their mRNA levels in rare minnow (Gobiocypris rarus) gills. Aquat Toxicol 88:183–190
Liu H, Yuan B, Li S (2012) Altered quantities and in vivo activities of cholinesterase from Daphnia magna in sub-lethal exposure to organophosphorus insecticides. Ecotoxicol Environ Saf 80:118–125
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 25:402–408
Machera K (1995) Developmental toxicity of cyproconazole, an inhibitor of fungal ergosterol biosynthesis, in the rat. Bull Environ Contam Toxicol 54:363–369
Marotta F, Tiboni GM (2010) Molecular aspects of azoles-induced teratogenesis. Expert Opin Drug Metab Toxicol 6:461–482
Matés JM (2000) Effects of antioxidant enzymes in the molecular control of reactive oxygen species toxicology. Toxicology 153:83–104
Menegola E, Broccia ML, Di Renzo F, Massa V, Giavini E (2005) Craniofacial and axial skeletal defects induced by the fungicide triadimefon in the mouse. Birth Defects Res B Dev Reprod Toxicol 74:185–195
Menegola E, Broccia ML, Di Renzo F, Giavini E (2006) Postulated pathogenic pathway in triazole fungicide induced dysmorphogenic effects. Reprod Toxicol 22:186–195
Mu X, Pang S, Sun X, Gao J, Chen J, Chen X, Li X, Wang C (2013) Evaluation of acute and developmental effects of difenoconazole via multiple stage zebrafish assays. Environ Pollut 175:147–157
Nagel R (2002) DarT: the embryo test with the zebrafish Danio rerio - a general model in ecotoxicology and toxicology. ALTEX 19:38–48
Papis E, Bernardini G, Gornati R, Prati M (2006) Triadimefon causes branchial arch malformations in Xenopus laevis embryos. Environ Sci Pollut Res Int 13:251–255
Qun-Fang Z, Gui-Bin J, Ji-Yan L (2002) Effects of sulethal levels of tributyltin chloride in a new toxicity test organism: the Chinese rare minnow (Gobiocypris rarus). Arch Environ Contam Toxicol 42:332–337
Ramsden CS, Henry TB, Handy RD (2013) Sub-lethal effects of titanium dioxide nanoparticles on the physiology and reproduction of zebrafish. Aquat Toxicol 126:404–413
Raudonis L, Surviliene E, Valiuskaite A (2004) Toxicity of pesticides to predatory mites and insects in apple-tree site under field conditions. Environ Toxicol 19:291–295
Rea SL, Wu D, Cypser JR, Vaupel JW, Johnson TE (2005) A stress-sensitive reporter predicts longevity in isogenic populations of Caenorhabditis elegans. Nat Genet 37:894–898
Robinson JF, Tonk EC, Verhoef A, Piersma AH (2012) Triazole induced concentration-related gene signatures in rat whole embryo culture. Reprod Toxicol 34:275–283
Seth V, Banerjee BD, Chakraborty AK (2001) Lipid peroxidation, free radical scavenging enzymes, and glutathione redox system in blood of rats exposed to propoxur. Pestic Biochem Phys 71:133–139
Shi XJ, Du YB, Lam PK, Wu RS, Zhou BS (2008) Developmental toxicity and alteration of gene expression in zebrafish embryos exposed to PFOS. Toxicol Appl Pharmacol 230:23–32
Soengas MS, Alarcón RM, Yoshida H, Giaccia AJ, Hakem R, Mak TW, Lowe SW (1999) Apaf-1 and caspase-9 in p53-dependent apoptosis and tumor inhibition. Science 284:156–159
Tiboni GM, Marotta F, Carletti E (2009) Fluconazole alters CYP26 gene expression in mouse embryos. Reprod Toxicol 27:199–202
Voelker D, Vess C, Tillmann M, Nagel R, Otto GW, Geisler R, Schirmer K, Scholz S (2007) Differential gene expression as a toxicant-sensitive endpoint in zebrafish embryos and larvae. Aquat Toxicol 81:355–364
Wang J, Liu X, Wang H, Wu T, Hu X, Qin F, Wang Z (2010) Expression of two cytochrome P450 aromatase genes is regulated by endocrine disrupting chemicals in rare minnow Gobiocypris rarus juveniles. Comp Biochem Physiol C Toxicol Pharmacol 152:313–320
Wang C, Wu Q, Wu C, Wang Z (2011) Application of dispersion-solidification liquid-liquid microextraction for the determination of triazole fungicides in environmental water samples by high-performance liquid chromatography. J Hazard Mater 185:71–76
Woźny M, Brzuzan P, Łuczyński MK, Góra M, Wolińska L, Bukowski R, Podlasz P (2010) CYP1A expression in liver and gills of rainbow trout (Oncorhynchus mykiss) after short-term exposure to dibenzothiophene (DBT). Chemosphere 79:110–112
Yamashita M (2003) Apoptosis in zebrafish development. Comp Biochem Physiol B Biochem Mol Biol 136:731–742
Yang L, Zha J, Li W, Li Z, Wang Z (2010) Atrazine affects kidney and adrenal hormones (AHs) related genes expressions of rare minnow (Gobiocypris rarus). Aquat Toxicol 97:204–211
Yang S, Liu S, Ren Z, Jiao X, Qin S (2014) Induction of oxidative stress and related transcriptional effects of perfluorononanoic acid using an in vivo assessment. Comp Biochem Physiol C Toxicol Pharmacol 160:60–65
Zarn JA, Bruschweiler BJ, Schlatter JR (2003) Azole fungicides affect mammalian steroidogenesis by inhibiting sterol 14 alpha-demethylase and aromatase. Environ Health Perspect 111:255–261
Zha J, Wang Z, Wang N, Ingersoll C (2007) Histological alternation and vitellogenin induction in adult rare minnow (Gobiocypris rarus) after exposure to ethynylestradiol and nonylphenol. Chemosphere 66:488–495
Zhang X, Zha J, Li W, Yang L, Wang Z (2008) Effects of 2,4-dichlorophenol on the expression of vitellogenin and estrogen receptor genes and physiology impairments in Chinese rare minnow (Gobiocypris rarus). Environ Toxicol 23:694–701
Zhao X, Wang S, Wu Y, You H, Lv L (2013) Acute ZnO nanoparticles exposure induces developmental toxicity, oxidative stress and DNA damage in embryo-larval zebrafish. Aquat Toxicol 136–137:49–59
Zhong XP, Xu Y, Liang Y, Liao T, Wang JW (2005) The Chinese rare minnow (Gobiocypris rarus) as an in vivo model for endocrine disruption in freshwater teleosts: a full life-cycle test with diethylstilbestrol. Aquat Toxicol 71:85–95
Zhu B, Wu ZF, Li J, Wang GX (2011) Single and joint action toxicity of heavy metals on early developmental stages of Chinese rare minnow (Gobiocypris rarus). Ecotoxicol Environ Saf 74:2193–2202
Zhu B, Liu TQ, Wang GX (2013) Developmental toxicity of 3,4-dichloroaniline on rare minnow (Gobiocypris rarus) embryos and larvae. Chemosphere 90:1132–1139
Zhu B, Gong YX, Liu L, Li DL, Wang Y, Ling F, Wang GX (2014) Toxic effects of triazophos on rare minnow (Gobiocypris rarus) embryos and larvae. Chemosphere 108:46–54
Acknowledgments
We would like to acknowledge Jia Song for the technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Zhu, B., Liu, L., Gong, YX. et al. Triazole-induced toxicity in developing rare minnow (Gobiocypris rarus) embryos. Environ Sci Pollut Res 21, 13625–13635 (2014). https://doi.org/10.1007/s11356-014-3317-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3317-6