Abstract
Open-top chambers (OTCs) are widely used experimental warming devices in open-field ecosystems such as tundra and alpine heath. However, knowledge of their performance in temperate deciduous forest ecosystems is largely lacking. The application of OTCs in forests might become important in the future since the effects of climate warming on growth, reproduction, and future distribution of understorey forest herbs have rarely been investigated. Therefore, polycarbonate OTCs covered with (OTCs+GF) and without permeable polypropylene GardenFleece (OTCs−GF) were installed in a temperate deciduous forest to create an experimental warming gradient. Short-term responses in phenology, growth, and reproduction of a model understorey forest herb (Anemone nemorosa L.) to OTC installation were determined. In a second growing season, an in-depth study of multiple abiotic conditions inside OTCs−GF was performed. Both OTCs+GF and OTCs−GF raised air and soil temperature in a realistic manner (ca. +0.4°C to +1.15°C), but OTCs−GF only in the leafless period (up to +1.5°C monthly average soil temperature). The early flowering forest herb A. nemorosa also showed a clear phenotypic response to OTC installation. Based on these facts and the large ecological drawbacks associated with OTCs+GF (mostly in connection with a higher relative air humidity and a lower light quantity) and very modest abiotic changes in OTCs−GF, we encourage the use of OTCs−GF in deciduous forest ecosystems for evaluating climate-warming effects on early flowering understorey forest herbs. There is also a potential to use this warming method on later flowering species, but this needs further research.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
It has been demonstrated that global warming already induces altitudinal and latitudinal plant migration in different parts of the world (e.g., Malcolm et al. 2002; Parmesan and Yohe 2003). Despite the ecological significance of forest herbs for temperate forest ecosystem biodiversity, competitive interactions, linkage with overstorey, and ecosystem functioning in general (Gilliam 2007), knowledge about the effects of climate warming on their phenology, growth, reproduction, and future distribution is largely lacking (but see Farnsworth et al. 1995; Svenning and Skov 2006; Jochum et al. 2007; De Frenne et al. 2009). Since it has been shown that many forest herbs are slow colonizers (Verheyen et al. 2003; Hermy and Verheyen 2007), climate change may hamper possible migration. This might impose a serious threat to their survival (Honnay et al. 2002). In order to predict future response of forest herbs to climate warming, there is an urgent need to better understand the potential effects of warming on vegetative growth, seed production and germination for these species (e.g., Hovenden et al. 2008).
Various temperature manipulation systems have been applied worldwide to study the influence of climate warming on ecosystems (e.g., Marion et al. 1997; Kimball 2005; Netten et al. 2008). The use of open-top chambers (OTCs) is the most common, simplest, and cheapest approach. OTCs are small greenhouses made of translucent Plexiglas with inclined walls and an open top. They passively heat a small vegetation plot by capturing solar radiation and protecting against wind. Several studies report on the use of OTCs in open-field ecosystems such as tundra (Arft et al. 1999; Hollister and Webber 2000; Danby and Hik 2007), alpine meadows (Totland and Nylehn 1998; Totland 1999; Kudernatsch et al. 2008), and South African quartz-fields (Musil et al. 2005). We are, however, aware of only one single study using OTCs to create a rise in temperature in a forest ecosystem (a 60-year spruce plantation at 3,110 m a.s.l. in China) examining seedling growth and physiology of two conifers (Yin et al. 2008).
In the present study, we aimed to find out whether OTCs can be used in a deciduous temperate forest ecosystem to assess the impact of a rise in temperature on herbaceous understorey plant species. We examined Anemone nemorosa L. (Ranunculaceae) which may be considered a model species for (European) slow colonizing herbaceous forest plant species. The present study is, according to our knowledge, the first to use open-top chambers for warming purposes in deciduous forest. Since it can be expected that the efficiency of OTCs is lower in forests than in open-field ecosystems due to its specific properties (shading, moist soils, lower wind speed, etc.), half of the OTCs were additionally covered with a permeable polypropylene fleece allowing both air and moisture exchange, but minimizing turbulence and potentially increasing the temperature difference with the ambient controls.
We specifically addressed the following question from a purely methodological point of view: are OTCs useful devices to gain insight in the impact of a warming climate on herbaceous understorey plants in a temperate deciduous forest ecosystem? Hence, the OTCs must fulfill the following criteria: (1) temperature is increased adequately and in a realistic manner compared to the IPCC predictions (IPCC 2007) and (2) ecological abiotic and biotic drawbacks of the OTCs are limited. Furthermore, given the high degree of phenotypic plasticity of A. nemorosa (Shirreffs 1985; De Frenne et al. 2009), a certain response in a trait of this forest herb to OTC installation, increases the potential usefulness of this experimental warming method.
Materials and methods
Study species
Wood anemone, Anemone nemorosa L. (Ranunculaceae), is a perennial and vernal forest geophyte common in deciduous temperate European woodlands, but it can also be found in W-Asia. It grows mostly in ancient forest (sensu Hermy and Verheyen 2007) on rich, usually not too acidic soils, moist to wet and can sustain in open areas (Shirreffs 1985). Shoots emerge between February and May and flowering starts a few weeks later. Flowers (one per shoot) are typically white, hermaphrodite, and mostly self-incompatible and insect-pollinated, although selfing occurs (Müller et al. 2000). The nuts (usually 10–30 per ramet) containing a single seed are mainly gravity-dispersed between May and June, but myrmecochory occurs as well (Delatte and Chabrerie 2008). Seeds do not form a persistent seed bank (Eriksson 1995). Vegetative spread through rhizomes is common (Brunet and von Oheimb 1998; Philipp and Petersen 2007) but reproduction through seeds, however, is considered to be most important for population maintenance and spread (Brunet and von Oheimb 1998; Müller et al. 2000; Stehlik and Holderegger 2000). Anemone nemorosa has typically low colonization rates (Verheyen et al. 2003), which may impose a threat if migration would be necessary when climate changes. It is actually one of the most cited ancient forest plant species (e.g., Verheyen et al. 2003).
Study area
The Aelmoeseneie forest (50°58′N, 3°49′E, 16 m a.s.l.) is a temperate mixed deciduous forest near Ghent in central Belgium, ca. 60 km from the North Sea. The forest has a total area of 28 ha and the dominant trees are about 85 years old. Mean (1961–1990) annual precipitation amounts to 821 mm, and is evenly distributed throughout the year. Mean annual temperature is 9.7°C, mean temperature of the coldest and warmest month is 2.5 and 17.2°C, respectively (FAO 2005). The fenced study area is located in a forest stand dominated by ash (Fraxinus excelsior L.) and pedunculate oak (Quercus robur L.). The approximate time of canopy flush for Q. robur (expressed as the date when the first completely unfolded leaves are visible) was around 5 to 10 April in both 2008 and 2009. The forest soil has developed from a quaternary layer of sand loam (0.5–1 m) on a shallow impermeable clay and sand complex of tertiary origin and is classified as Gleyic Cambisol in the World Reference Base (ISS-ISRIC-FAO 1998). Anemone nemorosa is widely distributed, carpeting the forest soil and dominating the ground vegetation until June. Lamiastrum galeobdolon (L.) Ehrend. & Polatschek, Oxalis acetosella L., Polygonatum multiflorum (L.) All. and Hedera helix L. co-occur.
Experimental design
To simulate climate warming, six hexagonal open-top chambers (OTCs) (cf. Marion et al. 1997; Hollister and Webber 2000) were installed to passively warm small vegetation plots within the natural carpeting A. nemorosa community with >90% cover. The OTCs were 60 cm high; one side was 66.4 cm at the base and 34.6 cm at the top and covered a surface area of 1.15 m2. The OTCs were made of extruded polycarbonate (VIS & UV light transmission 86%, IR light transmission <5%, DIN 5036) (Quinn Plastics, Derrylin, Northern Ireland). The open-top design allows free air exchange and is said to minimize undesirable chamber effects as changed precipitation, gas and humidity concentrations, and exclusion of pollinators or herbivores (Marion et al. 1997; Kanerva et al. 2005; Netten et al. 2008). To create a warming gradient, half of the open-top chambers (n = 3) were additionally covered with permeable 17 g m−2 polypropylene GardenFleece (GF; light transmission is 90.3% between 250 and 800 nm wavelength; Freudenberg, Vliesstoffe KG, Kaiserslautern, Germany) allowing both air and moisture exchange with the environment, but minimizing turbulence. The GF was attached on the top edges of the OTCs, into a small, light, and hexagonal wooden frame which could be lifted to carry out the field work inside the plots.
In the near vicinity of each OTC, an 80 × 80 cm2 control plot (0.64 m2; n = 6) was established. The OTCs and paired control plots were randomly installed in areas with expected equal cover percentages of A. nemorosa on 1 February 2008 before emergence of the shoots. Hence, three treatments were applied: control plots (C), open-top chambers (OTC–GF) and open-top chambers covered with GF (OTC+GF). Measurements of the first growing season were finished on 8 May 2008.
Abiotic variables (Table 1)
To characterize the abiotic conditions in the OTCs in comparison with the control plots, soil surface temperature (in the litter layer; 0 cm depth) and air temperature (10 cm above ground level) were measured at 15-min intervals inside two OTCs−GF, two OTCs+GF and two control plots using Decagon ECT Temperature probes connected to Decagon Em50 dataloggers (Decagon Devices Inc., Pullman, WA, USA). Furthermore, daytime relative air humidity (10 cm above ground level; RH) was measured in all plots every 5 days with an Eijkelkamp Portable P4.01 logger (Eijkelkamp Agrisearch Equipment, Giesbeek, The Netherlands) in April 2008 (n = 6). To evaluate the effect of the permeable GF on precipitation amount, six manual rain gauges were installed in an open-field grassland area adjacent to the forest stand, of which half were covered with GF (n = 3). Cumulative precipitation was measured weekly between 20 March and 16 May 2008.
Biotic variables (Table 1)
To have an estimate of phenological development, the number of open flowers per plot was counted twice a week (every 3–5 days) from the date the first individual flowered (25 February 2008) until all flowers had disappeared (1 May 2008). Flowers were considered open when the anthers were visible and thus exposed to pollinators. To have an estimate of maximal plant growth, total plant height (incl. flowers) of the eight highest wood anemones per plot (evenly spread per quadrant) was measured weekly. Furthermore, at the stage of seed maturity (see further), we harvested and determined the total oven-dried (50°C) above-ground dry matter biomass per plot (TADM, kg ha−1).
Ten random individuals (above-ground ramets) per plot were selected for further analysis from the harvested material. These ramets were flat oven-dried to constant mass (50°C). Leaf area (LA, mm2) was measured with a Li-Cor Portable Area Meter Li-3000 (Li-Cor Biosciences, Nebraska, USA). Leaf dry matter (LDM, mg) was weighed to the nearest 0.1 mg. Specific leaf area (SLA, mm2 mg−1) for the ten ramets per plot was calculated by dividing LA by LDM. Finally, leaf samples were pooled per plot and analyzed for total N (modified method of Kjeldahl) and total estimated C (loss on ignition, 4 h at increasing temperature until 450°C, [%C = (100 − % of ashes residue)/2]). C/N ratio was calculated by dividing C by N.
At the stage of seed maturity (8 May 2008), we collected the nuts (hereafter referred to as seeds) of ten randomly chosen ramets per plot. Seeds were considered mature when natural dispersal began (Baskin and Baskin 1998), i.e., when seeds fell easily from the plant. The seeds were air-dried at room temperature for 2–3 weeks. For each ramet, all seeds were counted, weighted (to the nearest 0.1 mg), and total number of seeds and mean seed mass (total seed mass/total number of seeds) were calculated. Per plot, 20–50 seeds (number was dependent on the availability) were randomly distributed in three Petri dishes lined with moist filter paper (Macherey–Nagel 440B) and placed in temperature-controlled incubators. Since A. nemorosa seeds germinate in autumn (Mondoni et al. 2008), seeds were given 8 weeks warm stratification (23°C) and subsequent cold stratification (10°C). Filter papers were kept moist with distilled water during the experiment. The number of germinated seeds (seeds with emerged radicle) was recorded weekly for 16 weeks after transfer to 10°C until germination had ceased in all dishes.
In-depth study OTCs−GF
To give a well-documented judgment on the OTCs−GF, an in-depth study of these open-top chambers was performed to gain insight in other possibly co-varying abiotic conditions. Therefore, five OTCs−GF and five paired control plots (all characteristics of plots and OTCs were identical to the 2008 design) were installed in a similar site ca. 100 m from the site where the first growing season measurements were performed (measurements from 17 September 2008 to 8 May 2009).
Soil temperature (5 cm depth), soil surface temperature (in the litter layer, 0 cm depth), air temperature (10 cm above ground level), relative air humidity (10 cm above ground level, %) and soil moisture (0–5 cm depth) were measured at 40-min intervals between 17 September 2008 and 8 May 2009 inside two randomly selected OTCs−GF and two control plots using Decagon EHT RH/Temperature, ECT Temperature, and EC-5 Soil Moisture probes connected to Decagon Em50 dataloggers. Furthermore, changes in light quantity and CO2 concentration were quantified. Photosynthetically active radiation (PAR) was measured with a Spectrum Technologies (Illinois, USA) PAR 3668 sensor connected to a Watchdog 425 datalogger at a 1-min interval for 5 days in February 2009 (before canopy flush). The sensors were used randomly within the five OTCs and control plots. A Bruël and Kjaer (Veenendaal, The Netherlands) Multigasmonitor Type 1302 was used to measure CO2 concentration in the five OTCs−GF and control plots on 7 April 2009 (before canopy flush) at 10 cm above ground level (Table 1).
Data analysis
Phenological advancement of height growth was analyzed by a repeated-measures general linear model (GLM) with time as within-subjects factor and treatment as between-subjects factor. Flowering phenology from OTCs and controls within one time period was compared using a Kruskal–Wallis test.
To compare characteristics of A. nemorosa (see Table 1) between the OTCs and control plots, their effect size was calculated as the natural logarithm of the ratio of the experimental mean to the control mean (according to Arft et al. 1999; Hedges et al. 1999):
with E = effect size and x = means of response variables for the OTCs and control plots.
Use of the natural logarithm linearizes the metric ratio (it treats deviations in the numerator the same as deviations in the denominator) and provides a more normal sampling distribution in small samples (Hedges et al. 1999).
In the case of germination percentage, an adapted effect size E′ was calculated as
since some OTC plots had zero germination percentage.
Biotic data available on ramet level (Table 1) were first tested on assumptions of normality and homogeneity of variances and then analyzed with a GLM with plot as block factor nested within treatment (three levels: OTC+GF, OTC−GF, and control) and a Bonferroni post-hoc test. Because of limited sample size, effect sizes of data available on plot level (Table 1) were analyzed with a Wilcoxon signed rank test. All analyses were performed with SPSS 15.0 and S-Plus 8.0.
Results
Abiotic effects first growing season
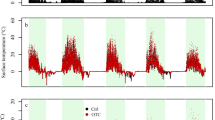
Between 1 February and 8 May 2008, average air and soil surface temperatures were higher in the OTCs compared to the control plots: in the OTCs−GF, average air and soil temperatures were +0.06 and +0.28°C higher, while in the OTCs+GF, air and soil temperatures increased by +0.44 and +1.19°C (Table 2). Daytime relative humidity was +10.6% [95% confidence interval: 7.2–14.1%] and +31.0% [27.9–34.2%] higher in the OTC−GF and the OTC+GF plots, respectively, compared to the control plots in April 2008 (for all time periods: Kruskal–Wallis Chi-square >9.57, df = 2, p < 0.01). Cumulative precipitation decreased by 13.4% in the rain gauges covered with GF compared to the open rain gauges (Wilcoxon rank-sum statistic Z = 1.964, n = 3, p = 0.050). Considering the fact that the variability of the precipitation flux in our study plot ranges between 40 and 80% of the open-field precipitation (Staelens et al. 2006), this 13% decreased precipitation flux in the OTC+GF-plots falls within the natural internal variation of our study plot.
Biotic effects
Both open-top chamber types altered some aspects of the growth and reproduction of A. nemorosa. While flower emergence was not affected, flower senescence was accelerated in the OTC+GF treatment compared to OTC−GF and the control plots (Fig. 1). Plant height was significantly increased (F = 5.88, df hypothesis = 14, df error = 208, p < 0.001) in both OTC treatments compared to the controls (Fig. 2). Total above-ground dry matter (TADM; signed-rank statistic Z = −0.53, p = 0.593, and Z = 0, p = 1, respectively, for OTCs+GF and OTCs−GF), specific leaf area (SLA; F = 0.784, df hypothesis = 2, df error = 9, p = 0.485), leaf N, leaf C, and leaf C/N ratio (all |Z| < 1.34, p > 0.180) did not change significantly in both OTC-types in comparison to the controls (Fig. 3).
Influence of open-top chamber installation on phenological advancement of A. nemorosa’s flowers expressed as a percentage of flowering ramets at a particular date to the maximum number of flowering ramets per plot (mean ± SE) in the control (C) and open-top chambers covered with (OTC+GF) and without (OTC−GF) GardenFleece. Significance levels from Kruskal–Wallis test: (*), p < 0.1; *, p < 0.05
Influence of open-top chamber installation on growth and chemical composition of the leaves of A. nemorosa (mean effect size ± SE). Total above-ground biomass (TADM; kg ha−1), specific leaf area (SLA, mm2 mg−1), leaf nitrogen (N), leaf carbon (C) and C/N ratio of A. nemorosa in the open-top chambers covered with (OTC+GF) and without (OTC−GF) GardenFleece compared to the control. No significant differences
A negative effect on number of seeds per individual in the OTCs+GF (F = 9.95, df hypothesis = 2, df error = 9.15, p = 0.005) was detected, while seed mass (F = 0.64, df hypothesis = 2, df error = 9.05, p = 0.550) and germination percentage (Z = −1.604, p = 0.109) were not significantly influenced (Fig. 4).
Influence of open-top chamber installation on reproduction of A. nemorosa. Seed number per individual, seed mass (mg), and germination percentage (%) (mean effect size ± SE) of A. nemorosa in the open-top chambers covered with (OTC+GF) and without (OTC−GF) GardenFleece compared to the control plots. Significance levels from Wilcoxon test for germination percentage (plot level) and Bonferroni post-hoc test for seed number and seed mass (ramet level): (ns), p > 0.1. **, p < 0.01
In-depth study OTCs−GF
Temperature differences between the OTCs−GF and control plots in the second study period were comparable to the first growing season and increased from the air (+0.38°C) over the surface (+0.41°C) to the soil temperature (+1.15°C). As in the first growing season, (1) temperature differences between the OTCs−GF and controls decreased after canopy flush, and (2) temperature differences increase from the air temperature, over the soil surface temperature to the soil temperature at 5 cm depth (Table 2).
Continuous RH measurements in the OTCs−GF gave a slight increase of +1.2% RH in the OTCs−GF compared to the control plots in the second growing season. Photosynthetically active radiation (PAR) decreased by −7.5% (from an average of 67.4 to 62.4 μmol m−2 s−1, respectively) in the OTCs−GF compared to the control plots before canopy flush. The concentration of CO2 gas in the OTCs−GF was not significantly different from the CO2 concentration in the control plots (466.4 ppm in the control plots vs. 470.0 ppm in the OTCs−GF; Wilcoxon’s Z = −1.273, n = 5, p = 0.203). Furthermore, volumetric soil moisture was +1.6% higher in the OTCs−GF compared to the control plots (18.1 vs. 16.5 vol%, respectively).
Discussion
Temperature elevation in OTCs
Mean air, soil surface, and soil temperature were found to increase by 0.4°C to 1.15°C in OTCs. The temperature increase in the OTCs−GF compared to the control plots is highest in the leafless period (February and March 2008 and October 2008 to March 2009), whereas the OTCs+GF maintained a similar temperature elevation after canopy flush. Although the IPCC (2007) predicts a median increase in annual temperature of +3.2°C by 2080–2099 in northern Europe, this increase will only be reached over a century. Hence, an abrupt increase of 3°C is more unrealistic than an increase of 1°C. Comparable temperature elevations were also reported by Yin et al. (2008) for OTCs placed in a 60-year spruce plantation in China wherein air temperature rose with 0.51°C and soil surface temperature with 0.34°C between April and September. Several studies, however, report on significantly higher temperature elevations when OTCs are placed in open-field ecosystems. Musil et al. (2005) installed OTCs in arid African quartz-fields and measured +5.5°C average warming. Also, Marion et al. (1997) report an average temperature elevation of 1.2–1.8°C in OTCs placed in tundra ecosystems around the world. In German alpine meadows, Kudernatsch et al. (2008) measured an increase of 0.7–1.4°C for air temperature and 0.2–0.8°C for soil temperature. The apparently lower temperature enhancements in forest ecosystems after canopy flush compared to open-field ecosystems are probably due to specific characteristics of forests: (1) the overstorey produces shade, (2) causes spectral shifting of penetrating solar radiation (Daws et al. 2002), which lowers the radiation-capturing capacity of the polycarbonate OTCs, (3) tempers average wind speed making the OTCs less effective and (4) moist litter layers furthermore tend to heat slower than for example dry African quartz-fields or mineral arctic soils (see also Yin et al. 2008).
Response of A. nemorosa to OTC installation
Several global meta-analyses and warming experiments document significant advancements of spring events such as flowering and fruiting (Fitter et al. 1995; Dunne et al. 2003; Parmesan and Yohe 2003; Sherry et al. 2007). In the present study, however, we did not observe a phenological acceleration of A. nemorosa flowers in the first spring after experimental warming was started. However, Tyler (2001) analyzed a 12-year flowering time series of A. nemorosa in Sweden and came to the conclusion that none of the temperature variables determined flowering of wood anemone. This might indicate that A. nemorosa may be one of the species whose phenology will not be affected by climate warming, maybe due to the role of photoperiodism in initiation of flowering (Yan and Wallace 1996), especially for spring species.
The accelerated flower senescence in the OTCs+GF plots cannot be caused by the absence of pollinators by the GF barrier (see further), since it is known that pollination accelerates senescence and cell death in the petals of flowers (Hadfield and Bennett 1997). The earlier senescence may be caused by two other (co-)occurring factors: (1) temperature elevation leads to faster development and senescence of flowers (Rubinstein 2000) and (2) relative air humidity elevation in the OTCs+GF may have caused growth of fungi and petal senescence is often hastened by wounding of the flower parts (Rubinstein 2000).
Both temperature treatments induced similar height growth of A. nemorosa, and may be caused by the fact that soil temperature at −5 cm increased more than soil surface temperature in open OTCs, which might be important for geophytes such as A. nemorosa. These results are consistent with other warming experiments that showed plant species to grow better or higher when there are no other limiting factors (nutrients, water, etc.) (e.g., Parsons et al. 1994; Totland and Nylehn 1998; Arft et al. 1999; Kudo and Suzuki 2003; Danby and Hik 2007; Yin et al. 2008).
Specific leaf area nor leaf C, N, or C/N ratio were affected in both treatments. Earlier studies found both increased (Loveys et al. 2002; Yin et al. 2008) and stable SLA (Suzuki and Kudo 1997; Lemmens et al. 2006) for different plant species following warming manipulations (for an overview, see Poorter et al. 2009). Despite the fact that individuals grew taller (see above), TADM (plot level) was not affected in both treatments as was the case in sub-arctic tundra vegetation warmed with OTCs in Abisko (Sweden) (Parsons et al. 1994). This might be caused by size-density compensation (Totland and Nylehn 1998).
To conclude: the vernal forest geophyte A. nemorosa showed a phenotypic response in certain plant traits to the rise in temperature before canopy flush. However, in this study we did not investigate a later flowering species. There is also a potential to use this experimental warming method to study later flowering species, but this needs further research as temperature differences decreased after canopy flush and hence OTCs−GF comprehend only a ‘pre-canopy flush’ warming treatment.
Biotic drawbacks of OTCs
Since A. nemorosa flowers are mostly self-incompatible and pollinated by hive bees (Apis mellifera L.) and other small insects (Shirreffs 1985), the GF imposed a near-impossibility of pollination of flowers. The only possible ways of pollination in the OTCs+GF were occasional (1) selfing, (2) wind cross-pollination, or (3) insect cross-pollination. Probably because of this phenomenon, plants grown in the OTCs+GF had a lower germination percentage.
The design of the present study makes it difficult to distinguish the effect of selfing from warming on regeneration processes. Richardson et al. (2000) studied the presence of Hemiptera inside and outside OTCs in a subarctic tundra ecosystem in Abisko (Sweden) and reported a significant barrier effect through which the density of Hemiptera was reduced. They also suggest that insects present on plants in the chamber may prolong their stay relative to insects on plants outside chambers. If this would also be the case in our study, and given the fact that genetic variation within A. nemorosa is small (Stehlik and Holderegger 2000) on the spatial scale of our plots (~1 m2), pollination by insects in our OTCs is presumably within genetically related individuals. It is generally accepted that self-fertilization in (mostly) self-incompatible plant populations causes an inbreeding depression (in e.g., germination percentage) in comparison to outcrossing (Brys et al. 2003; Busch 2005). Open-top chambers with GF totally exclude pollinators and herbivores. Hence, the lower germination percentage in the OTC+GF plots (mostly selfed individuals) is an indication that this might have happened. The lower germination percentage in the OTCs−GF may therefore also be, at least partly, caused by self-fertilization effects, which hampers distinguishing of direct warming effects. Since A. nemorosa’s seed production and germination growing along a 2,400-km latitudinal gradient from N-France to N-Sweden showed a strong positive response to increased temperature (De Frenne et al. 2009), the results obtained from the present OTC study will probably be biased by this selfing problem. Hence, quantifying this selfing effect inside open-top chambers—not only in forests but also in e.g., tundra (Richardson et al. 2000)—should be the scope of future research.
Abiotic drawbacks of OTCs
Open-top chambers seem very promising and useful in studying warming effects on early flowering herbaceous understory plants in temperate deciduous forests. Although the OTCs covered with GF appeared to be more effective in manipulating temperature than the open OTCs, we prefer the latter for two main reasons: (1) temperature increase was significant and realistic, and (2) abiotic and biotic drawbacks are smaller in OTCs−GF than in OTCs+GF.
-
1.
The open OTCs increased mean monthly temperature up to +1.5°C in the leafless period, which is during shoot emergence and vegetative growth of early flowering forest herbs. The IPCC (2007) also predicts a higher median temperature rise in December–January–February (+4.3°C) than in June–July–August (+2.7°C) for northern Europe by 2080–2099 compared to 1980–1999.
-
2.
The open OTCs changed other abiotic conditions (RH, CO2 concentration, PAR amount, soil moisture, precipitation, etc.) only to a small extent. Relative air humidity was increased with only 1.2% (continuous measurements 2009) or 10.6% (discrete daytime values 2008) in the OTCs−GF plots. A change of RH has a significant influence on plant growth and development (Leuschner 2002; Lendzion and Leuschner 2007, 2008). In the case of the OTCs−GF, however, RH elevations are limited, what makes us suppose that forest herbs are not or only marginally influenced, also since these plant species are already adapted to humid environments and small fluctuations in humidity under natural conditions (Leuschner 2002; Lendzion and Leuschner 2007). In the OTC+GF, however, RH was increased with 31.0% (discrete daytime values, 2008) adding a major drawback to this type of OTCs. Other abiotic variables (PAR, CO2 concentration, and soil moisture) showed no or only small differences between the OTCs−GF and controls.
Are OTCs useful warming devices in forest ecosystems?
A certain warming device may be considered ‘useful’ when it fulfills the following criteria: (1) it increases temperatures adequately and in a realistic manner, and (2) ecological abiotic and biotic drawbacks of the warming device are limited. According to our opinion, both criteria are fulfilled in case of the OTCs−GF. Furthermore, A. nemorosa responded clearly to installation of both OTC types. Hence, we recommend the use of OTCs−GF to gain insight into the response of early flowering forest herbs to global warming—if done so with caution and taking the ecological drawbacks into consideration, especially in connection with reproduction. In this way, open-top chambers can become a valuable tool for predicting the potential response of particularly early flowering forest herbs to climate warming in temperate forest ecosystems. There is also a potential to use this method for later flowering species, but this needs further research.
References
Arft AM, Walker MD, Gurevitch J, Alatalo JM, Bret-Harte MS, Dale M, Diemer M, Gugerli F, Henry GHR, Jones MH, Hollister RD, Jonsdottir IS, Laine K, Levesque E, Marion GM, Molau U, Molgaard P, Nordenhall U, Raszhivin V, Robinson CH, Starr G, Stenstrom A, Stenstrom M, Totland O, Turner PL, Walker LJ, Webber PJ, Welker JM, Wookey PA (1999) Responses of tundra plants to experimental warming: meta-analysis of the International Tundra Experiment. Ecol Monogr 69:491–511
Baskin CC, Baskin JM (1998) Seeds. Ecology, biogeography, and evolution of dormancy and germination. Academic Press, San Diego
Brunet J, Von Oheimb G (1998) Colonization of secondary woodlands by Anemone nemorosa. Nord J Bot 18:369–377
Brys R, Jacquemyn H, Endels P, Hermy M, De Blust G (2003) The relationship between reproductive success and demographic structure in remnant populations of Primula veris. Acta Oecol 24:247–253
Busch JW (2005) Inbreeding depression in self-incompatible and self-compatible populations of Leavenworthia alabamica. Heredity 94:159–165
Danby RK, Hik DS (2007) Responses of white spruce (Picea glauca) to experimental warming at a subarctic alpine treeline. Global Change Biol 13:437–451
Daws MI, Burslem DFRP, Crabtree LM, Kirkman P, Mullins CE, Dalling JW (2002) Differences in seed germination responses may promote coexistence of four sympatric Piper species. Funct Ecol 16:258–267
De Frenne P, Graae BJ, Kolb A, Brunet J, Chabrerie O, Cousins SA, Decocq G, Dhondt R, Diekmann M, Eriksson O, Heinken T, Hermy M, Jõgar Ü, Saguez R, Shevtsova A, Stanton S, Zindel R, Zobel M, Verheyen K (2009) Significant effects of temperature on the reproductive output of the forest herb Anemone nemorosa L. Forest Ecol Manag doi:10.1016/j.foreco.2009.04.038 (in press)
Delatte E, Chabrerie O (2008) Seed dispersal efficiency of forest herbaceous plant species by the ant Myrmica ruginodis. CR Biol 331:309–320
Dunne JA, Harte J, Taylor KJ (2003) Subalpine meadow flowering phenology responses to climate change: integrating experimental and gradient methods. Ecol Monogr 73:69–86
Eriksson O (1995) Seedling recruitment in deciduous forest herbs—the effects of litter, soil chemistry and seed bank. Flora 190:65–70
FAO (2005) NewLocClim, version 1.10. Food and Agriculture Organization of the United Nations, Rome
Farnsworth EJ, Nunez-Farfan J, Careaga SA, Bazzaz FA (1995) Phenology and growth of three temperate forest life forms in response to artificial soil warming. J Ecol 83:967–977
Fitter AH, Fitter RSR, Harris ITB, Williamson MH (1995) Relationships between 1st flowering date and temperature in the flora of a locality in Central England. Funct Ecol 9:55–60
Gilliam FS (2007) The ecological significance of the herbaceous layer in temperate forest ecosystems. Bioscience 57:845–858
Hadfield KA, Bennett AB (1997) Programmed senescence of plant organs. Cell Death Differ 4:662–670
Hedges LV, Gurevitch J, Curtis PS (1999) The meta-analysis of response ratios in experimental ecology. Ecology 80:1150–1156
Hermy M, Verheyen K (2007) Legacies of the past in the present-day forest biodiversity: a review of past land-use effects on forest plant species composition and diversity. Ecol Res 22:361–371
Hollister RD, Webber PJ (2000) Biotic validation of small open-top chambers in a tundra ecosystem. Global Change Biol 6:835–842
Honnay O, Verheyen K, Butaye J, Jacquemyn H, Bossuyt B, Hermy M (2002) Possible effects of habitat fragmentation and climate change on the range of forest plant species. Ecol Lett 5:525–530
Hovenden MJ, Wills KE, Chaplin RE, Schoor JKV, Williams AL, Osanai Y, Newton PCD (2008) Warming and elevated CO2 affect the relationship between seed mass, germinability and seedling growth in Austrodanthonia caespitosa, a dominant Australian grass. Global Change Biol 14:1633–1641
IPCC (2007) Climate change: the physical science basis. Cambridge University Press, Cambridge
ISS-ISRIC-FAO (1998) World reference base for soil resources, world soil resources report 84. Food and Agriculture Organization of the United Nations, Rome
Jochum GM, Mudge KW, Thomas RB (2007) Elevated temperatures increase leaf senescence and root secondary metabolite concentrations in the understory herb Panax quinquefolius (Araliaceae). Am J Bot 94:819–826
Kanerva T, Regina K, Ramo K, Karhu K, Ojanpera K, Manninen S (2005) Mesocosms mimic natural meadows as regards greenhouse gas fluxes and potential activities of nitrifying and denitrifying Bacteria. Plant Soil 276:287–299
Kimball BA (2005) Theory and performance of an infrared heater for ecosystem warming. Global Change Biol 11:2041–2056
Kudernatsch T, Fischer A, Bernhardt-Romermann M, Abs C (2008) Short-term effects of temperature enhancement on growth and reproduction of alpine grassland species. Basic Appl Ecol 9:263–274
Kudo G, Suzuki S (2003) Warming effects on growth, production, and vegetation structure of alpine shrubs: a five-year experiment in northern Japan. Oecologia 135:280–287
Lemmens CMHM, De Boeck HJ, Gielen B, Bossuyt H, Malchair S, Carnol M, Merckx R, Nijs I, Ceulemans R (2006) End-of-season effects of elevated temperature on ecophysiological processes of grassland species at different species richness levels. Environ Exp Bot 56:245–254
Lendzion J, Leuschner C (2007) Temperate woodland herbs are adapted to high air humidity. In: Abstracts of the 37th annual conference of the Ecological Society of Germany, Austria and Switzerland (GFÖ) 37:90
Lendzion J, Leuschner C (2008) Growth of European beech (Fagus sylvatica L.) saplings is limited by elevated atmospheric vapour pressure deficits. Forest Ecol Manage 256:648–655
Leuschner C (2002) Air humidity as an ecological factor for woodland herbs: leaf water status, nutrient uptake, leaf anatomy, and productivity of eight species grown at low or high VPD levels. Flora 197:262–274
Loveys BR, Scheurwater I, Pons TL, Fitter AH, Atkin OK (2002) Growth temperature influences the underlying components of Relative Growth Rate: an investigation using inherently fast- and slow-growing plant species. Plant Cell Environ 25:975–987
Malcolm JR, Markham A, Neilson RP, Garaci M (2002) Estimated migration rates under scenarios of global climate change. J Biogeogr 29:835–849
Marion GM, Henry GHR, Freckman DW, Johnstone J, Jones G, Jones MH, Levesque E, Molau U, Molgaard P, Parsons AN, Svoboda J, Virginia RA (1997) Open-top designs for manipulating field temperature in high-latitude ecosystems. Global Change Biol 3:20–32
Mondoni A, Probert R, Rossi G, Hay F, Bonomi C (2008) Habitat-correlated seed germination behaviour in populations of wood anemone (Anemone nemorosa L.) from northern Italy. Seed Sci Res 18:213–222
Müller N, Schneller JJ, Holderegger R (2000) Variation in breeding system among populations of the common woodland herb Anemone nemorosa (Ranunculaceae). Plant Syst Evol 221:69–76
Musil CF, Schmiedel U, Midgley GF (2005) Lethal effects of experimental warming approximating a future climate scenario on Southern African quartz-field succulents: a pilot study. New Phytol 165:539–547
Netten JJC et al (2008) Use of open-top chambers to study the effect of climate change in aquatic ecosystems. Limnol Oceanog Meth 6:223–229
Parmesan C, Yohe G (2003) A globally coherent fingerprint of climate change impacts across natural systems. Nature 421:37–42
Parsons AN, Welker JM, Wookey PA, Press MC, Callaghan TV, Lee JA (1994) Growth responses of 4 sub-arctic dwarf shrubs to simulated environmental change. J Ecol 82:307–318
Philipp M, Petersen PM (2007) Long-term study of dry matter allocation and rhizome growth in Anemone nemorosa. Plant Spec Biol 22:23–31
Poorter H, Niinemets Ü, Poorter L, Wright IJ, Villar R (2009) Causes and consequences of variation in leaf mass per area (LMA): a meta-analysis. New Phytol 182:565–588
Richardson SJ, Hartley SE, Press MC (2000) Climate warming experiments: are tents a potential barrier to interpretation? Ecol Entomol 25:367–370
Rubinstein B (2000) Regulation of cell death in flower petals. Plant Mol Biol 44:303–318
Sherry RA, Zhou XH, Gu SL, Arnone JA, Schimel DS, Verburg PS, Wallace LL, Luo YQ (2007) Divergence of reproductive phenology under climate warming. Proc Natl Acad Sci USA 104:198–202
Shirreffs DA (1985) Anemone nemorosa L. J Ecol 73:1005–1020
Staelens J, De Schrijver A, Verheyen K, Verhoest NEC (2006) Spatial variability and temporal stability of throughfall water under a dominant beech (Fagus sylvatica L.) tree in relationship to canopy cover. J Hydrol 330:651–662
Stehlik I, Holderegger R (2000) Spatial genetic structure and clonal diversity of Anemone nemorosa in late successional deciduous woodlands of Central Europe. J Ecol 88:424–435
Suzuki S, Kudo G (1997) Short-term effects of simulated environmental change on phenology, leaf traits, and shoot growth of alpine plants on a temperate mountain, northern Japan. Global Change Biol 3:108–115
Svenning JC, Skov F (2006) Potential impact of climate change on the northern nemoral forest herb flora of Europe. Biodivers Conserv 15:3341–3356
Totland O (1999) Effects of temperature on performance and phenotypic selection on plant traits in alpine Ranunculus acris. Oecologia 120:242–251
Totland O, Nylehn J (1998) Assessment of the effects of environmental change on the performance and density of Bistorta vivipara: the use of multivariate analysis and experimental manipulation. J Ecol 86:989–998
Tyler G (2001) Relationships between climate and flowering of eight herbs in a Swedish deciduous forest. Ann Bot Lond 87:623–630
Verheyen K, Honnay O, Motzkin G, Hermy M, Foster DR (2003) Response of forest plant species to land-use change: a life-history trait-based approach. J Ecol 91:563–577
Yan W, Wallace DH (1996) A model of photoperiod x temperature interaction effects on plant development. Crit Rev Plant Sci 15:63–96
Yin HJ, Liu Q, Lai T (2008) Warming effects on growth and physiology in the seedlings of the two conifers Picea asperata and Abies faxoniana under two contrasting light conditions. Ecol Res 23:459–469
Acknowledgments
We thank the Kempe Foundation for funding parts of this project. Luc Willems and Greet De bruyn are gratefully acknowledged for their field and laboratory assistance. This paper was written while PDF and ADS held a PhD and postdoctoral fellowship, respectively, from the Research Foundation—Flanders (FWO). WT held a PhD scholarship of the Institute for the Promotion of Innovation through Science and Technology in Flanders (IWT).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
De Frenne, P., De Schrijver, A., Graae, B.J. et al. The use of open-top chambers in forests for evaluating warming effects on herbaceous understorey plants. Ecol Res 25, 163–171 (2010). https://doi.org/10.1007/s11284-009-0640-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-009-0640-3