Abstract
The ZRT, IRT-like protein (ZIP) family plays an important role in the transport of zinc (Zn) and iron (Fe) across the cell membrane in many different species. However, studies on ZIP family are mainly limited in herbaceous species; hence, we investigated functional divergence of ZIP family in Populus trichocarpa. We identified 21 ZIP genes in P. trichocarpa and classified them into four groups based on phylogenetic analysis. Structural analyses revealed that most of the PtrZIP transporters have eight transmembrane domains (TMDs). PtrZIP members were unequally positioned in 19 P. trichocarpa linkage groups (LGs), with six tandem duplications and four segmental duplications. The promoter regions of PtrZIP genes contain Zn, Fe, copper (Cu), and other metal stress-related cis-elements. Additionally, tissue-specific expression of PtrZIP genes showed that most of them had relatively high expression levels in the root. Quantitative real-time RT-PCR (qRT-PCR) analysis revealed that the expression of PtrZIP genes were induced not only under deficiency or excess condition of Zn, Fe, Cu and manganese (Mn) but also under excess condition of cadmium (Cd) and lead (Pb) stress. These findings indicated that PtrZIP genes may have played potential roles in metal transporters. Genome-wide analysis of PtrZIP genes in P. trichocarpa provided more comprehensive insights on the structure and function of this gene family.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In plants, Zn, Fe, Cu, and Mn are micronutrients that are essential for a wide variety of physical processes, as they act as important cofactors for many enzymes and are components of transcription factors and other proteins (Puig et al. 2007). However, when present at high concentrations, along with nonessential metals such as Cd and Pb, essential metals can become extremely toxic, as they can cause oxidative damage or compete with other essential ions (Migeon et al. 2010). To maintain the balance of metal ions in the cell, plants have evolved regulatory mechanisms to ensure an adequate supply of essential nutrients and to tolerate heavy metal ion toxicity. One of most important mechanisms is the presence of metal transporters, which plays crucial roles in many aspects of the distribution of essential and toxic metals in plants (Migeon et al. 2010). In previous studies, many metal transporters have been identified in plants, including natural resistance-associated macrophage protein (NRAMP), cation diffusion facilitator (CDF), copper transporter (CTR), cation exchanger (CAX) and ZRT, IRT-like proteins (ZIP) (Colangelo and Guerinot 2006).
The divalent metal ZIP transporters are named for their sequence similarity to the Zn-regulated transporters (ZRT) and Fe-regulated transporters (IRT) from yeast (Eide et al. 1996; Zhao and Eide 1996a, 1996b). Large numbers of ZIP genes have been researched from virtually most of herbaceous plant, so far, there are 18, 16, 16, seven and nine members in Arabidopsis thaliana, Oryza sativa, Hordeum vulgare, Medicago truncatula, and Zea mays, respectively (Migeon et al. 2010; Tiong et al. 2015; Stephens et al. 2011; Mondal et al. 2014). For lower plant, 14, 4, 11, and 8 ZIP members are present in Chlamydomonas reinhardtii, Cyanidioschyzon merolae, Physcomitrella patens, and Selaginella moelendorfii, respectively (Ivanov and Bauer 2016; Hanikenne et al. 2005). The family is classified into four subfamilies, I, II, III, and IV on the basis of structural similarity (Gaither and Eide 2001). Most ZIP transporters contain eight transmembrane domains (TMDs) and have similar membrane topology. A variable region between TMDs III and IV contains histidine residues that bind divalent metal substrates and promote their transport (Eide et al. 1996; Zhao and Eide 1996a, 1996b).
The transport of Zn, Fe, Mn, Cu, and Cd by ZIP gene family members has been demonstrated in plants (Fu et al. 2017; Guerinot 2000; Wintz et al. 2003). AtIRT1 of A. thaliana is the first identified through functional complementation of a yeast mutant defective and transports Fe, Zn, Cd, and Mn (Eide et al. 1996; Fu et al. 2017; Wintz et al. 2003). AtIRT2 has been well studied due to its role in Fe uptake and transport in roots (Vert et al. 2009). Overexpressing of AtIRT3 increases the accumulation of Zn and Fe in shoots and roots, respectively. Moreover, AtIRT3 can complement Zn and Fe uptake-deficient yeast mutants (Lin et al. 2009). In A. thaliana, AtZIP5, AtZIP9, and AtZIP12 are expressed in roots in response to Zn deficiency (Kramer et al. 2007). OsZIP1 and OsZIP3 from rice can complement Zn-uptake deficient yeast mutant ZHY3. Additionally, OsZIP1, OsZIP2, OsZIP3, OsZIP4, OsZIP5, and OsZIP8 are upregulated by Zn deficiency (Ishimaru et al. 2005; Lee et al. 2010a, 2010b; Ramesh et al. 2003;). Overexpression of maize ZmIRT1 and ZmZIP3 enhances Fe and Zn accumulation in transgenic Arabidopsis (Li et al. 2015). Lin reported that expression of the ZNT1 of Noccaea caerulescens confers enhanced Zn and Cd tolerance and accumulation to A. thaliana (Lin et al. 2016). In woody trees, the ZIP transporter of Chengiopanax sciadophylloides takes part in Mn hyperaccumulation in the roots (Mizuno et al. 2008). In trifoliate orange, PtIRT1, PtZIP1, PtZIP2, PtZIP3, and PtZIP12 take part in Zn transport, PtIRT1 and PtZIP7 were Fe uptake transporter, and PtIRT1 was able to absorb Mn, suggesting their respective functions in Zn, Fe, and Mn transport (Fu et al. 2017).
As a model plant, Populus trichocarpa is a fast-growing forestry resource, making it an economically and ecologically important species (Taylor 2002). Although the ZIP genes have been studied extensively in herbaceous species, little research has been conducted in Populus, particularly on systematic analysis of the P. trichocarpa ZIP genes. In the present study, comprehensive analysis of the P. trichocarpa ZIP gene family was performed, and its phylogeny, chromosomal distribution, cis-element content, regulated miRNAs, gene ontology, and expression in response to metal stress are described. Our results provide a better understanding of the functions of the PtrZIP family members and the mechanisms of micronutrient uptake and heavy metal elimination in Populus.
Materials and Methods
Identification and Phylogenetic Analysis of ZIP Family
The sequences of the ZIP transporters in P. trichocarpa, P. sitchensis, C. reinhardtii, and S. moelendorfii were obtained from UniProt KB (http://www.uniprot.org), Phytozome (http://phytozome.net), and NCBI (http://www.ncbi.nlm) databases. ZIP transporters in A. thaliana, maize, and rice were identified by searching the TAIR (http://www.arabidopsis.org), MaizeGDB (http://www.maizegdb.org), and Rice Genome Annotation Program (http://rice.plantbiology.msu.edu) respectively. The existence of the ZIP domain (Pfam: PF02535) was assessed by the SMART program (http://smart.embl-heidelberg.de). Various biochemical parameters (molecular weight, isoelectric point (pI), signal peptide, and grand average of hydropathy (GRAVY) values) were determined using the ExPaSy server (www.expasy.org). MEGA5.0 was used to construct a phylogenetic tree of ZIP transporters from five species using the Dayhoff model in neighbor-joining (NJ) method, with 1000 bootstrap iterations (Tamura et al. 2011).
Exon/Intron Structure Analysis and Identification of Conserved Motifs
The CDS sequences were obtained from Phytozome, and genome sequences were obtained from NCBI. The Gene Structure Display Server (GSDS) (http://gsds1.cbi.pku.edu.cn) was used to analyze the gene structure and produce a schematic of the PtrZIP genes (Zhang et al. 2015). The Multiple Expectation Maximization for Motif Elucidation (MEME) system (http://meme-suite.org/tools/meme) was used to detect the conserved motifs (Bailey et al. 2006). The TMHMM Server v. 2.0 (http://www.cbs.dtu.dk/services/TMHMM/) was used to characterize predicted putative TMD motifs (Krogh et al. 2001). Multiple sequence alignments of the full-length PtrZIP amino acid sequences were performed with the BioEdit 7.1 software, as in previous studies (Wang et al. 2016). Adobe Illustrator CS 6.0 was used for the manual addition of motifs.
Chromosomal, Subcellular Locations and GO Annotation
The chromosomal locations of PtrZIP genes were found using the PopGenIE v3 database (http://www.popgenie.org/) (Sjödin et al. 2009). Genes separated by 100 kb of sequence or less were inferred to be tandem duplications (Soler et al. 2015). Genes were considered to have undergone segmental duplication if they were located on duplicated chromosomal segments (Hu et al. 2010). The MCScanX software (http://chibba.pgml.uga.edu/mcscan2/) was used to search for potential duplicated PtrZIP genes (Wang et al. 2012). An exception was made for genes falling into duplicated blocks. The rates of non-synonymous (Ka) and synonymous (Ks) substitutions were calculated using DnaSP tools (http://www.ub.edu/dnasp/). Subcellular targeting sites were identified using WoLF PSORT (https://wolfpsort.hgc.jp/). Blast2GO v3.0 was used to show the functional grouping of PtrZIP transporters and for the analysis of annotation data (Conesa and Götz 2008).
Promoter cis-Element Prediction
For predicting putative cis-elements, 2000 bp of promoter sequence upstream of the PtrZIP genes’ transcriptional start was obtained from Phytozome 10.2 (Liu et al. 2015). The PLACE database (Zhang et al. 2015) (http://www.dna.affrc.go.jp/htdocs/PLACE) and Plant Care database (Liu et al. 2015) (http://bioinformatics.psb.ugent.be) were used to identify cis-elements in the PtrZIP promoters. Promoter sequence alignment and analysis was carried out using the multiple alignment algorithm in the DNAMAN 7.0 program (http://www.lynnon.com).
Tissue-Specific Expression Profiles from PopGenIE v3
Expression data for PtrZIP genes from leaves, roots, and nodes were acquired from microarray data in PopGenIE v3.0 (http://popgenie.org/) (Sjödin et al. 2009) and from the NCBI Gene Expression Omnibus (GEO accession number GSE6422) (Yang et al. 2008). The gene expression information was illustrated using Cluster 3.0.
Plant Materials and Metal Stress Treatments
Clonally propagated P. trichocarpa (genotype Nisqually-1) was cultured in half-strength MS medium containing 1.5% (w/v) Sucrose and 0.8% (w/v) agar at 25 °C under a 16-h light/8-h dark photoperiod under illumination at 45 μmol m−2 s−1 with cool white fluorescent lights. Plants were grown in 250-ml Erlenmeyer flasks containing 100 ml medium and were subcultured at 4-week intervals. To test the expression levels of ZIP genes under metal stress, 3-week-old in vitro plants were floated on half-strength MS liquid medium with 600 μM ZnSO4, 300 μM FeCl3, 1.0 mM MnSO4, 20 μM CuSO4, 80 μM CdCl2, or 150 μM Pb(NO3)2, respectively. In a preliminary experiment, we evaluated the effect of different concentrations of metals on growth of in vitro plants. The results showed that the in vitro plants could significantly reduce but not abolish growth under above metal concentrations within 4 weeks. After 0, 1, 6, and 16 h of treatment, root samples were detached and collected. To induce Zn, Fe, Mn, or Cu deficiency, 3-week-old plants were grown in a Zn, Fe, Mn, or Cu-deprived half-strength MS liquid medium for 1 week. Three biological replicates were performed for each stress treatment. Each experiment was repeated at least three times independently. All samples were immediately frozen in liquid nitrogen and stored at −80 °C until analysis.
RNA Isolation and Real-Time Quantitative PCR
Total RNA was extracted using the cetyltrimethylammonium bromide (CTAB) method from roots of P. trichocarpa (Chang et al. 1993). Complementary DNA (cDNA) was obtained by reverse transcription using the TransScript™ II One-Step gDNA Removal and cDNA Synthesis SuperMix (TransGen Biotech, China). TransStart® Top Green qPCR SuperMix (TransGen Biotech, China) was used for qRT-PCR. The relative quantification method (2−ΔΔCT) was used to evaluate quantitative variation between replicates. Each independent sample was analyzed at least three times. The actin gene (XM_002298674) was used as the reference gene and it was showed to have stable expression in tissues of P. trichocarpa (Liu et al. 2015). Other details were described previously (Zhang et al. 2015). The PtrZIP gene-specific primers were designed in non-conserved regions using Primer Premier 5.0 and are presented in Supplementary Table S6.
Statistical Analysis
Data were analyzed using one-way ANOVA followed by Tukey’s test. Significance was defined as *P < 0.05, **P < 0.01.
Results and Discussion
Identification and Phylogenetic Analysis of P. trichocarpa ZIP Genes
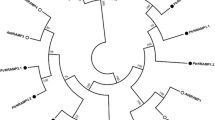
A total of 21 ZIP transporters containing ZIP domains were identified in P. trichocarpa, and these genes were named PtrZIP1.1–13.2 based on Phytozome (www.phytozome.net) (Table 1). Our paper shows one more gene which previous researches have not studied before (Migeon et al. 2010). The identified PtrZIP genes encoded proteins varying in length from 202 to 605 amino acids (AA), with an average length of 540 AA. The isoelectric points of PtrZIPs ranged from 5.68 to 9.07, and most of them were neutral. To examine the phylogenetic relationships, an unrooted phylogenetic tree was constructed from alignments of the full-length ZIP transporters from P. trichocarpa, A. thaliana, Z. mays, O. sativa, P. sitchensis, C. reinhardtii, and S. moelendorfii (Fig. 1). We obtained 18 ZIPs in A. thaliana (AtZIP1–14, AtIRT1–3, and AtIAR1), 10 ZIPs in Z. mays (ZmZIP1–9 and ZmIRT1), 17 ZIPs in O. sativa (OsZIP1–15 and OsIRT1–2), seven ZIPs in P. sitchensis (PsZIP1–7), 14 ZIPs in C. reinhardtii (CrZIP1–14 and CrZRT1–5), and eight ZIPs in S. moelendorfii (SmZIP1–7 and SmIAR1) (Supplementary Table S1). Sequences identified were confirmed in the PFAM database based on ZIP transmembrane domain. The ZIPs of A. thaliana and O. sativa were named by NCBI. The ZIPs of Z. mays, P. sitchensis, C. reinhardtii, and S. moelendorfii were named by previous researches (Ivanov and Bauer 2016; Mondal et al. 2014). According to the previous classification of A. thaliana (Migeon et al. 2010), we classified the 95 ZIP genes into classes I, II, III, and IV, and these four groups contained 56, 12, 7, and 20 members, respectively (Fig. 1). The four subfamilies were mixed plant groups. Among them, class I was the largest subgroup and contained most of woody and herbaceous species. The algal proteins of class I tended to cluster separately and this phenomenon also observed in previous studies (Hanikenne et al. 2005; Ivanov and Bauer 2016). It is interpreted as an indication of the independent expansion of the algal and other ZIP families. Class II consisted of fern, gymnosperm, woody, and herbaceous proteins. The phylogenetic tree also showed that the PtrZIPs were closely related to ZIPs from other plant species and they were orthologous to genes from other plant species (PtrZIP2, SmZIP2, PsZIP2, and AtZIP2; PtrZIP3, OsZIP3, ZmZIP3, and PsZIP3; PtrZIP6.2, PtrZIP6.3, AtZIP6, OsZIP6, ZmZIP6, and PsZIP6; PtrZIP9.1, PtrZIP9.2, ZmZIP9, and OsZIP9; PtrZIP11.1, PtrZIP11.2, and AtZIP11; PtrZIP13.1, PtrZIP13.2, and OsZIP13), while other orthologous genes were found (AtZIP1 and SmZIP1; AtIAR1 and SmIAR1; ZmZIP2 and OsZIP2; ZmIRT1 and OsIRT1; PsZIP1 and OsZIP1), indicating that these ZIPs may share a common evolutionary ancestor.
Phylogenetic analysis of the ZIP family members from various plant species. The phylogenetic tree shows four different clusters of ZIP transporters from various plant species inferred by the neighbor-joining (NJ) method of MEGA 5.0 with 1000 bootstrap replicates. The four plant-specific clusters were designated as I, II, III, and IV and are distinguished by color. The Populus trichocarpa ZIP proteins are written in a bold blue font to distinguish them from the other ZIP proteins (color figure online)
Gene Structure, Conserved Protein Motifs, and Structural Analysis
We compared the exon/intron organization in the full-length cDNAs and protein structures of each PtrZIP gene. To clarify the results, we first constructed a phylogenetic tree using the Populus protein sequences, which was in accordance with the phylogenetic tree mentioned above (Fig. 2a). Additionally, based on the phylogenetic analysis, we identified 10 sister pairs with strong bootstrap support for chromosomal location study (Supplementary Table S2). The coding sequences and corresponding genomic DNA sequences of PtrZIP genes were used to analyze the exon/intron organization, and the results are shown in Fig. 2b. The PtrZIP transporters in class I and class II shared a similar number of introns, ranging from one to three. However, in class III and class IV, the PtrZIP transporters had four to 11 introns. A joint analysis of the gene structures and phylogenetic trees indicated that the genes between classes I–II and classes III–IV have undergone a series of evolutionary events leading to intron insertions and thus may have different functions. The MEME tool was used for motif prediction, which resulted in the identification of 15 distinct motifs (Fig. 2c; Supplementary Table S3). PtrZIPs in class I and class II contained the highest number of motifs, while those in class III and class IV only contained seven motifs (Fig. 2c). Unique motifs were found in different subgroups, such as motifs 3, 5, and 7 in class I and motifs 13 and 15 in class II. These results indicated that most closely related members shared similar exon/intron structures and similar protein motifs and different subgroups have different exon/intron structures. The prediction of different structures indicated that these genes may have divergent functions. All PtrZIPs possessed from 3 to 13 putative TMDs (Table 1). The multiple sequence alignment of PtrZIP protein sequences is shown in Fig. 3 and illustrates the histidine-rich variable region that exists in most PtrZIP transporters between TMD III and TMD IV (Fig. 3). A well-conserved histidyl residue was identified in the AtIRT1, AtZRT1, and AtZRT2 proteins and suggested that this structure was a potential metal-binding domain (Eide et al. 1996; Gaither and Eide 2001; Mondal et al. 2014; Zhao and Eide 1996a, b) (Fig. 3). This result suggested that most PtrZIP transporters take part in metal transport via this metal-binding motif. Furthermore, two ZIP domains [LM] GIV [VS] HS VIIG [LVIM] SLG [AV] S and [SA] FH [QN] [VMLF] FEG [MIF] [GA] LGGCI were identified. The [LM] GIV [VS] HS VIIG [LVIM] SLG [AV] S motif was previously reported as a typical ZIP signature in rice (Pedas et al. 2008), maize (Mondal et al. 2014), and various other organisms (Eng et al. 1998). This typical motif was present in TMD IV of some class I PtrZIPs (PtrZIP1.1, PtrZIP1.2, PtrZIP3, PtrZIP4.4, and PtrZIP12), and domain mutants (one or two amino acid changes) were present in the remaining PtrZIPs in class I. The [SA] FH [QN] [VMLF] FEG [MIF] [GA] LGGCI motif has been reported as part of the maize ZIP structure in TMD V (Mondal et al. 2014). This motif was present in TMD V of all class I PtrZIPs. However, the two typical motifs were not present in class II, class III, and class IV. This result indicated that the class I PtrZIPs with both conserved motifs are functionally related to typical ZIPs from herbaceous plants. Class I PtrZIP motif mutants and class II–IV PtrZIPs may have functions that differ from ZIPs of herbaceous plants.
Phylogenetic tree, gene structure, and motif analysis for PtrZIP genes. An unrooted tree (a) was constructed from alignments of full-length PtrZIP sequences. Subgroup class I, class II, class III, and class IV are red, purple, green, and yellow, respectively. The exon–intron structures (b) were predicted based on sequences from the Phytozome database. The yellow boxes represent exons and the connecting lines indicate the introns. The PtrZIP genes motifs are represented in c. The motifs, numbered 1–15, are displayed in different colored boxes. The black lines represent the non-conserved sequences (color figure online)
Multiple alignments of the amino acid sequences deduced for all of PtrZIP transporters. The black lines indicate the predicted transmembrane (TMDI-VIII) regions. The red-colored bold box indicates the predicted domain (histidine-rich residues), two yellow-colored boxes indicate the structures [LM] GIV [VS] HS VIIG [LVIM] SLG [AV] S and [SA] FH [QN] [VMLF] FEG [MIF] [GA] LGGCI domains; some domains in brackets, for example, [LM] imply that the amino acid is L (Leucine) or M (methionine) (color figure online)
Chromosomal Location, Subcellular Location, and GO Annotation
The chromosomal locations illustrating gene duplication and genetic divergence between the PtrZIP genes are shown in Fig. 4. The results revealed that all of PtrZIP genes are distributed across chromosomes I, III, VI, VIII, IX, X, XVI, XVII, and XVIII (Fig. 4; Table 1) and that most of these genes were the result of duplication events. At least three duplications (genome-wide, segmental and tandem duplications) occurred in the Populus genome (Tuskan et al. 2006). According to previous studies (Chen et al. 2014), six tandem duplications and four segmental duplications occurred in P. trichocarpa and these are supported by our research (Fig. 4; Supplementary Table S2). These results provided strong evidence that tandem and segmental duplication have played an important role in PtrZIP gene expansion. The Ka/Ks ratio was calculated to determine the type of selective pressure acting on the PtrZIP motifs (Li et al. 1981). For instance, two homolog sequences have undergone purifying selection if the Ka/Ks value is less than 1.0; they are drifting neutrally if the Ka/Ks value is equal to 1.0; and if the value of Ka/Ks is more than 1.0, that pair of genes are under positive selection (Juretic et al. 2005). A summary of Ka/Ks ratios for 10 pairs of PtrZIP genes is shown in Supplementary Table S2. All Ka/Ks values were found to be less than 1.0. This result proved that all pairs of PtZIP genes have developed primarily under the influence of purifying selection.
The chromosomal distribution of the PtrZIP genes. Distribution of PtrZIP genes on LGI–XIX chromosomes. Homologous genome blocks are indicated by the same color. Tandemly duplicated genes are noted in the yellow boxes. For paralogs on different chromosomes the segmentally duplicated gene line is yellow (color figure online)
WoLF PSORT was used to predict the location of the transporters in P. trichocarpa (Table 1). The result showed that 15 PtrZIPs were predicted as plasma membrane proteins (Table 1), and at last, four as vacuolar membrane proteins and two as chloroplast membrane proteins (Table 1). In prior studies, for transport metal ions, most of ZIPs were localized to the plasma membrane (Barberon et al. 2014; Zhao and Eide 1996a, 1996b; Pedas et al. 2008), and some of them were an intracellular membrane protein whose role in the cellular response to metal deficiency (Vert et al. 2009). The predicted location of PtZIPs was similar to the results from previous studies; moreover, the results will be verified through experiment in further study (Vert et al. 2009).
The gene ontology (GO) annotations of PtrZIP transporters from Blast2GO v3.0, which consolidates specialized knowledge to ensure comprehensive and up-to-date biological information, contain biological processes, molecular functions, and cellular components (Fig. 5; Supplementary Table S4). The results revealed that the 21 PtrZIP genes took part in various biological processes, including transmembrane transport and ion transport. Specifically, four PtrZIP genes were predicted to response to starvation and nutrient levels, three PtrZIPs took part in fluid transport, and one involved in the vernalization response. All PtrZIPs had GO molecular functions associated with ion transport. Specifically, 11 PtrZIPs were predicted to involve in oxidoreductase activity with NAD/NADP as an acceptor; further research is needed to illuminate the conclusion. The related GO cellular component analysis indicated that one PtrZIP was predicted to be in an organelle and another in the cell and that all of them were localized at the membrane (Fig. 5; Supplementary Table S4). These predictions are consistent with the subcellular localization analysis by WoLF PSORT. From Supplementary Table S4, it can be seen that members of the ZIP family mainly transport Zn and Fe and take part in Zn and Fe uptake.
Promoter cis-Element Analysis
Promoter cis-elements play key roles in the tissue-specific or stress-responsive transcriptional regulation of genes in plants (Le et al. 2012). Using the PLACE and Plant Care databases, the putative cis-acting regulatory DNA elements in the PtrZIP genes were identified (Table 2 and Supplementary Table S5). In addition to the basic elements (TATA-box and CAAT-box), most PtrZIPs had several cis-elements that were related to environmental stress signaling and phytohormone responses (Table 2 and Supplementary Table S5), such as the TC-rich (stress and defense responsive), ABRE (abscisic acid responsive), ERE (ethylene-responsive element), CGTCA and TGACG (methyl jasmonate responsive), and TCA (salicylic acid responsive) motifs. It is well known that phytohormones such as salicylic acid (SA), jasmonic acid (JA), ethylene (ET), methyl jasmonate (MeJA), and abscisic acid (ABA) enable plants to adapt to abiotic stress. Thus, it is possible that most of these genes respond to abiotic stress (Santner and Estelle 2009). Moreover, a previous study indicated that the TCA-element is SA responsive and that SA appears to be an effective therapeutic agent in plant responses to heavy metal stress (Cd, Hg, and Pb) (Rivas-San Vicente and Plasencia 2011); thus, it is possible that SA mediates most PtrZIP genes response to metal stress. A previous study found that during Fe deficiency, the E-box in the promoter of IRT1 and IRT2, which is involved in interactions with helix-loop-helix (bHLH) transcription factors FIT (FER-like iron deficiency induced transcription factor), together with AtbHLH38 and AtbHLH39, transcriptionally regulated the expression of IRT1 and IRT2 (Wang et al. 2013; Wu et al. 2012; Yuan et al. 2008). All of the PtrZIP promoters were highly enriched with E-boxes (Table 2), which may interact with bHLH-like type transcription factors in Populus and are probably responsive to Fe deficiency. In response to Zn deficiency, the basic region leucine zipper (bZIP) family transcription factors bZIP19 and bZIP23 regulated the expression of AtZIP4 by binding to a Zn deficiency response element (ZDRE, ATGTCGACAC/G/T) that was located in the promoter of AtZIP4 (Assuncao et al. 2010; Claus and Chavarría-Krauser 2012). Using the DNAMAN multiple sequence alignment program, ZDRE was also found in the promoters of PtrZIP4.4, PtrZIP5, and PtrZIP9.2 (Table 2). Accordingly, it is possible that these genes may be regulated by the bZIP family during Zn deficiency. In previous studies, the squamosa promoter-binding protein-like 7 (SPL7) transcription factor regulated the expression of AtCOPT1 and AtCOPT2 during Cu deficiency by interacting with the GTAC motif in the promoter of AtCOPT1 and AtCOPT2 (Sanceno et al. 2003), indicating that the GTAC motifs were essential and sufficient for the response to copper deficiency. In our study, most of PtrZIP genes have a GTAC region (Table 2); therefore, these genes may also take part in mediating Cu mobilization. These results showed that the promoter of PtrZIP4.4 and PtrZIP5 harbor three typical metal stress response elements (E-box, ZDRE, and GTAC), which suggested that the PtrZIP gene family plays a role in the transport of multiple metal ions and likely has important roles in response to metal stress. Thus, more work is needed to test empirically the putative cis-element, subcellular localization or function of protein motifs in P. trichocarpa.
Expression Profiles in Different Tissues/Organs
Usually a high or preferential expression in tissues or organs suggested that these genes may play an important role (Liu et al. 2013, 2014). To understand the physiological function of the PtrZIP transporters in different tissues, gene expression data for the 21 PtrZIP genes across different tissues and organs of P. trichocarpa was obtained from an Affymetrix microarray (NCBI GEO accession GSE6422) (Yang et al. 2008) and visualized using Cluster 3.0. The microarray revealed that the PtrZIP genes had different expression patterns in various tissues under nutritional surpluses (Fig. 6). The public microarray expression profiles available for Populus showed that class I members (PtrZIP4.1, PtrZIP4.4, PtrZIP10.1, PtrZIP10.2, PtrZIP12, and PtrZIP13.2) and PtrZIP2 belonging to class II were upregulated in the roots, class I PtrZIPs (PtrZIP3, PtrZIP5, and PtrZIP13.1) and class II PtrZIPs (PtrZIP11.1 and PtrZIP11.2) had higher expression levels in mature leaves, and only PtrZIP3 had high expression in young leaves. In addition, three genes, PtrZIP1.1, PtrZIP1.2, and PtrZIP6.2, belonging to class I, were upregulated in the nodes, and class I genes (PtrZIP6.2 and PtrZIP6.3) and a class II gene (PtrZIP11.1) were highly expressed in the internodes. Among the PtrZIP family, class I and class II PtrZIPs had highest expression levels in various tissues, while class III and class IV members showed very low expression in these tissues. Interestingly, the GO results and microarray analysis indicated that class I and class II PtrZIPs mainly take part in transportation and absorption of metal ions during nutritional surpluses. Class III and class IV PtrZIP gene expression may be induced for metal ion transport during stress. In the gene structure and conserved protein motifs analysis of PtrZIP family, class I and class II shared similar numbers of introns and motifs which are different from the similar structure in class III and class IV (Fig. 2), As a result, it is interpreted as an indication of the structural variation may contribute to the different functions of ZIP family. These results also show that under sufficient nutritional conditions, the PtrZIP genes are mainly expressed in the roots. However, in maize, the maximum expression of ZmZIP genes was found in leaves (Mondal et al. 2014), reflecting differences between herbaceous and woody plants.
A heatmap showing the organ-specific expressions of PtrZIP genes. Roots, nodes, mature leaves, internodes, and young leaves were examined. The expression profiles of PtrZIP genes were acquired from PopGenIE v3.0 and the NCBI Gene Expression Omnibus (GEO) database. Red and green boxes indicate high and low transcript levels, respectively. The gene classification was in keeping with the phylogenetic tree mentioned previously, and each gene is indicated in every graph (color figure online)
Transcript Profiles Under Heavy Metal Stresses
Recently, functional analysis revealed that most of the ZIP genes in A. thaliana, rice, and maize play important roles in transporting Zn or Fe in the roots (Carvalho et al. 2015; Lin et al. 2009; Milner et al. 2013). Additionally, other studies have shown that ZIP genes can transport Cu, Mn, and Cd (Mizuno et al. 2008; Wintz et al. 2003). During essential micronutrient deficits and heavy metal stress, the root is the first organ to respond to changes of soil metal ion content. Roots participate in the uptake and transport of metal ions; thus, it is a very important organ for plants under metal stress. To gain insight into the functions of PtrZIPs, the change in their expression levels following some essential micronutrients (Zn, Fe, Cu, and Mn) and non-essential metal stresses (Cd and Pb) in the roots were analyzed by qRT-PCR (Figs. 7, 8, and 9).
The expression profiles of PtrZIP genes in roots under Zn and Fe deficiency and excess conditions. qRT-PCR expression analysis was applied to determine the expression profiles of PtrZIP genes. The standard deviations were obtained from three replicates of each metal ion stress experiment. Asterisks indicate a significant difference (*P < 0.05, **P < 0.01) between the stress treatment groups and the control group
The expression profiles of PtrZIP genes in roots under Mn and Cu deficiency and excess conditions. qRT-PCR expression analysis was applied to determine the expression profiles of PtrZIP genes. The standard deviations were obtained from three replicates of each metal ion stress experiment. Asterisks indicate a significant difference (*P < 0.05, * P < 0.01) between the stress treatment groups and the control group
The expression profiles of PtrZIP genes in roots under excessive amounts of Cd and Pb. qRT-PCR was used to analyze the expression profiles of PtrZIP genes. The standard deviations were obtained from three replicates of each metal ion stress experiment. Asterisks indicate a significant difference (*P < 0.05, **P < 0.01) between the stress treatment group and the control group
In the root, the expression of class Ι PtrZIP (PtrZIP1.1 and PtrZIP5), class II PtrZIP11.2, and class IV PtrZIP (PtrZIP9.2 and PtrZIP13.2) genes were induced during Zn deficiency. At the same time, these genes were decreased or unchanged with a Zn excess (Fig. 7), which is similar to the expression patterns of AtZIP1 and AtZIP3 in the roots of A. thaliana in response to Zn deficiency (Guerinot 2000). Furthermore, these PtrZIPs were predicted to be localized at the plasma membrane, which is also similar to A. thaliana IRT3 (Lin et al. 2009), suggesting that these genes may have the same function and can transport Zn from the soil into the cell. Among the PtrZIPs, PtrZIP5 is strongly induced with a shortage of Zn. Considering the presence of the ZDRE motif in its promoter, this gene may participate in Zn regulation downstream of bZIP in Populus. It is very similar to A. thaliana ZIP4 (Assuncao et al. 2010). The levels of PtrZIP5, PtrZIP7.1, PtrZIP8, PtrZIP11.2, and PtrZIP13.2 were upregulated in the root when Fe was limited and were unchanged when Fe was excessive (Fig. 7), indicating that these transporters might participate in Fe transport. Additionally, under Fe deficiency, Arabidopsis bHLH transcription factor FIT activates IRT1 through binding to the general bHLH transcription factor regulated motif E-box that is present in the promoter IRT1 (Yuan et al. 2008; Lemerciera et al. 1997). Previous studies found that five orthologs of AtbHLH were identified in P. trichocarpa and one of them, PtFIT (bHLH genes), was induced by Fe deficiency (Huang and Dai 2015). There were abundant E-box motifs in the promoters of PtrZIP genes, suggesting that these genes might be activated by bHLH transcription factor family member PtFIT, which helped the P. trichocarpa adapt to low Fe availability. PtrZIP4.4 and PtrZIP6.3 transcript levels were elevated in the roots (Fig. 8) during Mn deficiency, and these genes showed a decrease in transcript levels with excessive Mn, demonstrating that these transporters participate in responses to Mn availability. Furthermore, similar to IRT1 of A. thaliana, these PtrZIPs were predicted to be localized to the plasma membrane, which functions in Mn uptake and transport in the plasma membrane during Mn deficiency (Mizuno et al. 2008). In a previous study, AtIRT1 was reported to contain an important D100 amino acid residue that played an integral role in the selective transport of Fe and Mn (Lin et al. 2009). The alignment of the amino acid sequences of the AtIRT1 and PtrZIP genes that increased in response to Fe and Mn-deficiency is shown in Supplementary Fig. S1. Multiple sequence alignments identified the corresponding D100 residue in PtrZIP4.4, PtrZIP6.3, PtrZIP7.1, PtrZIP8, PtrZIP11.2, and PtrZIP13.2. Therefore, it is possible that these genes are more likely than other family members to transport Fe and Mn when the ion level is low. In the roots, the transcript levels of PtrZIP1.2 and PtrZIP3 were induced in the roots when Cu was limiting, and these genes were repressed after 16 h of exposure to excessive Cu (Fig. 8). This expression pattern is similar to AtZIP2 and AtZIP4, which are upregulated following Cu deficiency and downregulated in the presence of excess Cu (Guerinot 2000; Wintz et al. 2003), suggesting that PtrZIP1.2 and PtrZIP3 are involved in Cu transport. Additionally, there are some GTAC motifs (Sanceno et al. 2003) in the promoters of PtrZIP1.2 and PtrZIP3 genes, which indicated that these PtrZIP genes are regulated by SPL7 during Cu deficiency.
In terms of responses to non-essential heavy metals, the expression of PtrZIP3, PtrZIP4.1, PtrZIP9.1, and PtrZIP10.1 were induced in the root by Cd (Fig. 9), suggesting that PtrZIP genes might response to Cd stress. It was previously reported that IRT1 of A. thaliana can transport Cd during exposure to excessive Cd, and expression of the ZNT1 of N. caerulescens conferred enhanced Zn and Cd tolerance (Guerinot 2000; Lin et al. 2016). In our study, we also found that PtrZIPs might transport Cd. PtrZIP5, PtrZIP6.3, and PtrZIP11.2 genes were found to be induced by Pb exposure (Fig. 9). These results indicated that ZIPs may be functionally related to Pb transporters that have not been reported in herbaceous plants. Additionally, the structural analysis (Fig. 3) of class I PtrZIPs (PtrZIP5 and PtrZIP6.3) with mutations in the typical herbaceous ZIP motif ([LM] GIV [VS] HS VIIG [LVIM] SLG [AV] S) and class II PtrZIPs (PtrZIP11.2) that lacked both herbaceous ZIP motifs (Fig. 3) indicated that the structural variation may contribute to the different functions of ZIPs between herbaceous and woody plants. In addition, for the PtrZIP genes, the expression of a single gene was upregulated following a variety of heavy metal stresses. For example, PtrZIP5 was induced by Fe deficiency, Zn deficiency and excess Pb stress, which is similar to the responses of IRT1 for the transport of Zn, Fe, and Mn, as well as the toxic metal (Cd) (Guerinot 2000; Lin et al. 2009). Taken together, this indicates that the regulation of homeostasis of metal ions depends on numerous proteins interacting with each other. Further research is needed to illuminate the regulatory mechanism of PtrZIP transporters and their characteristics during metal stress.
Conclusions
In summary, the PtrZIP transporter family contains 21 candidate genes that are phylogenetically clustered into four distinct subfamilies. The predictions of subcellular localization showed that all of the members are membrane proteins. Microarray datasets showed that class I–II PtrZIPs had high expression levels in various tissues under sufficient nutritional conditions and are mainly expressed in roots, which differ from herbaceous plants. The expressions of PtrZIP1.1, PtrZIP1.2, PtrZIP3, PtrZIP4.4, PtrZIP5, PtrZIP6.3, PtrZIP7.1, PtrZIP8, PtrZIP9.2, and PtrZIP11.2 genes were induced by essential micronutrient (Cu, Fe, Zn, and Mn) deficiency, while those of PtrZIP3, PtrZIP4.1, PtrZIP9.1, and PtrZIP10.1 were induced by excess Cd stress, and those of PtrZIP5, PtrZIP6.3, and PtrZIP11.2 were induced by excess Pd stress. These results suggest that PtrZIPs may play important roles in balancing the concentrations of Zn, Fe, and other metals. This study sheds light on the nature of the ZIP gene family in P. trichocarpa. Also, it was used to produce Populus cultivars suitable for land with essential micronutrient deficiencies and toxic metal contamination.
References
Assuncao AG, Herrero E, Lin YF, Huettel B, Talukdar S, Smaczniak C, Immink RG, van Eldik M, Fiers M, Schat H et al (2010) Arabidopsis thaliana transcription factors bZIP19 and bZIP23 regulate the adaptation to zinc deficiency. Proc Natl Acad Sci U S A 107:10296–10301
Bailey TL, Williams N, Misleh C, Li WW (2006) MEME: discovering and analyzing DNA and protein sequence motifs. Nucleic Acids Res 34:369–373
Barberon M, Dubeaux G, Kolb C, Isono E, Zelazny E, Vert G (2014) Polarization of iron-regulated transporter 1 (IRT1) to the plant-soil interface plays crucial role in metal homeostasis. Proc Natl Acad Sci U S A 111:8293–8298
Carvalho S, Barreira da Silva R, Shawki A, Castro H, Lamy M, Eide D, Costa V, Mackenzie B, Tomás AM (2015) LiZIP3 is a cellular zinc transporter that mediates the tightly regulated import of zinc in Leishmania infantum parasites. Mol Microbiol 96:81–595
Chang SJ, Puryear J, Cairney J (1993) A simple and efficient method for isolating RNA from pine trees. Plant Mol Biol Rep 11:113–116
Chen X, Chen Z, Zhao HL, Zhao Y, Cheng BJ, Xiang Y (2014) Genome-wide analysis of soybean HD-Zip gene family and expression profiling under salinity and drought treatments. PLoS One 9:e87156
Claus J, Chavarría-Krauser A (2012) Modeling regulation of zinc uptake via ZIP transporters in yeast and plant roots. PLoS One 7:e37193
Colangelo EP, Guerinot ML (2006) Put the metal to the petal: metal uptake and transport throughout plants. Curr Opin Plant Biol 9:322–330
Conesa A, Götz S (2008) Blast2GO: a comprehensive suite for functional analysis in plant genomics. Int J Plant Genom 2008:619832
Eide D, Broderius M, Fett J, Guerinot ML (1996) A novel iron-regulated metal transporter from plants identified by functional expression in yeast. Proc Natl Acad Sci U S A 93:5624–5628
Eng BH, Guerinot ML, Eide D, Saier MH Jr (1998) Sequence analyses and phylogenetic characterization of the ZIP family of metal ion transport proteins. J Membr Biol 166:1–7
Fu XZ, Zhou X, Xing F, Ling LL, Chun CP, Cao L, Aarts MGM, Peng LZ (2017) Genome-wide identification, cloning and functional analysis of the zinc/iron-regulated transporter-like protein (ZIP) gene family in trifoliate orange (Poncirus trifoliata L. Raf.) Front Plant Sci 8:588
Gaither LA, Eide DJ (2001) Eukaryotic zinc transporters and their regulation. Biometals 14:251–270
Guerinot ML (2000) The ZIP family of metal transporters. Biochim Biophys Acta 1465:190–198
Hanikenne M, Krämer U, Demoulin V, Baurain D (2005) A comparative inventory of metal transporters in the green alga Chlamydomonas reinhardtii and the red alga Cyanidioschizon merolae. Plant Physiol 137:428–446
Hu R, Qi G, Kong Y, Kong D, Gao Q, Zhou G (2010) Comprehensive analysis of NAC domain transcription factor gene family in Populus trichocarpa. BMC Plant Biol 10:145
Huang D, Dai W (2015) Molecular characterization of the basic helix-loop-helix (bHLH) genes that are differentially expressed and induced by iron deficiency in Populus. Plant Cell Rep 34:1211–1224
Ishimaru Y, Suzuki M, Kobayashi T, Takahashi M, Nakanishi H, Mori S, Nishizawa NK (2005) OsZIP4, a novel zinc-regulated zinc transporter in rice. J Exp Bot 56:3207–3214
Ivanov R, Bauer P (2016) Sequence and coexpression analysis of iron-regulated ZIP transporter genes reveals crossing points between iron acquisition strategies in green algae and land plants. Plant Soil 2016:1–13
Juretic N, Hoen DR, Huynh ML, Harrison PM, Bureau TE (2005) The evolutionary fate of MULE-mediated duplications of host gene fragments in rice. Genome Res 15:1292–1297
Kramer U, Talke IN, Hanikenne M (2007) Transition metal transport. FEBS Let 581:2263–2272
Krogh A, Larsson B, Von Heijne G, Sonnhammer EL (2001) Predicting transmembrane protein topology with a hidden Markov model: application to complete genomes. J Mol Biol 305:567–580
Le DT, Nishiyama R, Watanabe Y, Vankova R, Tanaka M, Seki M, Ham LH, Yamaguchi-Shinozaki K, Shinozaki K, Tran LS (2012) Identification and expression analysis of cytokinin metabolic genes in soybean under normal and drought conditions in relation to cytokinin levels. PLoS One 7:e42411
Lee S, Jeong HJ, Kim SA, Lee J, Guerinot ML, An G (2010a) OsZIP5 is a plasma membrane zinc transporter in rice. Plant Mol Biol 73:507–517
Lee S, Kim SA, Lee J, Guerinot ML, An G (2010b) Zinc deficiency-inducible OsZIP8 encodes a plasma membrane-localized zinc transporter in rice. Mol Cells 29:551–558
Lemerciera C, Toa RQ, Swansonb BJ, Lyonsc GE, Koniecznya SF (1997) Mist1: a novel basic helix-loop-helix transcription factor exhibits a developmentally regulated expression pattern. Dev Biol 182:101–113
Li WH, Gojobori T, Nei M (1981) Pseudogenes as a paradigm of neutral evolution. Nature 292:237–239
Li SZ, Zhou XJ, Li HB, Liu YF, Zhu LY, Guo JJ, Liu XQ, Fan YL, Chen JT, Chen R (2015) Overexpression of ZmIRT1 and ZmZIP3 enhances iron and zinc accumulation in transgenic Arabidopsis. PLoS One 10:e0136647
Lin YF, Liang HM, Yang SY, Boch A, Clemens S, Chen CC, Wu JF, Huang JL, Yeh KC (2009) Arabidopsis IRT3 is a zinc-regulated and plasma membrane localized zinc/iron transporter. New Phytol 182:392–404
Lin YF, Zeshan H, Sangita T, Henk S, Aarts MGM (2016) Expression of the ZNT1 zinc transporter from the metal hyperaccumulator Noccaea caerulescens confers enhanced zinc and cadmium tolerance and accumulation to Arabidopsis thaliana. PLoS One 11:e0149750
Liu YK, Zhang D, Wang L, Li DQ (2013) Genome-wide analysis of mitogen-activated protein kinase gene family in maize. Plant Mol Biol Rep 31:1446–1460
Liu TK, Song XM, Duan WK, Huang ZN, Liu GF, Li Y, Hou XL (2014) Genome-wide analysis and expression patterns of NAC transcription factor family under different developmental stages and abiotic stresses in Chinese cabbage. Plant Mol Biol Rep 32:1041–1056
Liu QG, Wang ZC, Xu XM, Zhang HZ, Li CH (2015) Genome-wide analysis of C2H2 zinc-finger family transcription factors and their responses to abiotic stresses in poplar (Populus trichocarpa). PLoS One 10:e0134753
Migeon A, Blaudez D, Wilkins O, Montanini B, Campbell MM, Richaud P, Thomine S, Chalot M (2010) Genome-wide analysis of plant metal transporters, with an emphasis on poplar. Cell Mol Life Sci 67:3763–3784
Milner MJ, Seamon J, Craft E, Kochian LV (2013) Transport properties of members of the ZIP family in plants and their role in Zn and Mn homeostasis. J Exp Bot 64:369–381
Mizuno T, Hirano K, Kato S, Obata H (2008) Cloning of ZIP family metal transporter genes from the manganese hyperaccumulator plant Chengiopanax sciadophylloides, and its metal transport and resistance abilities in yeast. Soil Sci Plant Nutr 54:86–94
Mondal TK, Ganie SA, Rana MK, Sharma TR (2014) Genome-wide analysis of zinc transporter genes of maize (Zea mays). Plant Mol Biol Rep 32:605–616
Pedas P, Ytting CK, Fuglsang AT, Jahn TP, Schjoerring JK, Husted S (2008) Manganese efficiency in barley: identification and characterization of the metal ion transporter HvIRT1. Plant Physiol 148:455–466
Puig S, Andres-Colas N, Garcia-Molina A, Penarrubia L (2007) Copper and iron homeostasis in Arabidopsis: responses to metal deficiencies, interactions and biotechnological applications. Plant Cell Environ 30:271–290
Ramesh SA, Shin R, Eide DJ, Schachtman DP (2003) Differential metal selectivity and gene expression of two zinc transporters from rice. Plant Physiol 133:126–134
Rivas-San Vicente M, Plasencia J (2011) Salicylic acid beyond defence: its role in plant growth and development. J Exp Bot 62:3321–3338
Sanceno V, Puig S, Mira H, Thiele DJ, Penarrubia L (2003) Identification of a copper transporter family in Arabidopsis thaliana. Plant Mol Biol 51:577–587
Santner A, Estelle M (2009) Recent advances and emerging trends in plant hormone signaling. Nature 459:1071–1078
Sjödin A, Street NR, Sandberg G, Gustafsson P, Jansson S (2009) The Populus genome integrative explorer (PopGenIE): a new resource for exploring the Populus genome. New Phytol 182:1013–1025
Soler M, Camargo EL, Carocha V, Cassan-Wang H, San Clemente H, Savelli B, Hefer CA, Paiva JA, Myburg AA, Grima-Pettenati J (2015) The Eucalyptus grandis R2R3-MYB transcription factor family: evidence for woody growth-related evolution and function. New Phytol 206:1364–1377
Stephens BW, Cook DR, Grusak MA (2011) Characterization of zinc transport by divalent metal transporters of the ZIP family from the model legume Medicago truncatula. Biometals 24:51–58
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Taylor G (2002) Populus: arabidopsis for forestry. Do we need a model tree? Ann Bot 90:681–689
Tiong J, McDonald G, Genc Y, Shirley N, Langridge P, Huang CY (2015) Increased expression of six ZIP family genes by zinc (Zn) deficiency is associated with enhanced uptake and root-to-shoot translocation of Zn in barley (Hordeum vulgare). New Phytol 207:1097–1109
Tuskan GA, Difazio S, Jansson S, Bohlmann J, Grigoriev I, Hellsten U, Putnam N, Ralph S, Rombauts S, Salamov A et al (2006) The genome of black cottonwood, Populus trichocarpa (Torr. & Gray). Science 313:1596–1604
Vert G, Barberon M, Zelazny E, Seguela M, Briat JF, Curie C (2009) Arabidopsis IRT2 cooperates with the high-affinity iron uptake system to maintain iron homeostasis in root epidermal cells. Planta 229:1171–1179
Wang Y, Tang H, Debarry JD, Tan X, Li J, Wang X (2012) MCScanX: a toolkit for detection and evolutionary analysis of gene synteny and collinearity. Nucleic Acids Res 40:e49
Wang Z, Wu YL, Li LF, Su XD (2013) Intermolecular recognition revealed by the complex structure of human CLOCK-BMAL1 basic helix-loop-helix domains with E-box DNA. Cell Res 23:213–224
Wang MX, Yuan MQ, Fang Y (2016) Research on spatiotemporal spread of avian influenza A(H7N9) virus based on discrete geographic information. Article in Chinese 37:1491–1496
Wintz H, Fox T, Wu YY, Feng V, Chen W, Chang HS, Zhu T, Vulpe C (2003) Expression profiles of Arabidopsis thaliana in mineral deficiencies reveal novel transporters involved in metal homeostasis. J Biol Chem 278:47644–47653
Wu HL, Chen CL, Du J, Liu HF, Cui Y, Zhang Y, He YJ, Wang YQ, Chu CC, Feng ZY et al (2012) Co-overexpression FIT with AtbHLH38 or AtbHLH39 in Arabidopsis-enhanced cadmium tolerance via increased cadmium sequestration in roots and improved iron homeostasis of shoots. Plant Physiol 158:790–800
Yang X, Kalluri UC, Jawdy S, Gunter LE, Yin T, Tschaplinski TJ, Weston DJ, Ranjan P, Tuskan GA (2008) The F-box gene family is expanded in herbaceous annual plants relative to woody perennial plants. Plant Physiol 148:1189–1200
Yuan YX, Wu H, Wang N, Li J, Zhao WN, Du J, Wang DW, Ling HQ (2008) FIT interacts with AtbHLH38 and AtbHLH39 in regulating iron uptake gene expression for iron homeostasis in Arabidopsis. Cell Res 18:385–397
Zhang HZ, Yang JL, Wang WD, Li DD, Hu XQ, Wang H, Wei M, Liu QG, Wang ZC, Li CH (2015) Genome-wide identification and expression profiling of the copper transporter gene family in Populus trichocarpa. Plant Physiol Biochem 97:451–460
Zhao H, Eide D (1996a) The yeast ZRT1 gene encodes the zinc transporter protein of a high-affinity uptake system induced by zinc limitation. Proc Natl Acad Sci U S A 93:2454–2458
Zhao H, Eide D (1996b) The ZRT2 gene encodes the low affinity zinc transporter in Saccharomyces cerevisiae. J Biol Chem 271:23203–23210
Acknowledgements
This work were supported by the National Natural Science Foundation of China (No. 31400573) and the 111 project (B16010).
Author information
Authors and Affiliations
Contributions
Haizhen Zhang, Shicheng Zhao, Xuemei Xu, and Chenghao Li designed the experiments and wrote the manuscript. Haizhen Zhang and Dandan Li performed the experiments. Haizhen Zhang, Shicheng Zhao, and Xuemei Xu analyzed the data.
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Additional information
Data Archiving Statement
All identified amino acid of ZIP gene sequences were deposited into the NCBI database (http://www.ncbi.nlm.nih.gov/). The accession numbers are listed in Table 1.
Rights and permissions
About this article
Cite this article
Zhang, H., Zhao, S., Li, D. et al. Genome-Wide Analysis of the ZRT, IRT-Like Protein (ZIP) Family and Their Responses to Metal Stress in Populus trichocarpa . Plant Mol Biol Rep 35, 534–549 (2017). https://doi.org/10.1007/s11105-017-1042-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-017-1042-2