Abstract
Pancreatic ductal adenocarcinoma (PDAC) is one of the most lethal cancers in the world, and more molecular mechanisms should be illuminated to meet the urgent need of developing novel detection and therapeutic strategies. We analyzed the related microarray data to find the possible hub genes and analyzed their prognostic values using bioinformatics methods. The mRNA microarray datasets GSE62452, GSE15471, GSE102238, GSE16515, and GSE62165 were finally chosen and analyzed using GEO2R. The overlapping genes were found by Venn Diagrams, functional and pathway enrichment analyses were performed using the DAVID database, and the protein–protein interaction (PPI) network was constructed by STRING and Cytoscape. OncoLnc, which was linked to TCGA survival data, was used to investigate the prognostic values. In total, 179 differentially expressed genes (DEGs) were found in PDAC, among which, 130 were up-regulated genes and 49 were down-regulated. DAVID showed that the up-regulated genes were significantly enriched in extracellular matrix and structure organization, collagen catabolic and metabolic process, while the down-regulated genes were mainly involved in proteolysis, reactive oxygen species metabolic process, homeostatic process and cellular response to starvation. From the PPI network, the 21 nodes with the highest degree were screened as hub genes. Based on Molecular Complex Detection (MCODE) plug-in, the top module was formed by ALB, TGM, PLAT, PLAU, EGF, MMP7, MMP1, LAMC2, LAMA3, LAMB3, COLA1, FAP, CDH11, COL3A1, ITGA2, and VCAN. OncoLnc survival analysis showed that, high expression of ITGA2, MMP7, ITGB4, ITGA3, VCAN and PLAU may predict poor survival results in PDAC. The present study identified hub genes and pathways in PDAC, which may be potential targets for its diagnosis, treatment, and prognostic prediction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is one of the most lethal cancers in the world, being characterized as high aggressiveness, early metastasis, and insensitive to chemotherapy or radiotherapy [1], its 5-year survival is only 8% based upon the latest data [2], and little improvement has been seen over the past years [1, 3]. Hence, illumination of the molecular pathophysiology mechanisms and identification of the key signaling pathways and regulators is urgently needed to develop novel screening, diagnostic and therapeutic strategies.
Recently, the microarray technology has been extensively used to detect generally genetic alteration during tumorigenesis and cancer progression. With this technology, several gene expression profiling studies have shown hundreds of differentially expressed genes (DEGs) in PDAC carcinogenesis, which involved in various pathways, biological processes, and molecular functions. Comparative analysis of the overlapped DEGs may be more reliable when compared with a single expression profile. In this study, we used integrated bioinformatics methods to find the overlapped DEGs, analyzed the functional and pathway enrichment and protein–protein interaction (PPI) network to find the possible hub genes, and by using The Cancer Genome Atlas (TCGA) database to obtain the survival data and predict the prognostic values of the hub genes.
Methods
Collection of studies
We searched the GEO database (https://www.ncbi.nlm.nih.gov/geo/) for publics from inception to February 10, 2018, using the following strategy: “pancreatic cancer”, “homo sapiens” (Organism), “tissue” (Attribute Name), “RNA” (Sample Type), “expression profiling by array” (Study Type). Further inclusion criteria were as follows: (1) samples composed of both PDAC tissues and normal tissues, (2) gene expression profiling of mRNA, (3) sample count of each group are more than 10, and total count more than 30, and (4) sufficient information to perform the analysis. Five gene expression profiles (GSE62452, GSE15471, GSE102238, GSE16515, and GSE62165) were finally chosen.
Microarray data and data processing
GSE62452 datasets contained 69 tumor samples and 61 normal samples [4], GSE15471 consisted of 39 tumor samples and 39 normal samples [5], GSE102238 included 50 tumor samples and 50 normal samples [6], GSE16515 was composed of 36 tumor samples and 16 normal samples [7], and GSE62165 was formed by 118 tumor samples and 13 normal samples [8].
GEO2R (https://www.ncbi.nlm.nih.gov/geo/geo2r/) is an R programming languages-based tool to screen for DEGs [9]. By entering the series accession number, defining groups, assigning groups and clicking “Top 250”, the webpage could compare the differences between the groups. After saving the results, we picked the genes whose adjusted P-Values (adj. P) < 0.05 and |logFC|>1. Venn map (http://bioinformatics.psb.ugent.be/webtools/Venn/) was drawn to identify the overlapped genes.
Functional and pathway enrichment analysis
The Database for Annotation, Visualization and Integrated Discovery (DAVID) Version 6.8 (https://david.ncifcrf.gov/) is a comprehensive functional annotation tools to help us understand biological meaning behind the genes [10]. By Gene Ontology (GO) enrichment analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis, we found the potential relevant biological function annotation. P < 0.05 was considered statistically significant.
PPI network construction and module analysis
The STRING database Version 10.5 (http://string-db.org/) aims to collect and integrate interactions between proteins, including direct (physical) interactions and indirect (functional) interactions [11], and combined score > 0.4 was set as the cut-off criterion. Then we used Cytoscape Version 3.6.0 [12] to visualize the biomolecular interaction networks of the DEGs. Node degree ≥ 10 was set as the criterion of hub genes. Molecular Complex Detection (MCODE) plug-in was used to screen modules from the PPI network with degree cutoff 2, haircut on, node score cutoff 0.2, k-score 2, maximum depth 100, and nodes more than 8. The functional and pathway enrichment analysis was performed through DAVID in the modules.
Survival analysis of hub genes
OncoLnc (http://www.oncolnc.org/) is a tool for interactively exploring survival correlations, which contains survival data for 21 cancer studies performed by TCGA [13]. The PDAC patients were divided into two groups: low (expression lower than the first quartile) and high (expression higher than the third quartile), the overall survival of the two groups was assessed by Kaplan–Meier plots and log rank P-Value, log rank P-Value < 0.05 was the cut-off criterion. Since OncoLnc cannot provide with the hazard ratio (HR) with 95% confidence intervals (CI), we downloaded data from OncoLnc, and then used IBM SPSS Statistic Version 24.0.0.0 to perform the survival analysis and calculate the HR and 95% CI. At last, we also performed survival analysis of the hub genes using the data obtained from the GEO database of GSE62452 and GSE71729 to validate the results.
Results
Identification of DEGs
A total of 295, 1793, 2133, 1824, and 4063 genes were extracted from GSE62452, GSE15471, GSE102238, GSE16515, and GSE62165, respectively. Among them, 179 DEGs overlapped (Fig. 1), and 130 were up-regulated, 49 were down-regulated.
Functional and pathway enrichment analysis
GO biological process (BP) analysis indicated that the up-regulated DEGs were significantly enriched in extracellular matrix and structure organization, collagen catabolic and metabolic process, while the down-regulated genes were mainly involved in proteolysis, reactive oxygen species metabolic process, homeostatic process and cellular response to starvation. For GO cell component (CC), the up-regulated DEGs were significantly enriched in extracellular region part and matrix, proteinaceous extracellular matrix, and the down-regulated genes were mainly involved in extracellular space and region part. Upon molecular function (MF), the up-regulated DEGs were significantly enriched in extracellular matrix structural constituent, collagen binding, and integrin binding, while the down-regulated genes were mainly involved in exopeptidase activity, peptidase activity, and serine-type peptidase activity. Additionally, KEGG analysis proved that the up-regulated DEGs were significantly enriched in extracellular matrix (ECM)-receptor interaction, focal adhesion and phosphoinositide 3-kinase (PI3K)-Akt signaling pathway, while the down-regulated genes were mainly involved in pancreatic secretion, and complement and coagulation cascades (Fig. 2, if the terms enriched in this category were more than five, top five were chose according to P-Value).
PPI network construction and module analysis
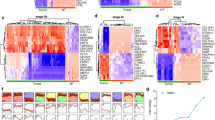
In total, 126 nodes and 327 edges were mapped in the PPI network of identified DEGs (Fig. 3a). Twenty-one genes with degree ≥ 10 were chosen as hub genes (Table 1). Through the MCODE plug-in, one significant module was selected with average MCODE score = 5.6, nodes = 16 and edges = 42 (Fig. 3b). Functional enrichment analysis indicated that the up-regulated genes in the significant module were enriched in extracellular matrix organization, extracellular structure organization, extracellular matrix disassembly, and collagen metabolic process, while the down-regulated genes were involved in platelet degranulation (Fig. 4).
a Protein–protein interaction (PPI) network of differentially expressed genes in pancreatic ductal adenocarcinoma. b A significant module selected from PPI network. Red nodes stand for up-regulated genes, while green nodes stand for down-regulated genes, and the darker color of the nodes stands for a larger |log FC|, the larger size of the nodes stands for a higher degree of connectivity. The lines represent interaction relationship between the nodes, and a wider line stands for a larger combined-score
Survival analysis of hub genes
OncoLnc predicted that among the selected hub genes, high mRNA expression of ITGA2, MMP7, ITGB4, ITGA3, VCAN and PLAU may be associated with poor survival of PDAC patients (P < 0.05). Survival analysis performed by SPSS was more conservative than OncoLnc, and the result showed that, high expression of MMP7 predicting poor survival may be debatable, as its P-Value is 0.053 (Fig. 5). The survival analysis of the hub genes using the data in GSE62452 validated that, high expression of ITGA2, ITGB4, and ITGA3 predicted poor survival. Date in GSE71729 showed that high expression of ITGB4 and PLAU had a poor survival (Table 2).
Discussion
PDAC, with a high mortality and short period of survival, is a malignancy that poses a serious threat to human health [1], unfortunately, the early diagnosis and efficient treatment of PDAC still remains as a huge problem due to the lack of understanding of the molecular mechanisms which drive the occurrence and development of PDAC. Therefore, it is of vital importance to have in-depth research into the factors and mechanisms, which might help in PDAC diagnosis and therapy [14]. With the development of bioinformatic and microarray technology, the precious and tremendous data of the patients could be shard, and it is much easier to determine the general genetic alterations in diseases occurrence, progression and prognosis, which may shed light on some hub genes or targets for clinical utility.
In this study, we identified 5 datasets comparing the differences in mRNA between tumor tissues and normal tissues. Eventually, a total of 179 DEGs were screened, including 130 up-regulated genes and 49 down-regulated genes. Functional and enrichment analysis revealed that the up-regulated DEGs were significantly enriched in extracellular organization, collagen catabolic and metabolic process, while the down-regulated genes were mainly involved in proteolysis, reactive oxygen species metabolic process, homeostatic process. Additionally, KEGG pathway analysis showed that the up-regulated DEGs were significantly enriched in ECM-receptor interaction, focal adhesion and PI3K-Akt signaling pathway, while the down-regulated genes were mainly involved in pancreatic secretion, and complement and coagulation cascades.
The results are in accordance with previous studies, which proved that PDAC was characterized by a dense stromal response, and stromal element contribute to its progression [15, 16], and Begum et al. showed that the ECM proteins increased PDAC tumor initiating potential, self-renewal and the frequency of cancer stem cells, indicating that the present method is effective in identifying hub genes. PI3K-Akt pathway is vital in various cancers [17, 18], dysregulation of it is common in PDAC [17], and up to 60% of PDAC cases had increased PI3K-Akt activity [17, 19, 20]. Liu et al. proved that inactivation of PI3K-Akt increased gemcitabine induced apoptosis in pancreatic cancer cells [21]. Hence, targets on this pathway might be potentially novel therapy for PDAC. Ebrahimi et al. summarized the agents targeting PI3K-Akt, only Wortmannin, LY294002, and Perifosine has been tested in pancreatic cancer, and the results might be optimistic [17].
By PPI network construction, we identified top 21 genes with high connectivity degrees, which include ALB, COL11A1, COL1A1, COL3A1, COL5A2, COL6A3, FBN1, FN1, COMP, EGF, ITGA2, ITGA3, ITGB4, MMP1, MMP7, PLAT, PLAU, THBS2, POSTN, SPARC, and VCAN, and among them, only ALB and EGF are down-regulated. Further survival analysis proved that, high expression of ITGA2, MMP7, ITGB4, ITGA3, VCAN and PLAU may predict poor survival. In validation analysis using the data from GSE62452 and GSE71729, high expression of ITGA3, ITGA2, ITGB4 and PLAU was proved to be associated with poor survival.
Till now, no research has showed the expression of ALB (albumin) in PDAC, there are only studies investigating the correlation between serum ALB and PDAC. Deng et al. developed a nomogram for predicting survival in PDAC patients, decrease level of ALB indicated poor survival [22]. Arima and Liu et al. also proved that high C-reactive protein (CRP)/ALB ratio also indicated poor survival [23]. However, the serum ALB level is influenced by the patient’s nutrition status, the liver function and other elements, and could not reflect the expression of ALB in the tissues, therefore, further studies are needed on this issue.
COL11A1, COL1A1, COL3A1, COL5A2, COL6A3, are all collagen genes. In Garcia-Pravia et al.’s research, the expression of COL11A1 is significantly increased in PDAC samples compared with normal and chronic pancreatitis (CP) tissues, and they further pointed out that, proCOL11A1 may be a powerful new marker for its diagnosis [24]. Araft et al. proved that PDAC tissues had significantly upregulated COL6A3 protein levels compared with paired adjacent tissues, and that presence of COL6A3 isoform and high protein levels appeared to correlate with tumor stage [25]. As for the rest of the collagen genes, no study had showed their relationship with PDAC for the moment, and maybe that’s what we can do next.
FBN1 (fibrillin 1) and FN1(fibronectin 1) are also ECMs, the study investigating FBN1 and PDAC is rare, only one pointed out that in the process of pancreatic islets progressed to angiogenic to insulinoma, FN1 and FBN1 were found in significantly higher abundance [26]. Hu et al. verified 25 protein biomarker candidates for PDAC prognosis, and they brought up that upregulated FN1 may predict poor survival [27]. Our survival analysis did not prove this, and the reason might lie in how we divided the group in our study. In their study, fold change ≥ 2 was regarded as upregulated, while in our study, expression higher than the third quartile was taken as highly expressed, and this may be the reason why the result does not accord.
COMP (cartilage oligomeric matrix protein) is a member of the thrombospondin family of ECM, and it was proved that, COMP was preferentially expressed in degenerating acinar cells in CP-like areas in pancreatic cancers and CP, indicating that this molecule is important in the course of acinar cell deterioration and dedifferentiation [28]. From their results, COMP could be a marker for PDAC with CP-like areas, but may not be of assistance in differentiating CP and PDAC.
The epidermal growth factor receptor (EGFR) signaling pathway is tightly related to tumorigenesis and progression [29]. Early studies [30,31,32] supported that overexpression of EGF (epidermal growth factor) and EGFR has been seen in pancreatic cancer samples compared with normal ones, in this point, our study is contrary to it. However, in another microarray analysis study, which used GSE16515 alone [33], the result was in accordance with ours, supporting that EGF was downregulated. This is really confusing, and the divergence may lie in that what we known about EGFR family is just the tip of the iceberg, and multiple members may participated in the aberrant autocrine and paracrine activation of this pathway [34]. On the other hand, Uegaki et al. thought that, the expression of EGF or EGFR alone does not reflect the prognosis of patients, their coexpression mattered [35], therefore new drugs blocking EGFR pathway still needs more exploration before advanced treatment shows up.
ITGA2, ITGA3, ITGB4 are all integrin subunits, which have important function in epithelial-mesenchymal transition (EMT). Nones et al. [36] proved that, in patients with PDAC, hypomethylation of ITGA2 correlated with high gene expression, which was related with poor survival, the result is the same as ours. There is also study showing that, ITGA3 was overexpressed in PDAC, and overexpression of ITGA3 correlated to poor survival [37], also the same as ours. Yamazaki and Masugi et al. firstly used microarray analysis, and identified ITG4 was upregulated in high-EMT xenografts derived from PDAC patients [38], additionally, they elucidated that, overexpression of ITGB4 promoted cell motility, and may be potential in regulating invasion and EMT [39]. Our study further believed that, high expression of ITGB4 was a risk factor for poor survival, and the results were validated both in GSE62452 and GSE71729, though no trial has been reported on this issue.
MMP1 and MMP7 are all metalloproteinases (MMPs), which have long been implicated for roles in cancer initiation and invasion [40]. Pancreatic cancer cells could induce alterations in MMPs in pancreatic stem cells (PSCs), including upregulation expression and activation of MMP1, and enhanced migration [41]. Fukuda et al. established that Stat3 signaling enforces MMP7 expression in pancreatic cancer cells, while MMP7 deletion restricts tumor size and metastasis in mice, and increased expression of MMP7 predicted shortened survival [42], our survival analysis of MMP7 is also in support of this. Even though data from GSE62452 and GSE71729 was not in support of this result, it may be due to that the sample size is not large enough in the two dataset, and more studies with larger sample-size are still needed to validate.
Plasminogen activator, tissue type (PLAT) and plasminogen activator, urokinase (PLAU) are both plasminogen activators. Bournet et al. used endoscopic ultrasound-guided fine needle aspiration biopsy samples to compare the different gene expression between advanced PDAC and pseudotumoural CP, and they demonstrated PLAT and PLAU were significantly overexpressed in cancer samples [43]. Besides, PLAU is highly expressed in more invasive pancreatic cells, and a combination of CDH3, LENG, and PLAU panels were significantly associated with poor survival [44]. In our study, high expression of PLAU also predicted poor survival, and data from GSE71729 further validated this result.
The basic research about THBS2 (thrombospondin-2) and PDAC is scarce, Kim et al. revealed that, the concentrations of plasma THBS2 discriminated among all stages of PDAC, and a new measurement combing both THBS2 and CA19-9 helped to increase the specificity to 98% in diagnosing PDAC [45], suggesting a combined blood marker panel may improve the detection of PDAC. POSTON (periostin) is a secretory protein function in cell adhesion, and was proved to drive the carcinogenic process, and furthermore, increase the chemoresistance to gemcitabine in pancreatic cancer cells [46]. Yu et al. elucidated that SPARC (secreted protein acidic and rich in cysteine) expressed differentially not only between PDAC samples and normal samples, but also showed difference in metastatic and normal lymph nodes, moreover, patients with positive POSTON expression had poor overall survival [47]. VCAN (versican) is a kind of proteoglycan, and was proved to be greatly increased in PDAC matrix, and disproportional increase of VCAN compared to another contradictory proteoglycan namely decorin may be associated with the aggressiveness of PDAC [48]. Survival analysis in our study pointed out high expression of VCAN was a risk factor for poor survival, but we failed to validate it with the data from GSE62452 and GSE71729, and no study has investigated the value of VCAN expression in the survival of PDAC for the moment, so this result still needs verification.
Conclusion
This study identified 179 DEGs, which include ALB, COL11A1, COL1A1, COL3A1, COL5A2, COL6A3, FBN1, FN1, COMP, EGF, ITGA2, ITGA3, ITGB4, MMP1, MMP7, PLAT, PLAU, THBS2, POSTN, SPARC, and VCAN. In addition, high expression of ITGA2, ITGA3, ITGB4, MMP7, PLAU, and VCAN may be predictors of poor survival. High-throughput technology, such as microarray analysis, and integrated bioinformatic analysis assist in the identification of hub genes in tumorigenesis and progression, and the results coordinate with previous studies well. The significant genes and pathways may open up brand-new possibilities for early detection and treatment of PDAC; however, further researches are still required for untangling the mechanism of PDAC occurrence and development.
References
Fu Y, Liu S, Zeng S, Shen H (2018) The critical roles of activated stellate cells-mediated paracrine signaling, metabolism and onco-immunology in pancreatic ductal adenocarcinoma. Mol Cancer 17:62
Siegel RL, Miller K, Ahmedin Jemal D (2018) Cancer statistics, 2018. CA 68:7–30
Li D, Xie K, Wolff R, Abbruzzese JL (2004) Pancreatic cancer. Lancet 363:1049–1057
Yang S, He P, Wang J, Schetter A, Tang W, Funamizu N, Yanaga K, Uwagawa T, Satoskar AR, Gaedcke J, Bernhardt M, Ghadimi BM, Gaida MM, Bergmann F, Werner J, Ried T, Hanna N, Alexander HR, Hussain SP (2016) A novel MIF signaling pathway drives the malignant character of pancreatic cancer by targeting NR3C2. Cancer Res 76:3838–3850
Badea L, Herlea V, Dima SO, Dumitrascu T, Popescu I (2008) Combined gene expression analysis of whole-tissue and microdissected pancreatic ductal adenocarcinoma identifies genes specifically overexpressed in tumor epithelia. Hepatogastroenterology 55:2016–2027
Sun Y, Zhang Z, Xue J (2017) Gene expression signatures associated with Perineural Invasion in pancreatic ductal adenocarcinoma. GEO Database, https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE102238
Pei H, Li L, Fridley BL, Jenkins GD, Kalari KR, Lingle W, Petersen G, Lou Z, Wang L (2009) FKBP51 affects cancer cell response to chemotherapy by negatively regulating Akt. Cancer Cell 16:259–266
Janky R, Binda MM, Allemeersch J, Van den Broeck A, Govaere O, Swinnen JV, Roskams T, Aerts S, Topal B (2016) Prognostic relevance of molecular subtypes and master regulators in pancreatic ductal adenocarcinoma. BMC Cancer 16:632
Barrett T, Wilhite SE, Ledoux P, Evangelista C, Kim IF, Tomashevsky M, Marshall KA, Phillippy KH, Sherman PM, Holko M, Yefanov A, Lee H, Zhang N, Robertson CL, Serova N, Davis S, Soboleva A (2013) NCBI GEO: archive for functional genomics data sets—update. Nucleic Acids Res 41:D991–D995
Huang DW, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4:44–57
Szklarczyk D, Morris JH, Cook H, Kuhn M, Wyder S, Simonovic M, Santos A, Doncheva NT, Roth A, Bork P, Jensen LJ, von Mering C (2017) The STRING database in 2017: quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res 45:D362–D368
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504
Anaya J (2016) OncoLnc: linking TCGA survival data to mRNAs, miRNAs, and lncRNAs. PeerJ Comput Sci. https://doi.org/10.7717/peerj-cs.67
Kretz AL, von Karstedt S, Hillenbrand A, Henne-Bruns D, Knippschild U, Trauzold A, Lemke J (2018) Should we keep walking along the trail for pancreatic cancer treatment? Revisiting TNF-related apoptosis-inducing ligand for anticancer therapy. Cancers (Basel) 10:77
Begum A, Ewachiw T, Jung C, Huang A, Norberg KJ, Marchionni L, McMillan R, Penchev V, Rajeshkumar NV, Maitra A, Wood L, Wang C, Wolfgang C, DeJesus-Acosta A, Laheru D, Shapiro IM, Padval M, Pachter JA, Weaver DT, Rasheed ZA, Matsui W (2017) The extracellular matrix and focal adhesion kinase signaling regulate cancer stem cell function in pancreatic ductal adenocarcinoma. PLoS ONE 12:e0180181
Ansari D, Carvajo M, Bauden M, Andersson R (2017) Pancreatic cancer stroma: controversies and current insights. Scand J Gastroenterol 52:641–646
Ebrahimi S, Hosseini M, Shahidsales S, Maftouh M, Ferns GA, Ghayour-Mobarhan M, Mahdi Hassanian S, Avan A (2017) Targeting the Akt/PI3K signaling pathway as a potential therapeutic strategy for the treatment of pancreatic cancer. Curr Med Chem 24:1321–1331
Wintzell M, Lofstedt L, Johansson J, Pedersen AB, Fuxe J, Shoshan M (2012) Repeated cisplatin treatment can lead to a multiresistant tumor cell population with stem cell features and sensitivity to 3-bromopyruvate. Cancer Biol Ther 13:1454–1462
Xu RH, Pelicano H, Zhou Y, Carew JS, Feng L, Bhalla KN, Keating MJ, Huang P (2005) Inhibition of glycolysis in cancer cells: a novel strategy to overcome drug resistance associated with mitochondrial respiratory defect and hypoxia. Cancer Res 65:613–621
Bhatt AN, Chauhan A, Khanna S, Rai Y, Singh S, Soni R, Kalra N, Dwarakanath BS (2015) Transient elevation of glycolysis confers radio-resistance by facilitating DNA repair in cells. BMC Cancer 15:335
Liu D, Zhang Y, Dang C, Ma Q, Lee W, Chen W (2007) siRNA directed against TrkA sensitizes human pancreatic cancer cells to apoptosis induced by gemcitabine through an inactivation of PI3K/Akt-dependent pathway. Oncol Rep 18:673–677
Deng QL, Dong S, Wang L, Zhang CY, Ying HF, Li ZS, Shen XH, Guo YB, Meng ZQ, Yu JM, Chen QW (2017) Development and validation of a nomogram for predicting survival in patients with advanced pancreatic ductal adenocarcinoma. Sci Rep 7:11524
Arima K, Yamashita YI, Hashimoto D, Nakagawa S, Umezaki N, Yamao T, Tsukamoto M, Kitano Y, Yamamura K, Miyata T, Okabe H, Ishimoto T, Imai K, Chikamoto A, Baba H (2017) Clinical usefulness of postoperative C-reactive protein/albumin ratio in pancreatic ductal adenocarcinoma. Am J Surg 216:111–115
Garcia-Pravia C, Galvan JA, Gutierrez-Corral N, Solar-Garcia L, Garcia-Perez E, Garcia-Ocana M, Del AJ, Menendez-Rodriguez P, Garcia-Garcia J, Del Amo-Iribarren J, Menéndez-Rodríguez P, García-García J, Juan R, Simón-Buela L (2013) Overexpression of COL11A1 by cancer-associated fibroblasts: clinical relevance of a stromal marker in pancreatic cancer. PLoS ONE 8:e78327
Arafat H, Lazar M, Salem K, Chipitsyna G, Gong Q, Pan TC, Zhang RZ, Yeo CJ, Chu ML (2011) Tumor-specific expression and alternative splicing of the COL6A3 gene in pancreatic cancer. Surgery 150:306–315
Naba A, Clauser KR, Mani DR, Carr SA, Hynes RO (2017) Quantitative proteomic profiling of the extracellular matrix of pancreatic islets during the angiogenic switch and insulinoma progression. Sci Rep 7:40495
Hu D, Ansari D, Pawlowski K, Zhou Q, Sasor A, Welinder C, Kristl T, Bauden M, Rezeli M, Jiang Y, Marko-Varga G (2018) Proteomic analyses identify prognostic biomarkers for pancreatic ductal adenocarcinoma. Oncotarget 9:9789–9807
Liao Q, Kleeff J, Xiao Y, Di Cesare PE, Korc M, Zimmermann A, Buchler MW, Friess H (2003) COMP is selectively up-regulated in degenerating acinar cells in chronic pancreatitis and in chronic-pancreatitis-like lesions in pancreatic cancer. Scand J Gastroenterol 38:207–215
Nandy D, Mukhopadhyay D (2011) Growth factor mediated signaling in pancreatic pathogenesis. Cancers (Basel) 3:841–871
Friess H, Berberat P, Schilling M, Kunz J, Korc M, Buchler MW (1996) Pancreatic cancer: the potential clinical relevance of alterations in growth factors and their receptors. J Mol Med (Berl) 74:35–42
Korc M, Chandrasekar B, Yamanaka Y, Friess H, Buchier M, Beger HG (1992) Overexpression of the epidermal growth factor receptor in human pancreatic cancer is associated with concomitant increases in the levels of epidermal growth factor and transforming growth factor alpha. J Clin Invest 90:1352–1360
Yamanaka Y, Friess H, Kobrin MS, Buchler M, Beger HG, Korc M (1993) Coexpression of epidermal growth factor receptor and ligands in human pancreatic cancer is associated with enhanced tumor aggressiveness. Anticancer Res 13:565–569
Long J, Liu Z, Wu X, Xu Y, Ge C (2016) Gene expression profile analysis of pancreatic cancer based on microarray data. Mol Med Rep 13:3913–3919
Friess H, Guo XZ, Nan BC, Kleeff J, Buchler MW (1999) Growth factors and cytokines in pancreatic carcinogenesis. Ann N Y Acad Sci 880:110–121
Uegaki K, Nio Y, Inoue Y, Minari Y, Sato Y, Song MM, Dong M, Tamura K (1997) Clinicopathological significance of epidermal growth factor and its receptor in human pancreatic cancer. Anticancer Res 17:3841–3847
Nones K, Waddell N, Song S, Patch AM, Miller D, Johns A, Wu J, Kassahn KS, Wood D, Bailey P, Fink L, Manning S, Christ AN, Nourse C, Kazakoff S, Taylor D, Leonard C, Chang DK, Jones MD, Thomas M, Watson C, Pinese M, Cowley M, Rooman I, Pajic M, Butturini G, Malpaga A, Corbo V, Crippa S, Falconi M, Zamboni G, Castelli P, Lawlor RT, Gill AJ, Scarpa A, Pearson JV, Biankin AV, Grimmond SM (2014) Genome-wide DNA methylation patterns in pancreatic ductal adenocarcinoma reveal epigenetic deregulation of SLIT-ROBO, ITGA2 and MET signaling. Int J Cancer 135:1110–1118
Zhu GH, Huang C, Qiu ZJ, Liu J, Zhang ZH, Zhao N, Feng ZZ, Lv XH (2011) Expression and prognostic significance of CD151, c-Met, and integrin alpha3/alpha6 in pancreatic ductal adenocarcinoma. Dig Dis Sci 56:1090–1098
Yamazaki K, Masugi Y, Effendi K, Tsujikawa H, Hiraoka N, Kitago M, Shinoda M, Itano O, Tanabe M, Kitagawa Y, Sakamoto M (2014) Upregulated SMAD3 promotes epithelial-mesenchymal transition and predicts poor prognosis in pancreatic ductal adenocarcinoma. Lab Invest 94:683–691
Masugi Y, Yamazaki K, Emoto K, Effendi K, Tsujikawa H, Kitago M, Itano O, Kitagawa Y, Sakamoto M (2015) Upregulation of integrin beta4 promotes epithelial-mesenchymal transition and is a novel prognostic marker in pancreatic ductal adenocarcinoma. Lab Invest 95:308–319
Knapinska AM, Estrada CA, Fields GB (2017) The roles of matrix metalloproteinases in pancreatic cancer. Prog Mol Biol Transl Sci 148:339–354
Tjomsland V, Pomianowska E, Aasrum M, Sandnes D, Verbeke CS, Gladhaug IP (2016) Profile of MMP and TIMP expression in human pancreatic stellate cells: regulation by IL-1alpha and TGFbeta and implications for migration of pancreatic cancer cells. Neoplasia 18:447–456
Fukuda A, Wang SC, Morris JT, Folias AE, Liou A, Kim GE, Akira S, Boucher KM, Firpo MA, Mulvihill SJ, Hebrok M (2011) Stat3 and MMP7 contribute to pancreatic ductal adenocarcinoma initiation and progression. Cancer Cell 19:441–455
Bournet B, Pointreau A, Souque A, Oumouhou N, Muscari F, Lepage B, Senesse P, Barthet M, Lesavre N, Hammel P, Levy P, Ruszniewski P, Cordelier P, Buscail L (2012) Gene expression signature of advanced pancreatic ductal adenocarcinoma using low density array on endoscopic ultrasound-guided fine needle aspiration samples. Pancreatology 12:27–34
Liu P, Weng Y, Sui Z, Wu Y, Meng X, Wu M, Jin H, Tan X, Zhang L, Zhang Y (2016) Quantitative secretomic analysis of pancreatic cancer cells in serum-containing conditioned medium. Sci Rep 6:37606
Kim J, Bamlet WR, Oberg AL, Chaffee KG, Donahue G, Cao XJ, Chari S, Garcia BA, Petersen GM, Zaret KS (2017) Detection of early pancreatic ductal adenocarcinoma with thrombospondin-2 and CA19-9 blood markers. Sci Transl Med 9:eaah5583
Liu Y, Li F, Gao F, Xing L, Qin P, Liang X, Zhang J, Qiao X, Lin L, Zhao Q, Du L (2016) Periostin promotes the chemotherapy resistance to gemcitabine in pancreatic cancer. Tumour Biol 37:15283–15291
Yu XZ, Guo ZY, Di Y, Yang F, Ouyang Q, Fu DL, Jin C (2017) The relationship between SPARC expression in primary tumor and metastatic lymph node of resected pancreatic cancer patients and patients’ survival. Hepatobiliary Pancreat Dis Int 16:104–109
Skandalis SS, Kletsas D, Kyriakopoulou D, Stavropoulos M, Theocharis DA (2006) The greatly increased amounts of accumulated versican and decorin with specific post-translational modifications may be closely associated with the malignant phenotype of pancreatic cancer. Biochim Biophys Acta 1760:1217–1225
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Lu, Y., Li, C., Chen, H. et al. Identification of hub genes and analysis of prognostic values in pancreatic ductal adenocarcinoma by integrated bioinformatics methods. Mol Biol Rep 45, 1799–1807 (2018). https://doi.org/10.1007/s11033-018-4325-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-018-4325-2