Abstract
Leaf rust (LR) and yellow rust (YR), caused by Puccinia triticina and Puccinia striiformis f. sp. tritici, respectively, are important diseases of wheat. Quaiu 3, a common wheat line developed at the International Maize and Wheat Improvement Center (CIMMYT), is immune to YR in Mexico despite seedling susceptibility to predominant races. Quaiu 3 also shows immunity to LR in field trials and is known to possess the race-specific gene Lr42. A mapping population of 182 recombinant inbred lines (RILs) was developed by crossing Quaiu 3 with susceptible Avocet-YrA and phenotyped with LR and YR in field trials for 2 years in Mexico. Quantitative trait loci (QTL) associated with YR and LR resistance in the RILs were identified using Diversity Arrays Technology and simple sequence repeat markers. A large-effect QTL on the long arm of chromosome 2D explained 49–54 % of the phenotypic variation in Quaiu 3 and was designated as Yr54. Two additional loci on 1BL and 3BS explained 8–17 % of the phenotypic variation for YR and coincided with previously characterized adult plant resistance (APR) genes Lr46/Yr29 and Sr2/Yr30, respectively. QTL on 1DS and 1BL corresponding to Lr42 and Lr46/Yr29, respectively, contributed 60–71 % of the variation for LR resistance. A locus on 3D associated with APR to both diseases explained up to 7 % of the phenotypic variance. Additional Avocet-YrA-derived minor QTL were also detected for YR on chromosomes 1A, 3D, 4A, and 6A. Yr54 is a newly characterized APR gene which can be combined with other genes by using closely linked molecular markers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Leaf or brown rust (LR), caused by Puccinia triticina is one of the most common and widely distributed wheat diseases. In recent decades it has caused significant losses in yield and quality and has produced epidemics in several regions globally (Dubin and Brennan 2009; Kolmer et al. 2009). Similarly, stripe or yellow rust (YR), caused by Puccinia striiformis f. sp. tritici, has reportedly caused significant yield losses in more than 60 countries (Stubbs 1985; Chen 2005). Recently, YR has become a serious threat to wheat, causing 50–100 % yield losses due to the breakdown of existing resistance genes and gradual adaptation of new strains in warmer regions, particularly the Central and West Asia and North Africa (CWANA) region (ICARDA 2011). The breakdown of the widely used race-specific genes (R-genes) Yr17 (Bayles et al. 2000; Wan et al. 2004; Chen 2007), Yr27 (Singh et al. 2004; Wan et al. 2004), Yr31 (Singh et al. 2011; Rosewarne et al. 2012), and Lr24 (Park et al. 2002) has created a serious challenge for wheat scientists and growers worldwide. Introducing genetic resistance into cultivars is the most economic and environmentally safe measure of rust disease management in wheat. To date, more than 71 LR and 53 YR resistance genes have been identified and catalogued in different Triticum and related species or genera (McIntosh et al. 2012; Singh et al. 2013; Xu et al. 2013). Most of the formally designated genes confer race-specific resistance, which often loses effectiveness within a few years of deployment because cultivars with single R-genes facilitate selection for virulent races (Bolton et al. 2008). Though most R-genes display high levels of resistance against the pathogen, and selection is therefore a relatively easy task, the longevity of those genes is only 3–5 years in a new cultivar (Singh 2012).
Many prominent APR genes, such as Lr34/Yr18/Sr57/Pm38 (Dyck1987; McIntosh 1992; Singh 1992; Spielmeyer et al. 2005; Lillemo et al. 2008; Krattinger et al. 2009), Lr46/Yr29/Sr58/Pm39 (Singh et al.1998; William et al. 2003; Lillemo et al. 2008), Lr67/Yr46/Sr55/Pm46 (Herrera-Foessel et al. 2011; Hiebert et al. 2011), Yr36 (Fu et al. 2009), Sr2/Yr30 (Singh et al. 2000; Suenaga et al. 2003), and Lr68 (Herrera-Foessel et al. 2012), have been well characterized. Similarly, during the last 15 years, several molecular mapping studies have reported dozens of quantitative trait loci (QTL) distributed throughout all 21 chromosomes for YR resistance, and at least in 20 chromosomes for LR resistance in hexaploid wheat germplasm (Naz et al. 2008; Basnet 2012; Singh 2012). Nonetheless, the identification of new sources of durable resistance and their molecular characterization is a continual process in ensuring genetic diversity in breeding programs that aim to develop advanced lines or cultivars with high and stable yield potential, besides having durable rust resistance and other desirable characteristics.
Quaiu 3, a high-yielding spring wheat line developed by the International Maize and Wheat Improvement Center (CIMMYT), displays a high level of resistance against LR and YR under field conditions. Recently, two sister lines of Quaiu 3 were released as the cultivars Koshan 09 (Afghanistan) and Gambo (Ethiopia) (CIMMYT Wheat Atlas: http://wheatatlas.cimmyt.org/country/varieties/AFG/0). Quaiu 3 does not seem to carry any effective seedling resistance genes for YR resistance, as it displays high infection type (IT) ratings in seedling screenings (IT 6–8 on the 0–9 scale, depending on Mexican P. striiformis races and the greenhouse environment). However, it carries a moderately effective race-specific LR resistance gene (Lr42), initially transferred from Aegilops tauschii to the winter wheat line KS91WGRC11 (Basnet et al. 2013). In a previous study, Basnet et al. (2013) also estimated a minimum of three genes conferring resistance to both LR and YR in Quaiu 3. In addition to Lr42, they also suggested that Quiau 3 carries two APR genes, Yr29 and Yr30, based on closely linked molecular markers. However, these known genes alone were not able to display near immune resistance to both LR and YR as in resistant parent Quaiu 3. Therefore, in this study we performed whole-genome scanning of molecular markers to map the resistance loci and investigate their individual as well as combined effects in Avocet-YrA/Quaiu 3 recombinant inbred lines (RILs).
The objectives of our study were to: (1) map genomic regions associated with APR to both LR and YR in the Avocet-YrA/Quaiu 3 RIL population using molecular markers, and (2) elucidate the usefulness of characterized genes in conferring APR and enhancing genetic diversity for durable rust resistance in wheat.
Materials and methods
Plant materials
The mapping population consisted of 182 randomly advanced F4-derived F5 RILs from a cross between Avocet-YrA and Quaiu 3 (hereafter Avocet and Quaiu, respectively). Quaiu (GID: 3822578, Pedigree: Babax/Lr42//Babax*2/3/Vivitsi) is a CIMMYT line developed in 2006. Based on international data, Quaiu has shown high levels of APR to LR and YR and a moderate level of APR to stem rust (SR), caused by Puccinia graminis f. sp. tritici, under field conditions. The parents Babax and Vivitsi are also CIMMYT spring wheat lines developed in 1992 and 2000, respectively, whereas the Lr42 source parent KS91WGRC11 (Century*3/Ae. tauschii accession TA2450) is a winter wheat line developed by UDSA-ARS and Kansas State University (Cox et al. 1994). The LR and YR susceptible parent Avocet is an Australian selection that was also used as a recurrent parent in developing a series of near-isogenic lines with YR genes (Wellings et al. 2004). The RIL population was developed using the single-spike descent method (Basnet et al. 2013).
Evaluation of resistance to LR and YR
The Avocet/Quaiu RIL population and parents were evaluated for responses to LR at CIMMYT’s Norman E. Borlaug Experimental Station (CENEB), near Ciudad Obregon, Mexico, for two crop seasons (2008–2009 and 2009–2010; abbreviated as LR09 and LR10, respectively). The same population and parents were also evaluated for YR responses at the CIMMYT Research Station near Toluca, Mexico, during the 2009 and 2010 crop seasons (YR09 and YR10 experiments).
Populations and parents were hand-sown in 1-m-long paired rows (approx. 60–80 plants per line) on top of 80-cm-wide raised beds. The susceptible variety Morocco was grown around the experimental field as spreader rows. To create homogeneous disease epidemics within each experimental unit, Morocco was also grown as a hill at one side of each plot in the middle of the 0.5-m-wide pathways. Artificial inoculations on spreader rows and hills were carried out twice (60 and 62 days after sowing in LR09, and 62 and 69 days after sowing in LR10) using hand-sprayers carrying an equal amount of urediniospores of two prevalent Mexican P. triticina races, MBJ/SP and MCJ/SP, suspended in Soltrol Oil (Phillips 66 Co., Bartlesville, OK, USA). Disease severity (DS) on parents and RILs was scored based on the modified Cobb Scale, where the percentage rusted tissues (0–100 %) was visually estimated according to Peterson et al. (1948). In LR09, DS was only measured once, when the susceptible check reached 100 % of severity, whereas in LR10, evaluation of DS was repeated 1 week after the first reading. For the repeated measurement, the area under disease progress curve (AUDPC) was calculated using the method suggested by Bjarko and Line (1988).
The planting scheme and plot sizes for YR phenotyping at Toluca were similar to those described for the CENEB experiments. Spreaders consisted of a mixture of six susceptible wheat lines possessing the defeated R-gene Yr27 and derived from the cross Avocet/Attila. The varying maturities of these lines ensured a continuous production of inoculum during the critical crop-growing period. An artificial epidemic was initiated by inoculating the 4-week-old spreaders three times, at 3- to 4-day intervals, with Mexican P. striiformis f. sp. tritici isolates MEX96.11 and MEX08.13. The first estimation of YR DS was conducted when the flag leaves of most susceptible RILs displayed 100 % DS, followed by two additional scorings at weekly intervals. The multiple DS data were converted into AUDPC scores for each line (Bjarko and Line 1988).
Molecular marker analysis and genetic linkage map construction
DNA from the parents Avocet and Quaiu was used to screen for 450 simple sequence repeat (SSR) markers (Röder et al. 1998; Pestsova et al. 2000; Sourdille et al. 2001; Song et al. 2002; Somers et al. 2004) to determine polymorphism. Information about the forward and reverse primer sequences and appropriate annealing temperatures for each marker were obtained from the GrainGenes database (http://wheat.pw.usda.gov/GG2/index.shtml). The polymorphic SSR markers were then used to genotype the entire mapping population of 182 RILs. After PCR, high-resolution allele separation was performed using an Applied Biosystems (Foster City, CA, USA) 3130xl genetic analyzer, followed by allele size determination with GeneMapper v3.7 software (Applied Biosystems). In addition to SSR marker genotyping, 30 μl of DNA samples (concentration 50 ng/μl) of parents and RILs were also sent to Triticarte Pty. Ltd., Yarrallumla, Australia for Diversity Arrays Technology (DArT) genotyping. An assay with Wheat PstI (TaqI) v2.3 was used to screen for 2,500 DArT markers in the parents and RILs (Jaccoud et al. 2001; Akbari et al. 2006). The alleles of polymorphic DArT loci were represented as 1 or 0, i.e. presence or absence of the marker sequence in a line, and designated as wPt followed by the clone number in the genomic representation.
After combining both marker types, a genetic map was constructed using QTL ICiMapping software (Li et al. 2008). A minimum logarithm of odds (LOD) score of 4.5 and maximum recombination frequency of 0.40 was set to make the linkage groups, while the Kosambi mapping function (Kosambi 1944) was used to convert the recombination frequency into map distances. To finalize the linkage map, marker ordering and rippling were performed using RECORD and COUNT algorithms, respectively.
QTL analysis of YR and LR responses
QTL analysis of DS and AUDPC for both YR and LR were performed using QTL ICiMapping (Li et al. 2008) and WinQTLCart v2.5 (Wang et al. 2011) mapping softwares. An inclusive composite interval mapping (ICIM) program with stepwise regression probability of 0.001 was employed in QTL ICiMapping, whereas composite interval mapping (CIM) with backward and forward regression was used in WinQTLCart. In both cases, the LOD threshold was calculated by 1,000 permutation tests. Moreover, QTL were reanalyzed by manually setting a LOD threshold of 2.5 in CIM to allow the detection of additional minor QTL. To further elucidate the effect of smaller QTL, analyses were performed in subtracted datasets after removing the RILs with large-effect QTL for YR or LR. Phenotypic distributions of YR and LR DS were also compared between two groups of RILs that were classified based on the presence or absence of a particular QTL. Finally, all the RILs were grouped into different QTL genotypic classes using flanking markers, and their mean performances and variances were analyzed using one-way ANOVA and pair-wise t tests.
Results
Disease evaluation
Substantial and uniform YR development occurred during both years of the Toluca trials. At the time of first evaluations in YR09 and YR10, the susceptible parent Avocet displayed 90 and 70 % severity, respectively, at flowering. Average YR severities of RILs ranged from 41.3 to 62.3 % in the two experiments across multiple evaluations. The distribution of RILs for YR DS and AUDPC was continuous but not normal across experiments. Similarly, excellent LR development was observed during both years at CENEB, where the susceptible parent displayed 100 % DS in both experiments during the early grain filling stage. The resistant parent Quaiu remained immune for both LR and YR in all experiments. Average LR severities of the RILs ranged from 22.1 to 33.8 % across the two experiments and at different stages of evaluation. As the population average DS for LR is significantly lower than the mid-parental value, the distribution of RILs was skewed towards resistance. Pearson’s correlation coefficients (r) for YR DS and AUDPC between the YR09 and YR10 experiments were highly significant (0.91–0.98, P < 0.001). Similarly, a high correlation between the LR09 and LR10 experiments was also observed for LR DS and AUDPC (0.86–0.96, P < 0.001). The correlation coefficients between YR and LR ranged from 0.21 to 0.32 (P < 0.01) across experiments. Likewise, high correlation (0.98–0.99, P < 0.001) was observed between DS recorded in different dates and corresponding AUDPC values for both YR and LR. The DS obtained from the first evaluation date, and AUDPC obtained from repeated measurements of each experiment, were used in subsequent genetic analyses. The DS at the first date of evaluation was probably the most representative phenotypic dataset, as it was recorded at maximum DS of the susceptible check (Peterson et al. 1948; Ren et al. 2012b).
Genetic linkage mapping and QTL analyses
Of 650 DArT and 130 SSR polymorphic markers, a total of 461 markers (359 DArT and 102 SSR) were placed in the final linkage map. Markers with a high level of redundancy, segregation distortion, and missing values were deleted. A total of 22 linkage groups were developed, representing all 21 chromosomes of hexaploid wheat. Only chromosome 5D had two linkage groups. The total genetic distance covered by all linkage groups was 2,654 cM, with an average distance of 5.8 cM between markers. The marker coverages of the A, B, and D genomes were 35, 43, and 22 %, respectively. Chromosomes 4D and 6D had the smallest linkage groups, with just three and six markers, respectively.
QTL analyses were performed using the final linkage map of Avocet/Quaiu for the DS and AUDPC values obtained from the LR and YR experiments. Apart from slim differences in the estimates of QTL effects, the mapping results given by both mapping softwares (QTL ICiMapping and WinQTLCart) were very similar. For consistency, only the results obtained from CIM in WinQTLCart are presented throughout this study. The LOD threshold values, which ranged from 2.9 to 3.8 based on 1,000 permutation tests for different traits, were auto-affixed during QTL analyses. In this study, QTL that were consistently detected above the LOD threshold across experiments are referred as major QTL, whereas those detected at sub-optimal LOD thresholds inconsistently across experiments are referred to minor QTL. As the DS and AUDPC data were highly correlated and the QTL results obtained with both of these datasets were similar, only the results obtained with DS datasets are presented in this study.
YR resistance QTL
Based on CIM, four major Quaiu-derived QTL were identified with DS in YR09 and YR10 and with average DS across experiments (YRAV; Table 1). The most significant QTL, QYr.tam-2D, was consistently identified in all experiments. Flanked by markers wpt-667162 and wpt-667054 on chromosome 2DL, QYr.tam-2D explained roughly 49–54 % of phenotypic variation (R 2) across experiments (Fig. 1). Similarly, QYr.tam-3B was flanked by markers Xgwm533 and wpt-7984 on chromosome 3BS (Online Resource 1) and explained 3–10 % of the phenotypic variation. A third QTL, QYr.tam-1B, explaining 6–7 % of the phenotypic variation was located on 1BL flanked by the markers wpt-668027 and csLV46G22 (Online Resource 1). The fourth QTL, QYr.tam-3D, located on chromosome 3D, explained 4–5 % of the total phenotypic variation and was flanked by markers wpt-672034 and Xbarc125 (Online Resource 1). The variation explained by the three smaller QTL substantially increased when analyses were performed in subtracted datasets, i.e. after excluding the RILs with QYr.tam-2D. In those analyses, QYr.tam-1B, QYr.tam-3B, and QYr.tam-3D explained up to 31, 20, and 11 % of the total phenotypic variation, respectively. Interestingly, two minor YR QTL, QYr.tam-6A and QYr.tam-6B, were also detected in subtracted datasets. QYr.tam-6A (R 2 = 5–7 %), detected in YR10 and YRAV DS, was derived from Avocet, whereas QYr.tam-6B (R 2 = 6–9 %), detected with YR09 and YR10 DS, was derived from Quaiu. Some Avocet-derived small-effect YR QTL were also detected at sub-optimal LOD thresholds (LOD = 2.8–3.2) with DS in YR10. These minor QTL (QYr.tam-1A, QYr.tam-3D.1, and QYr.tam-4A) were located on chromosomes 1A, 3D, and 4A, respectively, and explained up to 2 % of the phenotypic variation.
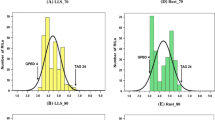
LOD profile of QTL QYr.tam-2D on chromosomes 2DL identified with disease severity in Toluca 2009 and 2010 experiments. The x and y axes correspond with LOD score and partial linkage map (distance measured in centimorgans) of the chromosome. The shaded chromosome segment represents the 2-LOD confidence interval of the QTL. The approximate location of the centromere is indicated with a solid arrowhead
LR resistance QTL
Two major QTL were identified for LR resistance, indicated by DS in LR09, LR10 and their average (LRAV) (Table 2). The first QTL, QLr.tam-1B (R 2 = 23–26 %), was flanked by markers wpt-668027 and csLV46G22 (Online Resource 1). The second QTL, QLr.tam-1D, at <1 cM from the marker Xwmc432, was flanked by wpt-666067 and wpt-667180 (Online Resource 1). This QTL explained up to 35 % of the phenotypic variation across experiments. A third QTL, QLr.tam-3D, with smaller effects, was identified on chromosome 3D, as measured by DS in LR10 and LRAV. For DS LR09 data, the LOD peak for QLr.tam-3D (LOD = 2.4) remained slightly below the significance threshold. This QTL, flanked by the markers wpt-672034 and Xbarc125, explained 4–7 % of the phenotypic variation (Table 2, Online Resource 1). After subtracting the RILs with QLr.tam-1D, QTL analysis considerably improved the variation attributed to QLr.tam-1B (41–55 %) and QLr.tam-3D (9–11 %).
Co-localization of QTL for dual APR
An overlap in the positions of QYr.tam-1BL and QLr.tam-1BL was observed (Online Resource 1). A similar overlap was observed on chromosome 3D between the QTL QYr.tam-3D and QLr.tam-3D (Online Resource 1). These co-localized disease resistance loci most probably represent pleiotropy or close linkage.
Effect of QTL on YR and LR severities
The distribution patterns of average LR and YR severities were observed among RILs in the presence (+QTL RILs) and absence (−QTL RILs) of different QTL, represented by parental alleles of closely linked markers. A discrete differentiation between +QYr.tam-2D and −QYr.tam-2D RILs was observed for average YR severity (Online Resource 2). However, a distorted segregation of Quaiu and Avocet marker alleles (χ2 P < 0.01) was evident within 1.2 cM of the QYr.tam-2D locus. RILs with QYr.tam-2D displayed an average DS range of 0–40 %, in contrast to 20–100 % for RILs lacking QYr.tam-2D. This observation indicated that QYr.tam-2D alone confers YR resistance up to a DS of 40 %. Similarly, the responses of RILs lacking QYr.tam-2D but displaying a continuous and approximately normal distribution for DS indicated the presence of multiple minor QTL, which when combined increased resistance to DS 20 %. After excluding the RILs possessing QYr.tam-2D, distributions of DS with and without QYr.tam-1B, QYr.tam-3B, and QYr.tam-3D were also compared. For all three QTL, the distribution of RILs lacking the respective QTL was shifted towards susceptibility, compared to the RILs possessing the QTL (data not shown). However, a few lines had high DS (90–100 %), despite carrying at least one of the three QTL based on linked markers.
For LR, the +QLr.tam-1D RILs were distributed towards resistance, with average DS ranging from 0 to 40 %, whereas the −QLr.tam-1D RILs displayed 10–100 % DS. A similar result was presented by classifying the Avocet/Quaiu RILs into +Lr42 and −Lr42 groups by Basnet et al. (2013). Similarly, in QLr.tam-1D subtracted datasets, +QLr.tam-1B and −QLr.tam-1B RILs displayed distinct distribution patterns, with DS ranging from 0 to 60 and 20 to 100 %, respectively (data not shown). However, distributions of +QLr.tam-3D and −QLr.tam-3D RILs distinctively overlapped, with DS ranging from 10 to 100 % in both categories (data not shown).
The RILs were divided into 16 and 8 genotypes based on the presence of four YR and three LR QTL, respectively. Resistance QTL in the RILs was defined by the presence of parental alleles of the closest marker loci for each QTL. The predicted means and associated standard errors of each genotype were calculated using the GLM procedure of SAS 9.2 (SAS Institute Inc., Cary, NC, USA) on DS datasets (YR/LR09, YR/LR10, and YR/LRAV) (Tables 3, 4).
With all YR DS datasets, significant differences were found between QTL genotypes (F prob. <0.001) based on one-way ANOVA. Following the ANOVA, pair-wise mean comparisons between all 16 QTL genotypes were carried out using least significant difference (LSD) statistics (Table 3). Among all QTL and datasets, QYr.tam-2D was the most effective in reducing DS (up to 50 %) compared to the no-QTL group. This QTL was observed to be as effective as the other three QTL combined. Similarly, QYr.tam-3B also significantly reduced DS in YR09 (up to 23 %) and YRAV (up to 17 %) datasets, compared to the no-QTL group. However, QYr.tam-3D and QYr.tam-1B were not significant in reducing DS in any of the three datasets. The effectiveness of QYr.tam-1B in reducing DS was significantly enhanced when combined with QYr.tam-3B or QYr.tam-3D. QYr.tam-2D expressed high resistance against YR when it was combined with one of the three QTL individually (~15 % DS) or all together (~5 % DS).
Similarly, when present individually, QLr.tam-1D and QLr.tam-1B effectively reduced LR DS (up to 65 and 70 %, respectively) in all three datasets, compared to the no-QTL group (Table 4). QLr.tam-3D significantly reduced DS only in LR10 (up to 40 %) and LRAV (up to 25 %) datasets. The effect of each LR QTL was more pronounced when in combination with others. RILs with all the three QTL combined displayed near immune response to LR.
Discussion
This study investigated the genetic control of APR to LR and YR in the CIMMYT spring wheat line Quaiu, using 2-year phenotypic data and molecular markers. QYr.tam-2D, which accounted for up to 54 % of variation in the RILs and played a major role in YR immunity to Quaiu, was designated as Yr54. QYr.tam-3B, with a relatively smaller effect, was mapped to the same position as the previously designated APR gene Yr30 (Singh et al. 2000; Suenaga et al. 2003). Similarly, QYr.tam-1B and QLr.tam-1B were co-located on the long arm of chromosome 1B, and most likely represent the well-characterized dual APR gene Lr46/Yr29 (Singh et al. 1998; William et al. 2003). Additionally, two new QTL, QYr.tam-3D and QLr.tam-3D, shared a common region on chromosome 3D, conferring YR and LR resistance and contributing to about 10 % of the phenotypic variation. QLr.tam-1DS represents the moderately effective R-gene Lr42, inherited in Quaiu from its Triticum tauschii progenitor (Sun et al. 2010; Basnet et al. 2013). This study further illustrates the complex architecture of APR gene expression when present alone or in combination with others, including a moderately effective R-gene.
Yr54, mapped on chromosome 2DL, was flanked by DArT markers wpt-667162 and wpt-667054 at a distance of <0.5 cM. Similarly, SSR marker Xgwm301, located on the distal end of 2DL in the wheat consensus map (Somers et al. 2004), was mapped 0.5 cM proximal to Yr54. Through cytogenetic analysis, the APR gene Yr16 was mapped near the centromere of chromosome 2D in Capelle–Desprez (Worland and Law 1986; Worland et al. 1988; Hart et al. 1993). Recently, Agenbag et al. (2012) mapped QYr.ufs-2D (Yr16) on chromosome 2DS in Capelle–Desprez. This QTL, defined by flanking markers Xgwm102 and wpt-664520, accounted for 10 % of the phenotypic variance. Several other studies have reported YR resistance near or within the same region as QYr.ufs-2D (Bariana et al. 2001; Mallard et al. 2005; Navabi et al. 2005; Melichar et al. 2008; Lu et al. 2009; Powell 2010). Small-effect YR QTL was also reported on 2DL by Suenaga et al. (2003) and Ren et al. (2012a) in cultivars Fukuho-komugi and Naxos, respectively. However, these QTL are unlikely to be similar to Yr54 because they were mapped approximately 20 cM from Yr54 and explained less variation (R 2 < 10 %), but, consistent with Yr54, Jagger et al. (2011) reported a QTL, QPst.jic-2D, derived from the German bread wheat cultivar Alcedo on chromosome 2DL. This QTL, defined by markers Xgwm320 and Xgwm301, explained 34–53 % of total YR variation measured by percentage infection and IT. The large proportion of variation explained by QPst.jic-2D and one of its interval defining markers was similar to that for Yr54, which explained 49–54 % of variation for DS and AUDPC. As there is no significant ancestral relationship between Quaiu and Alcedo (coefficient of % = 0.008), the allelic resemblance between Yr54 and QPst.jic-2D can only be speculated upon.
The YR QTL QYr.tam-3B was consistently detected with DS and AUDPC in both YR09 and YR10. This QTL was defined by markers Xgwm533 (2 cM), wpt-7984 (0.1 cM), and wpt-8446 (0.7 cM) on chromosome 3BS. The YR slow rusting gene Yr30, which is linked or pleiotropic to the SR slow rusting gene Sr2 and pseudo black chaff (PBC) phenotype, was reported on 3BS in several QTL mapping studies (Singh et al. 2000; Spielmeyer et al. 2003; Suenaga et al. 2003; Hayden et al. 2004; William et al. 2006; Khlestkina et al. 2007; Dedryver et al. 2009; Yu et al. 2010; Rosewarne et al. 2012). In those studies, 3BS loci (expected to be Sr2/Yr30) explained a wide range of variation (3.3–50 % for YR DS) and were mapped close to one or a few markers including Xgwm389 (2.7–5 cM), Xgwm533 (1.6–7 cM), Xgwm493 (2.5–6.6 cM), Xfba190 (1–2 cM), wpt-8446 (3 cM), and wpt-7984 (8.6 cM). All of those markers have been placed close to each other on 3BS across several wheat genetic maps. In our study, QYr.tam-3B explained 3–10 and 12–20 % of phenotypic variation in entire and Yr54-subtracted datasets, respectively. In addition, Quaiu displays a PBC phenotype, which has a strong association with Sr2 (Hare and McIntosh 1979). Based on this evidence, and the frequent occurrence of Sr2 and Yr30 in CIMMYT wheat germplasm, it is most likely that we mapped Yr30 as QYr.tam-3B in Quaiu. However, Lowe et al. (2011) and Bansal et al. (2010) reported other resistance loci, QYr.ucw-3BS and YrRub, which are closely linked with markers Xgwm533.1 and Xcfb3530, respectively, at the distal end of 3BS, and are reportedly different from Yr30. Further investigations with a larger population size and more molecular markers, which are beyond the scope of this study, are required to confirm the exact relationship between QYr.tam-3B, QYr.ucw-3BS, and YrRub.
The YR and LR resistance QTL, QYr.tam-1B and QLr.tam-1B, were detected on chromosome 1BL, in the region where the known APR gene Lr46/Yr29 is located (Singh et al. 1998; William et al. 2003). Both of these QTL were mapped 1 and 9 cM proximal to markers csLV46G22 and Xgwm140, respectively. In previous studies, Lr46/Yr29 was mapped within a maximum distance of 14 cM from the closely linked SSR markers Xgwm259, Xwmc044, and Xgwm140 in different bread wheat germplasm (Suenaga et al. 2003; Rosewarne et al. 2006; William et al. 2006; Lillemo et al. 2008; Melichar et al. 2008; Ren et al. 2012b). Moreover, strong associations of LR and YR responses with marker csLV46G22, a tightly linked Lr46/Yr29 marker developed and kindly provided by E. Lagudah, CSIRO, Australia, apparently support the presence of Lr46/Yr29 in Quaiu. The marker csLV46G22 was also used by Lillemo et al. (2011) to assess the effects of Lr46 with various additive YR resistance genes in Avocet/Saar and Avocet/Parula populations. Lillemo et al. (2011) suggested that the expression of Lr46/Yr29 is profoundly influenced by the genetic background of the host genotypes and their growing environments. Studies have shown different levels of phenotypic variance attributed to Lr46/Yr29 in different mapping populations (15–53 % for YR and 17–85 % for LR; William et al. 2003; Rosewarne et al. 2006, 2012; Lillemo et al. 2008). However, Lr46/Yr29 did not significantly reduce YR severity in cultivars Oligoculm (Suenaga et al. 2003) and Bainong 64 (Ren et al. 2012b). In this study, the maximum variation explained by QYr.tam-1B and QLr.tam-1B was 7 and 25 % in entire population datasets and 27 and 55 % after subtracting the large-effect QTL QYr.tam-2D and QLr.tam-1D, respectively.
Both QYr.tam-3D and QLr.tam-3D were defined by the proximally located markers wpt-672034 and Xbarc125 on chromosome 3DS based on integrated genetic and physical wheat maps (http://wheat.pw.usda.gov/ggpages/SSRclub/GeneticPhysical). Similar to QYr.tam-3D, Dedryver et al. (2009) have reported a small QTL, QYr.inra-3DS, flanked by markers Xbarc125 and Xgwm456 on 3DS, responsible for slow rusting resistance to YR. QYr.inra-3DS, derived from cultivar Recital, explained up to 12 % of the phenotypic variation for YR under field conditions. Similarly, Singh et al. (2000) and Boukhatem et al. (2002) reported YR resistance QTL linked to markers XksuA6 and Xbcd1532 that explained 12–14 % of the variation in the cultivar Opata 85/Synthetic population. In the wheat integrated map, marker Xbcd1532 is located 13–16 cM distal to SSR marker Xbarc125. Although it is possible that the 3DS QTL in Quaiu, Recital, and Opata might have common allelic origins, further investigation is required for confirmation. However, there is no report of a LR QTL in the region of QLr.tam-3D in Quaiu.
Recently, Sun et al. (2010) mapped Lr42 on the short arm of chromosome 1D, close to markers Xwmc432 (0.8 cM) and Xcfd15 (1.6 cM) in line KS93U50, a selection from KS91WGRC11. Based on LR seedling tests, Basnet et al. (2013) found that Quaiu inherited Lr42 that segregated in Avocet/Quaiu RILs in close proximity to the SSR marker Xwmc432. In our study, Lr42 was represented as QLr.tam-1D, which significantly reduced LR severity in the field and was mapped between DArT markers wpt-666067 (2 cM) and wpt-667180 (0.6 cM), and close to SSR marker Xwmc432 (1 cM). Other LR genes on chromosome 1DS, Lr21 (Huang et al. 2003) and Lr60 (Hiebert et al. 2008), are located approximately 23 and 16 cM, respectively, distal to QLr.tam-1D based on the consensus map of Somers et al. (2004). As virulence to Lr42 is not yet reported, use of this R-gene in conjunction with other LR APR might be useful in achieving a higher level of protection in the field.
The susceptible parent Avocet was found to contribute YR resistance that was detected inconsistently across experiments in sub-optimal LOD thresholds. Most of these minor QTL were detected in YR10, where Avocet displayed lower DS (70 %), compared to 100 % for the most susceptible RILs. It is likely that in the presence of other larger QTL, the Avocet QTL contributes only a small effect and can easily be missed, especially when disease pressure is very high. Previous studies showed that QTL that contribute <10 % to phenotypic variance are more prone to environmental influence or disease pressure, and hence their detection becomes more difficult across years and environments (Boukhatem et al. 2002). QYr.tam-1A was detected with DS and AUDPC in YR10 on chromosome 1AS. Based on common DArT markers, the location of QYr.tam-1A corresponds with a YR QTL, within the LDb1 region of 1AS, in the association study by Crossa et al. (2007). However, other 1A QTL reported by Ramburan et al. (2004), Bariana et al. (2010), and Prins et al. (2011) appear to be different from QYr.tam-1A, based on relative marker positions. QYr.tam-4A was detected in YR10 and mapped on 4AL, similar to that of the previously reported Avocet QTL, QYr.sgi-4A.2 (Ramburan et al. 2004; Prins et al. 2011). Similar QTL on 4AL have also been reported in other wheat germplasm by Crossa et al. (2007) and Vazquez et al. (2012). QYr.tam-6A, another Avocet-derived QTL on chromosome 6AL, was also previously reported (Singh et al. 2001; Singh et al. 2005; Lillemo et al. 2006; William et al. 2006; Prins et al. 2011). Prins et al. (2011) also mapped LR and SR resistance (most likely the Agropyrum elongatum-derived SR gene Sr26) within the same region as the YR QTL QYr.ufs-6A. Consistent with this finding, Singh et al. (2005) observed that the A. elongatum segment carrying Sr26 also reduced LR and YR severities in Avocet/Pavon and Avocet/Tonichi 81 populations. However, QYr.ufs-6A did not show any effect on LR resistance in our study. Another Avocet-derived small QTL, QYr.tam-3D.1, was evident in YR10 at a LOD peak of 2.8–3.0. It was placed between markers Xwmc552 and Xcfd223 on chromosome 3DL. This QTL might be a new one, as no previous reports on YR QTL similar to QYr.tam-3D.1 are available to date. It was interesting to see that several minor QTL were harbored by the susceptible parent Avocet, an Australian reselection lacking the R-gene YrA. Ramburan et al. (2004) argued that the APR to YR might have been retained during the selection of susceptible Avocet under conditions where reduced susceptibility could not be detected.
In contrast to other Avocet-derived minor QTL, QYr.tam-6B was from Quaiu and located on the long arm only when Yr54-subtracted datasets were used for analysis. Rosewarne et al. (2012) reported a 6BL QTL effective against both LR and YR in Pastor. The interval defining markers of Pastor 6BL (wpt-5176) and Quaiu QYr.tam-6B (wpt-4164) were placed within 2 cM in the Avocet/Quaiu genetic linkage map. Similarly, William et al. (2006) reported a QTL on 6BL that was effective against both LR and YR in Pavon76. In an association analysis, Crossa et al. (2007) also reported a linkage disequilibrium block at the distal end of chromosome 6B (LDb6), which shares some common DArT markers with QYr.tam-6B, significantly associated with YR resistance.
The disease response of RILs in the presence or absence of a particular QTL allowed assessment of its effects in conferring resistance, assuming that remaining QTL was randomly distributed. Among all QTL, Yr54 revealed a highly skewed distribution of RILs with maximum DS of 40 %. On the other hand, RILs with one of the other QTL (QYr.tam-1B, QYr.tam-3B, or QYr.tam-3D) were distributed across a wide range of DS (20–100 %), although skewed distributions were evident. Unexpectedly, a few RILs with these QTL displayed high DS (90–100 %). It is possible that either the lines were not discriminated properly compared to more susceptible ones under high disease conditions or were misclassified into the +QTL group by the markers. Similarly, for LR, the distributions of QLr.tam-1D and QLr.tam-1B RILs were distinctly skewed towards resistance, displaying a maximum DS of 40 %. However, the small-effect QTL QLr.tam-3D was not discriminated substantially among the RILs. Non-random assortment of undetected small-effect QTL might have caused the overlapping distribution between RILs with or without QLr.tam-3D. Similarly, grouping of RILs in QTL genotypes provided an assessment, in terms of means and variances, of each QTL or their combination whilst controlling for others (Tables 3, 4). When present alone, Yr54 was the only resistance gene that significantly reduced YR severity, compared to the no-QTL RILs (Online Resource 2). However, additive effects were observed, with significant disease reductions, when two or more QTL were combined together, except for the combination of QYr.tam-3B and QYr.tam-3D. It is worth mentioning that the grouping of RILs into different QTL genotypes was based on four consistently detected QTL. For this reason the average DS for the no-QTL group (81 %) was significantly lower than that of the most susceptible RILs, which displayed up to 100 % DS. Compared with 100 % as the control, the average DS of the three QTL genotypes, QYr.tam-1B (71 %), QYr.tam-3B (63 %), and QYr.tam-3D (66 %), were significantly lower. Similarly, QLr.tam-1D and QLr.tam-1B, when present alone, significantly reduced LR severity in all experiments. In contrast, QLr.tam-3D reduced DS significantly only in the LR10 and LRAV datasets. The additive effects among LR QTL were very pronounced on DS; the combination of two QTL in the RILs resulted in significantly lower DS (5–20 %), whereas all three QTL together led to near immunity.
A significant segregation distortion of markers around the Yr54 region might have resulted from unknown segregation distortion loci, which are normally associated with differential viability of gametes or zygotes (Lorieux et al. 1995; Xu 2008). Segregation distortion is a common phenomenon in the wheat genome, and has been reported to be closely linked with several genes and translocated segments (Kong et al. 2009; Tsilo et al. 2010; Cai et al. 2011). It is unlikely that the Yr54 region represents an alien translocation because significant numbers of true recombinants were observed between markers in the distorted region. Studies have suggested that segregation distortion does not cause significant impacts on QTL detection and their effect estimations, and hence can be ignored during QTL mapping (Xu 2008; Zhang et al. 2010).
In the Avocet/Quaiu RIL population, up to 60 and 70 % of the phenotypic variation was explained by detected LR and YR QTL, respectively. However, an enormous proportion of that variation was attributed to the large-effect loci QLr.tam-1D or Lr42 (25–35 %) and Yr54 (49–61 %). This study further demonstrates that, in the presence of large QTL, the effects of smaller ones are overshadowed and can remain undetected. QTL analysis using subtracted datasets is a better strategy for improving the detection of minor QTL. With Yr54-subtracted datasets, two additional QTL (QYr.tam-6A and QYr.tam-6B) were detected, along with the increased variation explained by the previously detected QTL. Similarly, increased variation of LR QTL was observed in QLr.tam-1D-subtracted datasets, although no new QTL was detected. Because 40 and 30 % of the variation of LR and YR responses, respectively, remained unexplained, it can be assumed that additional minor QTL were not detected in the Avocet/Quaiu population. Rosewarne et al. (2012) reported 8 and 13 QTL (maximum variation of 21 %) associated with LR and YR severity reduction, respectively, in the Avocet/Pastor population. Among the significant QTL, seven (four YR and three LR) were derived from the susceptible parent Avocet. Only one Avocet QTL from Avocet/Pastor, QYr.tam-6A, was common to the Avocet/Quaiu population. In previous studies, two loci similar to QYr.tam-6A and QYr.tam-6B were reported to be effective against both YR and LR (William et al. 2006; Rosewarne et al. 2012). Many factors affect QTL detection, and some that might have had an effect on the Avocet/Quaiu population include limited population size, few molecular markers, discrepancies in QTL effects, epistasis among QTL, overall environmental influences, and variation in aggressiveness or virulence in pathogen races used.
Good agronomic characteristics and high APR to LR and YR make Quiau and its derivatives important parents in the bread wheat improvement program at CIMMYT. Thus we can speculate that Yr54, QLr.tam-1B/QYr.tam-1B, and QLr.tam-3D/QYr.tam-3D should have a significant presence in advanced CIMMYT lines that are being distributed throughout the world. With the availability of defined molecular markers for each resistance locus, multiple APR pyramiding from the single source parent Quaiu should be easier and more efficient in the future. Two RILs (CIMMYT GID: 6032209 and 6032334), which possess only Yr54, are available upon request for research purposes.
References
Agenbag GM, Pretorius ZA, Boyd LA, Bender CM, Prins R (2012) Identification of adult plant resistance to stripe rust in the wheat cultivar Capelle–Desprez. Theor Appl Genet 125:109–120
Akbari M, Wenzl P, Caig V, Carling J, Xia L, Yang S, Uszynski G, Mohler V, Lehmensiek A, Kuchel H, Hayden M, Howes N, Sharp P, Vaughan P, Rathmell B, Huttner E, Kilian A (2006) Diversity arrays technology (DArT) for high-throughput profiling of the hexaploid wheat genome. Theor Appl Genet 113:1409–1420
Bansal UK, Bariana HS, Gill MB, Hayden MJ (2010) Chromosomal location of an uncharacterized stripe rust resistance gene in wheat. Euphytica 171:121–127
Bariana HS, Hayden MJ, Ahmed NU, Bell JA, Sharp PJ, McIntosh RA (2001) Mapping of durable adult plant and seedling resistance to stripe rust and stem rust diseases in wheat. Aust J Agric Res 52:1247–1255
Bariana HS, Bansal UK, Schmidt A, Lehmensiek A, Kaur J, Miah H, Howes N, McIntyre CL (2010) Molecular mapping of adult plant stripe rust resistance in wheat and identification of pyramided QTL genotypes. Euphytica 176:251–260
Basnet BR (2012) Molecular characterization of durable yellow and leaf rust resistance in two wheat populations. PhD dissertation, Texas A&M University, College Station, TX
Basnet BR, Singh RP, Herrera-Foessel SA, Ibrahim AMH, Huerta-Espino J, Calvo-Salazar V, Rudd JC (2013) Genetic analysis of adult plant resistance to yellow rust and leaf rust in common spring wheat ‘Quaiu 3’. Plant Dis 97:728–736
Bayles RA, Flath K, Hovmøller MS, de Vallavieille-Pope C (2000) Breakdown of the Yr17 resistance to yellow rust of wheat in northern Europe. Agronomie 20:805–811
Bjarko ME, Line RF (1988) Heritability and number of genes controlling leaf rust resistance in four cultivars of wheat. Phytopathology 78:457–461
Bolton MD, Kolmer JA, Garvin DF (2008) Wheat leaf rust caused by Puccinia triticina. Mol Plant Pathol 9:563–575
Boukhatem N, Baret PV, Mingeot D, Jacquemin JM (2002) Quantitative trait loci for resistance against yellow rust in two wheat-derived recombinant inbred line populations. Theor Appl Genet 104:111–118
Cai J, Zhang X, Wang B, Yan M, Qi Y, Kong L (2011) A genetic analysis of segregation distortion revealed by molecular markers in Lophopyrum ponticum chromosome 7E. J Genet 90:373–376
Chen XM (2005) Epidemiology and control of stripe rust [Puccinia striiformis f sp. tritici] on wheat. Can J Plant Pathol 27:314–337
Chen XM (2007) Challenges and solutions for stripe rust control in the United States. Aust J Agric Res 58:648–655
Cox TS, Raupp WJ, Gill BS (1994) Leaf rust-resistance genes Lr41, Lr42, and Lr43 transferred from Triticum tauschii to common wheat. Crop Sci 34:339–343
Crossa J, Burgueño J, Dreisigacker S, Vargas M, Herrera-Foessel SA, Lillemo M, Singh RP, Trethowan R, Warburton M, Franco J, Reynolds M, Crouch JH, Ortiz R (2007) Association analysis of historical bread wheat germplasm using additive genetic covariance of relatives and population structure. Genetics 177:1889–1913
Dedryver F, Paillard S, Mallard S, Robert O, Trottet M, Nègre S, Verplancke G, Jahier J (2009) Characterization of genetic components involved in durable resistance to stripe rust in the bread wheat ‘Renan’. Phytopathology 99:968–973
Dubin HJ, Brennan JP (2009) Combating stem and leaf rust of wheat: historical perspective, impacts, and lessons learned. International Food Policy Research Institute (IFPRI), Washington, DC. http://www.ifpri.org/sites/default/files/publications/ifpridp00910.pdf
Dyck PL (1987) The association of a gene for leaf rust resistance with the chromosome 7D suppressor of stem rust resistance in common wheat. Genome 29:467–469
Fu D, Uauy C, Distelfeld A, Blechl A, Epstein L, Chen X, Sela H, Fahima T, Dubcovsky J (2009) A kinase-START gene confers temperature-dependent resistance to wheat stripe rust. Science 323:1357–1360
Hare RA, McIntosh RA (1979) Genetic and cytogenetic studies of durable adult-plant resistance in ‘Hope’ and related cultivars to wheat rusts. Z Pflanzenzuchtg 83:35–367
Hart GE, Gale MD, McIntosh RA (1993) Linkage map of Triticum aestivum (hexaploid wheat, 2n = 42, genomes A, B and D) and T tauschii (2n = 14, genome D). In: O’Brien SJ (ed) Genetic maps: locus maps of complex genomes. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, pp 6204–6219
Hayden MJ, Kuchel H, Chalmers KJ (2004) Sequence tagged microsatellites for the Xgwm533 locus provide new diagnostic markers to select for the presence of stem rust resistance gene Sr2 in bread wheat (Triticum aestivum L.). Theor Appl Genet 109:1641–1747
Herrera-Foessel S, Lagudah E, Huerta-Espino J, Hayden M, Bariana H, Singh D, Singh R (2011) New slow-rusting leaf rust and stripe rust resistance genes Lr67 and Yr46 in wheat are pleiotropic or closely linked. Theor Appl Genet 122:239–249
Herrera-Foessel S, Singh R, Huerta-Espino J, Rosewarne G, Periyannan S, Viccars L, Calvo-Salazar V, Lan C, Lagudah E (2012) Lr68: a new gene conferring slow rusting resistance to leaf rust in wheat. Theor Appl Genet 124:1475–1486
Hiebert CH, Thomas JB, McCallum BD, Somers DJ (2008) Genetic mapping of the wheat leaf rust resistance gene Lr60 (LrW2). Crop Sci 48:1020–1026
Hiebert CW, Thomas JB, McCallum BD, Humphreys DG, Depauw RM, Hayden MJ, Mago R, Schnippenkoetter W, Spielmeyer W (2011) An introgression on wheat chromosome 4DL in RL6077 (Thatcher*6/PI 250413) confers adult plant resistance to stripe rust and leaf rust (Lr67). Theor Appl Genet 121:1083–1091
Huang L, Brooks SA, Li W, Fellers JP, Trick HN, Gill BS (2003) Map-based cloning of leaf rust resistance gene Lr21 from the large and polyploidy genome of bread wheat. Genetics 164:655–664
ICARDA (2011) Research to action—strategies to reduce the emerging wheat stripe rust disease. International wheat stripe rust symposium, Aleppo, Syria. ISBN: 92-9127-237-X. www.icarda.org/wheatrust
Jaccoud D, Peng K, Feinstein D, Kilian A (2001) Diversity arrays: a solid state technology for sequence information independent genotyping. Nucleic Acids Res 29:E25
Jagger L, Newell C, Berry S, MacCormack R, Boyd L (2011) The genetic characterization of stripe rust resistance in the German wheat cultivar Alcedo. Theor Appl Genet 122:723–733
Khlestkina EK, Röder MS, Unger O, Meinel A, Börner A (2007) More precise map position and origin of a durable non-specific adult plant disease resistance against stripe rust (Puccinina striiformis) in wheat. Euphytica 153:1–10
Kolmer JA, Chen X, Jin Y (2009) Diseases which challenge global wheat production—the wheat rusts. In: Carver BF (ed) Wheat science and trade. Wiley, Ames, pp 89–124
Kong L, Anderson JM, Ohm HW (2009) Segregation distortion in common wheat of a segment of Thinopyrum intermedium chromosome 7E carrying Bdv3 and development of a Bdv3 marker. Plant Breed 128:591–597
Kosambi D (1944) The estimation of map distances from recombination values. Ann Eugen 12:172–175
Krattinger SG, Lagudah ES, Spielmeyer W, Singh RP, Huerta-Espino J, McFadden H, Bossolini E, Selter LL, Keller B (2009) A putative ABC transporter confers durable resistance to multiple fungal pathogens in wheat. Science 323:1360–1363
Li H, Ribaut J-M, Li Z, Wang J (2008) Inclusive composite interval mapping (ICIM) for digenic epistasis of quantitative traits in biparental populations. Theor Appl Genet 116:243–260
Lillemo M, Skinnes H, Singh RP, van Ginkel M (2006) Genetic analysis of partial resistance to powdery mildew in bread wheat line Saar. Plant Dis 90:225–228
Lillemo M, Asalf B, Singh R, Huerta-Espino J, Chen X, He Z, Bjørnstad A (2008) The adult plant rust resistance loci Lr34/Yr18 and Lr46/Yr29 are important determinants of partial resistance to powdery mildew in bread wheat line Saar. Theor Appl Genet 116:1155–1166
Lillemo M, Singh RP, William M, Herrera-Foessel S, Huerta-Espino J, German S, Campos P, Chaves M, Madriaga R, Xia XC, Liang SS, Liu D, Li ZF, Lagudah ES (2011) Multiple rust resistance and gene additivity in wheat: lessons from multi-location case studies in cultivars Parula and Saar. In: McIntosh R (ed) Proc borlaug global rust initiative technical workshop, Saint Paul, Minnesota, USA, pp 111–120. ISBN: 978-0-615-54519-6
Lorieux M, Goffinet B, Perrier X, Gonzalez de Leon D, Lanaud C (1995) Maximum-likelihood models for mapping genetic markers showing segregation distortion. 1. Backross populations. Theor Appl Genet 90:70–80
Lowe I, Jankuloski L, Chao S, Chen X, See D, Dubcovsky J (2011) Mapping and validation of QTL which confer partial resistance to broadly virulent post-2000 North American races of stripe rust in hexaploid wheat. Theor Appl Genet 123:143–157
Lu Y, Lan C, Liang S, Zhou X, Liu D, Zhou G, Lu Q, Jing J, Wang M, Xia X, He Z (2009) QTL mapping for adult-plant resistance to stripe rust in Italian common wheat cultivars Libellula and Strampelli. Theor Appl Genet 119:1349–1359
Mallard S, Gaudet D, Aldeia A, Abelard C, Besnard AL, Sourdille P, Dedryver F (2005) Genetic analysis of durable resistance to yellow rust in bread wheat. Theor Appl Genet 110:1401–1409
Mcintosh RA (1992) Close genetic linkage of genes conferring adult-plant resistance to leaf rust and stripe rust in wheat. Plant Pathol 41:523–527
McIntosh RA, amazaki YY, Dubcovsky J, Rogers WJ, Morris C, Somers DJ, Appels R, Devos KM (2012) MacGene 2012: catalogue of gene symbols for wheat. http://www.shigen.nig.ac.jp/ wheat/komugi/genes/download.jsp. Accessed 4 Aug 2013
Melichar JPE, Berry S, Newell C, MacCormack R, Boyd LA (2008) QTL identification and microphenotype characterisation of the developmentally regulated yellow rust resistance in the UK wheat cultivar Guardian. Theor Appl Genet 117:391–399
Navabi A, Tewari JP, Singh RP, McCallum B, Laroche A, Briggs KG (2005) Inheritance and QTL analysis of durable resistance to stripe and leaf rusts in an Australian cultivar, Triticum aestivum ‘Cook’. Genome 48:97–107
Naz AA, Kunert A, Lind A, Pillen P, Léon J (2008) AB-QTL analysis in winter wheat: II. Genetic analysis of seedling and field resistance against leaf rust in a wheat advanced backcross population. Theor Appl Genet 116:1095–1104
Park RF, Bariana HS, Wellings CR, Wallwork H (2002) Detection and occurrence of a new pathotype of Puccinia triticina with virulence for Lr24 in Australia. Aust J Agric Res 53:1069–1076
Pestsova E, Ganal MW, Roder MS (2000) Isolation and mapping of microsatellite markers specific for the D genome of bread wheat. Genome 43:689–697
Peterson RF, Campbell AB, Hannah AE (1948) A diagrammatic scale for estimating rust severity on leaves and stems of cereals. Can J Res 26:496–500
Powell NM (2010) Phenotyping and genetic analysis of yellow rust resistance in the UK winter wheat cultivar Claire. PhD dissertation, University of East Anglia, Norwich, UK
Prins R, Pretorius Z, Bender C, Lehmensiek A (2011) QTL mapping of stripe, leaf and stem rust resistance genes in a Kariega × Avocet S doubled haploid wheat population. Mol Breed 27:259–270
Ramburan VP, Pretorius ZA, Louw JH, Boyd LA, Smith PH, Boshoff WH, Prins R (2004) A genetic analysis of adult plant resistance to stripe rust in the wheat cultivar Kariega. Theor Appl Genet 108:1426–1433
Ren Y, He Z, Li J, Lillemo M, Wu L, Bai B, Lu Q, Zhu H, Zhou G, Du J, Lu Q, Xia X (2012a) QTL mapping of adult plant resistance to stripe rust in a population derived from common wheat cultivars Naxos and Shanghai 3/Catbird. Theor Appl Genet 125:1211–1221
Ren Y, Li Z, He Z, Wu L, Bai B, Lan C, Wang C, Zhou G, Zhu H, Xia X (2012b) QTL mapping of adult-plant resistance to stripe rust and leaf rust in Chinese wheat cultivar Bainong 64. Theor Appl Genet 125:1253–1262
Röder MS, Korzun V, Wendehake K, Plaschke J, Tixier MH, Leroy P, Ganal MW (1998) A microsatellite map of wheat. Genetics 149:2007–2023
Rosewarne GM, Singh RP, Huerta-Espino J, William HM, Bouchet S, Cloutier S, McFadden H, Lagudah ES (2006) Leaf tip necrosis, molecular markers and β1-proteasome subunits associated with the slow rusting resistance genes Lr46/Yr29. Theor Appl Genet 112:500–508
Rosewarne G, Singh RP, Huerta-Espino J, Herrera-Foessel S, Forrest K, Hayden M, Rebetzke G (2012) Analysis of leaf and stripe rust severities reveals pathotype changes and multiple minor QTLs associated with resistance in an Avocet × Pastor wheat population. Theor Appl Genet 124:1283–1294
Singh RP (1992) Genetic association of leaf rust resistance gene Lr34 with adult-plant resistance to stripe rust in bread wheat. Phytopathology 82:835–838
Singh RP (2012) Pros and cons of utilizing major race-specific resistance genes versus partial resistance in breeding rust resistant wheat. In: The BGRI technical workshop, September 1–4, 2012, Beijing, China. http://www.globalrust.org/db/attachments/bgriworkshop/14/1/singh_web.pdf
Singh RP, Mujeeb-Kazi A, Huerta-Espino J (1998) Lr46: a gene conferring slow-rusting resistance to leaf rust in wheat. Phytopathology 88:890–894
Singh RP, Sorrells ME, Nelson JC (2000) Mapping of Yr28 and other genes for resistance to stripe rust in wheat. Crop Sci 40:1148–1155
Singh RP, Huerta-Espino J, William M (2001) Slow rusting gene based resistance to leaf and yellow rusts in wheat: genetics and breeding at CIMMYT. In: Proceedings of the 10th assembly of the Wheat Breeding Society of Australia Inc., Mildura, Australia, 16–21 September 2001, pp 103–108
Singh RP, Duvillier E, Huerta-Espino J (2004) Virulence to yellow rust resistance gene Yr27: In: A new threat to stable wheat production in Asia (Abs). Second regional yellow rust conference for CWANA, Islamabad, Pakistan 22–26 March, 2004
Singh RP, Huerta-Espino J, William HM (2005) Genetics and breeding for durable rust resistance to leaf and stripe rusts in wheat. Turk J Agric For 29:121–127
Singh RP, Huerta-Espino J, Bhavani S, Herrera-Foessel S, Singh D, Singh P, Velu G, Mason R, Jin Y, Njau P, Crossa J (2011) Race non-specific resistance to rust diseases in CIMMYT spring wheats. Euphytica 179:175–186
Singh D, Mohler V, Park RF (2013) Discovery, characterisation and mapping of wheat leaf rust resistance gene Lr71. Euphytica 190:131–136
Somers D, Isaac P, Edwards K (2004) A high-density microsatellite consensus map for bread wheat (Triticum aestivum L). Theor Appl Genet 109:1105–1114
Song QJ, Fickus EW, Cregan PB (2002) Characterization of trinucleotide SSR motifs in wheat. Theor Appl Genet 104:286–293
Sourdille P, Tavaud M, Charmet G, Bernard M (2001) Transferability of wheat microsatellite to diploid Triticeae species carrying the A, B and D genomes. Theor Appl Genet 103:346–352
Spielmeyer W, Lagudah ES, Sharp PJ (2003) Identification and validation of markers linked to broad-spectrum stem rust resistance gene Sr2 in wheat (Triticum aestivum L). Crop Sci 43:333–336
Spielmeyer W, McIntosh RA, Kolmer J, Lagudah ES (2005) Powdery mildew resistance and Lr34/Yr18 genes for durable resistance to leaf and stripe rust cosegregate at a locus on the short arm of chromosome 7D of wheat. Theor Appl Genet 111:731–735
Stubbs RW (1985) Stripe rust. In: Roelfs AP, Bushnell WR (eds) The cereal rusts, vol II. Academic Press, Orlando
Suenaga K, William HM, Huerta-Espino J, Singh RP (2003) Microsatellite markers for genes Lr34/Yr18 and other quantitative trait loci for leaf rust and stripe rust resistance in bread wheat. Phytopathology 93:881–890
Sun X, Bowden R, Carver BF, Bai G (2010) Molecular mapping of wheat leaf rust resistance gene Lr42. Crop Sci 50:59–66
Tsilo TJ, Jin Y, Anderson JA (2010) Identification of flanking markers for the stem rust resistance gene Sr6 in wheat. Crop Sci 50:1967–1970
Vazquez MD, Peterson CJ, Riera-Lizarazu O, Chen X, Heesacker A, Ammar K, Crossa J, Mundt CC (2012) Genetic analysis of adult plant, quantitative resistance to stripe rust in wheat cultivar ‘Stephens’ in multi-environment trials. Theor Appl Genet 124:1–11
Wan A, Zhao Z, Chen X, He Z, Jin S, Jia Q, Yao G, Yang J, Wang B, Li G, Bi Y, Yuan Z (2004) Wheat stripe rust epidemic and virulence of Puccinia striiformis f sp tritici in China in 2002. Plant Dis 88:896–904
Wang S, Basten CJ, Zeng Z-B (2011) Windows QTL cartographer v2.5. Department of Statistics, North Carolina State University, Raleigh, NC. http://statgen.ncsu.edu/qtlcart/WQTLCart.htm
Wellings CR, Singh RP, McIntosh RA, Pretorius ZA (2004) The development and application of near isogenic lines for the wheat stripe (yellow) rust pathosystem. In: Proceedings of the 11th cereal rusts and powdery mildews conference, Norwich, UK, August 2004, p 39
William M, Singh RP, Huerta-Espino J, Islas SO, Hoisington D (2003) Molecular marker mapping of leaf rust resistance gene Lr46 and its association with stripe rust resistance gene Yr29 in wheat. Phytopathology 93:153–159
William HM, Singh RP, Huerta-Espino J, Palacios G, Suenaga K (2006) Characterization of genetic loci conferring adult plant resistance to leaf and stripe rust in spring wheat. Genome 49:977–990
Worland AJ, Law CN (1986) Genetic analysis of chromosome 2D of wheat I: the location of genes affecting height, day-length insensitivity, hybrid dwarfism and yellow-rust resistance. Z Pflanzenzuchtg 96:331–345
Worland AJ, Petrovic S, Law CN (1988) Genetic analysis of chromosome 2D of wheat. Plant Breed 100:247–259
Xu S (2008) Quantitative trait locus mapping can benefit from segregation distortion. Genetics 180:2201–2208
Xu LS, Wang MN, Cheng P, Kang ZS, Hulbert SH, Chen XM (2013) Molecular mapping of Yr53, a new gene for stripe rust resistance in durum wheat accession PI 480148 and its transfer to common wheat. Theor Appl Genet 126:523–533
Yu L-X, Liu S, Anderson JA, Singh RP, Jin Y, Dubcovsky J, Brown-Guidera G, Bhavani S, Morgounov A, He Z, Huerta-Espino J, Sorrells ME (2010) Haplotype diversity of stem rust resistance loci in uncharacterized wheat lines. Mol Breed 26:667–680
Zhang L, Wang S, Li H, Deng Q, Zheng A, Li S, Li P, Li Z, Wang J (2010) Effects of missing marker and segregation distortion on QTL mapping in F2 populations. Theor Appl Genet 121:1071–1082
Acknowledgments
The authors thank Monsanto’s Beachell-Borlaug International Scholars Program (MBBISP) and SAGARPA-CONACYT of Mexico (Fondo Sectorial Project 146788) for funding. This study represents a part of the first author’s PhD dissertation researches completed at Texas A&M University, Texas, USA. Similarly, we thank Ms. Emma Quilligan for thorough English editing of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Basnet, B.R., Singh, R.P., Ibrahim, A.M.H. et al. Characterization of Yr54 and other genes associated with adult plant resistance to yellow rust and leaf rust in common wheat Quaiu 3. Mol Breeding 33, 385–399 (2014). https://doi.org/10.1007/s11032-013-9957-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11032-013-9957-2