Abstract
Diabetes mellitus is one of the major causes of ischemic and nonischemic heart failure. While hypertension and coronary artery disease are frequent comorbidities in patients with diabetes, cardiac contractile dysfunction and remodeling occur in diabetic patients even without comorbidities, which is referred to as diabetic cardiomyopathy. Investigations in recent decades have demonstrated that the production of reactive oxygen species (ROS), impaired handling of intracellular Ca2+, and alterations in energy metabolism are involved in the development of diabetic cardiomyopathy. AMP deaminase (AMPD) directly regulates adenine nucleotide metabolism and energy transfer by adenylate kinase and indirectly modulates xanthine oxidoreductase-mediated pathways and AMP-activated protein kinase-mediated signaling. Upregulation of AMPD in diabetic hearts was first reported more than 30 years ago, and subsequent studies showed similar upregulation in the liver and skeletal muscle. Evidence for the roles of AMPD in diabetes-induced fatty liver, sarcopenia, and heart failure has been accumulating. A series of our recent studies showed that AMPD localizes in the mitochondria-associated endoplasmic reticulum membrane as well as the sarcoplasmic reticulum and cytosol and participates in the regulation of mitochondrial Ca2+ and suggested that upregulated AMPD contributes to contractile dysfunction in diabetic cardiomyopathy via increased generation of ROS, adenine nucleotide depletion, and impaired mitochondrial respiration. The detrimental effects of AMPD were manifested at times of increased cardiac workload by pressure loading. In this review, we briefly summarize the expression and functions of AMPD in the heart and discuss the roles of AMPD in diabetic cardiomyopathy, mainly focusing on contractile dysfunction caused by this disorder.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
AMP deaminase (AMPD) plays important roles in energy metabolism, nucleotide pool size regulation, and amino acid metabolism [1,2,3] (Fig. 1). Deamination of AMP by AMPD, which lowers AMP level, keeps the adenylate kinase (AK) reaction proceeding in the direction of ATP generation from ADP (i.e., 2ADP \(\leftrightharpoons \) ATP + AMP), while it reduces the adenine nucleotide level. The AK reaction also maintains low ADP level, preserving a high ATP/ADP ratio and thus free energy of ATP hydrolysis (∆GATP = ∆G0ATP + RT ln([ADP][Pi]/[ATP]). AMPD competes with cytosolic 5’-nucleotidase for AMP, modulating the level of adenosine, which serves as a substrate of nucleotide synthesis and a ligand of P2 receptors. AMP deamination is an irreversible step in the purine nucleoside cycle (PNC), which supplies citric acid cycle intermediates and regenerates AMP from IMP. These functions of AMPD have been analyzed mostly in skeletal muscle in which AMPD activity is much higher than that in other tissues [4, 5]. AMPD shares its substrate, AMP, with AMP-activated protein kinase (AMPK), and there is an apparent reciprocal relationship between AMPD and AMPK activities [6,7,8,9,10,11,12,13,14]. AMPD in skeletal muscle has received attention for its roles in exercise endurance [3], myopathy [15], and muscle atrophy [16] and for its manipulation to improve insulin sensitivity via AMPK activation [17,18,19]. In contrast, the pathophysiological role of AMPD in the heart is poorly understood.
Overview of the metabolic pathways directly and indirectly regulated by AMP deaminase. Solid lines and dotted lines indicate metabolic pathways and regulatory pathways, respectively. A recent study suggests that there is mutual negative regulation between BCKDH and AMPD3 in cardiomyocytes [38]. AK adenylate kinase, AMPD AMP deaminase, AMPK AMP-activated protein kinase, ADSL adenylosuccinate lyase, ADSS adenylosuccinate synthase, BCKDH branched-chain α-keto acid dehydrogenase, 5′-NT 5′-nucleotidase, XOR xanthine oxidoreductase, Adeno adenosine, Ino inosine, sAMP adenylosuccinate. The figure was created with Biorender.com
Diabetes mellitus is characterized by dysfunctional insulin-mediated signaling and an altered profile of energy substrates to cells, which modulates mechanistic target of rapamycin complex (mTORC) signaling, leading to adaptation and/or maladaptation in energy production. As the shortage of ATP is sensed in cells through the elevation of AMP levels [20], it is conceivable that AMPD-mediated pathways are modified in diabetes. In fact, significantly increased levels of AMPD activity in the hearts of diabetic animal models were first reported more than 30 years ago [21], and the diabetes-induced change in AMPD activity in the heart and other organs was confirmed by animal models of type 1 and type 2 diabetes [9, 22,23,24] but not in a model of short-term diabetes [25] (Table 1). Unfortunately, the pathophysiological roles of AMPD upregulation in diabetes and insulin resistance have not been explored until recently [7, 9, 13, 24, 26, 27], while significant progress has been made in our knowledge regarding the mechanisms of diabetes-induced heart failure, fatty liver, and sarcopenia [28,29,30,31,32,33,34,35]. However, the results of a series of studies from this laboratory [23, 36,37,38,39] support the notion that AMPD upregulation plays an important role in diabetic cardiomyopathy as it does in diabetes-induced sarcopenia and fatty liver [7, 9, 13, 24, 26]. In this article, we briefly review the functions and regulations of AMPD and its contribution to diabetic cardiomyopathy. We mainly focused on the roles of AMPD in diastolic dysfunction at an early stage of diabetic cardiomyopathy since mechanisms in the early stage possibly trigger those that lead to subsequent systolic dysfunction and adverse cardiac remodeling.
Expression and function of AMPD in the heart
AMPD expression and activity regulation in the heart
There are three main isoforms of AMPD in human and rat tissues: isoform A (rat) or M (human) in skeletal muscle, isoform B (rat) or L (human) in the liver, and isoform C (rat) or E (human) in erythrocytes and the heart. While AMPD A/M isoform expression is primarily restricted to skeletal muscle, the B/L isoform and C/E isoform are expressed in many tissues. AMPD1, AMPD2, and AMPD3 genes encode AMPD A/M, AMPD B/L, and AMPD C/E, respectively, [40,41,42]. The AMPD isoforms have native tetrameric structures [41, 43, 44] and their assembly is thought to be driven by the relative abundance of polypeptides with no apparent preference for similar subunits [44]. Human AMPDs contain conserved C-terminal domains, where catalytic residues reside, and divergent N-terminal domains. The N-terminal domains of AMPD modulate binding capacities to actomyosin and cytoplasmic membranes, regulating intracellular distribution [45, 46] and catalytic activity [44, 47].
Western blot analysis for AMPD proteins shows full-length forms and N-terminal truncated forms [44, 48,49,50,51]. The N-terminal regions of AMPD proteins are very sensitive to proteolysis, and truncation of the N-terminal region is observed during purification and storage of the peptide at 4 ℃. The apparent Km values of N-terminal truncated forms of human AMPD1 and AMPD3 in absence of ATP were lower than those of the full-length forms [44], and truncated AMPD3 exhibited altered sensitivity to phosphoinositide compared with its full-length form [47]. Recently, Ronca and Raggi [52] discussed the physiological importance of AMPD1 truncation by a calpain-like protease and effects of the truncation on AMPD-troponin T interaction in skeletal muscle cells in a review article. However, whether such mechanisms apply to cardiomyocytes in vivo remains to be investigated.
In rodent hearts, hypoxia-inducible factor 1 (HIF-1) [53] and miR301b [36] were shown to regulate the transcription and translation of the AMPD3 gene, respectively. Foxo1/3/4 and HDAC3 in skeletal muscle [54, 55] and cyclin-dependent kinase-7 (CDK7)-mediated assembly of a super-enhancer in renal tubular cells [56] were also reported to be involved in regulation of AMPD3 expression, although their functions in the heart have not been confirmed. In addition, mutual regulation between AMPD3 and branched-chain α-keto acid dehydrogenase (BCKDH) in cardiomyocytes was revealed in a recent study [38] (Fig. 1); in rat neonatal cardiomyocytes, siRNA knockdown of BCKDH-E1α increased AMPD3 expression levels and activity, and conversely, knockdown of AMPD3 expression increased BCKDH activity without increasing BCKDH protein. Although the mechanism of mutual regulation remains to be further investigated, there is a possibility that suppression of BCKDH activity by upregulated AMPD contributes to impaired metabolism of branched-chain amino acids (BCAAs) in diabetic hearts as discussed in the section entitled AMPD and BCAA metabolism.
Studies using rats, rabbits, and piglets showed that cardiac AMPD is activated by ATP, ADP, adenosine, Ca2+, and NADH, while it is inhibited by GTP, inorganic phosphates, phosphoinositide and palmitoyl-CoA [57,58,59,60]. The regulation of cardiac AMPD by ATP (Fig. 1) is distinct from that of skeletal muscle AMPD, which is inhibited by ATP [52]. Protein kinase C (PKC) phosphorylates AMPD, which reduces the Km value of AMPD without affecting Vmax, and this posttranslational modification does not affect the allosteric response of AMPD to ATP and ADP [59, 61, 62]. In contrast, protein kinase A (PKA) does not appear to regulate AMPD activity in either the heart or skeletal muscle [62, 63]. Interaction with Ca2+-calmodulin has been shown to activate AMPD3 in erythrocytes [64], but the role of this interaction in the heart remains unknown.
Intracellular localization of AMPD in the heart
While AMPD has been thought to be present mostly in the cytosol, its localization in the sarcoplasmic reticulum (SR) as a member of the “cardiac SR-glycogenolytic complex” was proposed by Entman et al. [65,66,67,68,69] almost five decades ago. In addition, it was found that AMPD interacts with sarcomeric proteins, such as myosin, leading to a change in the level of soluble AMPD depending on the status of muscle contraction [42]. We recently postulated that AMPD may localize in mitochondria in addition to the SR and cytosol because AMPD is functionally linked with AK (Fig. 1), which plays a crucial role in high-energy phosphotransfer from ATP generation sites to ATP utilization sites as does the creatine kinase (CK) system. Although the majority of AKs are localized in the cytosol, their different isoforms have been found to be localized in mitochondria, nuclei, and plasma membranes [20]. In our study, AMPD3 signals were detected in cytosolic fractions, fractions of mitochondria-associated endoplasmic reticulum membrane (MAM), and fractions of endoplasmic reticulum (ER)/outer mitochondrial membrane and were barely in mitochondrial fractions derived from rat hearts [39]. AMPD3 proteins in the ER/SR and MAM appear to be involved in the regulation of BCAA metabolism and mitochondria-ER/SR contact formation, respectively, as discussed below (sections entitled AMPD and BCAA metabolism and AMPD and increased mitochondria-ER/SR contacts in diabetic cardiomyopathy). There were marked differences between the intracellular compartments in the proportions of signals corresponding to 90-kDa full-length AMPD3 and a signal of an approximately 78-kDa protein. In our previous studies [36,37,38,39], we interpreted the 78-kDa protein as a truncated form of AMPD3. However, the interpretation recently turned out to be wrong because our recent Western blot analysis using the same anti-AMPD3 antibody (Proteintech 23997-1-AP) and AMPD3-knockout mouse samples confirmed that the signal of the 90-kDa protein is specific to AMPD3 but that of the 78-kDa protein is not (Kouzu et al. 2023 unpublished observation). Nevertheless, Western blotting showed that full-length AMPD3 localizes not only in the cytosol but also in the SR and MAM in the rat heart. Immunogold labeling with electron microscopy revealed that AMPD1 was localized in myofibrils and the SR and nuclei in skeletal muscle [70, 71], but a similar analysis for cardiomyocytes has not been reported.
Role of the PNC in the heart
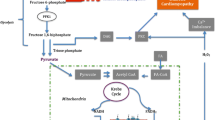
AMPD is one of the three enzymes that constitute the PNC (Fig. 1). AMPD catalyzes the conversion of AMP to IMP, which reacts with aspartate and GTP, generating adenylosuccinate by adenylosuccinate synthase (ADSS). The formed adenylosuccinate generates fumarate and AMP by adenylosuccinate lyase, completing the cycle. Since fumarate is not readily transported via the mitochondrial inner membrane, it is converted to malate before being taken up into the mitochondrial matrix. In skeletal muscle, elevation of workload stress increases the rate of the PNC, which is indicated by increased levels of citric acid cycle intermediates, decreased levels of aspartate, and increased ammonia release [72, 73].
In contrast to skeletal muscle, increased workload in isolated perfused rat hearts did not induce metabolite changes that reflect an increase in PNC-mediated metabolism [74]. In rat hearts in situ, an increase in ventricular afterload tended to elevate adenylosuccinate levels, but aspartate levels were also elevated, arguing against the possibility of an increased rate of the PNC [23]. The difference between skeletal muscle and cardiac muscle in response to increased workload might be attributable to lower ADSS levels and AMPD activity in cardiac muscle than in skeletal muscle [4, 5, 75, 76]. However, the PNC in the heart might play a protective role against ischemic injury. A recent study by Wu et al. [53] showed that treatment with hadacidin, an inhibitor of ADSS, significantly augmented both ATP and ADP depletion and AMP accumulation during ischemia in mouse hearts, suggesting that the PNC contributes to the preservation of energy charge in the ischemic myocardium.
AMPD and BCAA metabolism
Recent studies [3, 13, 38] have revealed that the PNC is not the only connection between AMPD and amino acid metabolism. Miller et al. [13] comprehensively examined the impact of AMPD3 overexpression on the metabolome and transcriptome of C2C12 cells. They found that BCAAs, glycolysis, and ceramide metabolic pathways were among the most highly enriched by overexpressed AMPD3 as were purine metabolism pathways. Overexpression of AMPD3 increased the intracellular levels of BCAAs, branched-chain α-keto acids (BCKAs), pyruvate, and α-ketoglutarate and increased the release of BCKAs and alanine into the culture medium. An increase in glutamine release into the culture media was not detectable possibly due to pre-existing glutamine in the culture medium. BCKDH activity was not directly determined in the study by Miller et al. [13], but the increase in intracellular BCKA levels was much larger than the increase in BCAA levels (i.e., 2–3-fold vs. 1.1–1.2-fold) in AMPD3-overexpressing C2C12 cells, and the levels of BCKAs in the culture media were also higher. Moreover, ATP was reduced with no change in ADP/ATP and AMP/ATP ratios. These findings might reflect reduced metabolic flux from BCKAs to short-chain acyl-CoA species by BCKDH in AMPD3-overexpressing cells.
While the role of BCAAs as energy substrates in the heart is small, their roles in the maintenance of normal function and morphology of the heart were indicated by the findings that inactivation of BCKDH worsened heart failure [77, 78] and that cardiomyocyte-selective deletion of BCKDH-E1α induced deterioration of cardiac function and adverse ventricular remodeling [79]. Cardiac BCKDH was found to localize not only in mitochondria but also in the ER where the E1α and E2 subunits of BCKDH physically interact with AMPD3 [38] (Fig. 2). As mentioned in the section titled AMPD expression and regulation in the heart, there is mutual negative regulation between BCKDH and AMPD3 functions (Fig. 1), and BCKDH activity was shown to be increased when AMPD3 expression was suppressed in cardiomyocytes in vitro. The physiological function of AMPD-mediated regulation of BCKDH is unclear. However, there is a possibility that, under the condition of sufficient ATP supply, AMPD activity, which is positively regulated by ATP, suppresses BCAA catabolism via BCKDH inhibition to spare BCAAs for protein synthesis. Interestingly, the suppression of BCKDH activity modifies the expression of enzymes relevant to fatty acid oxidation as well as AMPD expression in cardiomyocytes in vitro [38]. The relationship between BCKDH and AMPD3 may play a significant role in metabolic changes in diabetic hearts. In fact, reduced BCKDH expression in hearts of Otsuka Long-Evans Tokushima Fatty (OLETF) rats (a model of type 2 diabetes) was associated with upregulated AMPD3 activity and the accumulation of BCAAs and lipid droplets in cardiomyocytes [38, 80].
Current working hypothesis for the pathophysiological roles of upregulated AMPD in diabetic hearts. Upregulation of AMPD in diabetic hearts is possibly an adaptive response that supports AK-mediated high-energy phosphotransfer, compensates for impaired Ca2+ uptake in mitochondria by remodeling mitochondria-ER/SR contacts, and prevents excessive BCAA use for ATP synthesis. In the normal heart, the role of BCAAs as energy substrates in the heart is small, but their role might be larger during increased ATP demand in diabetic hearts, in which glucose oxidation is suppressed. When an increased cardiac workload raises the rate of ATP hydrolysis to a higher level, adverse actions of AMPD upregulation are exerted by enhancement of IMP generation and Ca2+ influx into mitochondria. AMPD AMP deaminase, IMP inosine monophosphate, Mito-ER contacts mitochondria-endoplasmic reticulum contacts, mPTP mitochondrial permeability transition pore, ROS reactive oxygen species, XOR xanthine oxidoreductase. The figure was created with Biorender.com
In contrast to the observations in C2C12 cells and OLETF rat hearts [13, 38], AMPD3 upregulation induced by HDAC3 deletion in skeletal muscle was associated with upregulation of enzymes participating in BCAA metabolism and increased oxidation of amino acids and fatty acids [3]. The reason for the different impacts of upregulated AMPD3 on amino acid and fatty acid oxidation between the studies [3, 13, 38] is unclear, but the PNC may function differently depending on the cell type and HDAC3 deletion may affect other targets besides AMPD3.
AMPK-mediated signaling pathways and AMPD
AMP functions as a primary sensor of the balance of energy supply/consumption in cells. Dzeja and Terzic [20] proposed an “adenylate kinase metabolic monitoring system” in which the change in the intracellular ADP level is amplified by AK to a larger change in AMP, leading to the activation of AMP-mediated processes that normalize the ADP/ATP balance. When ATP generation does not meet its demand, increased AMP activates AMPK by three mechanisms: promotion of Thr172 phosphorylation, inhibition of Thr172 dephosphorylation, and allosteric activation [81]. Activated AMPK promotes the catabolism of energy substrates (glucose uptake and glycolysis, fatty acid uptake and oxidation), except for lipolysis, and inhibits anabolic processes (gluconeogenesis, synthesis of glycogen, fatty acids, triglycerides, phospholipids, sterols, and proteins), enabling ATP synthesis to match ATP demand. In addition to AMP, LKB1- and CaMKKβ-mediated phosphorylation at Thr172 and Akt-mediated phosphorylation at Ser485 modulate AMPK activity.
A reciprocal relationship between AMPD and AMPK activity has been shown in some but not all studies in which AMPD was overexpressed or genetically deleted. In hepatic and renal cell lines in vitro, activation of AMPD suppressed AMPK activity [6,7,8,9,10,11,12,13,14]. Upregulation of AMPD2 in the liver in diabetic mice was associated with decreased AMPK phosphorylation and increased gluconeogenesis via the upregulated expression of phosphoenolpyruvate carboxykinase (PEPCK) and glucose-6-phosphatase, while the suppression of AMPK was mediated by increased uric acid levels [9]. AMPD3 overexpression for 48 h reduced AMPK-Thr172 phosphorylation and the mitochondrial oxygen consumption rate in skeletal muscle [13]. In a study by Davis et al. [12], ATP and ADP levels were reduced by AMPD3 overexpression for 48 h in C2C12 cells, but the AMP/ATP ratio and Thr172-phospho-AMPK level were unchanged. Both the contraction of skeletal muscle during tetanic contraction and its relaxation were impaired in AMPD1-deficient mice when the levels of AMP, AMP/ATP ratio, and phosphorylation of Thr172-AMPK were elevated [14]. Pharmacological inhibition of AMPD or knockout of AMPD1 increased tissue AMP and p-Thr172-AMPK levels during the stimulated contraction of skeletal muscles, although such impacts of AMPD suppression were not detected under resting conditions [10]. Taken together, these findings suggest that AMPD can participate in AMPK regulation by modulating the AMP/ATP ratio, although an increase in AMPD activity alone is not always sufficient to reduce the AMP/ATP ratio to a low enough level to inhibit AMPK. In other words, AMPD activity appears to elevate the threshold for activation of AMPK in response to mechanisms that increase ATP consumption and modify the AMP/ATP ratio.
Unfortunately, few data on AMPD–AMPK relationships in the heart have been reported. In studies by Rybakowska et al. [82, 83], changes in AMPD activity induced by a high-fat diet were not correlated with AMPK activity. In our study, there was no difference in Thr172-phospho-AMPK levels between control and OLETF rat hearts under baseline conditions despite two-fold higher AMPD levels in OLETF rat hearts. However, activation of AMPK/ULK1/mTORC signaling induced by ischemic heart failure was observed in control rats but not in OLETF rats [84]. The lack of AMPK activation in OLETF rats was associated with impaired autophagy in the myocardium and increased mortality due to heart failure. AMPK functions are downregulated in diabetic hearts [29] and whether the upregulated AMPD activity is involved in the suppression of the stress-induced activation of AMPK in diabetic hearts remains to be further investigated.
AMPD and SR/ER functions
ATP for Ca2+ uptake in the SR is supplied by glycolysis and two phosphotransfer systems, the CK system and the AK system. Several lines of evidence support the important contribution of glycolysis in the proximity of the SR to SR function. Mark Entman’s group [65,66,67,68,69] found that the SR is physically associated with glycogen and a series of enzymes involved in glycogenolysis (i.e., phosphorylase b kinase, phosphorylase b/a, debranching enzyme, adenylate cyclase, PKA), and they proposed that the “SR-glycogenolytic complex” regulates glycogenolysis in the SR. In a study by Xu et al. [85], the entire chain of glycolytic enzymes was associated with SR vesicles from both cardiac and skeletal muscles. The inhibition of glycolysis by iodoacetate, an inhibitor of GAPDH, suppressed Ca2+ uptake by SR vesicles, and the uptake was restored by phosphoenolpyruvate (PEP). The addition of a soluble hexokinase ATP trap eliminated Ca2+ uptake by ADP and PEP but had little effect on Ca2+ uptake induced by endogenous ATP, suggesting that glycolysis-derived ATP was preferentially used by the SR. Glycogen has been shown to be a preferential source for glucose oxidation in the working heart [86, 87]. Unfortunately, the role of AMPD in glycogenolysis or glycolysis in the heart has not been specifically investigated. However, it is possible that AMPD in the SR-glycogenolytic complex finely tunes phosphorylase b activity. AMP is a much more potent physiological activator of phosphorylase b than IMP [88] and thus IMP generation from AMP by AMPD could decrease phosphorylase b activation, potentially suppressing glycogenolysis in the SR.
AK isoforms localize in the cytosol, mitochondria, and/or nucleus, and AK1 is the major cytosolic AK isoform in humans [89]. The specific role of the AK system in SR function has not been characterized. However, circumstantial evidence suggests that AMPD in the SR/ER supports the adaptive increase in AK-mediated energy transfer to the SR in the heart under stress. The contribution of AK to high-energy phosphotransfer reactions is much smaller than that of CK (~ 10% vs. 89%) in healthy hearts [90]. However, in a dog model of pacing-induced heart failure, phosphotransfer via AK increased by 134%, contributing 21% to the total ATP turnover, while the contribution of CK was reduced from 89 to 40% [90]. In two murine models of heart failure (i.e., heart failure induced by transaortic constriction and that induced by myocardial infarction), CK activity was reduced, while AK activity remained unchanged [91]. As AMPD is important for the AK reaction to proceed in the direction of ATP formation, the role of AMPD would become larger when myocardial stress increases AK-mediated phosphotransfer reaction in ATP-consuming sites, including the SR.
Role of AMPD in diabetic cardiomyopathy
Diastolic dysfunction in diabetic cardiomyopathy
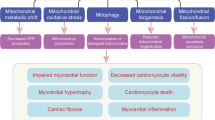
Diabetic cardiomyopathy progresses from an early stage with diastolic dysfunction alone to later stages with diastolic/systolic dysfunction, hypertrophy and loss of cardiomyocytes, reduced capillary density, and interstitial fibrosis [28, 29, 92]. The natural history of diabetic cardiomyopathy is explained by the sequential activation of multiple cardiac pathological mechanisms (increased generation of reactive oxygen species [ROS], impaired Ca2+ handling, altered Ca2+ sensitivity of myofilaments, mitochondrial dysfunction, remodeling of cytoskeletons and extracellular matrix, and impaired microcirculation) and extracardiac mechanisms (activation of neurohumoral systems, diabetic autonomic neuropathy, increased stiffness of peripheral arteries, and dysregulation of circulating blood volume due to nephropathy) [28,29,30,31,32]. Of these, increased generation of ROS, impaired Ca2+ handling in cardiomyocytes, and changes in myofilaments and their regulatory proteins [28, 31, 93,94,95] have been proposed to cause impaired diastolic function with preserved systolic function, a hallmark of the early stage of diabetic cardiomyopathy.
To gain insight into the role of AMPD in the mechanisms of diastolic dysfunction, we selected a model of type 2 diabetes, OLETF rat, for a series of studies [23, 36,37,38,39]. Although OLETF rats have not been frequently used for studies on diabetes in the literature, this model has several useful features. OLETF rats have a deficit in the cholecystokinin-1 (CCK1) receptor gene, causing the absence of the CCK1 receptor in the brain and gastrointestinal tract [96]. This genetic abnormality causes hyperphagia, obesity, and type 2 diabetes [97,98,99], and the time course of diabetes and its cardiovascular complications have been well characterized [99,100,101,102,103,104,105]. OLETF rats at ages of 29–35 weeks have features suitable for analysis of diastolic dysfunction due to impaired cardiomyocyte relaxation. At these ages, OLETF rats have hyperinsulinemic diabetes, and ventricular pressure–volume relationship analyses show diastolic dysfunction with preserved systolic function; tau (the left ventricular diastolic pressure time constant) and left ventricular end-diastolic pressure (LVEDP) were only modestly increased at baseline [23, 37, 106]. Ventricular pressure loading induces a larger upward shift in the end-diastolic pressure–volume relationship (EDPVR) in OLETF rats than in their nondiabetic control (Long-Evans Tokushima rats, LETO rats), although indices of systolic function were unchanged by the increased loading. Importantly, there was no difference in pressure–volume relationships between OLETF and LETO rats when cardiac arrest was executed with a cardioplegic solution in vitro [23], indicating that diastolic dysfunction shown in vivo is due to the impaired relaxation of the myocardium but not due to the increased stiffness of the myocardium. In this model of diabetes, both AMPD activity and protein levels were increased by approximately two-fold, while PKC-α activation and inorganic phosphate level reduction were not detected, suggesting that the change in the AMPD activity is mainly due to its increased protein level [23, 36,37,38,39].
Increased AMPD activity in diabetic hearts
Although an increase in cardiac AMPD activity has been observed in different models of diabetes (Table 1), its mechanism is poorly understood. In the OLETF heart, in which the AMPD3 protein level was elevated without a change in its mRNA level, increased translation of the AMPD3 gene by repression of miR301b was suggested to be involved [36]. In addition, the downregulation of BCKDH in diabetes might contribute to the upregulation of AMPD. The expression of most enzymes needed for BCAA metabolism, including BCKDH, is suppressed in diabetic hearts, adipose tissues, and the liver [107,108,109]. The reduction of BCKDH-E1α expression by siRNA increased AMPD3 mRNA and protein level by more than five-fold in rat cardiomyocytes in vitro, although the underlying mechanism remains unclear [38].
AMPD and dysregulation of intracellular Ca2+ in diabetic cardiomyopathy
Impaired Ca2+ handling in cardiomyocytes, which causes delay in cytosolic Ca2+ transient, has been observed in different models of early diabetes [28, 29, 31, 93, 106]. Its proposed mechanisms include downregulated protein expression and/or reduced activity of SERCA2a. In the alterations in SERCA2a protein and activity levels, a reduction in SERCA2a mRNA expression, enhanced SERCA2a degradation by elevated ER stress, and modification of SERCA2a by advanced glycation end products (AGEs), O-GlcNAcylation, and ROS have been suggested to be involved. Upregulated AMPD is unlikely to be involved in the change in the protein level of SERCA2a as overexpression of AMPD3 did not alter levels of SERCA2a, GRP78, or GRP94 in HEK293 cells [39]. However, AMPD activity potentially affects SR Ca2+ uptake during increased cardiac workload by reducing intracellular ATP levels via three mechanisms: depletion of adenine nucleotides, enhanced suppression of BCKDH activity, and inhibition of ATP synthesis in mitochondria [23, 37,38,39].
Upregulated AMPD promoted metabolic flux from AMP to the IMP/inosine/hypoxanthine/xanthine pathway and potentially reduced ATP generation by suppressing glycogenolysis and BCAA oxidation and inhibiting mitochondrial respiration [23, 37,38,39]. Upregulation of AMPD3 expression was found to reduce BCKDH activity, limiting the supply of BCAA-derived short-chain acyl-CoA species to the tricarboxylic cycle in cardiomyocytes [38], and to suppress mitochondrial state-3 respiration by inhibiting complex I in HEK293 cells [39]. Increased generation of ROS during pressure loading in OLETF rats was closely associated with mitochondrial dysfunction (as discussed in the following section) [37]. Collectively, these findings support the notion that upregulated AMPD expression is causally related to both increased AMP depletion and insufficient ATP synthesis in OLETF rat hears [23, 37, 38]. In contrast to diastolic function, systolic function was preserved during pressure loading despite the reduced tissue ATP level, which can be explained by the higher energy demand of SERCA2a (52 kJ/mol) than that of actomyosin ATPase (45–50 kJ/mol) [110].
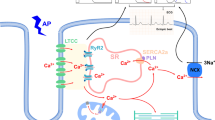
Ca2+ regulation in mitochondria is also impaired by diabetes. Mitochondrial Ca2+ uptake is reduced in cardiomyocytes subjected to high concentrations of glucose and in animal models of type 1 and type 2 diabetes [111,112,113,114,115,116]. Several mechanisms have been proposed for the reduction in mitochondrial Ca2+ levels by diabetes: lowered peak of cytosolic Ca2+ transient [117,118,119], which is induced by reduced Ca2+ stores in the SR and/or impaired SR functions, reduced Ca2+ transfer from the ER/SR to mitochondria [115], impaired function of the mitochondrial Ca2+ uniporter (MCU) complex (MCUC) [113, 114, 120], and increased Ca2+ efflux via mitochondrial Na2+–Ca2+ exchange [116]. Mitochondrial matrix Ca2+ activates the pyruvate dehydrogenase complex (PDC) by an allosteric mechanism and by its dephosphorylation via activation of PDC phosphatases. In addition, Ca2+ stimulates the regeneration of NADPH, which provides electrons for the production of reduced glutathione from oxidized glutathione. Thus, a reduction in mitochondrial matrix Ca2+ uptake in diabetic hearts is associated with suppressed glucose oxidation, reciprocally upregulated fatty acid oxidation, and increased generation of ROS in mitochondria [27, 112].
AMPD and increased mitochondria-ER/SR contacts in diabetic cardiomyopathy
Recently, we found that AMPD3 protein expression is increased not only in the cytosol but also in MAMs in OLETF rat hearts compared with that in nondiabetic control, LETO rat hearts [39]. Assessment of the extent of mitochondria-ER/SR contacts by electron microscopy showed that areas of mitochondria-ER/SR contact are significantly increased in OLETF rat cardiomyocytes. The causal relationship between the upregulated AMPD expression and the increase in mitochondria-ER/SR contacts is supported by the observations that the overexpression of AMPD3 increased mitochondria-ER/SR contacts in HEK293 cells and that this structural change was inhibited by knockdown of GRP78, a protein that tethers mitochondria to the ER/SR [39]. However, it is not clear whether AMPD3 proteins localized in the MAM primarily mediate the remodeling of mitochondria-ER/SR contacts.
As expected from the change in mitochondria-ER/SR contacts, the increase in mitochondrial Ca2+ levels in response to pressure loading was larger in OLETF rat hearts than in LETO rat hearts, while there was no difference in the levels at baseline [39]. These findings support the notion that AMPD modulates Ca2+ regulation in mitochondria by remodeling mitochondria-ER/SR contacts. It is possible to speculate that increased mitochondria-SR/ER contacts by AMPD upregulation is an adaptive mechanism to compensate for impaired Ca2+ uptake in mitochondria in diabetes, although it increases the risk of mitochondrial Ca2+ overload during increased cardiac workload and during pathological elevation of cytosolic Ca2+ levels.
AMPD and generation of ROS in diabetic cardiomyopathy
The role of ROS in the pathogenesis of diabetic cardiomyopathy has been extensively studied, and it has been proposed that excessive generation of ROS occurs before the development of diastolic dysfunction [31, 121]. In cardiomyocytes, there are multiple ROS-generating mechanisms and antioxidant defense mechanisms, both of which are reported to be modified in diabetes mellitus or hyperglycemia. ROS generated by NADPH oxidase (NOX) [122,123,124,125], mitochondria [126,127,128,129,130], uncoupled nitric oxide synthase [131,132,133], and/or xanthine oxidoreductase (XOR) [37, 134,135,136] are involved in diabetic cardiomyopathy, depending on the type or stage of diabetes mellitus models. Reported alterations in the ROS-mediated injury defense system in diabetic hearts include upregulation of superoxide dismutase (SOD) and catalase expression, which appears to be adaptive [137,138,139,140,141,142], and downregulation of peroxiredoxins [143, 144], SIRT3 [145, 146], NF-E2-related factor 2 (NRF2) [147, 148], and nicotinamide phosphoribosyltransferase (NAMPT) [149] expression, which augments ROS-induced injury. However, alterations in the antioxidant defense systems are also different between models of diabetes and their roles in pathogenesis of diabetic cardiomyopathy remain to be further investigated.
Our recent studies showed that increased AMPD activity in diabetes contributes to ROS-mediated injury via augmentation of ROS generation by XOR [37]. Hypoxanthine and xanthine are metabolites downstream of AMP deamination by AMPD, and it is therefore expected that upregulated AMPD increases XOR-derived ROS when the ATP turnover rate is increased, for example, by increased cardiac workload. In fact, while baseline levels of tissue hypoxanthine, xanthine and uric acid were similar in OLETF rats and their nondiabetic control, LETO rats, ventricular pressure loading increased tissue xanthine and uric acid to higher levels in OLETF rats [37]. In addition to its substrates, the activity of XOR was found to be increased by 20–30% during pressure loading via a mechanism mediated by accumulated inosine, while there was no activation of XOR by CKD5-mediated XOR-Thr222 phosphorylation [150] or increased intracellular iron levels [151, 152]. The causal role of XOR-derived ROS in diastolic dysfunction in OLETF rats was confirmed by the findings that a specific XOR inhibitor, topiroxostat, suppressed the elevation of tissue biomarkers of ROS (levels of 4-HNE and carbonylated proteins) and improved diastolic function during pressure loading [37].
An XOR-independent mechanism might also be involved in the generation of ROS by upregulated AMPD (Fig. 2). Inhibition of XOR in the OLETF rat myocardium to a level lower than that in the nondiabetic control did not completely restore state-3 respiration of mitochondria [37]. On the other hand, state-3 respiration on pyruvate/malate, but not that on succinate, in mitochondria was inhibited in AMPD3-overexpressing cells, indicating complex I inhibition by AMPD upregulation [39]. Inhibition of complex I induces generation of ROS [153, 154], and mitochondrial Ca2+ stimulates the production of ROS and greatly enhances the detrimental effect of ROS on the mitochondrial respirasome [155]. Thus, there is the possibility that increased mitochondrial ROS generation by enhanced mitochondrial Ca2+ influx via remodeled MAMs partly contributes to the excessive generation of ROS in AMPD3-upregulated hearts.
AMPD and increased vulnerability of cardiomyocytes to cell death
Cardiomyocyte death is one of the mechanisms underlying the irreversible progression of chronic heart failure [156, 157]. At late stages of diabetic cardiomyopathy, multiple mechanisms, including ischemia caused by insufficient microcirculation and inflammatory cytokines, have been proposed to trigger the programmed and unprogrammed cell death of cardiomyocytes [28, 31, 158]. Excessive generation of ROS and impaired Ca2+ handling, two important mechanisms of contractile dysfunction in diabetic cardiomyopathy, increase the propensity for mitochondrial permeability transition pore (mPTP) opening. mPTPs are nonselective channels in the inner mitochondrial membrane and open either in reversible low conductance mode or in irreversible high conductance mode, mediating redox signals or cell death, respectively [159,160,161]. The threshold for irreversible mPTP opening is regulated by multiple factors including the interaction of signal molecules, intracellular pH and Ca2+, and ATP levels in the matrix. Toxic levels of ROS and/or mitochondrial Ca2+ overload trigger irreversible opening of mPTPs, leading to necrotic cell death. As diabetes induces excessive production of ROS and dysregulation of intracellular Ca2+ homeostasis in cardiomyocytes, it is conceivable that the opening of mPTPs underlies progressive cardiomyocyte death in diabetic cardiomyopathy. This hypothesis is difficult to directly test because of technical difficulty in quantifying dead cardiomyocytes present at very small numbers in the heart. However, an increase in myocardial susceptibility to infarction after ischemia/reperfusion and a lowered threshold for mPTP opening in diabetic hearts have been reported in many, but not all, animal models of diabetes [114, 162,163,164,165].
As upregulated AMPD promotes generation of ROS and elevates mitochondrial Ca2+ levels during increased cardiac workload (as discussed above), it is expected that the level of AMPD expression correlates with the propensity for mPTP opening. In fact, the Ca2+ retention capacity (CRC) of isolated mitochondria (i.e., mitochondria with some extent of ER/SR contacts in crude mitochondrial fractions) was reduced by AMPD3 overexpression in H9c2 cardiomyoblasts and HEK293 cells, while the levels of mPTP regulatory proteins, cyclophilin D, GSK-3β, and Ser9-phospho-GSK-3β, in the mitochondrial fractions were unchanged [39]. Furthermore, ROS-induced mPTP opening, which was determined by the loss of mitochondrial membrane potential, was augmented by the overexpression of AMPD3 and conversely suppressed by the knockdown of AMPD3 expression in H9c2 cardiomyoblasts [39]. Consistent with these findings in cell lines in vitro, the CRC of mitochondria isolated from OLETF rat hearts was significantly lower than that of mitochondria from nondiabetic control hearts, although the change in CRC is likely partially due to a reduced level of Ser9-GSK-3β phosphorylation in mitochondria [162]. Future studies using a specific inhibitor of AMPD [18] or deletion of AMPD genes are necessary to elucidate the relative importance of AMPD among other regulatory factors in mPTP regulation. Nevertheless, it is important to note that detrimental effects exerted by upregulated AMPD on adenine nucleotides [23], tissue biomarkers of ROS [37], and mitochondrial Ca2+ levels [39] were undetectable under the baseline condition but were detected after an increase in cardiac workload. Thus, the impact of AMPD upregulation on the propensity for mPTP-mediated cell death is likely to depend on factors modulating cardiac work.
AMPD in human heart diseases
Mutations in AMPD2, which is predominantly expressed in the heart, brain, and liver, were found to cause pontocerebellar hypoplasia without gross abnormalities in the heart [166, 167]. In contrast, the C34T (Glu12Stop) mutation in exon 2 of human AMPD1 was found to be associated with lower cardiac AMPD activity in heart failure patients; cardiac AMPD activity in patients with CT or TT genotypes was approximately 50% lower than that in patients with the wild-type CC genotype [168, 169]. Possible explanations for the lower cardiac AMPD activity include the heteromeric composition of cardiac AMPD and/or a change in the expression pattern induced by heart failure since the expression of AMPD1 and AMPD3 transcripts can be detected in human hearts [170]. Whether the CT or TT genotype is resistant to diabetic cardiomyopathy is an interesting question that, if answered, can provide insight into the role of AMPD in the human diabetic heart.
Although the concept of diabetic cardiomyopathy in the human heart has not been established as that in animal models, diabetes is clearly a major risk factor of both heart failure with preserved ejection fraction and heart failure with reduced ejection fraction. In light of complex mechanisms that have been shown in animal models of diabetic cardiomyopathy and human diabetic hearts [28,29,30,31,32], multiple approaches to different targets are currently employed in prevention and management of heart failure in diabetic patients [171, 172]. Whether a therapy targeting AMPD significantly protects hearts from diabetic cardiomyopathy remains to be further investigated.
Summary and conclusion
Multiple AMPD genes are expressed in the heart, while AMPD3 and AMPD2 are predominantly expressed in the rat heart and human heart, respectively. In the rat heart, AMPD3 was found to localize in the cytosol, SR/ER, and MAM, while its presence in mitochondria appears to be very low or negligible. The localization of AMPD overlaps with that of AK, which is consistent with the functional relationship between AMPD and AK. Circumstantial evidence suggests that AMPD negatively regulates BCAA-BCKA catabolism by suppressing BCKDH activity in the ER and AMPK signaling pathways when not only AMP levels but also AMP/ATP ratios are increased in cardiomyocytes. The physiological significance of the two AMPD functions is unclear. However, because ATP is an activator of AMPD, it is possible that the suppression of BCKDH and AMPK by activated AMPD, leading to reduced catabolism of energy substrates, is a feedback mechanism for fine-tuning the rate of ATP synthesis while sparing BCAAs for protein synthesis.
Upregulation of AMPD activity in myocardial tissue has been observed in different models of type 1 and type 2 diabetes. However, its pathophysiological role has been characterized only in one model of type 2 diabetes, OLETF rats. In this model, AMPD protein levels were increased in the cytosol, ER, and MAM but not in mitochondria. AMPD upregulation was associated with enlarged areas of mitochondria-ER/SR contact, which might be a compensatory response to the reduction in mitochondrial Ca2+ uptake in diabetic hearts. Diastolic dysfunction induced by ventricular pressure loading in OLETF rats was correlated with depletion of ATP and adenine nucleotides, increased generation of ROS, and augmented elevation of mitochondrial Ca2+ levels. Most of the detrimental effects of AMPD upregulation are attributable to increased XOR-derived ROS production as a result of increased flux from AMP to uric acid and depletion of adenine nucleotides, although enhanced mitochondrial Ca2+ influx via remodeled MAMs is possibly an additional mechanism (Fig. 2). Increased generation of ROS and modulation of mitochondrial Ca2+ handling by AMPD upregulation increase the propensity for necrotic cell death by lowering the threshold for mPTP opening (Fig. 2). As a major limitation, the proposed roles of upregulated AMPD in diabetic cardiomyopathy are based on findings in cultured cells with manipulated AMPD expression and metabolomic analyses in OLETF rat hearts. The findings need to be further confirmed by the use of specific inhibitors of AMPD and AMPD knockout mice, which would provide insight into a role of AMPD as a therapeutic target in the diabetic heart. However, cardiac-specific knockout of a single AMPD gene may provide results that are difficult to interpret, as did the effect of AMPD1 knockout on the contractile function of skeletal muscle [10, 14, 173], because of the compensatory function of co-expressed AMPD genes. Nevertheless, it is important to note that significant roles of AMPD in a rat model of diabetic cardiomyopathy were undetectable at the baseline condition and manifested in conditions of increased cardiac work. Thus, levels of physical activity and cardiac workload would need to be taken into account to characterize the roles of AMPD in human heart diseases in future investigations.
Data availability
Not applicable.
References
Hancock CR, Brault JJ, Terjung RL (2006) Protecting the cellular energy state during contractions: role of AMP deaminase. J Physiol Pharmacol 57(Suppl 10):17–29
Zabielska MA, Borkowski T, Slominska EM, Smolenski RT (2015) Inhibition of AMP deaminase as therapeutic target in cardiovascular pathology. Pharmacol Rep 67(4):682–688. https://doi.org/10.1016/j.pharep.2015.04.007
Hong S, Zhou W, Fang B, Lu W, Loro E, Damle M, Ding G, Jager J, Zhang S, Zhang Y, Feng D, Chu Q, Dill BD, Molina H, Khurana TS, Rabinowitz JD, Lazar MA, Sun Z (2017) Dissociation of muscle insulin sensitivity from exercise endurance in mice by HDAC3 depletion. Nat Med 23(2):223–234. https://doi.org/10.1038/nm.4245
Purzycka J (1962) AMP and adenosine aminohydrolases in rat tissues. Acta Biochim Pol 9:83–93
Moriwaki Y, Yamamoto T, Higashino K (1999) Enzymes involved in purine metabolism–a review of histochemical localization and functional implications. Histol Histopathol 14(4):1321–1340. https://doi.org/10.14670/HH-14.1321
Ouyang J, Parakhia RA, Ochs RS (2011) Metformin activates AMP kinase through inhibition of AMP deaminase. J Biol Chem 286(1):1–11. https://doi.org/10.1074/jbc.M110.121806
Lanaspa MA, Cicerchi C, Garcia G, Li N, Roncal-Jimenez CA, Rivard CJ, Hunter B, Andrés-Hernando A, Ishimoto T, Sánchez-Lozada LG, Thomas J, Hodges RS, Mant CT, Johnson RJ (2012) Counteracting roles of AMP deaminase and AMP kinase in the development of fatty liver. PLoS ONE 7(11):e48801. https://doi.org/10.1371/journal.pone.0048801
Plaideau C, Liu J, Hartleib-Geschwindner J, Bastin-Coyette L, Bontemps F, Oscarsson J, Hue L, Rider MH (2012) Overexpression of AMP-metabolizing enzymes controls adenine nucleotide levels and AMPK activation in HEK293T cells. FASEB J 26(6):2685–2694. https://doi.org/10.1096/fj.11-198168
Cicerchi C, Li N, Kratzer J, Garcia G, Roncal-Jimenez CA, Tanabe K, Hunter B, Rivard CJ, Sautin YY, Gaucher EA, Johnson RJ, Lanaspa MA (2014) Uric acid-dependent inhibition of AMP kinase induces hepatic glucose production in diabetes and starvation: evolutionary implications of the uricase loss in hominids. FASEB J 28(8):3339–3350. https://doi.org/10.1096/fj.13-243634
Plaideau C, Lai YC, Kviklyte S, Zanou N, Löfgren L, Andersén H, Vertommen D, Gailly P, Hue L, Bohlooly-Y M, Hallén S, Rider MH (2014) Effects of pharmacological AMP deaminase inhibition and Ampd1 deletion on nucleotide levels and AMPK activation in contracting skeletal muscle. Chem Biol 21(11):1497–1510. https://doi.org/10.1016/j.chembiol.2014.09.013
Tandelilin AA, Hirase T, Hudoyo AW, Cheng J, Toyama K, Morisaki H, Morisaki T (2015) AMPD1 regulates mTORC1-p70 S6 kinase axis in the control of insulin sensitivity in skeletal muscle. BMC Endocr Disord 15:11. https://doi.org/10.1186/s12902-015-0010-9
Davis PR, Miller SG, Verhoeven NA, Morgan JS, Tulis DA, Witczak CA, Brault JJ (2020) Increased AMP deaminase activity decreases ATP content and slows protein degradation in cultured skeletal muscle. Metabolism 108:154257. https://doi.org/10.1016/j.metabol.2020.154257
Miller SG, Hafen PS, Law AS, Springer CB, Logsdon DL, O’Connell TM, Witczak CA, Brault JJ (2021) AMP deamination is sufficient to replicate an atrophy-like metabolic phenotype in skeletal muscle. Metabolism 123:154864. https://doi.org/10.1016/j.metabol.2021.154864
Hafen PS, Law AS, Matias C, Miller SG, Brault JJ (2022) Skeletal muscle contraction kinetics and AMPK responses are modulated by the adenine nucleotide degrading enzyme AMPD1. J Appl Physiol 133(5):1055–1066. https://doi.org/10.1152/japplphysiol.00035.2022
Fischer S, Drenckhahn C, Wolf C, Eschrich K, Kellermann S, Froster UG, Schober R (2005) Clinical significance and neuropathology of primary MADD in C34-T and G468-T mutations of the AMPD1 gene. Clin Neuropathol 24(2):77–85
Lecker SH, Jagoe RT, Gilbert A, Gomes M, Baracos V, Bailey J, Price SR, Mitch WE, Goldberg AL (2004) Multiple types of skeletal muscle atrophy involve a common program of changes in gene expression. FASEB J 18(1):39–51. https://doi.org/10.1096/fj.03-0610com
Cheng J, Morisaki H, Toyama K, Sugimoto N, Shintani T, Tandelilin A, Hirase T, Holmes EW, Morisaki T (2014) AMPD1: a novel therapeutic target for reversing insulin resistance. BMC Endocr Disord 14:96. https://doi.org/10.1186/1472-6823-14-96
Admyre T, Amrot-Fors L, Andersson M, Bauer M, Bjursell M, Drmota T, Hallen S, Hartleib-Geschwindner J, Lindmark B, Liu J, Löfgren L, Rohman M, Selmi N, Wallenius K (2014) Inhibition of AMP deaminase activity does not improve glucose control in rodent models of insulin resistance or diabetes. Chem Biol 21(11):1486–1496. https://doi.org/10.1016/j.chembiol.2014.09.011
Hudoyo AW, Hirase T, Tandelillin A, Honda M, Shirai M, Cheng J, Morisaki H, Morisaki T (2017) Role of AMPD2 in impaired glucose tolerance induced by high fructose diet. Mol Genet Metab Rep 13:23–29. https://doi.org/10.1016/j.ymgmr.2017.07.006
Dzeja P, Terzic A (2009) Adenylate kinase and AMP signaling networks: metabolic monitoring, signal communication and body energy sensing. Int J Mol Sci 10(4):1729–1772. https://doi.org/10.3390/ijms10041729
Jenkins RL, McDaniel HG, Atkins L (1991) Changes in AMP deaminase activities in the hearts of diabetic rats. Biochim Biophys Acta 1077(3):379–384. https://doi.org/10.1016/0167-4838(91)90554-d
Podgorska M, Kocbuch K, Grden M, Szutowicz A, Pawelczyk T (2006) Prevalence of unidirectional Na+-dependent adenosine transport and altered potential for adenosine generation in diabetic cardiac myocytes. Basic Res Cardiol 101(3):214–222. https://doi.org/10.1007/s00395-005-0578-8
Kouzu H, Miki T, Tanno M, Kuno A, Yano T, Itoh T, Sato T, Sunaga D, Murase H, Tobisawa T, Ogasawara M, Ishikawa S, Miura T (2015) Excessive degradation of adenine nucleotides by up-regulated AMP deaminase underlies afterload-induced diastolic dysfunction in the type 2 diabetic heart. J Mol Cell Cardiol 80:136–145. https://doi.org/10.1016/j.yjmcc.2015.01.004
Castro MC, Villagarcía HG, Schinella G, Massa ML (1868) Francini F (2023) Mechanism of preventive effects of exendin-4 and des-fluoro-sitagliptin in a murine model of fructose-induced prediabetes. Biochim Biophys Acta Mol Cell Biol Lipids 9:159363. https://doi.org/10.1016/j.bbalip.2023.159363
Pawelczyk T, Sakowicz M, Szczepanska-Konkel M, Angielski S (2000) Decreased expression of adenosine kinase in streptozotocin-induced diabetes mellitus rats. Arch Biochem Biophys 375(1):1–6. https://doi.org/10.1006/abbi.1999.1548
Yang H, Wang Q, Xi Y, Yu W, Xie D, Morisaki H, Morisaki T, Cheng J (2023) AMPD2 plays important roles in regulating hepatic glucose and lipid metabolism. Mol Cell Endocrinol 577:112039. https://doi.org/10.1016/j.mce.2023.112039
Andres-Hernando A, Cicerchi C, Garcia GE, Orlicky DJ, Stenvinkel P, Johnson RJ, Lanaspa MA (2023) Phosphate depletion in insulin-insensitive skeletal muscle drives AMPD activation and sarcopenia in chronic kidney disease. Science 26(4):106355. https://doi.org/10.1016/j.isci.2023.106355
Miki T, Yuda S, Kouzu H, Miura T (2013) Diabetic cardiomyopathy: pathophysiology and clinical features. Heart Fail Rev 18(2):149–166. https://doi.org/10.1007/s10741-012-9313-3
Jia G, Hill MA, Sowers JR (2018) Diabetic cardiomyopathy: an update of mechanisms contributing to this clinical entity. Circ Res 122(4):624–638. https://doi.org/10.1161/CIRCRESAHA.117.311586
Maack C, Lehrke M, Backs J, Heinzel FR, Hulot JS, Marx N, Paulus WJ, Rossignol P, Taegtmeyer H, Bauersachs J, Bayes-Genis A, Brutsaert D, Bugger H, Clarke K, Cosentino F, De Keulenaer G, Dei Cas A, González A, Huelsmann M, Iaccarino G, Lunde IG, Lyon AR, Pollesello P, Rena G, Riksen NP, Rosano G, Staels B, van Laake LW, Wanner C, Farmakis D, Filippatos G, Ruschitzka F, Seferovic P, de Boer RA, Heymans S (2018) Heart failure and diabetes: metabolic alterations and therapeutic interventions: a state-of-the-art review from the Translational Research Committee of the Heart Failure Association-European Society of Cardiology. Eur Heart J 39(48):4243–4254. https://doi.org/10.1093/eurheartj/ehy596
Ritchie RH, Abel ED (2020) Basic mechanisms of diabetic heart disease. Circ Res 126(11):1501–1525. https://doi.org/10.1161/CIRCRESAHA.120.315913
Rupee S, Rupee K, Singh RB, Hanoman C, Ismail AMA, Smail M, Singh J (2023) Diabetes-induced chronic heart failure is due to defects in calcium transporting and regulatory contractile proteins: cellular and molecular evidence. Heart Fail Rev 28(3):627–644. https://doi.org/10.1007/s10741-022-10271-5
Cusi K, Sanyal AJ, Zhang S, Hartman ML, Bue-Valleskey JM, Hoogwerf BJ, Haupt A (2017) Non-alcoholic fatty liver disease (NAFLD) prevalence and its metabolic associations in patients with type 1 diabetes and type 2 diabetes. Diabetes Obes Metab 19(11):1630–1634. https://doi.org/10.1111/dom.12973
Memaj P, Jornayvaz FR (2022) Non-alcoholic fatty liver disease in type 1 diabetes: prevalence and pathophysiology. Front Endocrinol (Lausanne) 13:1031633. https://doi.org/10.3389/fendo.2022.1031633
Tack W, De Cock AM, Dirinck EL, Bastijns S, Ariën F, Perkisas S (2023) Pathophysiological interactions between sarcopenia and type 2 diabetes: a two-way street influencing diagnosis and therapeutic options. Diabetes Obes Metab. https://doi.org/10.1111/dom.15321
Tatekoshi Y, Tanno M, Kouzu H, Abe K, Miki T, Kuno A, Yano T, Ishikawa S, Ohwada W, Sato T, Niinuma T, Suzuki H, Miura T (2018) Translational regulation by miR-301b upregulates AMP deaminase in diabetic hearts. J Mol Cell Cardiol 119:138–146. https://doi.org/10.1016/j.yjmcc.2018.05.003
Igaki Y, Tanno M, Sato T, Kouzu H, Ogawa T, Osanami A, Yano T, Kuno A, Miki T, Nakamura T, Miura T (2021) Xanthine oxidoreductase-mediated injury is amplified by upregulated AMP deaminase in type 2 diabetic rat hearts under the condition of pressure overload. J Mol Cell Cardiol 154:21–31. https://doi.org/10.1016/j.yjmcc.2021.01.002
Ogawa T, Kouzu H, Osanami A, Tatekoshi Y, Sato T, Kuno A, Fujita Y, Ino S, Shimizu M, Toda Y, Ohwada W, Yano T, Tanno M, Miki T, Miura T (2023) Downregulation of extramitochondrial BCKDH and its uncoupling from AMP deaminase in type 2 diabetic OLETF rat hearts. Physiol Rep 11(4):e15608. https://doi.org/10.14814/phy2.15608
Osanami A, Sato T, Toda Y, Shimizu M, Kuno A, Kouzu H, Yano T, Ohwada W, Ogawa T, Miura T, Tanno M (2023) Adenosine monophosphate deaminase in the endoplasmic reticulum-mitochondria interface promotes mitochondrial Ca2+ overload in type 2 diabetes rat hearts. J Diabetes Investig 14(4):560–569. https://doi.org/10.1111/jdi.13982
Ogasawara N, Goto H, Yamada Y, Watanabe T (1978) Distribution of AMP-deaminase isozymes in rat tissues. Eur J Biochem 87(2):297–304. https://doi.org/10.1111/j.1432-1033.1978.tb12378.x
Ogasawara N, Goto H, Yamada Y, Watanabe T, Asano T (1982) AMP deaminase isozymes in human tissues. Biochim Biophys Acta 714(2):298–306. https://doi.org/10.1016/0304-4165(82)90337-3
Sabina RL, Mahnke-Zizelman DK (2000) Towards an understanding of the functional significance of N-terminal domain divergence in human AMP deaminase isoforms. Pharmacol Ther 87(2–3):279–283. https://doi.org/10.1016/s0163-7258(00)00040-1
Thakkar JK, Janero DR, Yarwood C, Sharif H, Hreniuk D (1993) Isolation and characterization of AMP deaminase from mammalian (rabbit) myocardium. Biochem J 290(2):335–341. https://doi.org/10.1042/bj2900335
Mahnke-Zizelman DK, Tullson PC, Sabina RL (1998) Novel aspects of tetramer assembly and N-terminal domain structure and function are revealed by recombinant expression of human AMP deaminase isoforms. J Biol Chem 273(52):35118–35125. https://doi.org/10.1074/jbc.273.52.35118
Mahnke-Zizelman DK, Sabina RL (2001) Localization of N-terminal sequences in human AMP deaminase isoforms that influence contractile protein binding. Biochem Biophys Res Commun 285(2):489–495. https://doi.org/10.1006/bbrc.2001.5180
Mahnke-Zizelman DK, Sabina RL (2002) N-terminal sequence and distal histidine residues are responsible for pH-regulated cytoplasmic membrane binding of human AMP deaminase isoform E. J Biol Chem 277(45):42654–42662. https://doi.org/10.1074/jbc.M203473200
Sims B, Mahnke-Zizelman DK, Profit AA, Prestwich GD, Sabina RL, Theibert AB (1999) Regulation of AMP deaminase by phosphoinositides. J Biol Chem 274(36):25701–25707. https://doi.org/10.1074/jbc.274.36.25701
Mahnke-Zizelman DK, D’cunha J, Wojnar JM, Brogley MA, Sabina RL (1997) Regulation of rat AMP deaminase 3 (isoform C) by development and skeletal muscle fibre type. Biochem J 326(2):521–529. https://doi.org/10.1042/bj3260521
Barshop BA, Frieden C (1984) Analysis of the interaction of rabbit skeletal muscle adenylate deaminase with myosin subfragments. A kinetically regulated system. J Biol Chem 259(1):60–66
Marquetant R, Sabina RL, Holmes EW (1989) Identification of a noncatalytic domain in AMP deaminase that influences binding to myosin. Biochemistry 28(22):8744–8749. https://doi.org/10.1021/bi00448a010
Chilson OP, Kelly-Chilson AE, Siegel NR (1997) AMP-deaminases from chicken and rabbit muscle: partial primary sequences of homologous 17-kDa CNBr fragments: autorecognition by rabbit anti-[chicken AMPD]. Comp Biochem Physiol B Biochem Mol Biol 116(3):371–377. https://doi.org/10.1016/s0305-0491(96)00270-2
Ronca F, Raggi A (2023) Role of the interaction between troponin T and AMP deaminase by zinc bridge in modulating muscle contraction and ammonia production. Mol Cell Biochem. https://doi.org/10.1007/s11010-023-04763-7
Wu J, Bond C, Chen P, Chen M, Li Y, Shohet RV, Wright G (2015) HIF-1α in the heart: remodeling nucleotide metabolism. J Mol Cell Cardiol 82:194–200. https://doi.org/10.1016/j.yjmcc.2015.01.014
Milan G, Romanello V, Pescatore F, Armani A, Paik JH, Frasson L, Seydel A, Zhao J, Abraham R, Goldberg AL, Blaauw B, DePinho RA, Sandri M (2015) Regulation of autophagy and the ubiquitin-proteasome system by the FoxO transcriptional network during muscle atrophy. Nat Commun 6:6670. https://doi.org/10.1038/ncomms7670
Brocca L, Toniolo L, Reggiani C, Bottinelli R, Sandri M, Pellegrino MA (2017) FoxO-dependent atrogenes vary among catabolic conditions and play a key role in muscle atrophy induced by hindlimb suspension. J Physiol 595(4):1143–1158. https://doi.org/10.1113/JP273097
Mi Z, Song Y, Cao X, Lu Y, Liu Z, Zhu X, Geng M, Sun Y, Lan B, He C, Xiong H, Zhang L, Chen Y (2020) Super-enhancer-driven metabolic reprogramming promotes cystogenesis in autosomal dominant polycystic kidney disease. Nat Metab 2(8):717–731. https://doi.org/10.1038/s42255-020-0227-4
Barsacchi R, Ranieri-Raggi M, Bergamini C, Raggi A (1979) Adenylate metabolism in the heart. Regulatory properties of rabbit cardiac adenylate deaminase. Biochem J 182(2):361–366. https://doi.org/10.1042/bj1820361
Spychala J, Marszalek J (1991) Regulatory properties of AMP deaminases from rat tissues. Int J Biochem 23(10):1155–1159. https://doi.org/10.1016/0020-711x(91)90158-j
Thakkar JK, Janero DR, Yarwood C, Sharif HM (1993) Modulation of mammalian cardiac AMP deaminase by protein kinase C-mediated phosphorylation. Biochem J 291(2):523–527. https://doi.org/10.1042/bj2910523
Thakkar JK, Janero DR, Sharif HM, Hreniuk D, Yarwood C (1994) Cardiac adenylate deaminase: molecular, kinetic and regulatory properties under phosphate-free conditions. Biochem J 300(2):359–363. https://doi.org/10.1042/bj3000359
Hu B, Altschuld RA, Hohl CM (1993) Adenosine stimulation of AMP deaminase activity in adult rat cardiac myocytes. Am J Physiol 264(1 Pt 1):C48-53. https://doi.org/10.1152/ajpcell.1993.264.1.C48
Hohl AM, Hohl CM (1999) Isolation and regulation of piglet cardiac AMP deaminase. Mol Cell Biochem 201(1–2):151–158. https://doi.org/10.1023/a:1007083000564
Tovmasian EK, Hairapetian RL, Bykova EV, Severin SE Jr, Haroutunian AV (1990) Phosphorylation of the skeletal muscle AMP-deaminase by protein kinase C. FEBS Lett 259(2):321–323. https://doi.org/10.1016/0014-5793(90)80037-j
Mahnke DK, Sabina RL (2005) Calcium activates erythrocyte AMP deaminase [isoform E (AMPD3)] through a protein-protein interaction between calmodulin and the N-terminal domain of the AMPD3 polypeptide. Biochemistry 44(14):5551–5559. https://doi.org/10.1021/bi048121p
Entman ML, Goldstein MA (1976) Schwartz A (1976) The cardiac sarcoplasmic reticulum—glycogenolytic complex, an internal beta-adrenergic receptor. Life Sci 19(11):1623–1630. https://doi.org/10.1016/0024-3205(76)90066-7
Entman ML, Kanike K, Goldstein MA, Nelson TE, Bornet EP, Futch TW, Schwartz A (1976) Association of gylcogenolysis with cardiac sarcoplasmic reticulum. J Biol Chem 251(10):3140–3146
Entman ML, Bornet EP, Van Winkle WB, Goldstein MA, Schwartz A (1977) Association of glycogenolysis with cardiac sarcoplasmic reticulum: II. Effect of glycogen depletion, deoxycholate solubilization and cardiac ischemia: evidence for a phorphorylase kinase membrane complex. J Mol Cell Cardiol 9(7):515–528. https://doi.org/10.1016/s0022-2828(77)80367-2
Entman ML, Bornet EP, Barber AJ, Schwartz A, Levey GS, Lehotay DC, Bricker LA (1977) The cardiac sarcoplasmic reticulum-glycogenolytic complex. A possible effector site for cyclic AMP. Biochim Biophys Acta 499(2):228–237. https://doi.org/10.1016/0304-4165(77)90005-8
Goldstein MA, Murphy DL, van Winkle WB, Entman ML (1985) Cytochemical studies of a glycogen-sarcoplasmic reticulum complex. J Muscle Res Cell Motil 6(2):177–187. https://doi.org/10.1007/BF00713059
Ashby B, Frieden C, Bischoff R (1979) Immunofluorescent and histochemical localization of AMP deaminase in skeletal muscle. J Cell Biol 81(2):361–373. https://doi.org/10.1083/jcb.81.2.361
Mattii L, Bianchi F, Falleni A, Frascarelli S, Masini M, Alì G, Chiellini G, Sabbatini ARM (2020) Ultrastructural localization of histidine-rich glycoprotein in skeletal muscle fibers: colocalization with AMP deaminase. J Histochem Cytochem 68(2):139–148. https://doi.org/10.1369/0022155419897573
Aragón JJ, Lowenstein JM (1980) The purine-nucleotide cycle. Comparison of the levels of citric acid cycle intermediates with the operation of the purine nucleotide cycle in rat skeletal muscle during exercise and recovery from exercise. Eur J Biochem 110(2):371–377. https://doi.org/10.1111/j.1432-1033.1980.tb04877.x
Graham TE, MacLean DA (1992) Ammonia and amino acid metabolism in human skeletal muscle during exercise. Can J Physiol Pharmacol 70(1):132–141. https://doi.org/10.1139/y92-020
Taegtmeyer H (1985) On the role of the purine nucleotide cycle in the isolated working rat heart. J Mol Cell Cardiol 17(10):1013–1018. https://doi.org/10.1016/s0022-2828(85)80082-1
Davey CL (1961) The amination of inosine monophosphate in skeletal muscle. Arch Biochem Biophys 95:296–304. https://doi.org/10.1016/0003-9861(61)90149-7
Dow JW, Bowditch J, Nigdikar SV, Brown AK (1987) Salvage mechanisms for regeneration of adenosine triphosphate in rat cardiac myocytes. Cardiovasc Res 21(3):188–196. https://doi.org/10.1093/cvr/21.3.188
Sun H, Olson KC, Gao C, Prosdocimo DA, Zhou M, Wang Z, Jeyaraj D, Youn JY, Ren S, Liu Y, Rau CD, Shah S, Ilkayeva O, Gui WJ, William NS, Wynn RM, Newgard CB, Cai H, Xiao X, Chuang DT, Schulze PC, Lynch C, Jain MK, Wang Y (2016) Catabolic defect of branched-chain amino acids promotes heart failure. Circulation 133(21):2038–2049. https://doi.org/10.1161/CIRCULATIONAHA.115.020226
Li T, Zhang Z, Kolwicz SC Jr, Abell L, Roe ND, Kim M, Zhou B, Cao Y, Ritterhoff J, Gu H, Raftery D, Sun H, Tian R (2017) Defective branched-chain amino acid catabolism disrupts glucose metabolism and sensitizes the heart to ischemia-reperfusion injury. Cell Metab 25(2):374–385. https://doi.org/10.1016/j.cmet.2016.11.005
Yu JY, Cao N, Rau CD, Lee RP, Yang J, Flach RJR, Petersen L, Zhu C, Pak YL, Miller RA, Liu Y, Wang Y, Li Z, Sun H, Gao C (2023) Cell-autonomous effect of cardiomyocyte branched-chain amino acid catabolism in heart failure in mice. Acta Pharmacol Sin 44(7):1380–1390. https://doi.org/10.1038/s41401-023-01076-9
Mizuno M, Kuno A, Yano T, Miki T, Oshima H, Sato T, Nakata K, Kimura Y, Tanno M, Miura T (2018) Empagliflozin normalizes the size and number of mitochondria and prevents reduction in mitochondrial size after myocardial infarction in diabetic hearts. Physiol Rep 6(12):e13741. https://doi.org/10.14814/phy2.13741
Lin SC, Hardie DG (2018) AMPK: sensing glucose as well as cellular energy status. Cell Metab 27(2):299–313. https://doi.org/10.1016/j.cmet.2017.10.009
Rybakowska I, Slominska EM, Romaszko P, Lipiński M, Zukowska P, Smolenski RT (2014) Activity of AMP-regulated protein kinase and AMP-deaminase in the heart of mice fed high-fat diet. Nucleosides Nucleotides Nucleic Acids 33(4–6):347–352. https://doi.org/10.1080/15257770.2014.880480
Rybakowska IM, Slominska EM, Romaszko P, Olkowicz M, Kaletha K, Smolenski RT (2015) AMP-regulated protein kinase activity in the hearts of mice treated with low- or high-fat diet measured using novel LC-MS method. Mol Cell Biochem 404(1–2):5–10. https://doi.org/10.1007/s11010-015-2360-z
Murase H, Kuno A, Miki T, Tanno M, Yano T, Kouzu H, Ishikawa S, Tobisawa T, Ogasawara M, Nishizawa K, Miura T (2015) Inhibition of DPP-4 reduces acute mortality after myocardial infarction with restoration of autophagic response in type 2 diabetic rats. Cardiovasc Diabetol 14:103. https://doi.org/10.1186/s12933-015-0264-6
Xu KY, Zweier JL, Becker LC (1995) Functional coupling between glycolysis and sarcoplasmic reticulum Ca2+ transport. Circ Res 77(1):88–97. https://doi.org/10.1161/01.res.77.1.88
Goodwin GW, Ahmad F, Taegtmeyer H (1996) Preferential oxidation of glycogen in isolated working rat heart. J Clin Invest 97(6):1409–1416. https://doi.org/10.1172/JCI118561
Goodwin GW, Ahmad F, Doenst T, Taegtmeyer H (1998) Energy provision from glycogen, glucose, and fatty acids on adrenergic stimulation of isolated working rat hearts. Am J Physiol 274(4):H1239–H1247. https://doi.org/10.1152/ajpheart.1998.274.4.H1239
Katz A (2022) A century of exercise physiology: key concepts in regulation of glycogen metabolism in skeletal muscle. Eur J Appl Physiol 122(8):1751–1772. https://doi.org/10.1007/s00421-022-04935-1
Panayiotou C, Solaroli N, Karlsson A (2014) The many isoforms of human adenylate kinases. Int J Biochem Cell Biol 49:75–83. https://doi.org/10.1016/j.biocel.2014.01.014
Dzeja PP, Vitkevicius KT, Redfield MM, Burnett JC, Terzic A (1999) Adenylate kinase-catalyzed phosphotransfer in the myocardium: increased contribution in heart failure. Circ Res 84(10):1137–1143. https://doi.org/10.1161/01.res.84.10.1137
Aksentijević D, Lygate CA, Makinen K, Zervou S, Sebag-Montefiore L, Medway D, Barnes H, Schneider JE, Neubauer S (2010) High-energy phosphotransfer in the failing mouse heart: role of adenylate kinase and glycolytic enzymes. Eur J Heart Fail 12(12):1282–1289. https://doi.org/10.1093/eurjhf/hfq174
Prandi FR, Evangelista I, Sergi D, Palazzuoli A, Romeo F (2023) Mechanisms of cardiac dysfunction in diabetic cardiomyopathy: molecular abnormalities and phenotypical variants. Heart Fail Rev 28(3):597–606. https://doi.org/10.1007/s10741-021-10200-y
Jaquenod De Giusti C, Palomeque J, Mattiazzi A (2022) Ca2+ mishandling and mitochondrial dysfunction: a converging road to prediabetic and diabetic cardiomyopathy. Pflugers Arch 474(1):33–61. https://doi.org/10.1007/s00424-021-02650-y
Jenkins MJ, Pearson JT, Schwenke DO, Edgley AJ, Sonobe T, Fujii Y, Ishibashi-Ueda H, Kelly DJ, Yagi N, Shirai M (2013) Myosin heads are displaced from actin filaments in the in situ beating rat heart in early diabetes. Biophys J 104(5):1065–1072. https://doi.org/10.1016/j.bpj.2013.01.037
Waddingham MT, Edgley AJ, Astolfo A, Inagaki T, Fujii Y, Du CK, Zhan DY, Tsuchimochi H, Yagi N, Kelly DJ, Shirai M, Pearson JT (2015) Chronic Rho-kinase inhibition improves left ventricular contractile dysfunction in early type-1 diabetes by increasing myosin cross-bridge extension. Cardiovasc Diabetol 14:92. https://doi.org/10.1186/s12933-015-0256-6
Funakoshi A, Miyasaka K, Shinozaki H, Masuda M, Kawanami T, Takata Y, Kono A (1995) An animal model of congenital defect of gene expression of cholecystokinin (CCK)-A receptor. Biochem Biophys Res Commun 210(3):787–796. https://doi.org/10.1006/bbrc.1995.1728
Kawano K, Hirashima T, Mori S, Saitoh Y, Kurosumi M, Natori T (1992) Spontaneous long-term hyperglycemic rat with diabetic complications. Otsuka Long-Evans Tokushima Fatty (OLETF) strain. Diabetes 41(11):1422–1428. https://doi.org/10.2337/diab.41.11.1422
Bi S, Moran TH (2016) Obesity in the Otsuka Long Evans Tokushima fatty rat: mechanisms and discoveries. Front Nutr 27(3):21. https://doi.org/10.3389/fnut.2016.00021
Yagi K, Kim S, Wanibuchi H, Yamashita T, Yamamura Y, Iwao H (1997) Characteristics of diabetes, blood pressure, and cardiac and renal complications in Otsuka Long-Evans Tokushima Fatty rats. Hypertension 29(3):728–735. https://doi.org/10.1161/01.hyp.29.3.728
Mizushige K, Yao L, Noma T, Kiyomoto H, Yu Y, Hosomi N, Ohmori K, Matsuo H (2000) Alteration in left ventricular diastolic filling and accumulation of myocardial collagen at insulin-resistant prediabetic stage of a type II diabetic rat model. Circulation 101(8):899–907. https://doi.org/10.1161/01.cir.101.8.899
Yu Y, Ohmori K, Kondo I, Yao L, Noma T, Tsuji T, Mizushige K, Kohno M (2002) Correlation of functional and structural alterations of the coronary arterioles during development of type II diabetes mellitus in rats. Cardiovasc Res 56(2):303–311. https://doi.org/10.1016/s0008-6363(02)00513-8
Hosomi N, Noma T, Ohyama H, Takahashi T, Kohno M (2002) Vascular proliferation and transforming growth factor-beta expression in pre- and early stage of diabetes mellitus in Otsuka Long-Evans Tokushima fatty rats. Atherosclerosis 162(1):69–76. https://doi.org/10.1016/s0021-9150(01)00683-9
Saito F, Kawaguchi M, Izumida J, Asakura T, Maehara K, Maruyama Y (2003) Alteration in haemodynamics and pathological changes in the cardiovascular system during the development of Type 2 diabetes mellitus in OLETF rats. Diabetologia 46(8):1161–1169. https://doi.org/10.1007/s00125-003-1156-y
Sakata S, Lebeche D, Sakata Y, Sakata N, Chemaly ER, Liang LF, Padmanabhan P, Konishi N, Takaki M, del Monte F, Hajjar RJ (2006) Mechanical and metabolic rescue in a type II diabetes model of cardiomyopathy by targeted gene transfer. Mol Ther 13(5):987–996. https://doi.org/10.1016/j.ymthe.2006.01.002
Sakata S, Lebeche D, Sakata N, Sakata Y, Chemaly ER, Liang LF, Tsuji T, Takewa Y, del Monte F, Peluso R, Zsebo K, Jeong D, Park WJ, Kawase Y, Hajjar RJ (2007) Restoration of mechanical and energetic function in failing aortic-banded rat hearts by gene transfer of calcium cycling proteins. J Mol Cell Cardiol 42(4):852–861. https://doi.org/10.1016/j.yjmcc.2007.01.003
Takada A, Miki T, Kuno A, Kouzu H, Sunaga D, Itoh T, Tanno M, Yano T, Sato T, Ishikawa S, Miura T (2012) Role of ER stress in ventricular contractile dysfunction in type 2 diabetes. PLoS ONE 7(6):e39893. https://doi.org/10.1371/journal.pone.0039893
She P, Van Horn C, Reid T, Hutson SM, Cooney RN, Lynch CJ (2007) Obesity-related elevations in plasma leucine are associated with alterations in enzymes involved in branched-chain amino acid metabolism. Am J Physiol Endocrinol Metab 293(6):E1552–E1563. https://doi.org/10.1152/ajpendo.00134.2007
Herman MA, She P, Peroni OD, Lynch CJ, Kahn BB (2010) Adipose tissue branched chain amino acid (BCAA) metabolism modulates circulating BCAA levels. J Biol Chem 285(15):11348–11356. https://doi.org/10.1074/jbc.M109.075184
Wiklund P, Zhang X, Pekkala S, Autio R, Kong L, Yang Y, Keinänen-Kiukaanniemi S, Alen M, Cheng S (2016) Insulin resistance is associated with altered amino acid metabolism and adipose tissue dysfunction in normoglycemic women. Sci Rep 6:24540. https://doi.org/10.1038/srep24540
Kammermeier H (1987) High energy phosphate of the myocardium: concentration versus free energy change. Basic Res Cardiol 82(Suppl 2):31–36. https://doi.org/10.1007/978-3-662-11289-2_3
Hu Y, Suarez J, Fricovsky E, Wang H, Scott BT, Trauger SA, Han W, Hu Y, Oyeleye MO, Dillmann WH (2009) Increased enzymatic O-GlcNAcylation of mitochondrial proteins impairs mitochondrial function in cardiac myocytes exposed to high glucose. J Biol Chem 284(1):547–555. https://doi.org/10.1074/jbc.M808518200
Diaz-Juarez J, Suarez J, Cividini F, Scott BT, Diemer T, Dai A, Dillmann WH (2016) Expression of the mitochondrial calcium uniporter in cardiac myocytes improves impaired mitochondrial calcium handling and metabolism in simulated hyperglycemia. Am J Physiol Cell Physiol 311(6):C1005–C1013. https://doi.org/10.1152/ajpcell.00236.2016
Ji L, Liu F, Jing Z, Huang Q, Zhao Y, Cao H, Li J, Yin C, Xing J, Li F (2017) MICU1 alleviates diabetic cardiomyopathy through mitochondrial Ca2+-dependent antioxidant response. Diabetes 66(6):1586–1600. https://doi.org/10.2337/db16-1237
Suarez J, Cividini F, Scott BT, Lehmann K, Diaz-Juarez J, Diemer T, Dai A, Suarez JA, Jain M, Dillmann WH (2018) Restoring mitochondrial calcium uniporter expression in diabetic mouse heart improves mitochondrial calcium handling and cardiac function. J Biol Chem 293(21):8182–8195. https://doi.org/10.1074/jbc.RA118.002066
Dia M, Gomez L, Thibault H, Tessier N, Leon C, Chouabe C, Ducreux S, Gallo-Bona N, Tubbs E, Bendridi N, Chanon S, Leray A, Belmudes L, Couté Y, Kurdi M, Ovize M, Rieusset J, Paillard M (2020) Reduced reticulum-mitochondria Ca2+ transfer is an early and reversible trigger of mitochondrial dysfunctions in diabetic cardiomyopathy. Basic Res Cardiol 115(6):74. https://doi.org/10.1007/s00395-020-00835-7
Velmurugan S, Liu T, Chen KC, Despa F, O’Rourke B, Despa S (2023) Distinct effects of mitochondrial Na+/Ca2+ exchanger inhibition and Ca2+ uniporter activation on Ca2+ sparks and arrhythmogenesis in diabetic rats. J Am Heart Assoc 12(14):e029997. https://doi.org/10.1161/JAHA.123.029997
Yu Z, Quamme GA, McNeill JH (1994) Depressed [Ca2+]i responses to isoproterenol and cAMP in isolated cardiomyocytes from experimental diabetic rats. Am J Physiol 266(6 Pt 2):H2334–H2342. https://doi.org/10.1152/ajpheart.1994.266.6.H2334
Lagadic-Gossmann D, Buckler KJ, Le Prigent K, Feuvray D (1996) Altered Ca2+ handling in ventricular myocytes isolated from diabetic rats. Am J Physiol 270(5 Pt 2):H1529–H1537. https://doi.org/10.1152/ajpheart.1996.270.5.H1529
Belke DD, Swanson EA, Dillmann WH (2004) Decreased sarcoplasmic reticulum activity and contractility in diabetic db/db mouse heart. Diabetes 53(12):3201–3208. https://doi.org/10.2337/diabetes.53.12.3201
Garbincius JF, Elrod JW (2022) Mitochondrial calcium exchange in physiology and disease. Physiol Rev 102(2):893–992. https://doi.org/10.1152/physrev.00041.2020
Kaludercic N, Di Lisa F (2020) Mitochondrial ROS formation in the pathogenesis of diabetic cardiomyopathy. Front Cardiovasc Med 7:12. https://doi.org/10.3389/fcvm.2020.00012
Privratsky JR, Wold LE, Sowers JR, Quinn MT, Ren J (2003) AT1 blockade prevents glucose-induced cardiac dysfunction in ventricular myocytes: role of the AT1 receptor and NADPH oxidase. Hypertension 42(2):206–212. https://doi.org/10.1161/01.HYP.0000082814.62655.85
Zhang M, Kho AL, Anilkumar N, Chibber R, Pagano PJ, Shah AM, Cave AC (2006) Glycated proteins stimulate reactive oxygen species production in cardiac myocytes: involvement of Nox2 (gp91phox)-containing NADPH oxidase. Circulation 113(9):1235–1243. https://doi.org/10.1161/CIRCULATIONAHA.105.581397
Roe ND, Thomas DP, Ren J (2011) Inhibition of NADPH oxidase alleviates experimental diabetes-induced myocardial contractile dysfunction. Diabetes Obes Metab 13(5):465–473. https://doi.org/10.1111/j.1463-1326.2011.01369.x
Varga ZV, Kupai K, Szűcs G, Gáspár R, Pálóczi J, Faragó N, Zvara A, Puskás LG, Rázga Z, Tiszlavicz L, Bencsik P, Görbe A, Csonka C, Ferdinandy P, Csont T (2013) MicroRNA-25-dependent up-regulation of NADPH oxidase 4 (NOX4) mediates hypercholesterolemia-induced oxidative/nitrative stress and subsequent dysfunction in the heart. J Mol Cell Cardiol 62:111–121. https://doi.org/10.1016/j.yjmcc.2013.05.009
Anderson EJ, Kypson AP, Rodriguez E, Anderson CA, Lehr EJ, Neufer PD (2009) Substrate-specific derangements in mitochondrial metabolism and redox balance in the atrium of the type 2 diabetic human heart. J Am Coll Cardiol 54(20):1891–1898. https://doi.org/10.1016/j.jacc.2009.07.031
Mariappan N, Elks CM, Sriramula S, Guggilam A, Liu Z, Borkhsenious O, Francis J (2010) NF-kappaB-induced oxidative stress contributes to mitochondrial and cardiac dysfunction in type II diabetes. Cardiovasc Res 85(3):473–483. https://doi.org/10.1093/cvr/cvp305
Bombicino SS, Iglesias DE, Mikusic IAR, D’Annunzio V, Gelpi RJ, Boveris A, Valdez LB (2016) Diabetes impairs heart mitochondrial function without changes in resting cardiac performance. Int J Biochem Cell Biol 81(Pt B):335–345. https://doi.org/10.1016/j.biocel.2016.09.018
Koncsos G, Varga ZV, Baranyai T, Boengler K, Rohrbach S, Li L, Schlüter KD, Schreckenberg R, Radovits T, Oláh A, Mátyás C, Lux Á, Al-Khrasani M, Komlódi T, Bukosza N, Máthé D, Deres L, Barteková M, Rajtík T, Adameová A, Szigeti K, Hamar P, Helyes Z, Tretter L, Pacher P, Merkely B, Giricz Z, Schulz R, Ferdinandy P (2016) Diastolic dysfunction in prediabetic male rats: Role of mitochondrial oxidative stress. Am J Physiol Heart Circ Physiol 311(4):H927–H943. https://doi.org/10.1152/ajpheart.00049.2016
Jeong EM, Chung J, Liu H, Go Y, Gladstein S, Farzaneh-Far A, Lewandowski ED, Dudley SC Jr (2016) Role of mitochondrial oxidative stress in glucose tolerance, insulin resistance, and cardiac diastolic dysfunction. J Am Heart Assoc 5(5):e003046. https://doi.org/10.1161/JAHA.115.003046
Pacher P, Beckman JS, Liaudet L (2007) Nitric oxide and peroxynitrite in health and disease. Physiol Rev 87(1):315–424. https://doi.org/10.1152/physrev.00029.2006
Soliman H, Gador A, Lu YH, Lin G, Bankar G, MacLeod KM (2012) Diabetes-induced increased oxidative stress in cardiomyocytes is sustained by a positive feedback loop involving Rho kinase and PKCβ2. Am J Physiol Heart Circ Physiol 303(8):H989–H1000. https://doi.org/10.1152/ajpheart.00416.2012
Lin C, Guo Y, Xia Y, Li C, Xu X, Qi T, Zhang F, Fan M, Hu G, Zhao H, Zhao H, Liu R, Gao E, Yan W, Tao L (2021) FNDC5/Irisin attenuates diabetic cardiomyopathy in a type 2 diabetes mouse model by activation of integrin αV/β5-AKT signaling and reduction of oxidative/nitrosative stress. J Mol Cell Cardiol 160:27–41. https://doi.org/10.1016/j.yjmcc.2021.06.013
Rajesh M, Mukhopadhyay P, Bátkai S, Mukhopadhyay B, Patel V, Haskó G, Szabó C, Mabley JG, Liaudet L, Pacher P (2009) Xanthine oxidase inhibitor allopurinol attenuates the development of diabetic cardiomyopathy. J Cell Mol Med 13(8B):2330–2341. https://doi.org/10.1111/j.1582-4934.2008.00564.x
Battelli MG, Bolognesi A (1842) Polito L (2014) Pathophysiology of circulating xanthine oxidoreductase: new emerging roles for a multi-tasking enzyme. Biochim Biophys Acta 9:1502–1517. https://doi.org/10.1016/j.bbadis.2014.05.022
Faria A, Persaud SJ (2017) Cardiac oxidative stress in diabetes: mechanisms and therapeutic potential. Pharmacol Ther 172:50–62. https://doi.org/10.1016/j.pharmthera.2016.11.013
Kakkar R, Kalra J, Mantha SV, Prasad K (1995) Lipid peroxidation and activity of antioxidant enzymes in diabetic rats. Mol Cell Biochem 151(2):113–119. https://doi.org/10.1007/BF01322333
Vincent HK, Powers SK, Dirks AJ, Scarpace PJ (2001) Mechanism for obesity-induced increase in myocardial lipid peroxidation. Int J Obes 25(3):378–388. https://doi.org/10.1038/sj.ijo.0801536
Boudina S, Sena S, Theobald H, Sheng X, Wright JJ, Hu XX, Aziz S, Johnson JI, Bugger H, Zaha VG, Abel ED (2007) Mitochondrial energetics in the heart in obesity-related diabetes: direct evidence for increased uncoupled respiration and activation of uncoupling proteins. Diabetes 56(10):2457–2466. https://doi.org/10.2337/db07-0481
Ye G, Metreveli NS, Donthi RV, Xia S, Xu M, Carlson EC, Epstein PN (2004) Catalase protects cardiomyocyte function in models of type 1 and type 2 diabetes. Diabetes 53(5):1336–1343. https://doi.org/10.2337/diabetes.53.5.1336
Shen X, Zheng S, Thongboonkerd V, Xu M, Pierce WM Jr, Klein JB, Epstein PN (2004) Cardiac mitochondrial damage and biogenesis in a chronic model of type 1 diabetes. Am J Physiol Endocrinol Metab 287(5):E896-905. https://doi.org/10.1152/ajpendo.00047.2004
Herlein JA, Fink BD, O’Malley Y, Sivitz WI (2009) Superoxide and respiratory coupling in mitochondria of insulin-deficient diabetic rats. Endocrinology 150(1):46–55. https://doi.org/10.1210/en.2008-0404
Turko IV, Murad F (2003) Quantitative protein profiling in heart mitochondria from diabetic rats. J Biol Chem 278(37):35844–35849. https://doi.org/10.1074/jbc.M303139200
Umbarkar P, Singh S, Arkat S, Bodhankar SL, Lohidasan S, Sitasawad SL (2015) Monoamine oxidase-A is an important source of oxidative stress and promotes cardiac dysfunction, apoptosis, and fibrosis in diabetic cardiomyopathy. Free Radic Biol Med 87:263–273. https://doi.org/10.1016/j.freeradbiomed.2015.06.025
Sun Y, Tian Z, Liu N, Zhang L, Gao Z, Sun X, Yu M, Wu J, Yang F, Zhao Y, Ren H, Chen H, Zhao D, Wang Y, Dong S, Xu C, Lu F, Zhang W (2018) Exogenous H2S switches cardiac energy substrate metabolism by regulating SIRT3 expression in db/db mice. J Mol Med (Berl) 96(3–4):281–299. https://doi.org/10.1007/s00109-017-1616-3
Sun Y, Teng Z, Sun X, Zhang L, Chen J, Wang B, Lu F, Liu N, Yu M, Peng S, Wang Y, Zhao D, Zhao Y, Ren H, Cheng Z, Dong S, Lu F, Zhang W (2019) Exogenous H2S reduces the acetylation levels of mitochondrial respiratory enzymes via regulating the NAD+-SIRT3 pathway in cardiac tissues of db/db mice. Am J Physiol Endocrinol Metab 317(2):E284–E297. https://doi.org/10.1152/ajpendo.00326.2018
Tan Y, Ichikawa T, Li J, Si Q, Yang H, Chen X, Goldblatt CS, Meyer CJ, Li X, Cai L, Cui T (2011) Diabetic downregulation of Nrf2 activity via ERK contributes to oxidative stress-induced insulin resistance in cardiac cells in vitro and in vivo. Diabetes. https://doi.org/10.2337/db10-1164
Luo J, Yan D, Li S, Liu S, Zeng F, Cheung CW, Liu H, Irwin MG, Huang H, Xia Z (2020) Allopurinol reduces oxidative stress and activates Nrf2/p62 to attenuate diabetic cardiomyopathy in rats. J Cell Mol Med 24(2):1760–1773. https://doi.org/10.1111/jcmm.14870
Oka SI, Byun J, Huang CY, Imai N, Ralda G, Zhai P, Xu X, Kashyap S, Warren JS, Alan Maschek J, Tippetts TS, Tong M, Venkatesh S, Ikeda Y, Mizushima W, Kashihara T, Sadoshima J (2021) Nampt potentiates antioxidant defense in diabetic cardiomyopathy. Circ Res 129(1):114–130. https://doi.org/10.1161/CIRCRESAHA.120.317943
Kim BS, Serebreni L, Fallica J, Hamdan O, Wang L, Johnston L, Kolb T, Damarla M, Damico R, Hassoun PM (2015) Cyclin-dependent kinase five mediates activation of lung xanthine oxidoreductase in response to hypoxia. PLoS ONE 10(4):e0124189. https://doi.org/10.1371/journal.pone.0124189
Ghio AJ, Kennedy TP, Stonehuerner J, Carter JD, Skinner KA, Parks DA, Hoidal JR (2002) Iron regulates xanthine oxidase activity in the lung. Am J Physiol Lung Cell Mol Physiol 283(3):L563–L572. https://doi.org/10.1152/ajplung.00413.2000
Martelin E, Lapatto R, Raivio KO (2002) Regulation of xanthine oxidoreductase by intracellular iron. Am J Physiol Cell Physiol 283(6):C1722–C1728. https://doi.org/10.1152/ajpcell.00280.2002
Petrosillo G, Matera M, Moro N, Ruggiero FM, Paradies G (2009) Mitochondrial complex I dysfunction in rat heart with aging: critical role of reactive oxygen species and cardiolipin. Free Radic Biol Med 46(1):88–94. https://doi.org/10.1016/j.freeradbiomed.2008.09.031
Chen CL, Zhang L, Jin Z, Kasumov T, Chen YR (2022) Mitochondrial redox regulation and myocardial ischemia-reperfusion injury. Am J Physiol Cell Physiol 322(1):C12–C23. https://doi.org/10.1152/ajpcell.00131.2021
Jang S, Javadov S (2017) Association between ROS production, swelling and the respirasome integrity in cardiac mitochondria. Arch Biochem Biophys 630:1–8. https://doi.org/10.1016/j.abb.2017.07.009
Moe GW, Marín-García J (2016) Role of cell death in the progression of heart failure. Heart Fail Rev 21(2):157–167. https://doi.org/10.1007/s10741-016-9532-0
Del Re DP, Amgalan D, Linkermann A, Liu Q, Kitsis RN (2019) Fundamental mechanisms of regulated cell death and implications for heart disease. Physiol Rev 99(4):1765–1817. https://doi.org/10.1152/physrev.00022.2018
Jia G, DeMarco VG, Sowers JR (2016) Insulin resistance and hyperinsulinaemia in diabetic cardiomyopathy. Nat Rev Endocrinol 12(3):144–153. https://doi.org/10.1038/nrendo.2015.216
Zorov DB, Juhaszova M, Sollott SJ (2014) Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev 94(3):909–950. https://doi.org/10.1152/physrev.00026.2013
Bauer TM, Murphy E (2020) Role of mitochondrial calcium and the permeability transition pore in regulating cell death. Circ Res 126(2):280–293. https://doi.org/10.1161/CIRCRESAHA.119.316306
Bernardi P, Carraro M, Lippe G (2022) The mitochondrial permeability transition: recent progress and open questions. FEBS J 289(22):7051–7074. https://doi.org/10.1111/febs.16254
Miki T, Miura T, Hotta H, Tanno M, Yano T, Sato T, Terashima Y, Takada A, Ishikawa S, Shimamoto K (2009) Endoplasmic reticulum stress in diabetic hearts abolishes erythropoietin-induced myocardial protection by impairment of phospho-glycogen synthase kinase-3beta-mediated suppression of mitochondrial permeability transition. Diabetes 58(12):2863–2872. https://doi.org/10.2337/db09-0158
Hotta H, Miura T, Miki T, Togashi N, Maeda T, Kim SJ, Tanno M, Yano T, Kuno A, Itoh T, Satoh T, Terashima Y, Ishikawa S, Shimamoto K (2010) Angiotensin II type 1 receptor-mediated upregulation of calcineurin activity underlies impairment of cardioprotective signaling in diabetic hearts. Circ Res 106(1):129–132. https://doi.org/10.1161/CIRCRESAHA.109.205385
Miki T, Itoh T, Sunaga D, Miura T (2012) Effects of diabetes on myocardial infarct size and cardioprotection by preconditioning and postconditioning. Cardiovasc Diabetol 11:67. https://doi.org/10.1186/1475-2840-11-67
Itoh T, Kouzu H, Miki T, Tanno M, Kuno A, Sato T, Sunaga D, Murase H, Miura T (2012) Cytoprotective regulation of the mitochondrial permeability transition pore is impaired in type 2 diabetic Goto-Kakizaki rat hearts. J Mol Cell Cardiol 53(6):870–879. https://doi.org/10.1016/j.yjmcc.2012.10.001
Kortüm F, Jamra RA, Alawi M, Berry SA, Borck G, Helbig KL, Tang S, Huhle D, Korenke GC, Hebbar M, Shukla A, Girisha KM, Steinlin M, Waldmeier-Wilhelm S, Montomoli M, Guerrini R, Lemke JR, Kutsche K (2018) Clinical and genetic spectrum of AMPD2-related pontocerebellar hypoplasia type 9. Eur J Hum Genet 26(5):695–708. https://doi.org/10.1038/s41431-018-0098-2
Gilboa T, Elefant N, Meiner V, Hacohen N (2023) Delineating the phenotype and genetic basis of AMPD2-related pontocerebellar hypoplasia. Neurogenetics 24(1):61–66. https://doi.org/10.1007/s10048-022-00706-4
Kalsi KK, Yuen AH, Rybakowska IM, Johnson PH, Slominska E, Birks EJ, Kaletha K, Yacoub MH, Smolenski RT (2003) Decreased cardiac activity of AMP deaminase in subjects with the AMPD1 mutation–a potential mechanism of protection in heart failure. Cardiovasc Res 59(3):678–684. https://doi.org/10.1016/s0008-6363(03)00497-8
Kalsi KK, Yuen AH, Johnson PH, Birks EJ, Yacoub MH, Smolenski RT (2005) AMPD1 C34T mutation selectively affects AMP-deaminase activity in the human heart. Nucleos Nucleot Nucl Acids 24(4):287–288. https://doi.org/10.1081/NCN-59721
Smolenski RT, Rybakowska I, Turyn J, Romaszko P, Zabielska M, Taegtmeyer A, Słomińska EM, Kaletha KK, Barton PJ (2014) AMP deaminase 1 gene polymorphism and heart disease-a genetic association that highlights new treatment. Cardiovasc Drugs Ther 28(2):183–189. https://doi.org/10.1007/s10557-013-6506-5
Marx N, Federici M, Schütt K, Müller-Wieland D, Ajjan RA, Antunes MJ, Christodorescu RM, Crawford C, Di Angelantonio E, Eliasson B, Espinola-Klein C, Fauchier L, Halle M, Herrington WG, Kautzky-Willer A, Lambrinou E, Lesiak M, Lettino M, McGuire DK, Mullens W, Rocca B, Sattar N, ESC Scientific Document Group (2023) 2023 ESC guidelines for the management of cardiovascular disease in patients with diabetes. Eur Heart J 44(39):4043–4140. https://doi.org/10.1093/eurheartj/ehad192.Erratum.In:EurHeartJ.2023;44(48):5060
Abovich A, Matasic DS, Cardoso R, Ndumele CE, Blumenthal RS, Blankstein R, Gulati M (2023) The AHA/ACC/HFSA 2022 heart failure guidelines: changing the focus to heart failure prevention. Am J Prev Cardiol 15:100527. https://doi.org/10.1016/j.ajpc.2023.100527
Cheng J, Morisaki H, Sugimoto N, Dohi A, Shintani T, Kimura E, Toyama K, Ikawa M, Okabe M, Higuchi I, Matsuo S, Kawai Y, Hisatome I, Sugama T, Holmes EW, Morisaki T (2014) Effect of isolated AMP deaminase deficiency on skeletal muscle function. Mol Genet Metab Rep 1:51–59. https://doi.org/10.1016/j.ymgmr.2013.12.004
Funding
TM is funded by Grant 21K08035 from the Japan Society for the Promotion of Science, Tokyo, Japan and a Grant for Scientific Research from Hokkaido University of Science, Sapporo, Japan. HK is funded by Grant 23K07481 from the Japan Society for the Promotion of Science, Tokyo, Japan. MT is funded by Grant 21K08110 from the Japan Society for the Promotion of Science, Tokyo, Japan. AK is funded by Grant 23K06152 from the Japan Society for the Promotion of Science, Tokyo, Japan.
Author information
Authors and Affiliations
Contributions
TM conceptualized the topic and wrote the first draft of the manuscript. HK, MT, YK, and AK commented on previous versions of the manuscript. All figures were prepared by TM. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have not disclosed any competing interests.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Miura, T., Kouzu, H., Tanno, M. et al. Role of AMP deaminase in diabetic cardiomyopathy. Mol Cell Biochem (2024). https://doi.org/10.1007/s11010-024-04951-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11010-024-04951-z