Abstract
Lakes, the main entities of lacustrine environments, are a rich archive of environmental and geogenic changes in terms of compositional variation of water and sediment. Water and sediment samples (N = 173) were collected during 2013–2014 from the Wular Lake, one of the important fresh lakes within the Indian landmass. The study provides insights on the solutes acquisition mechanism and provenance of ionic constituents within the lake water and the sediments. Besides, the impact of catchment attributes on the lake system was in addition assessed. The hydrochemical results suggest that the chemical weathering of silicate and carbonates within the catchment shapes the lake water chemistry and characterizes the facies pattern into a hybrid type. The geochemical results of the lake sediments demonstrate that the improved abrasion rates and ensuant settling of detritus into the lake are closely linked with the prominent physical weathering over chemical weathering. The new finding of the present study is that sediments represent an unweathered basalt compositional trend, plausible provenance from mafic rocks, experiencing low to moderate degree of chemical weathering. The study found that increased encroachment within the lake catchment due to continued anthropogenic forcing is the primary source contributing the organic matter (OM) as well as the higher levels of Cl, NO3, SO4, and P to the lake. These findings corroborate with the land use-land cover changes (from the last 50 years) within the lake catchment in significantly deteriorating the lake system. The study recommends that the ongoing conversion of lake peripheral areas into urban settlement and agro-horticulture land by filling activities should be restricted.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Lakes are the main constituents of the lacustrine environments, which archive the temporal environmental changes in terms of compositional variation of water and sediment (Müller et al., 2021; Bhang et al., 2019; Dubois et al., 2018; Luque & Julià, 2002). They are considered because they link to hydrological systems on local and transboundary scales, provide water resources, and support a variety of ecosystem services (Bhateria & Jain, 2016; Hoverman & Johnson, 2012; Ballatore & Muhandiki, 2002). Recent evidence suggest that these aquatic systems respond promptly to any external variable activity than marine, hence are more imperilled (Adrian et al., 2016; Cohen, 2003). Since 68% of the world’s freshwater is contained in large lakes (Beeton et al., 2008; Reid & Beeton, 1992; Stephens et al., 2020), it is imperative that the health of these freshwater ecosystems be continuously monitored and assessed.
Despite the fact that lakes are found around the world (Downing et al., 2006; Meybeck, 1995), India is among the few countries with an optimal distribution of lakes. Wular, Dal, Loktak, Kolleru, Pulicat, Bhimtal, and Gurdungmar are some major natural lakes in India, offering great ecological and societal benefits. The Kashmir Valley in India is richly endowed with abundant freshwater resources, manifested in the many lakes that dot the valley’s landscape (Pandit, 1999; Badar et al., 2013; Saleem et al., 2015). Among them, Wular Lake represents the foremost diverse hydrological features, acting as a huge sink that absorbs the high annual floods of major streams of the Jhelum Basin (Jeelani & Shah, 2006; Sarah et al., 2011). The Wular is the major freshwater lake within the Indian subcontinent (Sheikh et al., 2014; Saini et al., 2008) being designated as a wetland of Global Importance (Wetlands International, 2007). The lake is taken into account among a couple of natural lakes that bear tremendous ecological and socio-economic importance, yielding its 60% (even more) fish production of the region (Rumysa et al., 2012).
Over the past few years, there is a drastic reduction in the storage capacity of the Wular Lake due to excessive sedimentation triggered by erosion and increasing illegal encroachments within the lake catchment. As a result, water quality deteriorated in the lake and eutrophication increased, which is certainly a growing concern. Few noteworthy work conducted within the study region (e.g., Hassan et al., 2015; Shah et al., 2020; Sheik et al., 2014) come with a conclusion that natural mineralization from host rocks and human settlement within the lake periphery govern the ionic characteristics of lake water and detritus. Considering the lake as an open system with its close link to peripheral catchment, the importance of updated knowledge about the lake system cannot be overemphasized. Additionally, due to its strategic importance within the Indian landmass for being a major freshwater lake, assessing the impact of catchment attributes on the lake system becomes critical. The present study aims to better understand (1) the solute acquisition mechanism and provenance of ionic constituents in water and the sediment (2) to gain insights into the denudation processes within the Wular lacustrine. The study will help the policymakers in developing long-term future management plans by restricting the anthropogenic forcing within the lake catchment so that freshwater resources from the lake can be effectively protected and conserved.
Study area
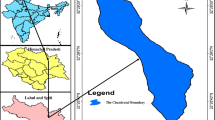
The testing site (Wular Lake) is a lowland part of the Jhelum Basin within Himalayan Synclinorium. With latitudes 34.87374 and 34.87827 and longitudes 74.3341 and 74.4402 (Fig. 1), it is bounded by the mountains on both the western and eastern sides and covers an area of 189 km2 (Hassan et al., 2015). Although the Wular Lake catchment is principally linked with the whole Jhelum Basin, comprising 24 watersheds, however, the perineal Erin and Madhumati streams are the prominent ones feeding the lake directly. Rugged rock outcrops and permanent snow and glacier fields characterize the upper elevation (> 3500) area of the lake catchment. The LULC characteristics of the study region (Fig. 1) show that forests (sparse and dense) are the main components covering 43% of the catchment basin as compared to agriculture (20%) and horticulture land (9%) (Mushtaq & Pandey, 2014). The lake is made within the intermountain setting and hence, it receives an outsized number of sediments from the encompassing hills comprising mafic igneous rocks, Quaternary sediments, and associated Triassic carbonates.
Material and methods
Sampling program
A total (N = 173) samples (i.e., lake water: 130, sediment: n = 43) were collected from seven sites within the lake body and along its peripheral catchments on different frequencies employing a grid pattern (Fig. 1, Table 1). Water samples were taken in two labelled and well-drained high-density polyethyene bottles. The primary sampling set (1-L) meant for the cation measurement was acidified with concentrated nitric acid (HNO3) and filtered by a 0.45 µm nucleopore membrane, while the second unfiltered sampling set (1-L) was used for anion measurement. Similarly, fresh deposited bottom sediment samples were collected at different transects with the assistance of a scoop from shallow areas (< 1 m) and from deeper areas (1–6 m) by the Ekman-Birge Dredge. Besides, some samples were also collected from the lake periphery. All the collected samples were packed in polythene envelops and were dried in an oven at 5 °C before their analysis.
Analytical technique
In situ parameters like pH and electric conductivity (EC) were measured by a portable water testing kit. Lake water samples were analyzed for their ionic constituents at Aquatic Ecology Laboratory, Department of Environment Sciences, University of Kashmir, by following the prescribed standard methods (APHA, 1999). The anions and cations were determined by standard methods including titration, flame, and spectrometry methods. Ca2+ and Mg2+ were measured by ethylene diamine tetraacetate titration while the Cl− and HCO3− concentrations were determined by AgNO3− (0.02 N) and HCl (0.01 N) titrating methods, respectively. The concentrations of Na+ and K+ from the water samples were measured by a flame emission photometer (Systronics-130), while SO4−, F−, Fe2+, and NO3− concentrations were measured with a UV–VIS spectrophotometer (EI-1371) at wavelengths of 420, 570, 508, and 220 nm. Within the majority of the water samples, the entire cation charge (TZ+ in meq L−1) balanced that of the entire anions (TZ− in meq L−1) within analytical uncertainties ± 8%.
The Wular Lake sediment samples were powdered to −200 mesh. The Sequential X-ray Spectrometer (SIEMENS-SRS3000) at the Wadia Institute of Himalayan Geology, Dehradun, India, in 2014 analyzed sediments for elemental concentrations. Few samples were also analyzed at the Central Institute of Mining and Fuel Research, Dhanbad, Jharkhand using the X-RF technique. Several in-house (e.g., DG-H and MB-H) and international standards including JG-1, JG-2, SCO-1, and GSD-9 were used during the analysis. The analytical precision for major elements is within 1% while for trace elements, it ranged from 5 to 10%. Similarly, organic carbon, nitrogen, hydrogen, and phosphate were measured at the Centre of Research and Development (CORD), University of Kashmir. Organic carbon was measured by Black and Wackley methods (Wackley & Black, 1934), nitrogen by the Kjeldahl method (Kjeldahl, 1883), and phosphorus by ammonium molybdate method (Stockdale, 1958).
Results
Hydrochemical characteristics
Considerable variation within the physical and ionic constituents of lake water was observed (Fig. 2; Table 2). The character of lake water was found alkaline with a pH starting from 7.1 to 9. Among TZ+, the calcium (Ca2+) dominates the cation budget (TZ+) followed magnesium (Mg2+), sodium (Na+), and potassium (K+), whereas bicarbonate (HCO3−) dominates of anion budget (TZ−) trailed by chloride (Cl−), sulfate (SO4−), nitrate (NO3−), and phosphorus (P−). Ca2+ and Mg2+ concentrations within the lake water ranged from 38 to 107 mg L−1 and 5 to 45 mg L−1, both being higher at the site I and lower at sites III, IV, and V. Na+ and K+ concentrations, ranging from 2–18 mg L−1 and 1–5 mg L−1 were reported higher at sites II and VI, respectively. Similarly, the concentration of HCO3− and SO4− varied from 60 to 245 mg L−1 and 1 to 20 mg L−1, being higher at sites IV and V, and lower at site VII. The concentrations of Cl−, NO3−, and P− within the lake waters varied from 6 to 28 mg L−1, 330 to 2110 µg L−1, and 60 to 393 µg L−1, respectively. The higher amounts of Cl−, NO3−, SO4−, and P− were noted near the peripheral areas of the lake. Further, solutes in lake waters showed the effect of dilution with higher concentration during the lean flow in winter and lower values during the higher flow in summer/autumn (Fig. 2).
Sediment geochemical characteristics
Significant variations are observed within the major oxides (SiO2, CaO, Fe2O3, Al2O3, and MgO) content with the foremost abundant are SiO2 (53–60%) and Al2O3 (11–16%) followed by Fe2O3 (5.1–10.2%), CaO (1.4–6.8%), and MgO (2.7–4.9%). However, smaller variation (< 2%) is found within the elements like K2O, TiO2, and Na2O (Fig. 4a, b). Trace elemental abundances ranged from Zn (29 to 171 ppm), Cu (4 to 55 ppm), Ni (6 to 68 ppm), Co (3 to 23 ppm), and Pb (6 to 32 ppm). The statistical summary of all the ionic constituents in sediments samples of the Wular Lake is presented in Table 3. Compared to average Panjal basalt, no significant changes were found in the Si and Al concentrations in lake sediments; however, there is a notable depletion of Ca and Na (Fig. 4b). Even though Fe acts in the same way as the above-mentioned elements, its decrease has some additional inferences owing to its reacting nature. The minor change in MnO and P2O5 content of the lake sediments can be observed when compared to source rocks signifying the assessable display of these elemental contents from source to sink. The trace elements also show notable compositional variation in contrast to upper continental crust (UCC) and average Panjal basalts (Fig. 6). The Al2O3 content of the Wular Lake sediments displays a significant negative correlation SiO2 (R2 = 0.6) with SiO2, TiO2 (R2 = −0.4), CaO (R2 = −0.72), Na2O (R2 = −0.8), and MgO (R2 = −0.81), while there is significant positive correlation of K2O (R2 = 0.91), Fe2O3 (R2 = 0.92), Co (R2 = 0.73), Ni (R2 = 0.77), and Cu (R2 = 0.78), with Al2O3. We also observed a negative correlation between SiO2 and Fe2O3 (R2 = − 0.53).
Discussion
Weathering and provenance
Hydrochemical data plotted as Ca + Mg vs. HCO3 + SO4, Na+ + K+ vs. TZ+, TDS vs. Na / (Na + Ca), and Ca + Mg-HCO3-SO4 vs. Na-Cl (Fig. 3a-d) along with Piper trilinear and Langlair-Ludwig diagrams (Fig. 3e, f) suggests that lake water chemistry is governed by chemical weathering of silicate and carbonates, besides the inputs from anthropogenic sources. The chemical weathering of hosted rocks within the immediate catchment is additionally reflected by the higher concentration of HCO3 (245 mg L−1) and pH > 9. Within the Ca + Mg vs. HCO3 + SO4 plot (Fig. 3a), most of the points fall above the aquiline and hence, clearly suggests the contribution from silicate and carbonate dissolution in regulating the lake water chemistry. The dominance of Ca and Mg over HCO3 and SO4 (Fig. 3a) evinces the interaction of water with silicate lithology because the carbonate-derived Ca2+ and Mg2+ is not equal to the carbonate-derived bicarbonate (HCO3−). This observation of silicate weathering is further evinced from Na+ + K+ vs. TZ+ and Ca + Mg-HCO3-SO4 vs. Na-Cl plots (Fig. 3b, c). A majority (85%) of samples showed Na/Cl ratio > 1, thus, indicating the release of sodium during silicate weathering. Furthermore, it had been observed that majority of the samples lie near the Na+ + K+ = 0.2 TZ+ line in Na+ + K+ vs. TZ+ plot (Fig. 3b). The calculated Na+ + K+/TZ+ ratio of 0.05–0.19 yields meaningful results of silicate dissolution (Shah & Jeelani, 2016). The processes of cation–anion exchange within the lake basin were clearly observed by the Ca + Mg-HCO3-SO4 vs. Na-Cl scatter plot (Fig. 3c) where most of the data points showed a linear relationship between Na-Cl and Ca + Mg-HCO3-SO4 with a slope of ~ 100% towards the positive trend, suggesting that Ca, Mg, and Na are interrelated through cation–anion exchange reaction. The Ca-Mg-HCO3 water type (refer, Fig. 3e, f) suggests that lake water is being fed with freshwaters, and this further evinces the involvement of multiple lithologies in bringing alkali and alkaline-earth metal to characterize the facies pattern (Fig. 4).
Even though there is notable depletion of Ca and Na in lake sediments, but the insignificant change in the Si and Al concentrations could be related to the breakdown of principal minerals like pyroxenes, amphiboles, and plagioclases present in the source rocks. Additionally, such elements after subsequent weathering could even remain in solution in the form of soluble minerals as carbonate and chloride (Garzanti et al., 2010; Rashid et al., 2015). The inflowing of these dissolved salts into the lake through different transporting agencies is accountable for increasing pH in the lake water. The large variation in the abundances of trace element metals such as Ni and Co in the lake sediments reflects the impact of cation exchange. Seybold et al. (2005) suggest that higher levels of pH, clay content, and organic matter increase the cation exchange capacity (CEC). The significant negative correlation of Al2O3 content of sediments with SiO2 (R2 = 0.6), TiO2 (R2 = −0.4), CaO (R2 = −0.72), Na2O (R2 = −0.8), and MgO (R2 = −0.81) evinces that they are not connected to Al–bearing minerals, and associated minerals like feldspars are not influenced from the detrital source, while the significant positive correlation of K2O (R2 = 0.91), Fe2O3 (R2 = 0.92), Co (R2 = 0.73), Ni (R2 = 0.77), and Cu (R2 = 0.78), with Al2O3, indicates their source from pyroxenes, hornblende, garnet, biotite, and spinel, present in the hosted lithologies. Further, the significant negative correlation between SiO2 and Fe2O3 (R2 = −0.53) content infers the presence of hematite and goethite in the form of silica/iron oxides and hydroxides in the lake sediments. Iron after entering into solution as carbonate is converted to iron carbonate, which then oxidizes as hematite (Wilson, 2004; Firth & Garden, 2008). This produces convolution of rocks and persists as insoluble deposits at a particular place for some period, developing some portion of regolith, that are eventually being transported by various agencies to the lake basin. The lack of this correlation could also be due to the influence of anthropogenic forcing within the catchment.
To understand the weathering intensity of the sediments, the A-CN-K and A-CN-K-FM diagrams and chemical index of alteration (CIA) are plotted (Fig. 5). The A-CN-K and A-CN-K-FM diagrams (Gaillardet et al., 1999; Garzanti et al., 2013) and CIA (Dinis et al., 2020; Nesbitt & Young, 1984) are widely accepted and give an accurate estimate of weathering and provenance (Ryan & Williams, 2007; Rashid & Ganai, 2018). The calculated CIA values (50–74) in the Wular Lake sediments reflect that sediments experienced a low to moderate degrees of chemical weathering. The weathering intensity and effect of K-metasomatism were also assessed by A-CN-K and A-CN-K-FM triangular diagrams (Fig. 5a, b). The clustering of samples parallel to basaltic and andesitic trend lines of triangles (Fig. 5a, b) also corroborate the low to moderate degree of chemical weathering. Except Al (which is retained in the weathered residue), both alkali and alkaline-earth elements are leached out into solution during the course of weathering. Ca- and Na-rich mineral phases like the amphiboles and the plagioclase have a tendency to dissolute faster than potassium-rich minerals such as orthoclase and micas (Brantley, 2003; Wilson, 2004). The continued weathering of the main source rock may lead the composition upwards, and parallel to A-CN join of the A-CN-K plot (Fig. 5a). Consequently, highly weathered rocks will have higher Al content, and hence plotting towards “A apex” on this diagram. The shift in the positions samples is mainly due to the mobilization of Ca and Na, and a shift closer to the “A” end would suggest greater loss of Na and Ca in them. Considering the mafic mineral component in rock weathering, the studied sediments are plotted in the A-CNK-FM plot (Fig. 5b) which depicts the weathering and provenance-related changes involving Fe–Mg–bearing minerals. In the A-CNK-FM diagram (Fig. 5b), the Wular Lake sediments exhibit a trend parallel to basalt and andesite moving towards the A-FM join. The moving away from CNK and FM infers the mobilization of Ca, Na, K, Fe, and Mg in all suites of sediments and enrichment of Al in finer ones. Most of the samples plot well beneath the representative shale values. The mixing of various source components of variable compositions could be the plausible reason for this trend. This is also supported by the fact that the deep-water sediments exhibit high clay content (66–81%) characterized with leptokurtic (0.3–2.5), positive skewness (0.22 to − 0.6), and higher values of phi (1–8).
a A-CN-K and b A-CNK-FM diagram (after Nesbitt & Young, 1996) showing predicted weathering trends, provenance, and chemical index of alteration (CIA)
To constrain the provenance of the Wular Lake sediments, geochemistry data were plotted in discriminant diagrams (Fig. 6a, b) where most of the data points plot in the mafic and intermediate igneous provenance fields and only a few samples shifted towards the quartzose sedimentary provenance field (Fig. 6a). As discriminant function is based on most mobile major elements, the sedimentary and post-deposition phenomena including weathering, diagenesis, and metamorphism govern variability in the mobile elemental concentration, and hence, does not support to discriminant between samples from the key tectonic provenances (Garzanti et al., 2010; Hou et al., 2018; Nwaila et al., 2017). The Al2O3/TiO2 ratios of the clastic rocks are principally used to deduce the source rock compositions (Rashid et al., 2015; Taylor & McLennan, 1985). In the studied lake sediments, Al2O3/TiO2 ratios ranged from 8 to 27 with an average of 12, suggesting the mafic igneous and intermediate rock source. Hayashi et al. (1997) suggest that Al2O3/TiO2 ratios between 3 and 8 indicate the mafic source, while the values between 8 and 21, and > 21 reflect intermediate and felsic igneous source, respectively. Further, to identify the significance of Al and Ti in provenance studies, and to deduce the origin of siliciclastic rocks, the geochemistry data was plotted as Al2O3 versus TiO2 (Fig. 6b). In this plot, majority of the data points plot along the 3granite + 1basalt trend line, which gives the inference that most of the minerals have been converted to clays by chemical weathering. The CIA values between 50 and 74 also corroborate the involvement of chemical weathering. Sheikh et al. (2014) revealed that the Wular Lake sediments have chief source rock contribution from the carbonates despite the lake catchment is dominated by the Panjal Traps. Our results suggest that Wular Lake sediments exhibit striking similarity with the average Panjal basalts when plotted with multi-elemental spiders (Fig. 4b) and discrimination diagrams (Fig. 6a, b). The results substantially infer that the Panjal Traps, which are basaltic to andesitic in nature, supplied the majority of detritus to the lake.
Impact of catchment attributes on lake
Terrestrial input by fluvial systems, and aquatic fauna, contributes significantly to the organic matter (OM) in the lake ecosystem (Dean, 2006; Li et al., 2018). In Wular Lake sediments, the higher levels of OM (25.2%) (Table 3) are due to the high algal and aquatic plant growth within the lake, along with significant input from terrestrial sources like floating gardens grown by the local population for their livehood within the lake. According to Kristensen et al. (2008) and Zan et al. (2012), aquatic fauna, vegetation growing inside the lakes, and human forcing all control the amount of organic material in lacustrine environments. The carbon and nitrogen ratio (C/N) of the OM is chiefly retrained by the accessibility of OM and fine clayey fractions, offering a significant clue to evaluate the comparative source contributing to terrestrial plants as well as planktonic OM in the sedimentary basins (Perdue & Koprivnjak, 2007; Kumar et al., 2016). The C/N ratio in the Wular Lake sediments ranges from 5 to 18 (average = 18), suggesting that the main source of the OM is managed by autochthonous algal matter and terrestrial OM mixing. In their studies, Meyers and Teranes (2001) and Achyuthan et al. (2020) suggest that C/N ratios between 4 and 10 suggest an algal source, while those above 10 suggest a mixed source from algal and vascular land plants.
The waste-water discharge through the ephemeral streams and a direct influx of sewage from the households near the lake periphery are responsible for a higher C/N ratio and water quality deterioration. This suggests that reduction of the lake catchment area due to urbanization, tourism, water discharge, and horticulture activities (Fig. 7) is the principal source to contribute OM to the lake sediments. In addition, during the summer and monsoon times, the lake is flooded by the higher recharge from inflow streams wherein the excess terrestrial inputs are being flushed out during periods of higher lake levels. Higher OM concentration and lower C/N ratio in the lake sediments noticeably disclose the high phytoplankton activity in the lake. Similarly, the higher values of total nitrogen (TN) in the lake are mainly linked with agro-horticultural practices and habitational sites. The margins of the lake are occupied by agricultural and horticultural fields where the use of nitrogen fertilizers is higher (Fig. 7). The land use and land cover changes within the Walur Lake during the last 40 years (Fig. 7) suggested that there is a drastic and continuous increase in the buildup, wetland, and barren land throughout the observation period. Analysis of the available data sets shows that there is a complex interplay among different identified and delineated land categories within Walur Lake. The conversion of open water area into aquatic vegetation, and then into horticultural activities, which ultimately is transformed into buildup by humans, induced events within the peripheries of the lake. A continuation of such environmental damage to the lake catchment would have an adverse effect on the hydraulically connected groundwater system by deteriorating the water quality and flux magnitude (e.g., Dong et al., 2010). By disseminating the knowledge about the importance of aquatic ecosystems among the local people, a sustainable relationship between humans and their natural habitat can be established, which is possible for the studied lake.
Conclusion and future prospects
Increased LULC changes witnessed in the Wular Lake catchment over the past few decades have influenced the lake dynamics and its water quality has remained a great concern. Towards this, water and sediment samples were collected monthly following grid pattern from the lake basin during 2013–2014. The study found that Ca2+ dominates the cation budget (TZ+) and HCO3− dominates the anion budget (TZ−). The study suggests that the concentration of all the ionic constituents in lake waters exhibited nearly the same seasonal pattern, being higher during the lean flow in winter and lower values during the peak flow in summer/autumn, owing to the dilution effect. The hydrochemical results suggest that chemical weathering of silicates and carbonates within the catchment shapes the lake water chemistry, and characterizes the facies pattern into CA-Mg-HCO3. The study documented compositional variations in sediments in terms of major oxides (SiO2, CaO, Fe2O3, Al2O3, and MgO) and trace elemental content (Zn, Cu, Ni, Co, Pb) with minor fluctuations (< 2%) in K2O, TiO2, and Na2O. Further, it was found that improved abrasion rates and subsequent settling of detritus into the lake were a dominant process, suggesting prominent physical weathering over chemical weathering. The results evince that Walur Lake sediments exhibit striking similarity with the average Panjal basalts, which substantially infer that the Panjal Traps supplied the majority of detritus to the lake. Moreover, the study suggested that waste-water discharge and the direct influx of sewage from the households near the lake margins, land other land use-land cover modification within the lake catchment is the principal source to contribute higher amounts of organic matter, and C/N ratios to the lake. The study recommends that a significant decrease by conversion of peripheral areas into urban settlement and agro-horticulture land by filling of soil should be restricted. This can be done by spreading the knowledge about the importance of lake ecosystems among local people so that a sustainable relationship between humans and their natural environment can be established, which is possible for the studied lake. The study will assist the policymakers in developing long-term future management plans by restricting the anthropogenic forcing within the lake catchment so that freshwater resources from the lake can be effectively protected and conserved.
Data availability statement
Data is available from the corresponding author upon reasonable request.
Change history
14 January 2022
A Correction to this paper has been published: https://doi.org/10.1007/s10661-022-09758-3
References
Achyuthan, H., Lone, A. M., Shah, R. A., & Fousiya, A. A. (2020). Climate, C/N ratio and organic matter accumulation: an overview of examples from Kashmir Himalayan lakes. In A. Dimri, B. Bookhagen, M. Stoffel, & T. Yasunari (Eds.), Himalayan weather and climate and their impact on the environment (pp. 185–203). Switzerland: Springer Nature.
Adrian, R., Hessen, D. O., Blenckner, T., Hillebrand, H., Hilt, S., Jeppesen, E., & Trolle, D. (2016). Environmental impacts lake ecosystems. In S. Ichtiaque Rasool (Ed.), North Sea Region Climate Change Assessment (pp. 315–340). Switzerland: Springer International Publishing.
APHA. (1999). Standard methods for the examination of water and waste water. American Public Health Association, (20th ed., pp1268), Washington DC.
Badar, B., Romshoo, S. A., & Khan, M. A. (2013). Modelling catchment hydrological responses in a Himalayan Lake as a function of changing land use and land cover. Journal of Earth System Science, 122(2), 433–449.
Ballatore, T. J., & Muhandiki, V. S. (2002). The case for a world lake vision. Hydrological Processes, 16(11), 2079–2089.
Beeton, A., Hecky, R., & Stewart, K. M. (2008). Environmental trends and potential future states of large freshwater lakes. In Polunin, N.V.C (Eds.), Aquatic ecosystems trends and global prospects, (pp., 81–93). Cambridge University Press, Cambridge CB2 8RU, UK.
Bhang, K. J., Schwartz, F. W., Lee, H. W., & Park, S. S. (2019). Scaling effect of lake distribution on power law by coastal area simulation. Journal of Coastal Research, 301–305.
Bhateria, R., & Jain, D. (2016). Water quality assessment of lake water: A review. Sustain. Water Resources Management, 2, 161–173.
Brantley, S. L. (2003). Reaction kinetics of primary rock-forming minerals under ambient conditions. In Surface and ground water, weathering, and soils, 5–9, 73–117. Elsevier Inc.
Cohen, A. S. (2003). Paleolimnology: The history and evolution of lake systems (p. 500p). Oxford University Press.
Dinis, P.A., Garzanti, E., Hahn, A., Vermeesch, P., & Cabral-Pinto, M. (2020). Weathering indices as climate proxies. A step forward based on Congo and SW African river muds. Earth Science Reviews, 201, 103039.
Dean, W. E. (2006). Characterization of organic matter in lake sediments from Minnesota and Yellowstone National Park. US Geological Survey (open File Report), 1053, 40p.
Dong, J. D., Zhang, Y. Y., Zhang, S., Wang, Y. S., Yang, Z. H., & Wu, M. L. (2010). Identification of temporal and spatial variations of water quality in Sanya Bay, China by three-way principal component analysis. Environmental Earth Sciences, 60(8), 1673–1682.
Downing, J. A., Prairie, Y. T., Cole, J. J., Duarte, C. M., Tranvik, L. J., Striegl, R. G., et al. (2006). The global abundance and size distribution of lakes, ponds, and impoundments. Limnology and Oceanography, 51(5), 2388–2397.
Dubois, N., Saulnier-Talbot, É., Mills, K., Gell, P., Battarbee, R., Bennion, H., Chawchai, S., Dong, X., Francus, P., Flower, R., & Gomes, D. F. (2018). First human impacts and responses of aquatic systems: A review of palaeolimnological records from around the world. The Anthropocene Review, 5(1), 28–68.
Firth, A. R., & Garden, J. F. (2008). Interactions between magnetite oxidation and flux calcination during iron ore pellet induration. Metallurgical and Materials Transactions Bulletin, 39(4), 524–533.
Gaillardet, J., Dupré, B., & Allègre, C. J. (1999). Geochemistry of large river suspended sediments: Silicate weathering or recycling tracer. Geochimica Et Cosmochimica Acta, 63(23–24), 4037–4051.
Garzanti, E., Andò, S., France-Lanord, C., Vezzoli, G., Censi, P., Galy, V., & Najman, Y. (2010). Mineralogical and chemical variability of fluvial sediments: 1. Bedload sand (Ganga–Brahmaputra, Bangladesh). Earth and Planetary Science Letters, 299(3–4), 368–381.
Garzanti, E., Padoan, M., Setti, M., Najman, Y., Peruta, L., & Villa, I. M. (2013). Weathering geochemistry and Sr-Nd fingerprints of equatorial upper Nile and Congo muds. Geochemistry, Geophysics, Geosystem, 14(2), 292–316.
Hassan, U. Z., Shah, J. A., Kanth, T. A., & Pandit, A. K. (2015). Influence of land use/land cover on the water chemistry of Wular Lake in Kashmir Himalaya (India). Ecological Processes, 4(1), 1–11.
Hayashi, K. I., Fujisawa, H., Holland, H. D., & Ohmoto, H. (1997). Geochemistry of ∼1.9 Ga sedimentary rocks from northeastern Labrador, Canada. Geochimica et cosmochimica acta, 61(19), 4115–4137.
Hou, Q., Mou, C., Wang, Q., & Tan, Z. (2018). Provenance and tectonic setting of the early and middle Devonian Xueshan formation, the north Qilian Belt. China. Geological Journal, 53(4), 1404–1422.
Hoverman, J. T., & Johnson, P. T. J. (2012). Ponds and lakes: A journey through the life aquatic. Nature Education Knowledge, 3(6), 17.
Jeelani, G. H., & Shah, A. Q. (2006). Geochemical characteristics of water and sediment from the Dal Lake, Kashmir Himalaya: Constraints on weathering and anthropogenic activity. Environmental Geology, 50(1), 12–23.
Kjeldahl, J. G. C. T. (1883). A new method for the determination of nitrogen in organic matter. Zeitschrift Für Analytische Chemie, 22(1), 366–382.
Kristensen, E., Bouillon, S., Dittmar, T., & Marchand, C. (2008). Organic carbon dynamics in mangrove ecosystems: a review. Aquatic Botany, 89, 201–219.
Kumar, V., Tiwari, M., Nagoji, S., & Tripathi, S. (2016). Evidence of anomalously low δ 13 C of marine organic matter in an Arctic fjord. Scientific Reports, 6(1), 1–9.
Li, Z., Xu, X., Ji, M., Wang, G., Han, R., Ma, J., & Liu, J. (2018). Estimating sedimentary organic matter sources by multi-combined proxies for spatial heterogeneity in a large and shallow eutrophic lake. Journal of Environmental Management, 92(3), 554–562.
Luque, J. A., & Julià, R. (2002). Lake sediment response to land-use and climate change during the last 1000 years in the oligotrophic Lake Sanabria (northwest of Iberian Peninsula). Sedimentary Geology, 148(1–2), 343–355.
Meybeck, M. (1995). Global distribution of lakes. In A. Lerman, D. M. Imboden, & J. R. Gat (Eds.), Physics and chemistry of lakes (pp. 1–35). Springer.
Meyers, P. A., & Terranes, J. L. (2001). Sediment Organic Matter. Tracking environmental changes using lake sediment – Vol. 2. In Last W.M., Smol J. P. (Eds.), Physical and Geochemical Methods (pp 239-270). Kluwer Academic, Dordrecht, Netherlands.
Müller, D., Tjallingii, R., Płóciennik, M., Luoto, T. P., Kotrys, B., Plessen, B., Ramisch, A., Schwab, M. J., Błaszkiewicz, M., Słowiński, M., & Brauer, A. (2021). New insights into lake responses to rapid climate change: The Younger Dryas in Lake Gościąż, central Poland. Boreas, 50(2), 535–555.
Mushtaq, F., & Pandey, A. C. (2014). Assessment of land use/land cover dynamics vis-à-vis hydrometeorological variability in Wular Lake environs Kashmir Valley, India using multitemporal satellite data. Arabian Journal of Geosciences, 7(11), 4707–4715.
Nesbitt, H. W., & Young, G. M. (1984). Prediction of some weathering trends of plutonic and volcanic rocks based on thermodynamic and kinetic considerations. Geochimica Et Cosmochimica Acta, 48(7), 1523–1534.
Nesbitt, H. W., & Young, G. M. (1996). Petrogenesis of sediments in the absence of chemical weathering: Effects of abrasion and sorting on bulk composition and mineralogy. Sedimentology, 43(2), 341–358.
Nwaila, G., Frimmel, H. E., & Minter, W. E. (2017). Provenance and geochemical variations in shales of the Mesoarchean Witwatersrand Supergroup. The Journal of Geology, 125(4), 399–422.
Pandit, A. K. (1999). Freshwater ecosystems of the Himalaya (p. 207p). Parthenon Publishing New York.
Perdue, E. M., & Koprivnjak, J. F. (2007). Using the C/N ratio to estimate terrigenous inputs of organic matter to aquatic environments. Estuarine, Coastal and Shelf Science, 73(1–2), 65–72.
Rashid, S. A., & Ganai, J. A. (2018). Depositional environments, provenance and paleoclimatic implications of Ordovician siliciclastic rocks of the Thango Formation, Spiti Valley, Tethys Himalaya, northern India. Journal of Asian Earth Sciences, 157, 371–386.
Rashid, S. A., Ganai, J. A., Masoodi, A., & Khan, F. A. (2015). Major and trace element geochemistry of lake sediments, India: Implications for weathering and climate control. Arabian Journal of Geosciences, 8(8), 5677–5684.
Reid, D. F., & Beeton, A. M. (1992). Large lakes of the world: A global science opportunity. GeoJournal, 28(1), 67–72.
Rumysa, K., Sharique, A. A., Tariq, Z., Farooq, M., Bilal, A., & Pinky, K. (2012). Physico chemical status of Wular Lake in Kashmir. Journal of Chemistry, Biology and Physics Science, 31, 631–636.
Ryan, K. M., & Williams, D. M. (2007). Testing the reliability of discrimination diagrams for determining the tectonic depositional environment of ancient sedimentary basins. Chemical Geology, 242, 103–125.
Saini, R. K., Swain, S., Patra, A., Khanday, G. J., Gupta, H., Purushothaman, P., & Chakrapani, G. J. (2008). Water chemistry of three Himalayan Lakes: Dal (Jammu & Kashmir), Khajjiar (Himachal Pradesh) and Nainital (Uttarakhand). Himalayan Geology, 29(1), 63–72.
Saleem, M., Jeelani, G., & Shah, R. A. (2015). Hydrogeochemistry of Dal Lake and the potential for present, future management by using facies, ionic ratios, and statistical analysis. Environmental Earth Sciences, 74(4), 3301–3313.
Sarah, S., Jeelani, G. H., & Ahmed, S. (2011). Assessing variability of water quality in a groundwater-fed perennial lake of Kashmir Himalayas using linear geostatistics. Journal of Earth System Science, 120(3), 399–411.
Seybold, C. A., Grossman, R. B., & Reinsch, T. G. (2005). Predicting cation exchange capacity for soil survey using linear models. Soil Science Society of America Journal, 69(3), 856–863.
Shah, R. A., & Jeelani, G. (2016). Vulnerability of karst aquifer to contamination: A case study of Liddar catchment, Kashmir Himalayas. Journal of Himalayan Ecology and Sustainable Development, 11, 58–72.
Shah, R. A., Achyuthan, H., Lone, A. M., Lone, S. A., & Malik, M. S. (2020). Environmental risk assessment of lake surface sediments using trace elements: A case study, the Wular Lake. Journal of the Geological Society of India, 95(2), 145–151.
Sheikh, J. A., Jeelani, G., Gavali, R. S., & Shah, R. A. (2014). Weathering and anthropogenic influences on the water and sediment chemistry of Wular Lake. Kashmir Himalaya. Environmental Earth Sciences, 71(6), 2837–2846.
Shellnutt, J. G., Bhat, G. M., Wang, K. L., Brookfield, M. E., Jahn, B. M., & Dostal, J. (2014). Petrogenesis of the flood basalts from the Early Permian Panjal Traps, Kashmir, India: Geochemical evidence for shallow melting of the mantle. Lithos, 204, 159–171.
Stephens, G. L., Slingo, J. M., Rignot, E., Reager, J. T., Hakuba, M. Z., Durack, P. J., Worden, J., & Rocca, R. (2020). Earths water reservoirs in a changing climate. Proceedings of the Royal Society America, 476(2236), 20190458.
Stockdale, D. (1958). The molybdate method for the determination of phosphorus, particularly in basic slag and in steel. The Analyst, 83(982), 24–36.
Taylor, S. R., & McLennan, S. M. (1985). The continental crust: Its composition and evolution (p. 312p). Blackwell Scientific.
Walkley, A., & Black, I. A. (1934). An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Science, 37(1), 29–38.
Wetlands International. (2007). Comprehensive management action plan for Wular Lake, Kashmir, wetlands international-South Asia, A-25, (2nd Floor), Defence Colony New Delhi, 110024.
Wilson, M. J. (2004). Weathering of the primary rock-forming minerals: Processes, products and rates. Clay Minerals, 39(3), 233–266.
Zan, F., Huo, S., Xi, B., Zhang, J., Liao, H., Wang, Y., & Yeager, K. M. (2012). A 60-year sedimentary record of natural and anthropogenic impacts on Lake Chenghai, China. Journal of Environmental Sciences, 24(4), 602–609.
Acknowledgements
Authors are thankful to the Director, Wadia Institute of Himalayan Geology (WIHG), Dehra-dun, for the permission to publish this work wide letter no. TCPME/March. -2021/02 dated 12/03/2021. The assigned identification number of the manuscript is WIHG/0129.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There is no conflict of interest to declare. Authors certify that the submission is original work and is not under consideration elsewhere. Also, there is no financial interest to report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shah, R.A., Ganaie, J.A., Yaseen, S. et al. Aquatic geochemistry of a major freshwater lake in the Kashmir Himalaya: solute acquisition and denudation process in the lacustrine system. Environ Monit Assess 193, 835 (2021). https://doi.org/10.1007/s10661-021-09623-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-021-09623-9