Abstract
The aim of this research is to assess the effects of oil palm plantations on stream habitat and their fish assemblage diversity. We hypothesize that streams which drain through oil palm plantations tend to be less heterogeneous, limiting the occurrence of many species, than streams that drain through forest fragments, which support higher fish diversity. A total of 17 streams were sampled; eight in forest fragments and nine in oil palm plantations. Environmental and biological variables were sampled along 150 m stretch in each stream. Of the 242 environmental variables measured, ten were considered important to assess the condition of structural habitat, and out of these variables, four were considered relevant in the distinction between streams in oil palm plantations and forest fragments. A total of 7245 fishes were collected, belonging to 63 species. Unlike our original hypothesis, the species richness did not differ between forest fragment and oil palm plantations streams, showing that it is not a good divert measure in streams disturbance assessment. However, fish assemblages differed in species composition, and 56 species were recorded in oil palm plantation streams, while 44 species were recorded in forest fragments streams. Some species were identified as indicators of either altered (Aequidens tetramerus and Apistogramma agassizii) or undisturbed areas (Helogenes marmoratus). Overall, oil palm plantations were proven to change stream habitat structure and fish species distribution, corroborating other studies that have evidenced changes in patterns of biological community structure due to impacts by different land uses.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With a cultivated area of approximately 16.3 million hectares of oil palm (Elaeis guineensis Jacq) in the world, this monoculture is one of the most rapidly growing agricultural land uses in the tropical region (FAO 2013). The increasing demand for oil palm for production of foodstuff derivatives, cosmetics, and biofuels is one of the main reasons for its expansion (Da Silva 2013). Currently, South East Asia concentrates 60% of oil palm plantations worldwide; however, the high cost of creating new plantation areas and the new restrictions adopted by environmental politics have made it difficult to expand this activity in the region (Kongsager and Reenberg 2012). Therefore, the Brazilian Amazon has been considered an area with high potential for oil palm expansion (Persson and Azar 2010), with 140,000 ha of already cultivated land, and another 190,000 with soil conditions and ideal climate. Thus, approximately 330,000 ha in the Brazilian Amazon are expected to be used for oil palm plantation by 2020 (Ramalho-Filho et al. 2010).
Higher carbon stock rate and economic profitability compared to other cash crop activities are amongst the main arguments in favor of oil palm expansion in the Amazon (Müller et al. 2006). On the other hand, characteristics such as lower structural complexity, smaller canopy cover than in forested areas, climatic instability, use of fertilizers and pesticides, the need for total replacement of plantations, which takes from 25 to 30 years, and the impact of these changes in biodiversity (Fitzherbert et al. 2008; Savilaakso et al. 2014; Luke et al. 2016) indicate that oil palm plantation could be a new environmental threat to the Amazon (Butler and Laurence 2009). However, there are still few studies on the effects of this activity on the Amazon biodiversity (Correa et al. 2015; Lees et al. 2015; Almeida et al. 2016), and knowledge is even more limited regarding aquatic ecosystems (Cunha et al. 2015; Shimano and Juen 2016).
Habitat homogenization deriving from the deployment of monoculture and from native vegetation suppression has been considered an important threat to stream ecological integrity (Allan 2004). Changes in land use throughout catchment result in the loss of structure complexity, which affects stream habitat structure (Poff and Ward 1990; Clapcott et al. 2012), both directly and indirectly, due to margin erosion, silting, or to changes in the type of energy source in the system (from allochthonous to autochthonous) (Karr 1981; Dayang-Norwana et al. 2011; Senior et al. 2013). Therefore, understanding the mechanisms of habitat simplification and indicating which variables generate this condition is an effective way to measure environmental impacts. This approach can effectively measure changes in channel morphology, hydrodynamics, shelter availability for living organisms, adjacent vegetation density, substrate composition, among others, thus yielding a complex and comprehensive analysis of waterbody features (Barbour et al. 1999; Kaufmann et al. 1999).
Changes in stream habitat structure may also result in environmental conditions changes, which are considered important selective forces of the adaptive features of species in a given environment (‘Habitat Templet Theory’ Southwood 1977, 1988). In a reduced time scale, such changes might work as a possible environmental filters that affect species distribution, particularly in habitat specialist species, resulting in changes in biological community structure (Scarsbrook and Townsend 1993). Among aquatic organisms, fishes have proven to be important indicators of stream ecological integrity (Mendonça et al. 2005); they comprise individuals of different trophic levels, occupy different micro-habitats, are resistant to different levels of anthropic changes, and have relatively well known both life history and taxonomy compared to other aquatic organisms (Fausch et al. 1990; Harris 1995; Scott and Hall Jr 1997).
In the Amazon River Basin, small first- and second-order streams according to Strahler’s scale (Strahler 1957) may account for greater than 80% of total channel length in meso-scale, and the rapid expansion of cash crop in this area is one of the main threats to these systems (McClain and Elsenbeer 2001). Despite of this close relationship between the streams ecological integrity and the landscape use along the catchments, features such as the high dimension, structural complexity, and high biological diversity of the Amazon Basin have hindered the understanding on how these anthropogenic changes affect stream biodiversity. Consequently, these features also hinder the development of management, monitoring and conservation strategy of these system.
Therefore, our study aimed to assess how the presence of oil palm cultivation around the streams affect their habitat structure and fish assemblage structure in relation to streams that drain through forested areas present along of this anthropogenic landscape. We hypothesize that streams draining oil palm plantations have lower habitat heterogeneity leading to a fish assemblage composition destabilization with lower richness and increase of more generalist species abundance when compared to streams draining forest fragments.
Material and methods
Study area
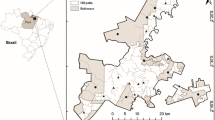
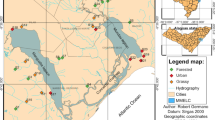
We sampled wadable streams in the Acará and Mojú Rivers basin, located in Northeastern mesoregion of Pará state (2°13′00”S / 2°43′00”S and 48°54′00”W / 48°28′00”W), in a geographic unit denominated Belém Endemism Area (AEB) (Fig. 1). This unit was proposed based in their biodiversity endemism and geographic peculiarities, and has being suggested as a basic unit for development of conservation strategy for Amazon (Almeida and Vieira 2013). The AEB has approximately 243,000 km2, contemplating 27 protected areas, 14 indigenous land areas (Almeida and Vieira 2013) and concentrates the majority of oil palm production in the Amazon (Müller et al. 2006), thus comprising an extensive mosaic of both preserved and altered lands.
The vegetation in the study area is comprised by 28% of ombrophilous forest and 82% converted into several land uses, among which one the most significant is oil palm plantation (Almeida and Vieira 2013). The climate in the region is tropical humid, subtype “Af”, according to Köppen’s classification adapted by Peel et al. (2007), with a rainy season from December to May and a dry season from June to November. Mean annual rainfall in the region is 2344 mm3, reaching a monthly maximum of 427 mm3 in March, and a minimum of 54 mm3 in September (Albuquerque et al. 2012). Mean temperature in the region is 26 °C and mean air humidity reaches up to 85% (Oliveira et al. 2002).
Data sampling
We sampled 17 streams, eight of them within preserved forested area and nine in oil palm plantations areas. Streams were selected according to the following criteria: a) river source inside the treatment (oil palm or forest); b) areas used for planting at similar times, and; c) absence of other impacts, such as urbanization and short-cycle monocultures, not deriving from the activities within the scope of this paper.
In each stream, we selected a 150 m stretch that was subdivided in ten 15 m long transects. In each transect, we performed the characterization of the habitat and sampled fish specimens. All streams were sampled during the dry season to minimize seasonal effects on habitat structure and on fish assemblages. Habitat structure was assessed using the Field Operations Manual for Wadeable Streams proposed by Peck et al. (2006) and adapted by Callisto et al. (2014), with data reduction and metric calculation following Kaufmann et al. (1999). This methodology result in a set of variables, which includes information on the channel morphology, substrate, hydraulic features, flow currents types, channel declivity and sinuosity, canopy cover, riparian vegetation structure, availability of refuges for fish and presence of human impact. Water physical-chemical characteristics, such as pH, conductivity (μS cm−1), turbidity (NTU), temperature (°C), and dissolved oxygen (mg L−1), were measured at three equidistant points in the sampling stretch using a Horiba U50 multiparameter. Detailed description about how environmental variables were measured can be found in Juen et al. (2016). This field operation has been widely used in international (Kaufmann and Hughes 2006; Bryce et al. 2010) and national studies (Macedo et al. 2014a; Cunha et al. 2015; Prudente et al. 2016) involving stream ecosystems.
Fish assemblages were sampled using two hand nets with 55 cm diameter (3 mm mesh) during three hours (18 min in each long transect). This method is considered as an efficient technique for estimating fish assemblage structure in low order streams (Uieda and Castro 1999) and has been frequently used to assess the fish assemblage in small amazon streams (Prudente et al. 2016). Fishes were tagged separately by transect, preserved in 10% formalin for approximately 48 h, transferred to 70% ethanol, and deposited in the ichthyological collection of Museu Paraense Emilio Goeldi (MPEG) in Belém, Pará (Brazil).
Data analyses
Each stream was considered an independent sample, totaling 17 samples in the analyses. The environmental variables measured were submitted to a pre-selection process, and all variables with zero value in more than 80% of the samples and with coefficients of variation lower than 10% were initially excluded. Selected variables were assessed regarding their capacity to discriminate forested areas from oil palm plantations by analyzing the overlap between interquartile ranges in the box-and-whisker plots (Barbour et al. 1996). Variables with no overlap between quartiles or small overlaps between quartiles and no overlaps between medians were considered sensitive. Variable sensitivity was confirmed through a Student t-test for independent samples with a 5% significance level. Lastly, a Spearman correlation test was used to evaluate the redundancy between selected environmental variables. In the case of redundant metrics (r2 > 0.75, p < 0.05), those pointed out in literature as important in structuring stream fish assemblages were maintained (Fernandes et al. 2012; Macedo et al. 2014b; Giam et al. 2015; Prudente et al. 2016).
Environmental variables selected were standardized and submitted a principal component analysis (PCA) based on a Euclidean distance matrix (Legendre and Legendre 1998) to check which variables contributed the most to distinguish the stream habitat between forest fragments and oil palm plantation streams. PCA axes were selected through the broken-stick criterion where a given axis may be retained when their observed eigenvalues exceed the expected eigenvalues generated by the broken stick (Jackson 1993). Variables with loading values higher than 0.60 were maintained for PCA axes interpretation.
Since the fish abundance in oil palm plantation streams was higher than in forested streams, the species richness for each sample was assessed using an individual-based rarefaction method considering the smaller abundance value recorded (n = 230) (Gotelli and Colwell 2001). The richness value was tested between the treatments using a Student t-test for independent samples with a 5% significance level. The hypothesis that fish assemblage composition is different between forest fragments streams and oil palm plantation streams was assessed using the non-metric multidimensional scaling method (NMDS), based on a distance matrix calculated from the Bray-Curtis similarity index. Potential differences in this composition were tested using abundance log values, which were submitted to a permutation multivariate analysis (PERMANOVA - Permutation Multivariate Analyses of Variance). The PERMANOVA probability value was obtained through a Monte Carlo randomization method, based on 9999 randomizations (Anderson 2005; Anderson et al. 2008).
We used a Threshold Indicator Taxa Analysis (TITAN) to detect change points in the fish assemblages in response to an environmental gradient (Baker and King 2010, 2013). In this study, the PCA axes selected by the broken stick method represented the environmental gradient. This analysis identifies individual taxon contribution and optimal values of predictor variable based on binary partition of samples by indicator value scores (IndVal; Dufrêne and Legendre 1997), which integrate occurrence frequency and relative abundance of each taxon. Just as in classification and regression trees, this analysis use a deviance reduction measure to partition sample units at a given predictor variable value (De’ath and Fabricius 2000).
The TITAN analysis considered purity and reliability properties measured in the calculation of indication value (IndVal; Dufrêne and Legendre 1997), together with Change Point Analysis - nCPA (King and Richardson 2003). Results were verified with 500 bootstraps yielding confidence intervals and change points at which the assemblage responded negatively [sum(z-)] and positively [sum(z+)] to predictor variable (Baker and King 2010). Following the recommendations of Baker and King (2010), we excluded taxa that occurred at less than three sites and with fewer than five individuals (36 retained species). Abundances maintained were logarithmically transformed (log(x + 1)) to reduce the influence of highly variable taxa on indicator score calculations in each data set, which was particularly important for taxa with low occurrence frequencies. Species shall be significantly associated to a higher or lower habitat condition if IndVal <0.05, purity >0.95, and reliability >0.95 (Baker and King 2010; Cardoso et al. 2013). The TITAN interpretation was carried out based in environmental variables selected for each PCA axis interpretation, based in their loadings values.
All analyses were performed with the R software (R Development Core Team 2003) using the packages FactoMineR (Lê et al. 2008), TITAN2 (Baker and King 2010) and vegan (Oksanen et al. 2016).
Results
Sampled streams had a mean width of 3.42 m, Standard Deviation ±1.10 (with an average of 3.39 m ± 1.08 in plantation areas, and 3.45 m ± 1.20 in forested areas), and a mean depth of 0.34 m ± 0.14 (0.38 m ± 0.14 in plantation areas and 0.30 m ± 0.14 in forested areas). The water in these streams was predominantly acidic with an average pH of 4.8 ± 0.24 (4.9 ± 0.15 in plantation areas and 4.7 ± 0.25 in forested areas), with mean dissolved oxygen of 6.96 mg/l ± 1.40 (6.71 ± 0.83 in plantation areas and 7.23 ± 1.88 in forested areas), and mean temperature of 25.8 °C ± 0.88 (26.2 °C ± 0.67 in plantation areas and 25.4 °C ± 0.96 in forested areas).
Of the 242 variables measured using the habitat assessment protocol, 44 were removed from the analysis because they had zero values in more than 80% of the samples, and 21 were removed because they had a coefficient of variation lower than 10%. The visual assessment of interquartile overlap, followed by confirmation through a Student t-test, resulted in the exclusion of 127 variables. Finally, the correlation analysis indicated only ten variables for evaluating environmental conditions of structural habitat (Table 1, Additional figure is given in online resource 1). The environmental variables selected were: thalweg mean depth; % fine substrate (silt/clay/mud; size <0.6 mm of diameter); riparian canopy cover by thin trees estimation (> 5 m high and <0.3 m of diameter); large woody debris in active channel (pieces/reach >0.3 m of diameter); large woody debris above active channel (pieces/reach >0.3 m of diameter); large woody debris volume in and above active channel (m3/reach >0.8 m of diameter); fish cover by overhanging vegetation; fish cover by undercut banks; proportion of riparian non-agricultural human disturbance; Oxidation Reduction Potential of water (Additional figure is given in online resource 1).
The principal component analysis showed that the variables selected had a significant contribution to the distinction of stream habitat between forested and oil palm plantations areas. Only the first principal component was selected for the interpretation of results, explaining 37.04% of the variation in the habitat structure of streams. Plantation areas were characterized by a high proportion of non-agricultural human impact (HNOAG) and higher percentage of fine substrate (PCT_FN), while forested areas had a higher large woody debris volume in and above active channel (LWDVC), as well as high values of water oxidation and reduction (ORP) (Table 1, Fig. 2).
Ordinations resulting from the principal components analysis (PCA) representing the habitat variables (vectors) measured in stream reaches (symbols) sampled located in oil palm plantations and forested areas in northeastern Pará state, Brazil. Open circle (○) represents streams in oil palm plantation area and closed circles (●) represent streams in forested area
A total of 7245 fish specimens were sampled; 4333 (56 species) in oil palm plantation streams and 2912 (42 species) in forest fragments streams (Table 2). However, the richness based on individual-based rarefaction method did not differ between these treatments (t = 2.62, df = 1, p > 0.05). The total richness was 63 species, allocated in six orders and 24 families. A higher abundance was observed in the order Characiformes (21 species; 60.10% of the specimens sampled), followed by Siluriformes (18; 16.08%) and Perciformes (7; 14.45%). The most abundant species were Microcharacidium weitzmani (37.2%), Apistogramma gr. regani (9.41%), Trichomycterus hasemani (8.30%), and Hyphessobrycon heterorhabdus (8.26%) (Table 2). The ordination yielded by NMDS evidenced a distinction between species composition of oil palm plantations and forest fragments, and it was corroborated by the PERMANOVA result ( pseudo F = 2.82; p < 0.01) (Fig. 3). Of the total species captured, 21 occurred only in oil palm plantation streams, with highlight to the exclusiveness of families Poeciliidae (N = 4, occurring in a single sample), Polycentridae (N = 1, one sample), Auchenipteridae (N = 2, two samples), and Doradidae (N = 49, three samples). In forested areas, four exclusive species were observed, Megalechis thoracata, Batrochoglanis raninus, Ammocriptocharax elegans, and Denticetopsis epa, with emphasis on the exclusiveness of families Callichthyidae and Pseudopimelodidae (Table 2).
Ordination resulting from the non-metric multi-dimensional scaling analysis (NMDS) based on species composition of fishes collected in oil palm plantation and forest fragments streams in northeastern Pará state, Brazil. Open circle (○) represents streams in oil palm plantation area and closed circles (●) represent streams in forested area
TITAN (Threshold Indicator Taxa Analysis) indicated a small difference in species ecological limits associated to variation in habitat condition (z- = 1.52 and sum z + = −1.22). In addition, of the 36 analyzed species, Aequidens tetramerus, Apistogramma agassizii and Microcharacidium weitzmani were positively associated to first PCA axis, that based on axis interpretation, means an occurrence increase of these species followed by increase in percentage of fine substrate and proportion of non-agricultural human impact (Fig. 4). On the other hand, only Helogenes marmoratus was negatively associated to first PCA axis, that mean an occurrence increase followed by increase in large woody debris volume in and above active channel and Oxireduction Potential of water (Table 3, Fig. 4).
Change points and 90% confidence limits of significant indicator taxa along the first PCA axis (Threshold Indicator Taxa Analysis, p < 0.05, purity >0.95, reliability >0.95 for p < 0.05, 500 bootstrap and 1000 permutation replicates). Change points are sized in proportion to the magnitude of the response (z scores)
Discussion
In the present study, oil palm plantations have proven to be an important modifying agent of stream ecosystem structures, since the presence of this activity resulted in changes both in habitat structure, and in water physical-chemical characteristics, which reflected in stream fish assemblages. In the Eastern Amazon, streams draining oil palm plantations showed an increase in percentage of fine substrate and in the proportion of non-agricultural human impact, along with a decrease in large woody debris volume in and above active channel (LWDVC) and in water oxidation and reduction. We also recorded changes in the fish assemblage composition, where generalist species were predominant and general richness and abundance of species were higher when compared with streams of forested areas.
The relationship between stream habitat structure and cash crops has been widely described in literature (Moerke and Lamberti 2006; Hrodey et al. 2009; Santos et al. 2015; Luke et al. 2016). Changes in land use along the catchment, such as agriculture, affect negatively the structure of remaining riparian vegetation (Heartsill-Scalley and Aide 2003). Consequently, these changes affect the stream habitat due to a higher incidence of light, reduction of allochthonous organic matter input (Fernandes et al. 2012), changes in chemical elements of water (De Souza et al. 2013), and reducing the offer of shelter for organisms (Crook and Robertson 1999; Wrigth and Flecker 2004). The network of dirt roads used for cash crops has also been considered an important modifying agent of stream habitat, mainly contributing with the siltation of this system, which affects the water physical-chemical feature and biodiversity (McClain and Elsenbeer 2001; Wantzen and Mol 2013). In the present study, we believe that both changes in riparian vegetation structure and the presence of dirt roads, contributed to stream habitat changes in oil palm plantation areas. Besides, the Brazilian environmental laws (Law n° 12.651, May 25, 2012), consider dirt roads and bridges as a low impact disturbance and their construction is permitted in permanent preservation areas (APP – Área de Preservação Permanente in Portuguese), which includes a 30 m strip of riparian vegetation area in both side of streams less than ten meters wide. This situation tend to contribute even more to the input of sediments and organic matter in the streams, as well as with changes in the physical and chemical characteristics of water.
The changes observed in the habitat corroborate the results presented by Dosskey et al. (2010), who considers the riparian vegetation as responsible for maintaining the physical-chemical properties of streams draining. Changes in stream habitat was also observed by Cunha et al. (2015) in a study conducted in Amazonia, who found that the highest proportion of non-agricultural human impact was caused by changes in habitat integrity of streams due to land use for oil palm plantation. With lower amount of woody debris, and substrates with smaller diameter in oil palm plantation streams, which are thus characterized as sites with lower heterogeneity and lower offer of habitats. These metrics were sensitive to compare and distinguish forest fragments from oil palm plantations, and are directly related to the preservation state and configuration of riparian vegetation, which are key factors to the good functioning of the natural conditions of aquatic ecosystems (Pusey and Arthington 2003). On the other hand, a higher canopy cover was observed in the oil palm plantation, however, this riparian vegetation is composed by thin trees (<30 cm), suggesting a regenerating or recent vegetation. In addition, the significant number of thin trees is driven by the absence of larger trees, which are considered to be strong contenders for resources since they present a greater demand (Coomes and Allen 2007).
Following the same reasoning, the increase in sedimentation found in oil palm plantation streams might be associated to changes in riparian vegetation, which works as a filter for water volume and for the amount of transported sediment coming from the adjacent forest fragments and into the streams (Pusey and Arthington 2003; De Souza et al. 2013). In streams surrounded by less dense vegetation resulting from oil palm plantations, the volume of water input in the streams is higher, as is sediment load, corroborating Restrepo et al. (2015), who found changes in the sedimentation process of water bodies in Rio Magdalena basin, Colombia, resulting from the suppression of primary vegetation over the years. In addition, according to Bonachea et al. (2010), in a process similar to the sedimentation of water bodies, habitat loss deriving from homogenization caused by changes in the native vegetation is also another relevant consequence of disorganized human activities as it decreases the offer of shelter, foraging sites, and reproductive sites in several aquatic organisms (Casatti et al. 2012).
On the other hand, biodiversity also suffers the impacts of palm monoculture. However, most studies address terrestrial organisms, e.g., the study by Lees et al. (2015) in the Eastern Amazon, who observed differences in avifauna richness between oil palm plantation, primary forest, secondary forest, and pasture. Oil palm plantation samples had the lowest richness in the study. In another study carried out in the same area, Correa et al. (2015) recorded higher anuran richness in forest fragments than in oil palm plantations; however, they found no difference in the abundance of these animals. Also, regarding terrestrial fauna, in a study conducted in Malaysia, Turner and Foster (2009) evidenced that terrestrial invertebrate abundance and biomass decrease in oil palm plantations compared to primary forest fragments and logged forest fragments. Regarding aquatic organisms, some studies have indicated relationships similar to those observed for terrestrial organisms. Rawi et al. (2013) observed a lower diversity of aquatic insects in streams next to oil palm plantations compared to primary forest fragments streams and streams near highways. In another study carried out in the Amazon, Cunha et al. (2015) observed a lower richness of Heteroptera in oil palm plantation streams compared to primary forest fragments streams. However, there were no differences in the abundance of these insects.
Although the state of knowledge regarding changes caused by oil palm plantations in the natural community structure is significant, studies about the effects on ichthyofauna are still scarce. In a pioneer study, conducted in Indonesia, Giam et al. (2015) observed differences in fish assemblage structure; higher species richness was found in streams that drain through continuous forests and riparian vegetation fragments, compared to oil palm plantation streams, where a loss of approximately 42% in fish richness is estimated.
Unlike the hypothesized in the present study, the fish assemblage’s richness did not differ between forest fragments and oil palm plantations streams. Similar pattern has also been observed and discussed in tropical streams submitted to a different type of environmental disturbances (Teresa and Casatti 2012), which was manly attributed to a higher evenness in the distribution of species within these streams and the fact that disturbed areas provide condition that favor the frequency increase of species that previously inhabited these areas. In some cases, the species richness in streams may also increase after disturbances (McCabe and Gotelli 2000). According to Huston’s dynamic-equilibrium model, considered a good predictor model to streams communities, the species richness peak can be obtained in different disturbance intensity according to the rates of competitive exclusion and population (Huston 1979). In this sense, it is important to reinforce that species richness must be carefully evaluated and never used as the only measure of diversity streams disturbances assessment.
Regarding fish species composition, the results obtained indicate differences between streams in forest fragments and oil palm plantations. This composition difference may be related to environmental changes, since environmental features are determining factors for the occurrence of species in a given area (Jackson et al. 2001; Terra et al. 2015). Despite that, the fish species composition of one sampled stream in forest fragments was similar to streams in oil palm plantations in the NMDS ordination, this pattern may be leading because this site is surrounded by oil palm plantations, both downstream and upstream, but the stream source is located in forest fragments. However, this influence (distance of plantations) was not measured in our study and we suggest for a future landscape ecology studies to assess this relation and elucidate this question.
Therefore, fish species might be used as indicators of preserved or degraded areas (Ferreira and Cassati 2006). In the present study, the increase in abundance of Aequidens tetramerus and Apistogramma agassizii in oil palm plantations streams may be attributed to high tolerance and generality of many species of this family (Cichlidae) to lower environmental conditions (Burress 2015). In addition, the wide distribution of these two species in the Amazon basin (Kullander 2003) make them possible environmental indicators of areas disturbed by oil palm plantation. On the other hand, Helogenes marmoratus is associated to higher environmental conditions of structural heterogeneity (forest fragment streams), since their perpetuation depends on more specific environments, e.g., foliage bed and woody debris, deriving from riparian forests, which are deposited in the stream substrate, serving as shelter for these specie (Sazima et al. 2006).
Overall, changes in the patterns of biological community structure are common in face of the impacts caused by different land uses (Iwata et al. 2003; Deegan et al. 2011). In addition, the maintenance of riparian vegetation is essential to mitigate these impacts, since small streams depend on the structure of this marginal vegetation to maintain their natural features (Pusey and Arthington 2003; Casatti et al. 2012; De Souza et al. 2013). Therefore, monitoring studies on structural analyses (e.g.: amount of woody debris) of aquatic ecosystems have been carried out in the United States, Europe and Asia with the purpose of understanding their functioning and of creating conservation measures (Metzger and Casatti 2006; Li et al. 2010). In Brazil, however, these methodologies are still scarce.
Studies that add ecosystem structural information to biological information comprise an effective biodiversity monitoring tool used in temperate regions (Jaramillo-Villa and Caramaschi 2008); all the same, they can be widely adapted and practiced in tropical and subtropical regions such as the Brazilian Amazon. Hence, the results shown here indicate that the use of data on fish assemblage composition combined with structural habitat condition approach is effective in the assessment of streams and might subsidize strategies for the preservation and conservation of these ecosystems, as land use for oil palm plantations has been impacting the Amazon biodiversity. The conservation and monitoring of riparian vegetation corridors along all catchments in the studied area are of utmost importance. In addition, with a decrease in habitat quality, changes in species composition might occur, leading to the loss of more sensitive and forest-specialist species.
References
Albuquerque MF, Souza EB, Oliveira MCF, Souza-Júnior JA (2012) Precipitação nas mesorregiões do Estado do Pará: climatologia, variabilidade e tendências nas últimas décadas (1978-2008). Rev Bras Clim 6(6):151–168
Allan JD (2004) Landscapes and riverscapes: the influence of land use on stream ecosystems. Annu Rev Ecol Syst 35(1):257–284. https://doi.org/10.1146/annurev.ecolsys.35.120202.110122
Almeida AS, Vieira ICG (2013) Sumário Executivo: Cenários para a Amazônia – Área de Endemismo Belém. Museu Paraense Emílio Goeldi, Belém
Almeida SM, Silva LC, Cardoso MR, Cerqueira PV, Juen L, Santos MP (2016) The effects of oil palm plantations on the functional diversity of Amazonian birds. J Trop Ecol 5:1–16
Anderson MJ (2005) PERMANOVA: a FORTRAN computer program for permutational multivariate analysis of variance. University of Auckland, New Zealand, Department of Statistics
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA+ for PRIMER: guide to software and statistical methods. PRIMER-E, Plymouth
Baker ME, King RS (2010) A new method for detecting and interpreting biodiversity and ecological community thresholds. Methods Ecol Evol 1(1):25–37. https://doi.org/10.1111/j.2041-210X.2009.00007.x
Baker ME, King RS (2013) Of TITAN and straw men: an appeal for greater understanding of community data. Freshw Sci 32(2):489–506. https://doi.org/10.1899/12-142.1
Barbour MT, Gerritsen J, Griffith GE, Frydenborg R, McCarron E, White JS, Bastian ML (1996) A framework for biological criteria for Florida streams using benthic macroinvertebrates. J N Am Benthol Soc 15(2):185–211. https://doi.org/10.2307/1467948
Barbour MT, Gerritsen J, Snyder BD, Striblings JB (1999) Rapid bioassessment protocols for use in streams and Wadeable rivers: periphyton, benthic macroinvertebrates and fish, 2nd edn. EPA 841-B-99-002. U.S. Environmental Protection Agency; Office of Water; Washington, D.C
Bonachea J, Viola M, Bruschi MA, Hurtado L, Forte LM, da Silva M et al (2010) Natural and human forcing in recent geomorphic change; case studies in the Rio de la Plata basin. Sci Total Environ 408(13):2674–2695. https://doi.org/10.1016/j.scitotenv.2010.03.004
Bryce SA, Lomnicky GA, Kaufmann PR (2010) Protecting sediment-sensitive aquatic species in mountain streams through the application of biologically based streambed sediment criteria. J N Am Benthol Soc 29(2):657–672. https://doi.org/10.1899/09-061.1
Burress ED (2015) Cichlid fishes as models of ecological diversification: patterns, mechanisms, and consequences. Hydrobiologia 748(1):7–27. https://doi.org/10.1007/s10750-014-1960-z
Butler RA, Laurence WF (2009) Is the oil palm the next threat to the Amazon? Trop Conserv Sci 2(1):1–10
Callisto M, Alves CBM, Lopes JM, Castro MA (2014) Condições ecológicas em bacias hidrográficas de empreendimentos hidrelétricos. CEMIG, Belo Horizonte
Cardoso P, Rigal F, Fattorini S, Terzopoulou S, Borges PA (2013) Integrating landscape disturbance and indicator species in conservation studies. PLoS One 8(5):1–10. https://doi.org/10.1371/journal.pone.0063294
Casatti L, Teresa FB, Gonçalves-Souza T, Bessa E, Manzotti AR, Gonçalves CDS, Zeni JDO (2012) From forests to cattail: how does the riparian zone influence stream fish? Neotrop Ichthyol 10(1):205–214. https://doi.org/10.1590/S1679-62252012000100020
Clapcott JE, Collier KJ, Death RG, Goodwin EO, Harding JS, Kelly D, Leathwick JR, Yong RG (2012) Quantifying relationships between land-use gradients and structural and functional indicators of stream ecological integrity. Freshw Biol 57(1):74–90. https://doi.org/10.1111/j.1365-2427.2011.02696.x74
Coomes DA, Allen RB (2007) Effects of size, competition and altitude on tree growth. J Ecol 95(5):1084–1097. https://doi.org/10.1111/j.1365-2745.2007.01280.x
Correa FS, Juen L, Rodrigues LC, Silva-Filho HF, Santos-Costa MC (2015) Effects of oil palm plantations on anuran diversity in the eastern Amazon. Anim Biol 65(3–4):321–335. https://doi.org/10.1163/15707563-00002481
Crook DA, Robertson AI (1999) Relationship between riverine fish and woody debris: implications for lowland rivers. Mar Freshw Res 50(8):941–953. https://doi.org/10.1071/MF99072
Cunha EJ, Montag LFA, Juen L (2015) Oil palm crops effects on environmental integrity of Amazonian streams and Heteropteran (Hemiptera) species diversity. Ecol Indic 52:422–429. https://doi.org/10.1016/j.ecolind.2014.12.024
Da Silva JA (2013) Avaliação do programa nacional de produção e uso do biodiesel no Brasil – PNPB. Rev Pol Agric 22(3):18–31
Dayang-Norwana AAB, Kunjappan R, Chin M, Schoneveld G, Potter L, Andriani R (2011) The local impacts of oil palm expansion in Malaysia: an assessment based on a case study in Sabah state. Working paper 78. CIFOR, Bogor, Indonesia
De Souza ALT, Fonseca DG, Libório RA, Tanaka MO (2013) Influence of riparian vegetation and forest structure on the water quality of rural low-order streams in SE Brazil. For Ecol Manag 298:12–18. https://doi.org/10.1016/j.foreco.2013.02.022
De’Ath G, Fabricius KE (2000) Classification and regression trees: a powerful yet simple technique for ecological data analysis. Ecology 81(11):3178–3192. https://doi.org/10.1890/0012-9658(2000)081[3178:CARTAP]2.0.CO;2
Deegan LA, Neill C, Haupert CL, Ballester MVR, Krusche AV, Victoria RL, Thomas SM, de Moor E (2011) Amazon deforestation alters small stream structure, nitrogen biogeochemistry and connectivity to larger rivers. Biogeochemistry 105(1):53–74. https://doi.org/10.1007/s10533-010-9540-4
Dosskey MG, Vidon P, Gurwick NP, Allan CJ, Duval TP, Lowrance R (2010) The role of riparian vegetation in protecting and improving chemical water quality in streams. J Am Water Resour Assoc 46(2):261–277. https://doi.org/10.1111/j.1752-1688.2010.00419.x
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67(3):345–366. https://doi.org/10.1890/0012-9615(1997)067[0345:SAAIST]2.0.CO;2
FAO (2013) FAOSTAT database food and agriculture Organization of the United Nations, Rome, Italy. http://www.faostat.fao.org. Accessed 16 Jan 2016
Fausch KD, Lyons J, Karr JR, Angermeier PL (1990) Fish communities as indicators of environmental degradation. Am Fish Soc Symp 8:123–144
Fernandes IM, Lourenço LS, Ota RP, Moreira MMM, Zawadzki CH (2012) Effects of local and regional factors on the fish assemblage structure in meridional Amazonian streams. Environ Biol Fish 96(7):837–848. https://doi.org/10.1007/s10641-012-0079-1
Ferreira CP, Cassati L (2006) Stream biotic integrity assessed by fish assemblages in the upper Rio Paraná basin. Biota Neotrop 6(3):0–0. https://doi.org/10.1590/S1676-06032006000300002
Fitzherbert EB, Struebig MJ, Morel A, Danielsen F, Brühl CA, Donald PF, Phalan B (2008) How will oil palm expansion affect biodiversity. Trends Ecol Evol 23(10):538–545. https://doi.org/10.1016/j.tree.2008.06.012
Giam X, Hadiaty RK, Tan HH, Parenti LR, Wowor D, Sauri S, Chong KY, Yeo DCJ, Wilcove DS (2015) Mitigating the impact of oil-palm monoculture on freshwater fishes in Southeast Asia. Conserv Biol 29(5):1357–1367. https://doi.org/10.1111/cobi.12483
Gotelli NJ, Colwell RK (2001) Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of species richness. Ecol Lett 4(4):379–391. https://doi.org/10.1046/j.1461-0248.2001.00230.x
Harris JH (1995) The use of fish in ecological assessments. Aust J Ecol 20(1):65–80. https://doi.org/10.1111/j.1442-9993.1995.tb00523.x
Heartsill-Scalley T, Aide TM (2003) Riparian vegetation and stream condition in a tropical agriculture-secondary forest mosaic. Ecol Appl 13(1):225–234. https://doi.org/10.1890/1051-0761(2003)013[0225:RVASCI]2.0.CO;2
Hrodey PJ, Sutton TM, Frimpong EA, Simon TP (2009) Land-use impacts on watershed health and integrity in Indiana warmwater streams. Am Midl Nat 161(1):76–95. https://doi.org/10.1674/0003-0031-161.1.76
Huston M (1979) A general hypothesis of species diversity. Am Nat 113(1):81–101. https://doi.org/10.1086/283366
Iwata T, Nakano S, Inoue M (2003) Impacts of past riparian deforestation on stream communities in a tropical rain forest in Borneo. Ecol Appl 13(2):461–473. https://doi.org/10.1890/1051-0761(2003)013[0461:IOPRDO]2.0.CO;2
Jackson DA (1993) Stopping rules in principal components analysis: a comparison of heuristical and statistical approaches. Ecology 74(8):2204–2214. https://doi.org/10.2307/1939574
Jackson DA, Peres-Neto PR, Olden JD (2001) What controls who is where in freshwater fish communities - the roles of biotic, abiotic, and spatial factors. Can J Fish Aquat Sci 58(1):157–170. https://doi.org/10.1139/f00-239
Jaramillo-Villa U, Caramaschi EP (2008) Índices de integridade biótica usando peixes de água doce: Uso nas regiões tropical e subtropical. Oecol Bras 12(3):442–462
Juen L, Cunha EJ, Carvalho FG, Ferreira MC, Begot TO, Andrade AL, Shimano Y, Leão H, Pompeu OS, Montag LFA (2016) Effects of oil palm plantations on the habitat structure and biota of streams in eastern Amazon. River Res Appl 32(10):2081–2094. https://doi.org/10.1002/rra.3050
Karr JR (1981) Assessment of biotic integrity using fish communities. Fisheries 6(6):21–27. https://doi.org/10.1577/1548-8446(1981)006<0021:AOBIUF>2.0.CO;2
Kaufmann PR, Hughes RM (2006) Geomorphic and anthropogenic influences on fish and amphibians in Pacific northwest coastal streams. In: Hughes RM, Wang L, Seelback PW (eds) Landscape influence on stream habitat and biological assemblages, vol 48. American Fisheries Society Symposium, Bethesda, pp 429–455
Kaufmann PR, Levine P, Robison GE, Seeliger C, Peck DV (1999) Quantifying physical habitat in wadeable streams. U. S. Environmental Protection Agency, EPA/620/R-99/003, Washington, D.C
King RS, Richardson CJ (2003) Integrating bioassessment and ecological risk assessment: an approach to developing numerical water-quality criteria. Environ Manag 31(6):795–809. https://doi.org/10.1007/s00267-002-0036-4
Kongsager R, Reenberg A (2012) Contemporary land-use transitions: the global oil palm expansion. GLP report no. 4. GLP-IPO, Copenhagen
Kullander SO (2003) Check list of the freshwater fishes of south and central America. EDIPUCRS, Porto Alegre
Lê S, Josse J, Husson F (2008) FactoMineR: an R package for multivariate analysis. J Stat Softw 25(1):1–18. https://doi.org/10.18637/jss.v025.i01
Lees AC, Moura NG, Almeida AS, Vieira ICG (2015) Poor prospects for avian biodiversity in Amazonian oil palm. PLoS One 10(5):1–17. https://doi.org/10.1371/journal.pone.0122432
Legendre P, Legendre L (1998) Numerical Ecology, 2nd edn. Elsevier, Amsterdam
Li T, Li W, Qian Z (2010) Variations on ecosystem services value in response to land use changes in Shenzhen. Ecol Econ 69(7):1427–1435. https://doi.org/10.1016/j.ecolecon.2008.05.018
Luke SH, Barclay H, Bidin K, Vun Khen, C, Ewers RM, Foster WA et al (2016) The effects of catchment and riparian forest quality on stream environmental conditions across a tropical rainforest and oil palm landscape in Malaysian Borneo. Ecohydrology 10(4):1–14. https://doi.org/10.1002/eco.1827
Macedo DR, Hughes RM, Ligeiro R, Ferreira WR, Castro MA, Junqueira NT, Oliveira DR, Firmiano KR, Kaufmann PR, Pompeu PS, Callisto M (2014a) The relative influence of catchment and site variables on fish and macroinvertebrate richness in cerrado biome streams. Landsc Ecol 29(6):1001–1016. https://doi.org/10.1007/s10980-014-0036-9
Macedo DR, Pompeu PS, Morais L, Castro MA, Alves CBM, França JS et al (2014b) Sampling site selection, land use and cover, field reconnaissance, and sampling. In: Callisto M, Hughes RM, Lopes JM, Castro MA (eds) Ecological conditions in hydropower basins. Companhia Energética de Minas Gerais, Belo Horizonte, pp 61–83
McCabe DJ, Gotelli NJ (2000) Effects of disturbance frequency, intensity, and area on assemblages of stream macroinvertebrates. Oecol 124(2):270–279. https://doi.org/10.1007/s004420000
McClain ME, Elsenbeer H (2001) Terrestrial inputs to Amazon streams and internal biogeochemical processing. In: McClain ME, Victoria RL, Richey JE (eds) The biogeochemistry of the Amazon Basin. Oxford University Press, Oxford, pp 185–208
Mendonça FP, Magnusson WE, Zuanon J (2005) Relationships between habitat characteristics and fish assemblages in small streams of Central Amazonia. Copeia 2005(4):751–764. https://doi.org/10.1643/0045-8511(2005)005[0751:RBHCAF]2.0.CO;2
Metzger JP, Casatti L (2006) From diagnosis to conservation: the state of the art of biodiversity conservation in the BIOTA/FAPESP program. Biota Neotrop 6(2):0–0. https://doi.org/10.1590/S1676-06032006000200002
Moerke AH, Lamberti GA (2006) Relationships between land use and stream ecosystems: a multi stream assessment in southwestern Michigan. In: Hughes RM, Wang L, Seelbach PW (eds) Landscape influences on stream habitats and biological assemblages, vol 48. American Fisheries Society Symposium, Maryland, pp 323–338
Müller AA, Furlan-Junior J, Celestino-Filho P (2006) Embrapa Amazônia Oriental e o agronegócio do dendê no Pará. Embrapa Amazônia Oriental, Belém
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara RB et al (2016) Vegan: community ecology package. R package version 2.3–3. http://CRAN.R-project.org/package=vegan. Accessed 10 Jan 2016
Oliveira LL, Fontinhas RL, Lima AMM, Lima RJS (2002) Mapas dos parâmetros climatológicos do Estado do Pará: umidade, temperatura e insolação, médias anuais. Anais do XIII Congresso Brasileiro de Meteorologia. Sociedade Brasileira de Meteorologia, Fortaleza http://www.cbmet.com/cbm-files/22-762cad766c70d3a4452c4afd29decb7b.doc. Accessed 14 Dec 2015
Peck DV, Herlihy AT, Hill BH, Hughes RM, Kaufmann PR, Klemm DJ et al (2006) Environmental monitoring and assessment program-surface water western pilot study: field operations manual for wadeable streams. U.S. Environmental Protection Agency, Office of Research and Development, Washington, D.C
Peel MC, Finlayson BL, McMahon TA (2007) Updated world map of the Köppen-Geiger climate classification. Hydrol Earth Syst Sci 11(5):1633–1644. https://doi.org/10.5194/hess-11-1633-2007
Persson UM, Azar C (2010) Preserving the world's tropical rorests - a price on carbon may not do. Environ Sci Technol 44(1):210–215. https://doi.org/10.1021/es902629x
Poff NL, Ward JV (1990) Physical habitat template of lotic systems: recovery in the context of historical pattern of spatiotemporal heterogeneity. Environ Manag 14(5):629–645. https://doi.org/10.1007/BF02394714
Prudente BS, Pompeu PS, Juen L, Montag LF (2016) Effects of reduced-impact logging on physical habitat and fish assemblages in streams of eastern Amazonia. Freshw Biol 62(2):303–316
Pusey BJ, Arthington AH (2003) Importance of the riparian zone to the conservation and management of freshwater fish: a review. Mar Freshw Res 54(1):1–16. https://doi.org/10.1071/MF02041
R Development Core Team (2003) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ramalho-Filho A, Motta PEF, Freitas PL, Teixeira WG (2010) Zoneamento agroecológico, produção e manejo para a cultura da palma de óleo na Amazônia. Embrapa Solos, Rio de Janeiro
Rawi CSM, Al-Shami SA, Madrus MR, Ahmad AH (2013) Local effects of forest fragmentation on diversity of aquatic insects in tropical forest streams: implications for biological conservation. Aquat Ecol 47(1):75–85. https://doi.org/10.1007/s10452-012-9426-8
Restrepo JD, Kettner AJ, Syvitski JPM (2015) Recent deforestation causes rapid increase in river sediment load in the Colombian Andes. Anthropocene 10:13–28. https://doi.org/10.1016/j.ancene.2015.09.001
Santos FB, Ferreira FC, Esteves KE (2015) Assessing the importance of the riparian zone for stream fish communities in a sugarcane dominated landscape (Piracicaba River basin, southeast Brazil). Environ Biol Fish 98(8):1895–1912. https://doi.org/10.1007/s10641-015-0406-4
Savilaakso S, Garcia C, Garcia-Ulloa J, Ghazoul J, Groom M, Guariguata MR, Laumonier Y, Nasi R, Petrokofsky G, Snaddon J, Zrust M (2014) Systematic review of effects on biodiversity from oil palm production. Eviron Evid 3(1):4. https://doi.org/10.1186/2047-2382-3-4
Sazima I, Carvalho LN, Mendonça FP, Zuanon J (2006) Fallen leaves on the water-bed: diurnal camouflage of three night active fish species in an Amazonian streamlet. Neotrop Ichthyol 4(1):119–122. https://doi.org/10.1590/S1679-62252006000100013
Scarsbrook MR, Townsend CR (1993) Stream community structure in relation to spatial and temporal variation: a habitat templet study of two contrasting new Zeland streams. Freshw Biol 29(3):395–410. https://doi.org/10.1111/j.1365-2427.1993.tb00774.x
Scott MC, Hall LW Jr (1997) Fish assemblages as indicators of environmental degradation in Maryland coastal plain streams. Trans Am Fish Soc 126(3):349–360. https://doi.org/10.1577/1548-8659(1997)126<0349:FAAIOE>2.3.CO;2
Senior MJ, Hamer KC, Bottrell S, Edwards DP, Fayle TM, Lucey JM et al (2013) Trait-dependent declines of species following conversion of rain forest to oil palm plantations. Biodivers Conserv 22(1):253–268. https://doi.org/10.1007/s10531-012-0419-7
Shimano Y, Juen L (2016) How oil palm cultivation is affecting mayfly assemblages in Amazon streams. Ann Limnol Int J Limnol 52:35–45. https://doi.org/10.1051/limn/2016004
Southwood TRE (1977) Habitat, the templet for ecological strategies? J Anim Ecol 46:337–365
Southwood TRE (1988) Tatics, strategies and templets. Oikos 52(1):3–18. https://doi.org/10.2307/3565974
Strahler AN (1957) Quantitative analysis of watershed geomorphology. Trans Am Geophys Union 38(6):913–920. https://doi.org/10.1029/TR038i006p00913
Teresa FB, Casatti L (2012) Influence of forest cover and mesohabitat types on functional and taxonomic diversity of fish communities in Neotropical lowland streams. Ecol Freshw Fish 21(3):433–442. https://doi.org/10.1111/j.1600-0633.2012.00562.x
Terra BF, Hughes RM, Araújo FG (2015) Fish assemblages in Atlantic Forest streams: the relative influence of local and catchment environments on taxonomic and functional species. Ecol Freshw Fish Early view 25(4):527–544. https://doi.org/10.1111/eff.12231
Turner EC, Foster WA (2009) The impact of forest conversion to oil palm on arthropod abundance and biomass in Sabah, Malaysia. J Trop Ecol 25(1):23–30. https://doi.org/10.1017/S0266467408005658
Uieda VS, Castro RMC (1999) Coleta e fixação de peixes de riachos. In: Caramaschi EP, Mazzoni R, Peres-Neto PR (eds) Ecologia de Peixes de Riachos, Série Oecologia Brasiliensis, vol VI. PPGE-UFRJ, Rio de Janeiro, pp 1–22
Wantzen KM, Mol JH (2013) Soil erosion from agriculture and mining: a threat to tropical stream ecosystems. Agriculture 3(4):660–683. https://doi.org/10.3390/agriculture3040660
Wrigth JP, Flecker AS (2004) Deforesting the riverscape: the effects of wood on fish diversity in a Venezuelan piedmont stream. Biol Conserv 120(3):443–451. https://doi.org/10.1016/j.biocon.2004.02.022
Acknowledgements
The authors are grateful to financial support of Coordination for the Improvement of Higher Education Personnel (CAPES) and National Counsel of Technological and Scientific Development (CNPq); and logistic support for field research of the Conservation International (CI-Brazil) and Agropalma Group.
Funding
Ferreira MC has received Granting Scholarship by Coordination for the Improvement of Higher Education Personnel (CAPES). Prudente BS has received Research Grant by National Councel of Technological and Scientific Development (CNPq) through the Programa de Capacitação Internacional (PCI). Juen L. and Montag LFA has received Research Grants by Coordination for the Improvement of Higher Education Personnel (CAPES) / Pró-Reitoria de Pesquisa e Pós-Graduação (PROPESP/UFPA) and Productivity Grants by National Counsel of Technological and Scientific Development (CNPq).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(GIF 2148 kb)
Rights and permissions
About this article
Cite this article
Ferreira, M.C., Begot, T.O., da Silveira Prudente, B. et al. Effects of oil palm plantations on habitat structure and fish assemblages in Amazon streams. Environ Biol Fish 101, 547–562 (2018). https://doi.org/10.1007/s10641-018-0716-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-018-0716-4