Abstract
Landscape connectivity has traditionally been studied for animal species rather than for plants, especially under a multispecies approach. However, connectivity can be equally critical for both fauna and flora and, thus, an essential point in the selection of key management areas and measures. This paper explores a spatially explicit framework to assess the contribution of habitat patches in the conservation and enhancement of plant functional connectivity and habitat availability in a multispecies context. It relies on graph theory and a habitat availability index and differentiates between two management scenarios: (i) conservation; and (ii) restoration, by considering current and potential species distribution based on species distribution models together with a vegetation survey. The results mapped at high spatial resolution priority target areas to apply management measures. We found that intervening in a small proportion of the study area may lead to double the average overall landscape connectivity of the studied species. This study aimed at proposing an innovative methodology that allows studying connectivity for multiple plant species at landscape scale while integrating their individual characteristics. The proposed framework is a step toward incorporating connectivity concerns into plant biodiversity management, based on a better understanding of landscape structure and functionality. Here, we illustrated its significant potential for local conservation and restoration planning and resource optimization.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The extent of certain plant species has greatly decreased due to historical and recent climate and land-use changes (Lovejoy and Wilson 2019), causing the fragmentation (Honnay et al. 2002) and extintion (Humphreys et al. 2019) of plant populations. To mitigate these effects and ensure the long-term persistence of species populations, we must preserve remaining key ecosystems and restore degraded landscapes and their biodiversity (Brudvig 2011; Beale et al. 2013; Mori et al. 2017). In this regard, biodiversity can be promoted through landscape connectivity (Correa Ayram et al. 2015), which allows colonization of new potential habitats and increases species ability to respond to climate and land-use changes (Rubio et al. 2012; Saura et al. 2014, 2018). Therefore, the protection and promotion of landscape connectivity have become an important aspect of long-term biodiversity plans (Lindborg and Eriksson 2004; Opdam and Wascher 2004).
Landscape connectivity has two components: structural and functional (Tischendorf and Fahrig 2000; Crooks and Sanjayan 2006). Structural connectivity is independent of particular responses of species to the landscape and only focuses on the landscape structure. On the other hand, functional connectivity accounts for the landscape structure and the behavioral response of organisms to it. One of the main attributes considered in functional connectivity assessments is species’ dispersal ability, which can be especially difficult to determine for plant species. Tracking seeds trajectories is challenging due to the large number of factors intervening in their movement and constraints in the identification of the mother plant, especially for long-distance dispersal (Wang et al. 2002; Murphy and Lovett-Doust 2004; Carlo et al. 2013; Fajardo et al. 2019). Even if dispersal capacities have been studied for several plant species, there is a knowledge gap regarding species-specific movement capacities for whole plant communities (Vittoz and Engler 2007; Damschen et al. 2008). When planning to restore or conserve plant diversity, we should consider multiple plant species that together imply functional diversity, rather than focusing on single genera or species (Roberge and Angelstam 2004). Thus, studying simultaneously connectivity for a group of species while considering their individual dispersal abilities is vital (Damschen et al. 2006, 2008; Beier et al. 2008).
Given these limitations, we present a framework to conduct plant connectivity analyses. We propose a novel methodology to guide decision making to preserve and improve plant biodiversity by evaluating landscape connectivity. In this study, we adopted a multispecies approach that integrates the analysis of several plant species to identify key biodiversity areas considering the characteristics of each species (dispersal abilities and habitat preferences).
Connectivity can be enhanced through (i) increasing the number or quality of functional links or corridors between habitat patches; (ii) improving the surrounding landscape matrix to ease species dispersal; or (iii) increasing the amount, size, or suitability of habitat patches available for the target species (Brudvig 2011; de la Fuente et al. 2018; Dondina et al. 2018). This study focuses on the third alternative and includes: (a) assessing the role played by current habitat patches providing connectivity, and (b) quantifying connectivity upgrade through the inclusion of new habitat patches. These potentially additional patches may increase habitat availability for the species and serve as starting points for colonization movements (Saura et al. 2014). We used species distribution models (SDM, Guisan et al. 2017) to assess habitat suitability for the focus species, which together with a vegetation survey allowed to discriminate species current and potential distributions at the landscape scale and precise spatial resolution (25 m). Potential functional connectivity (Calabrese and Fagan 2004; Crooks and Sanjayan 2006) among these areas was analyzed by considering the potential seed dispersal of each species and using a graph theory approach (Urban and Keitt 2001; Urban et al. 2009) together with a habitat availability metric (Pascual-Hortal and Saura 2006; Saura 2007), the Probability of Connectivity index (PC, Saura and Rubio 2010). This metric simultaneously allows the quantification of the overall connectivity of landscapes, the individual contribution of each patch to it, and the spatially explicit prioritization of areas with the greatest current and potential contribution to connectivity and habitat availability for the focal plant species.
This study aimed to develop a spatially explicit framework to optimize decision making through the evaluation of landscape connectivity to, ultimately, preserve and improve plant diversity. We studied connectivity of multiple species at landscape scale while integrating their individual characteristics. This methodology would allow a more efficient identification of areas to promote the diversification and expansion of woody plant species, together with improving their resilience to climate or other landscape changes. It is expected to become especially useful to make better use of resources in scenarios with limited funds allocated to management actions.
Materials and methods
Study area
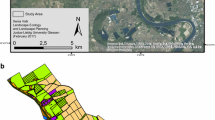
The study was carried out in the Biosphere Reserve of Sierra del Rincón (41° 03′ N 3° 29′ W, Madrid’s North Range, central Spain) (Fig. 1). We particularly focused on this area because we counted on (i) a great amount of data availability; (ii) extensive previous work and knowledge on present woody species; and (iii) the need for connectivity enhancement actions, as traditional land uses (such as livestock, coppicing for fuel, or reforestations) have generated large homogeneous areas (Mateo et al. 2018) with decelerated dynamics due to impediments to seed arrival.
The study area occupies 186 km2 within a mountainous area (850–2083 m above sea level) crisscrossed by four main low-flow streams. This physiographic variability, coupled with the corresponding climatic variation (annual precipitation ranging from 600 to 1050 mm, and mean monthly temperature from 6.3 to 12.5 °C), allow a particularly rich flora, integrated into a mosaic of forests, shrublands, pastures, and croplands. The most representative forest communities are Pyrenean oak (Quercus pyrenaica) coppice with standards forests; holm oak (Quercus rotundifolia) stands and afforested pine groves of Pinus sylvestris, Pinus uncinata, and Pinus pinaster. The focus species of this study are listed in Table 1. The study area is also very homogeneous in terms of lithology (medium to fine grained metamorphic materials: schists, quartzites and slates), soil pH (always acidic soils), and texture (balanced textures ranging from sandy loam to sandy clay loam).
The current configuration of the reserve landscape is closely related to past and present human activities. Livestock, agriculture, and forest activities (coppicing for fuel, logging, and afforestation) have been the main triggers for the current spatial distribution of vegetation, with large homogeneous areas covered with just a few species (such as monospecific Pinus sp. plantations). Several of these uniform spaces show decelerated dynamics of vegetation due to impediments to seed arrival. Additionally, some of these areas exhibit a partial loss of functionality after the abandonment of traditional uses.
Vegetation survey
Two sampling strategies were carried out in the study area: systematic and opportunistic (Mateo et al. 2018). The systematic sampling (132 circular plots) followed a regular grid over the study area with vertices separated by 1000 m. The opportunistic sampling (302 circular plots) was carried out along roads and forest tracks with a separation of 1 km, and plots located 20 m downhill to reduce any potential edge effect. A minimum distance of 300 m between plots was imposed to prevent spatial autocorrelation (Dormann et al. 2007). The presence/absence of all woody species (trees and shrubs) were gathered in circular plots with a ten-meter radius. Plots on pastures, inaccessible or without natural vegetation (rocks, crops, villages, etc.) were discarded.
Species distribution modeling
Species distribution models were generated for each species following an ensemble procedure (Araújo and New 2006) that integrates three statistical techniques: generalized linear models (McCullagh and Nelder 1989), boosted regression trees (Friedman 2001), and random forests (Breiman 2001). The models were generated using the biomod2 R package (Thuiller et al. 2008) with the default parameters. All three techniques were trained only with presence/absence data from the opportunistic sampling (302), and only those species with a minimum of 10 presences were considered (27 species, Table 1). Models were fitted using bioclimatic and environmental variables with a Pearson correlation value lower than 0.8 (Dormann et al. 2007) as predictors. Two climatic variables were selected: May precipitation and August maximum temperature, available at a resolution of 30 arc-seconds (~ 1 km2 at the equator) in WorldClim 1.3 (Hijmans et al. 2005). Climatic data were downscaled to 25 m spatial resolution employing the methodology proposed by Mateo et al (2019b). Five additional predictors were selected as environmental variables and processed also at 25 m resolution from a digital elevation model: (1) linear aspect (orientation transformed into a linear variable), (2) least surface distance to rivers, (3) heat load index (McCune and Keon 2002) to account for the varying temperatures of southwest and southeast facing slopes, (4) slope (an indicator of the local topography and an indirect measure of soil characteristics and microclimatic conditions), and (5) solar radiation in August (for details, see Mateo et al. 2019a, b). The opportunistic sampling was randomly divided ten times into two parts: one containing 70% of the data to train the models, and another one with the remaining 30% to evaluate them with the Area Under the Curve (AUC) statistic (Fan et al. 2006). Thus, ten models were created for each statistical technique (generalized linear models, boosted regression trees, and random forests) and species, but only those with an AUC greater than 0.8 were used. These models were combined to generate the ensemble models for each species: mean of the different runs weighted by their AUC value. Lastly, each species’ SDM was evaluated with the independent systematic sampling dataset (132 plots, testing AUC).
The obtained SDMs provided a suitability index for every pixel of the study area for each species, at 25 m resolution. Therefore, suitability maps were produced for each of the 27 species, and they, together with the inventory sampling, were used as a basis for the connectivity analyses.
Definition of habitat patches
Habitat patches are defined as relatively homogeneous areas that provide high habitat suitability for a species and are surrounded by a landscape matrix of lower suitability (Correa Ayram et al. 2015). They were individually delineated for the 27 focal species from the suitability maps derived from the SDMs. Although plants respond to a gradient of resource quality (Murphy and Lovett-Doust 2004), a binary perception of the landscape was necessarily assumed for this study, selecting a threshold of the suitability index. We used a threshold derived from the Receiver Operational Curve plot (ROC) (Freeman and Moisen 2008) for each SDM. This threshold separates pixels with suitability enough to be occupied by the species from places less suitable that theoretically do not have suitability enough or the required characteristics to be inhabited by that specific species.
The resulting suitable pixels from the binary maps were broken down into areas of current and potential distribution based on the vegetation surveys. Current patches were composed of adjacent high-suitability pixels closer to an observed presence than to an observed absence from both systematic and opportunistic samplings. On the other hand, potential patches represented areas that theoretically meet the suitability characteristics and yet are not currently inhabited by the particular species. Pixels within a potential patch are closer to an observed absence than to an observed presence according to the vegetation survey. These potential sites represent areas that might be colonized by more species or may be suitable for the introduction of plant species during ecological restoration. Both current and potential patches had a minimum area of 1 ha. Hereinafter, the total area covered by current or potential distributions will be referred to as plausible area.
Connectivity analyses
Habitat network definition
The landscape was represented for the 27 focal species through graph theory as a set of nodes, representing habitat patches, and the potential connections between them as links (Urban and Keitt 2001; Urban et al. 2009). Nodes were characterized by an associated attribute, corresponding to the mean suitability value of each habitat patch (extracted from SDM), weighted (i.e., multiplied) by their area. This attribute allows measuring the intrinsic value of patches as habitat for each species, aside from their spatial location (Saura 2007). Potential connections between habitat patches were featured with Euclidean distances and calculated with Conefor Inputs (Saura and Torné 2009) extension for ArcGIS. Thus, the links or connections of each pair of nodes were identified as the shortest paths between them and characterized by their length (Saura et al. 2011, 2018).
Global connectivity, as well as the contribution of each individual patch, were quantified through Probability of Connectivity index (PC, for further details, see S3 in SM, Saura and Pascual-Hortal 2007; Saura and Rubio 2010). PC is a widely recognized indicator of landscape connectivity (Rubio et al. 2012; Engelhard et al. 2017; Rincón et al. 2017; Dondina et al. 2018), based on the habitat availability concept (Pascual-Hortal and Saura 2006). PC measures connectivity as a function of the dispersal capacity of each species (see S1 and Table S2 in SM), the distribution of landscape elements, and a considered attribute of the nodes (mean habitat suitability weighted by the area in this case). We used the command-line version of Conefor software (Saura and Torné 2009) to calculate the overall PC index for each species and the individual contribution (i.e., importance) of every single patch to it.
When evaluating connectivity for each species, we studied two possible scenarios: (i) current scenario, to understand the current state of our study area and to locate the priority areas for conservation measures; and (ii) restoration scenario, where we analyzed the effects of management actions that lead to species introduction in their potential patches, such as direct planting or seeding the focal species, and managing surrounding features to foster species settlement and survival. In this latest scenario, we identified areas where active restoration measures might imply a greater increase in habitat availability and connectivity.
Ranking the current importance of habitat patches
We calculated the current degree of overall connectivity (overall PC) and the individual contribution of current patches in each network. The individual patch importance was calculated by the variation in PC (\(\mathrm{dPC}\)) when removing each patch (S3 in SM, and Saura and Rubio 2010). High-importance patches (i.e., high dPC) are considered critical for maintaining the overall landscape connectivity. Afterward, the dPC value of each patch was transferred to its pixels, i.e., pixels within current patches were characterized by the importance of the container patch per area unit (dPC/patch area). Pixels outside current patches did not have a dPC value, indicating null importance for the conservation of the species.
Once pixels were characterized separately for the 27 focal species, we determined the areas with higher relevance for the largest number of species. For this purpose, we normalized (min–max scaling) and summed the individual importance value for all the species in each pixel. In this case study, we treated all species equally, however, the importance of each species could be weighted in other conceptually related studies. As a result, we obtained an integrated estimate of the relevance for the conservation of all the analyzed species, the so-called general conservation importance value. A higher value of this indicator could be due to a large individual contribution to connectivity for one or more species, or a slighter individual contribution for several species. We selected 10% of the pixels with the highest values of general conservation importance and aggregated them into priority patches for conservation, so that contiguous valuable pixels generated a unique patch.
Evaluating the restoration scenario
Simultaneously, we simulated the addition of the new potential patches to the current set of patches and assessed the contribution to the global PC of each added patch for each species. Elements whose addition promoted a higher increase in connectivity (i.e., higher absolute dPC value, see S3 in SM) were considered the priority areas for restoration.
As in the previous section, a raster map of general relevance for landscape management was created by integrating the individual importance values of all species into a general restoration importance value. In this way, pixels with a higher general restoration importance value identified areas with a higher potential to accommodate a greater diversity, because of its substantial connectivity importance or the large number of species that could live in the area but are currently not present.
Subsequently, the pixels with the highest potential contribution (pixels with the 10% highest values of general restoration importance) were aggregated into priority patches for ecological restoration efforts. The 10% threshold was selected as a feasible value to separate priority restoration patches from other potential patches that would contribute less to overall connectivity. Nonetheless, a different threshold value could be selected depending on the restoration budget or intensity intended.
Finally, we assumed the implementation of active restoration measures (e.g., plantation of focal species) to recover all the studied species that could potentially inhabit these priority areas. These measures might turn the priority areas from potential to current patches, changing, therefore, the habitat network. We recalculated the landscape PC value for every species to quantify the improvement in connectivity after the restoration efforts.
Results
A total of 77 woody species were detected in the vegetation survey, but only 27 had more than 10 presences. The SDMs (Fig. S4, SM) showed a mean independent testing AUC of 0.784, with a standard deviation of 0.086 (Table S5, SM). The binarization of the SDMs separated the suitable habitat areas from the landscape matrix, originating a mean of 126 suitable patches per species (standard deviation of 77.8). The suitable area greatly depends on the species, ranging from 2.5% (Genista florida) to 51% (Erica arborea) of the study area.
The subsequent differentiation between current and potential patches (Fig. S6, SM) indicated that most of the suitable area (mean 56% and standard deviation 14%) is still unoccupied. Species had on average 32 current and 94 potential patches (standard deviation 25.8 and 52 respectively). This difference in the number of patches is balanced by their size, as for most species, patches are larger in the current than in the potential scenario. All results related to the number and size of patches for each species in the current and potential scenarios are listed in Table S7 in SM.
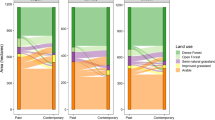
The priority areas for conservation measures (Fig. 2) (i.e., the areas with the highest general conservation importance value) occupied a total of 1659 ha (9% of the total study area). Priority conservation areas encompassed a mean of 18% of each species current distribution (ranging from 71 to 1% among the different focal species).
General conservation importance on the left (General importance for habitat availability and connectivity conservation in the current scenario) and general restoration importance on the right (potential improvement). The general importance was calculated as the sum of every species importance (normalized dPC). Priority target areas for management measures are pointed out with green lines in the conservation scenario and maroon lines in the potential one
Similarly, 73 patches were selected as the target for active restoration measures due to their greater increase in connectivity for a larger number of species (Fig. 2). They occupied a total area of 1582 ha (8% of the total study area). Restoring these priority restoration areas may cause a mean increase of 12% (9.79 standard deviation) of the distribution area currently covered by each species (Table 1). In this way, the mean total plausible area occupied by the species rises (proportion of suitable classified as current rather than potential habitat) from 44 to 57% after the proposed restoration.
This restoration would imply a mean increase in PC of 138% (Table 2). Hence, the restoration of only 8% of the landscape by introducing the lacking species may entail over twice the mean level of connectivity than in the current situation for all the focal species. Additionally, there was a decrease in the individual importance (dPC value) of the current existing patches after the incorporation of the selected priority patches, meaning a lower dependence on individual patches for the functionality of the entire habitat network.
Priority restoration patches were concentrated in the center and south of the study area, while conservation patches were mainly located in the northern half (Fig. 2). There was 14 ha of intersection between relevant conservation and restoration areas. Even when these places displayed a considerable current contribution, the introduction of the lacking species would boost their value. The results suggested a greater improvement after restoration for species with a lower ratio of current to potential area. That is to say, the increase in species distribution area and PC seems to be larger for species with a low proportion of its total plausible area currently occupied (Table S8 in SM).
Discussion
We presented an approach to include connectivity analysis in landscape plant biodiversity management, based on a better understanding of landscape structure and functionality. The results show the potential of the adopted approach to spatially explicitly guide management decisions that aim to increase connectivity in a multispecies context. Applying this framework to landscapes with reduced plant diversity, could highly increase species distribution and connectivity, thereby improving ecosystem resilience and promoting populations persistence (Knoke et al. 2008; Mori et al. 2017). Moreover, the exposed methodology allows selecting priority target areas, focal species, and quantifying the benefits (in terms of habitat availability, Pascual-hortal and Saura 2006) of applying diverse measures (conservation and restoration) in different areas at landscape scale. Thus, this framework helps maximize ecological benefits from management measures under biodiversity targets, which is especially important in scarce resource scenarios. On the other hand, the accuracy of this method depends on the data resolution. The climatic data and the digital elevation model used in the SDM are usually available at similar resolutions worldwide, however, high-resolution vegetation surveys are rarely available, as they usually are very expensive to obtain, especially in large or inaccessible areas. However, the potential benefits of applying this framework in conservation and restoration planning may justify the cost of conducting high-resolution surveys in other low-diversity landscapes.
Multispecies approach
Considering a set of species rather than a single taxon is a key aspect when managing biodiversity and promoting ecosystem functionality (Maes and Van Dyck 2005; Damschen et al. 2008). Additionally, detailed characteristics of each considered species (e.g., preference in habitat selection) should be accounted for. In fact, connectivity assessments require dealing with species-specific dispersal capacities, which become particularly challenging to address for plants. These difficulties are accentuated in large study areas with numerous species. Consequently, little literature has been reported to assess multispecies functional connectivity (Brodie et al. 2014; Petsas et al. 2020), especially for plants (Phillips et al. 2008; Aquilué et al. 2021). This work takes a step forward to study simultaneously connectivity for multiple plant species at landscape scale while integrating their individual characteristics. The large working scale, together with a precise resolution and detailed information for multiple species, result in ecologically based suitable and feasible management measures.
Accounting for forest potentiality
It is crucial that the areas where conservation measures are implemented actually meet the adequate bioclimatic and environmental characteristics to result in ecologically feasible actions. Therefore, the selection of target conservation or restoration areas should be based on a comprehensive ecological understanding of the landscape (Rodríguez et al. 2007; Cianfrani et al. 2010). This study relies on SDMs predictions that consider the bioclimatic and environmental characteristics of the landscape and identify areas that could actually host the focus plant species due to their physiological characteristics. The binarization of SDM and the vegetation survey allowed separating (i) realized distribution areas, currently occupied by the species, and (ii) potential distribution areas. In fact, the latter may be of the greatest importance, as areas with a high potential could provide a much larger diversity after restoration measures. Nevertheless, plants SDM generally only consider the current distribution of species or do not differentiate between realized and potential distributions (Vetaas 2002; Booth 2017; Pecchi et al. 2019). The proposed methodology is especially useful for determining the potential diversity of the study landscape and to propose achievable and ecologically meaningful measures.
The landscape uniformity (homogeneous flora composition in space) of the study area and the high-resolution vegetation survey led us to classify habitat patches into current or potential nodes under the assumption of proximity to a survey plot with presence or absence of the species. We assumed that if habitat cells (i.e., with high suitability for a species) were closer to a presence, the plant species was present in that cell, whereas, if they were closer to an absence, the habitat cell was a potential one. This assumption could mean that species present in certain cells were not detected in the survey, leading to an underestimation of the current distribution area. This error could appear when either the closest survey plot did not have suitable conditions to be inhabited by the species (unlike the focal point or habitat cell) or when the species was present in the area at low densities. On the other hand, it is possible to find habitat cells without a species presence closer to a survey plot with presence of the species. As the maximum distance among adjacent survey plots is 1 km and consequently, the longest distance from any point in the study area to the closer plot is 707 m, we assumed that the distance to a current presence was small enough to be also accepted as a current presence. Even when the focal species was not currently present in the habitat cell, it could colonize this area without human assistance due to the high suitability of the environmental conditions and the short distance to an actual presence. In more heterogeneous landscapes with a greater variability in flora composition, a higher sampling intensity would be advisable to avoid making unreasonable assumptions.
The identification of nodes (Table S7, SM) showed that the species only occupied on average 44% of their suitable habitat, leaving the remaining 56% with lower diversity than the maximum achievable according to the microclimatic and ecological characteristics of the landscape. This suggests that species in the study area could potentially experience a considerable expansion. The low presence of focal species might be due to site-level factors not considered in SDMs (biotic and abiotic variables such as depredation, competition, human actions, and soil characteristics), historical contingencies (e.g., species arrival order, or land use), and landscape-scale factors (e.g., connectivity with other presence sites, allowing seed arrival) (Brudvig 2011; Guisan et al. 2019). Restoration efforts may adjust these factors to increase species biodiversity and ecosystem services to society (Mori et al. 2017).
Moreover, the classification of patches into current and potential allowed a proper assessment of two management strategies according to the type of measures considered: (i) conservation scenario; and (ii) restoration scenario. Generally, these two strategies are not differentiated (Holl and Aide 2011; Chapa-Vargas and Monzalvo-Santos 2012; Beale et al. 2013; Alagador et al. 2014; Engelhard et al. 2017; Aquilué et al. 2021), focusing on either of them or considering that target areas are suitable for both conservation and restoration measures. In contrast, our results show that priority target areas for both scenarios only intersect in 14 ha (less than 10% of the intervention areas arranged for conservation or restoration measures).
Prioritization in the different scenarios
The PC index did not only allow the identification of priority patches to apply management measures. It additionally allowed quantifying and ranking the current and potential contribution of every pixel to plant diversity in terms of habitat availability and connectivity. This ranking can be combined with other landscape characteristics or indices in the search of the best restoration or conservation areas. Another benefit of the proposed methodology is its flexibility to adapt to different areas, necessities, or resources. Importantly, it considers different management strategies, and additionally, it allows setting a different threshold value to separate priority patches from less important ones, depending on the budget or intensity of actions expected. dPC together with nodes classification between current and potential patches allowed the prioritization of the target areas and the reliable quantification of the benefits resulting from the two management scenarios.
Conservation scenario
The quantification of current pixel contribution (general conservation importance) helped to find the priority areas to preserve biodiversity. These are key areas with a current higher contribution to habitat connectivity and availability for more species, and hence, should be the first and foremost in the conservation ranking. The degradation, loss, or isolation of these priority areas could drastically restrict the capacity of species networks to maintain their functionality, decreasing gene flow, and possibly leading to local species loss (Gibbs 2001; Neel 2008).
Restoration scenario
The General restoration importance generated a list of priority areas to be restored, as well as the potential species that could be added to them to increase plant diversity. The inclusion of these species in the priority target areas implied a greater increase in habitat availability and connectivity for all the species than in any other place. Although these priority restoration areas only covered 8% of the study surface, their restoration would entail a substantial gain in the PC, more than twice as great as in the current scenario (Table 2). Furthermore, the mean area currently covered by each species would increase from 44 to 57%. Increasing the distribution area of the species would boost taxonomic biodiversity in the restored patches and enable the genetic flow with previous populations, along with the colonization of new potential areas (assisted colonization, Sutherland et al. 2019). Moreover, increasing species richness inside connected target patches is shown to benefit biodiversity in surrounding non-target habitats (Brudvig et al. 2009). The landscape might therefore benefit from a combination of active and passive restoration measures.
Species patches had a mean size of 77.9 ha before restoration efforts, and 42.4 ha after (Tables S7 and 1). The ecological restoration of the priority areas might cause a shift in the ecological net from a set of large areas to an intricated net of smaller patches, but with a higher total distribution area and an improved capacity for interpatch species flux (larger PC value after restoration, Table 2). Furthermore, the individual importance (dPC value) of the current patches decreased after the hypothetical addition of the selected priority restoration patches. This means that the loss of a patch would have less important consequences (in terms of connectivity) than in a non-intervention scenario. Therefore, species would benefit after restoration efforts from a broader and sparser habitat network with reinforced habitat functionality and forest stability against possible alterations. Generally, species with a low ratio between current and potential area showed a greater improvement in their area and connectivity after restoration efforts (Table S8, SM). Thus, this method might favor species with greater potential for improvement.
Further improvements and future research
This framework is an important step toward the introduction of connectivity concerns into plant conservation strategies. However, landscapes and connectivity are very complex entities, and this framework could be considerably improved to further account for this complexity. Ideally, the influence of the landscape on the dispersal of species should be considered. It is known that movement among habitat patches does not only depend on the spatial distribution and dispersal ability of the studied organisms, but it also depends on the resistance to movement posed by the landscape matrix (Tischendorf and Fahrig 2000; Rico et al. 2012; Zeller et al. 2012; Auffret et al. 2017). However, landscape resistances to species dispersal were not considered in this study. We only accounted for the type of dispersal vector and some morphological characteristics of species (S1 and Table S2 in SM) to determine their movement capacity, even though it is also dependent on the abundance and behavior of the dispersal vectors (Damschen et al. 2008). Dispersal vectors and plants movements may be affected by physical or ecological characteristics, such as wind, slope, tree cover density, and animal seed dispersers. For this reason, it would be interesting to add information about the resistance offered by the landscape to seed movement, yet measures of the effect of a heterogeneous matrix on dispersal are not easy to estimate for organisms such as plants, which rely on a variety of other organisms and agents to disperse (Murphy and Lovett-Doust 2004). Consequently, little work has been reported to determine these resistances (especially for whole plant communities) or how they influence patch prioritization. However, further research on this topic would be highly useful to understand the error associated with ignoring landscape resistance in plant connectivity studies.
Landscape transformations affect species differently: they commonly disfavor specialist species (Clavel et al. 2011) and thus, conservation studies usually focus on them when managing biodiversity. Depending on the goals pursued, it might be interesting to focus on endangered species, with low representation in the study area, or with a specific characteristic (e.g. high trophic value for fauna). Some adjustments could be made to adapt the approach we presented to specific cases. One possible refinement could be to weigh the importance of the study species by their interest for the specific objectives or study landscape. Thus, when calculating the general importance for conservation or restoration, the contribution to connectivity and habitat availability for some species would have a larger influence than that of others.
Moreover, it could also be worthwhile to take into account the costs of restoration. Even though the restoration of one patch would report great improvements, its management may be excessively expensive. Instead, it could be advisable to restore several patches that together imply the same connectivity improvements but are less expensive. On the other hand, several patches could imply greater connectivity improvements than the single priority patch under the same budget. Thus, it is important to optimize not only connectivity and biodiversity levels, but also the implied costs.
Finally, it could be interesting to incorporate future projections of SDMs to determine the potential future distribution of the species under a climate change perspective (Beltrán et al. 2014). This addition would allow identifying critical areas to connect current and future distributions, thus favoring species persistence over time, and therefore long-term plant biodiversity.
Data and codes availability
The data and codes supporting the findings of this study are available with the identifier(s) at the private link https://figshare.com/s/0a0673531da840809af3.
References
Alagador D, Cerdeira JO, Araújo MB (2014) Shifting protected areas: scheduling spatial priorities under climate change. J Appl Ecol 51:703–713. https://doi.org/10.1111/1365-2664.12230
Aquilué N, Messier C, Martins KT et al (2021) A simple-to-use management approach to boost adaptive capacity of forest to global uncertainty. For Ecol Manag 481:1–11
Araújo MB, New M (2006) Ensemble forecasting of species distributions. Trends Ecol Evol 22:42–47. https://doi.org/10.1016/j.tree.2006.09.010
Auffret AG, Rico Y, Bullock JM et al (2017) Plant functional connectivity—integrating landscape structure and effective dispersal. J Ecol 105:1648–1656. https://doi.org/10.1111/1365-2745.12742
Beale CM, Baker NE, Brewer MJ, Lennon JJ (2013) Protected area networks and savannah bird biodiversity in the face of climate change and land degradation. Ecol Lett 16:1061–1068. https://doi.org/10.1111/ele.12139
Beier P, Majka DR, Spencer WD (2008) Forks in the road: choices in procedures for designing wildland linkages. Conserv Biol 22:836–851. https://doi.org/10.1111/j.1523-1739.2008.00942.x
Beltrán BJ, Franklin J, Syphard AD et al (2014) Effects of climate change and urban development on the distribution and conservation of vegetation in a Mediterranean type ecosystem. Int J Geogr Inf Sci 28:1561–1589. https://doi.org/10.1080/13658816.2013.846472
Booth TH (2017) Assessing species climatic requirements beyond the realized niche: some lessons mainly from tree species distribution modelling. Clim Change 145:259–271. https://doi.org/10.1007/s10584-017-2107-9
Breiman L (2001) Random Forests. Mach Learn 45:5–32. https://doi.org/10.1007/9781441993267_5
Brodie JF, Giordano AJ, Dickson B et al (2014) Evaluating multispecies landscape connectivity in a threatened tropical mammal community. Conserv Biol. https://doi.org/10.1111/cobi.12337
Brudvig LA (2011) The restoration of biodiversity: where has research been and where does it need to go? Am J Bot 98:549–558. https://doi.org/10.3732/ajb.1000285
Brudvig LA, Damschen EI, Tewksbury JJ et al (2009) Landscape connectivity promotes plant biodiversity spillover into non-target habitats. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.0809658106
Calabrese JM, Fagan WF (2004) A comparison-shopper’s guide to connectivity metrics. Front Ecol Environ 2:529–536. https://doi.org/10.1890/1540-9295(2004)002[0529:ACGTCM]2.0.CO;2
Carlo TA, García D, Martíez D et al (2013) Where do seeds go when they go far? Distance and directionality of Reports R eports. Ecology 94:301–307
Chapa-Vargas L, Monzalvo-Santos K (2012) Natural protected areas of San Luis Potosí, Mexico: ecological representativeness, risks, and conservation implications across scales. Int J Geogr Inf Sci 26:1625–1641. https://doi.org/10.1080/13658816.2011.643801
Cianfrani C, Le Lay G, Hirzel AH, Loy A (2010) Do habitat suitability models reliably predict the recovery areas of threatened species? J Appl Ecol 47:421–430. https://doi.org/10.1111/j.1365-2664.2010.01781.x
Clavel J, Julliard R, Devictor V (2011) Worldwide decline of specialist species: toward a global functional homogenization? Front Ecol Environ 9:222–228. https://doi.org/10.1890/080216
Correa Ayram CA, Mendoza ME, Etter A, Pérez Salicrup DR (2015) Habitat connectivity in biodiversity conservation: a review of recent studies and applications. Prog Phys Geogr. https://doi.org/10.1177/0309133315598713
Crooks KR, Sanjayan M (2006) Connectivity conservation. In: Crooks KR, Sanjayan M (eds) Connectivity conservation. Cambridge University Press, Cambridge, pp 1–20
Damschen E, Haddad NM, Orrock JL et al (2006) Corridors increase plant species richness at large scales. Science 313:1284–1286. https://doi.org/10.1126/science.1130098
Damschen EI, Brudvig LA, Haddad NM et al (2008) The movement ecology and dynamics of plant communities in fragmented landscapes. Proc Natl Acad Sci USA 105:19078–1983. https://doi.org/10.1073/pnas.0802037105
de la Fuente B, Mateo-Sánchez MC, Rodríguez G et al (2018) Natura 2000 sites, public forests and riparian corridors: the connectivity backbone of forest green infrastructure. Land Use Policy 75:429–441. https://doi.org/10.1016/j.landusepol.2018.04.002
Dondina O, Saura S, Bani L, Mateo-Sánchez MC (2018) Enhancing connectivity in agroecosystems: focus on the best existing corridors or on new pathways? Landsc Ecol 33:1741–1756. https://doi.org/10.1007/s10980-018-0698-9
Dormann CF, Mcpherson JM, Araújo MB et al (2007) Methods to account for spatial autocorrelation in the analysis of species distributional data: a review. Ecography (Cop) 30:609–628. https://doi.org/10.1111/j.2007.0906-7590.05171.x
Engelhard SL, Huijbers CM, Stewart-Koster B et al (2017) Prioritising seascape connectivity in conservation using network analysis. J Appl Ecol 54:1130–1141. https://doi.org/10.1111/1365-2664.12824
Fajardo J, Mateo RG, Vargas P et al (2019) The role of abiotic mechanisms of long-distance dispersal in the American origin of the Galápagos flora. Glob Ecol Biogeogr 28:1610–1620. https://doi.org/10.1111/geb.12977
Fan J, Upadhye S, Worster A (2006) Understanding receiver operating characteristic (ROC) curves. Can J Emerg Med 8:19–20
Freeman EA, Moisen GG (2008) A comparison of the performance of threshold criteria for binary classification in terms of predicted prevalence and kappa. Ecol Model 217:48–58. https://doi.org/10.1016/j.ecolmodel.2008.05.015
Friedman JH (2001) greedy function approximation: a gradient boosting machine. Ann Stat 29:1189–1232. https://doi.org/10.1017/CBO9781107415324.004
Gibbs JP (2001) Demography versus habitat fragmentation as determinants of genetic variation in wild populations. Biol Conserv 100:15–20
Guisan A, Thuiller W, Zimmermann N (2017) Habitat suitability and distribution models: with applications in R. Cambridge University Press, Cambridge
Guisan A, Broennimann O, Buri A et al (2019) Climate change impacts on mountain biodiversity. In: Lovejou TE, Hannah L (eds) Biodiversity and climate change: transforming the biosphere. Yale University Press, New Haven, pp 221–233
Hijmans RJ, Cameron SE, Parra JL et al (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 1978:1965–1978. https://doi.org/10.1002/joc.1276
Holl KD, Aide TM (2011) When and where to actively restore ecosystems? For Ecol Manag 261:1558–1563. https://doi.org/10.1016/j.foreco.2010.07.004
Honnay O, Verheyen K, Butaye J et al (2002) Possible effects of habitat fragmentation and climate change on the range of forest plant species. Ecol Lett 5:525–530. https://doi.org/10.1046/j.1461-0248.2002.00346.x
Humphreys AM, Govaerts R, Ficinski SZ et al (2019) Global dataset shows geography and life form predict modern plant extinction and rediscovery. Nat Ecol Evol 3:1043–1047. https://doi.org/10.1038/s41559-019-0906-2
Knoke T, Ammer C, Stimm B, Mosandl R (2008) Admixing broadleaved to coniferous tree species: a review on yield, ecological stability and economics. Eur J For Res 127:89–101. https://doi.org/10.1007/s10342-007-0186-2
Lindborg R, Eriksson O (2004) Historical landscape connectivity affects present plant species diversity. Ecology 85:1840–1845
Lovejoy TE, Wilson EO (2019) Biodiversity and climate change: transforming the biosphere. Yale University Press, New Haven
Maes D, Van Dyck H (2005) Habitat quality and biodiversity indicator performances of a threatened butterfly versus a multispecies group for wet heathlands in Belgium. Biol Conserv 123:177–187. https://doi.org/10.1016/j.biocon.2004.11.005
Mateo RG, Gastón A, Aroca-fernández MJ et al (2018) Optimization of forest sampling strategies for woody plant species distribution modelling at the landscape scale. For Ecol Manag 410:104–113. https://doi.org/10.1016/j.foreco.2017.12.046
Mateo RG, Aroca-Fernández MJ, Gastón A et al (2019a) Looking for an optimal hierarchical approach for ecologically meaningful niche modelling. J Methods Ecol 409. https://doi.org/10.1016/j.ecolmodel.2019.108735
Mateo RG, Gastón A, José M et al (2019b) Hierarchical species distribution models in support of vegetation conservation at the landscape scale. J Veg Sci. https://doi.org/10.1111/jvs.12726
McCullagh P, Nelder JA (1989) Generalized linear models. Chapman & Hall, Boston
McCune B, Keon D (2002) Equations for potential annual direct incident radiation and heat load. J Veg Sci 13:603–606. https://doi.org/10.1111/j.1654-1103.2002.tb02087.x
Mori AS, Lertzman KP, Gustafsson L (2017) Biodiversity and ecosystem services in forest ecosystems: a research agenda for applied forest ecology. J Appl Ecol 54:12–27. https://doi.org/10.1111/1365-2664.12669
Murphy HT, Lovett-Doust J (2004) Context and connectivity in plant metapopulations and landscape mosaics: does the matrix matter? Oikos 105:3–14. https://doi.org/10.1111/j.0030-1299.2004.12754.x
Neel MC (2008) Patch connectivity and genetic diversity conservation in the federally endangered and narrowly endemic plant species Astragalus albens (Fabaceae). Biol Conserv 141:938–955. https://doi.org/10.1016/j.biocon.2007.12.031
Opdam P, Wascher D (2004) Climate change meets habitat fragmentation: linking landscape and biogeographical scale levels in research and conservation. Biol Conserv 117:285–297. https://doi.org/10.1016/j.biocon.2003.12.008
Pascual-Hortal L, Saura S (2006) Comparison and development of new graph-based landscape connectivity indices: towards the priorization of habitat patches and corridors for conservation. Landsc Ecol 21:959–967. https://doi.org/10.1007/s10980-006-0013-z
Pecchi M, Marchi M, Burton V et al (2019) Species distribution modelling to support forest management. A literature review. Ecol Model 411:108817. https://doi.org/10.1016/j.ecolmodel.2019.108817
Petsas P, Tsavdaridou AI, Mazaris AD (2020) A multispecies approach for assessing landscape connectivity in data-poor regions. Landsc Ecol 35:561–576. https://doi.org/10.1007/s10980-020-00981-2
Phillips SJ, Williams P, Midgley G, Archer A (2008) Optimizing dispersal corridors for the cape proteaceae using network flow. Ecol Appl 18:1200–1211
Rico Y, Boehmer HJ, Wagner HH (2012) Determinants of actual functional connectivity for calcareous grassland communities linked by rotational sheep grazing. Landsc Ecol 27:199–209. https://doi.org/10.1007/s10980-011-9648-5
Rincón G, Solana-Gutiérrez J, Alonso C et al (2017) Longitudinal connectivity loss in a riverine network: accounting for the likelihood of upstream and downstream movement across dams. Aquat Sci 79:573–585. https://doi.org/10.1007/s00027-017-0518-3
Roberge JM, Angelstam P (2004) Usefulness of the umbrella species concept as a conservation tool. Conserv Biol 18:76–85. https://doi.org/10.1111/j.1523-1739.2004.00450.x
Rodríguez JP, Brotons L, Bustamante J, Seoane J (2007) The application of predictive modelling of species distribution to biodiversity conservation. Divers Distrib 13:243–251. https://doi.org/10.1111/j.1472-4642.2007.00356.x
Rubio L, Rodriguez-Freire M, Mateo Sánchez MC et al (2012) Sustaining forest landscape connectivity under different land cover change scenarios. For Syst 21:223–235. https://doi.org/10.5424/fs/2012212-02568
Saura S (2007) A new habitat availability index to integrate connectivity in landscape conservation planning: comparison with existing indices and application to a case study. Landsc Urban Plan 83:91–103. https://doi.org/10.1016/j.landurbplan.2007.03.005
Saura S, Pascual-Hortal L (2007) A new habitat availability index to integrate connectivity in landscape conservation planning: comparison with existing indices and application to a case study. Landsc Urban Plan 83:91–103. https://doi.org/10.1016/j.landurbplan.2007.03.005
Saura S, Rubio L (2010) A common currency for the different ways in which patches and links can contribute to habitat availability and connectivity in the landscape. Ecography (Cop) 33:523–537. https://doi.org/10.1111/j.1600-0587.2009.05760.x
Saura S, Torné J (2009) Conefor Sensinode 2.2: a software package for quantifying the importance of habitat patches for landscape connectivity. Environ Model Softw 24:135–139. https://doi.org/10.1016/j.envsoft.2008.05.005
Saura S, Estreguil C, Mouton C, Rodríguez-Freire M (2011) Network analysis to assess landscape connectivity trends: application to European forests (1990–2000). Ecol Indic 11:407–416. https://doi.org/10.1016/j.ecolind.2010.06.011
Saura S, Bodin Ö, Fortin MJ (2014) Stepping stones are crucial for species’ long-distance dispersal and range expansion through habitat networks. J Appl Ecol 51:171–182. https://doi.org/10.1111/1365-2664.12179
Saura S, Bertzky B, Bastin L et al (2018) Protected area connectivity: shortfalls in global targets and country-level priorities. Biol Conserv 219:53–67. https://doi.org/10.1016/j.biocon.2017.12.020
Sutherland WJ, Fleishman E, Clout M et al (2019) Ten years on: a review of the first global conservation horizon scan. Trends Ecol Evol 34:139–153. https://doi.org/10.1016/j.tree.2018.12.003
Thuiller W, Albert C, Araújo MB et al (2008) Predicting global change impacts on plant species ’ distributions: future challenges. Perspect Plant Ecol Evol Syst 9:137–152. https://doi.org/10.1016/j.ppees.2007.09.004
Tischendorf L, Fahrig L (2000) On the usage and measurement of landscape connectivity. Oikos 90:7–19
Urban DL, Keitt T (2001) Landscape connectivity: a graph-theoretic perspective. Ecology 82:1205–1218
Urban DL, Minor ES, Treml EA, Robert S (2009) Graph models of habitat mosaics. Ecol Lett 12:260–273. https://doi.org/10.1111/j.1461-0248.2008.01271.x
Vetaas OR (2002) Realized and potential climate niches: a comparison of four Rhododendron tree species. J Biogeogr 29:545–554. https://doi.org/10.1046/j.1365-2699.2002.00694.x
Vittoz P, Engler R (2007) Seed dispersal distances: a typology based on dispersal modes and plant traits. Bot Helv 117:109–124. https://doi.org/10.1007/s00035-007-0797-8
Wang BC, Smith TB, Wang BC (2002) Closing the seed dispersal loop. Trends Ecol Evol 17:8–10
Zeller KA, McGarigal K, Whiteley AR (2012) Estimating landscape resistance to movement: a review. Landsc Ecol 27:777–797. https://doi.org/10.1007/s10980-012-9737-0
Acknowledgements
This study was supported by funding from the Comunidad de Madrid and the European Union through the BOSSANOVA project (P2013/MAE-2760), and the Universidad Politécnica de Madrid. We would like to thank Santiago Saura for his advice on the design of the study. We also appreciate the contribution of Gonzalo Chamorro and Eva Gavela who assisted in the collection of the vegetation survey.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. This work was supported by the Comunidad de Madrid and the European Union under the BOSSANOVA project (P2013/MAE-2760); and Universidad Politécnica de Madrid under Grant Programa Propio 2018.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Not applicable.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Daniel Sanchez Mata.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visithttp://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Goicolea, T., G. Mateo, R., Aroca-Fernández, M.J. et al. Considering plant functional connectivity in landscape conservation and restoration management. Biodivers Conserv 31, 1591–1608 (2022). https://doi.org/10.1007/s10531-022-02413-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-022-02413-w