Abstract
Global climate change is increasing its negative impact on agricultural production day by day. Abiotic stress factors brought about by climate change cause significant losses in yield, quality, and plant growth. In addition to increasing the effects of drought and salinity with global warming, changes in precipitation regimes and flooding stresses have recently negatively affected agricultural areas. To avoid the negative effects of stress, proline (Pro) and glycine betaine (GB) applications are the subject of research as important applications. This study, it was aimed to determine the effects of Pro and GB doses on growth, physiological and biochemical parameters applied under conditions artificially created ten-day flooding stress in green onions. One full irrigation subject (I100) and one stress subject (flooding), were created in the experiment. On the other hand, three different Pro doses (Pro1–1 μM, Pro2–2 μM, and Pro3–3 μM) and three different GB doses (GB50–50 μM, GB100–100 μM, and GB150–150 μM) administration and control were the other subjects of the study. Pro and GB applications were applied to the leaves as a spray twice. As a result of the study, flooding stress revealed 28% loss in leaf number, 43% plant fresh weight, and 32% plant dry weight loss. Malondialdehyde (MDA), hydrogen peroxidase (H2O2), and leaf temperature (T leaf) flooding stress conditions were found to be the parameters that provide the most significant increase and define the stress. It was observed that Pro2, Pro3, and GB50 applications supported plant growth under full irrigation conditions. Besides, it was revealed that Pro3 and GB50 doses reduced the negative effects of flooding stress. It has been concluded that appropriate doses of Pro and GB applications in terms of sustainable agriculture are important applications in reducing the negative effects of stress.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Since the discovery of agriculture by human beings, plants are exposed to many abiotic stress conditions such as floods, drought, salt stress, mineral malnutrition, and high-low temperatures throughout their lives (Teklić et al. 2021). Abiotic stress causes serious losses in agricultural production worldwide and significantly reduces the amount of agricultural land; It is one of the most important problems of today’s agriculture and causes up to 50% yield loss in plants under stress conditions (Rockström et al. 2017).
The prevalence of abiotic stress in large agricultural lands where climate change is effective and its negative effects on plants continue to increase day by day. With the effect of global climate change, it is seen that almost all of the world’s agricultural lands are affected positively or negatively, as well as the activities of abiotic stress factors have increased. The increase in surface temperatures as a result of the intense increase in greenhouse gases in the atmosphere as a result of global climate change has revealed significant changes in the precipitation regime (Gray and Brady 2016). Besides causing drought, surface temperatures adversely affected the intensity of precipitation and triggered flood formation. Studies have shown that the risk of flooding will increase day by day and its effects will negatively affect world life (Mirza 2011; Ghanbari et al. 2021; Boulange et al. 2021). For this reason, floods have become an important abiotic stress factor limiting agricultural production.
In addition to the fact that the frequency and duration of flooding stress are important in the formation, its effect on plants varies according to many factors such as the structure of the soil, the number of nutrients in the soil, species, and variety of characteristics (Seymen 2021). While a loss of 15–80% in yield occurs depending on the occurrence of flooding stress (Patel et al. 2014), there is a loss of up to 100% in terms of quality, depending on the plant type. As a result of flooding stress formation, aerobic root respiration is impaired, and consequently physiological and metabolic effects in plants (Ma et al. 2022; Martínez-Arias et al. 2022). The accumulation of excess water in the soil because of flooding causes a decrease in the soil redox potential (Ponting et al. 2021), and significant changes occur in the nutrient profile. When free oxygen is depleted, soil microorganisms use nitrogen as an alternative electron acceptor in respiration (Martín-Rodríguez 2022). In addition, because of the inhibition of gas exchange in the root zone, high partial pressures of CO2 have negative effects on soil flora and root growth (Patel et al. 2014). As a result, plants try to respond to stress by increasing metabolic enzymes as well as morphological, physiological, and biochemical changes (Greenway et al. 2006; Seymen 2021). Superoxide, hydrogen peroxide, singlet oxygen, and hydroxyl radicals formed as a result of oxygen deficiency prevent the development by accumulating reactive oxygen species (ROS) in plant cells (Das and Roychoudhury, 2014). Disruptions in the ROS balance cause damage to proteins, DNA, lipids, and cellular functions, as well as cause changes in metabolic events, affecting plants from restriction to death (Anjum et al. 2015).
Plants respond to flooding stress with their defense mechanisms, as well as using tolerant species and cultivars against flooding stress (Jackson et al. 2009; Ezin et al. 2010), making use of rootstocks in grafted plants (Choi et al. 2020; Haghighi 2022) and the use of some biostimulant (Castro-Duque et al. 2020). Researchers have shown that proline (Pro) and glycine betaine (GB), which act as biostimulants, contribute to plant growth in many stress factors (Zouari et al. 2018; Dikilitas et al. 2020; Kayak et al. 2022).
In addition to the Pro regulating the osmotic function in plants, it is involved in the defense mechanism of cells against stress and as a signal molecule that interacts with metabolic pathways (Kishor et al. 2005; Sharma et al. 2011). GB, on the other hand, is not directly involved in the scavenging of ROS under abiotic stress conditions (Chen and Murata 2008). GB plays an important role in the protection of photosynthetic activity by making important contributions to chloroplasts and thylakoid membranes, as well as protecting cells from oxidative damage under abiotic stress conditions (Valenzuela-Soto and Figueroa-Soto 2019).
Onion (Allium cepa L.) is one of the important vegetable species consumed fresh and dry in human nutrition. According to 2020 records, it has been reported that 4,452,347 tons of green onions are produced in 208,347 hectares of land worldwide. It is recorded that 129,023 tons of green onions are produced in 7797 hectares of land in Turkey (FAO 2022). In Turkey, high-quality fresh onions are produced in open field conditions in regions with a transitional climate. However, in Turkey, it can be cultivated in the form of greenhouse cultivation in winter, in open field conditions in winter periods in temperate climates, and in early spring and autumn periods in regions with a continental climate. In these periods, with the effect of global climate change, heavy rains negatively affect the onion-growing areas. For this reason, it is essential to know exactly the effects of flooding stress on yield and quality in plants, as well as physiological and biochemical changes. It has become more important to develop some strategies to reduce the negative effects of flooding stress (Seymen 2021). In the current study, Pro and GB applications, which are considered biological applications to plants exposed to flooding stress, are seen as an important approach. Studies have shown that Pro and GB applications have positive effects on many abiotic stress factors (Kayak et al. 2022). But, although the effects of flooding stress conditions have been determined in some species, studies have not been conducted or are limited to important vegetable species such as onions. For this purpose, it was aimed to determine the positive effects of plant growth, physiological and biochemical changes and applied Pro and GB doses in green onions under flooding stress.
Materials and Methods
Experimental Area
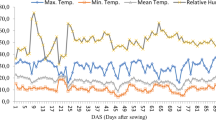
The research was carried out in the greenhouse belonging to the Department of Horticulture, Faculty of Agriculture, Selcuk University, between February 24 and May 19, 2022. The soil used in the research has a clay-loam structure and contains 2.9% organic matter. It was determined that the EC was 1.17 dS/m, pH 7.61 and the lime content was 7.7%. It has been determined that the soil is in good condition in terms of nutrients and field capacity and the wilting point is 29.6% and 13.4%, respectively. Some of the climatic parameters measured between the dates of the research were taken from the portable meteorology station (Davis Vantage Pro2) installed in the experimental area. The average temperature and average relative humidity values taken are presented in Fig. 1 and there was no restriction in onion cultivation.
Plant Material, Experimental Design, and Cultural Practices
BT-BUR-TOP, one of the most cultivated onion varieties in the market, was used as plant material. Plastic pots with a top diameter of 29 cm, a bottom diameter of 19 cm, and a height of 24 cm were used in the experiment. After placing the unperforated bags inside the pots, 12.5 kg of air-dry soil was placed in each of them. 10 seeds were planted in each pot and 1 liter of irrigation water was given. After the seeds germinated, thinning was done to leave five plants in each pot.
In the study, which was established according to the randomized plots trial design, two irrigation subjects were created, one full irrigation subject (I100) and one stress subject (flooding). On the other hand, three different Pro doses (Pro1–1 μM, Pro2–2 μM, and Pro3–3 μM), three different GB (GB50–50 μM, GB100–100 μM, and GB150–150 μM) administrations, and seven factors were created as control. The experiment has 3 replications, and each replica has a total of five pots. The amount of irrigation water applied to the pots was determined according to the gravimetric soil moisture measurement method. For this purpose, when the useful water capacity of the control I100 subject, which was chosen as the witness subject, fell to 40–45%, irrigation was carried out and the soil moisture reached the field capacity each time.
After the samples, which were calculated as three doses of Pro (L-Proline-CAS 147-85-3-Calbiochem, MERCK) and GB (Betaine CAS 107-43‑7 C9H11NO2, SIGMA), were dissolved in pure water, solutions were prepared in separate containers. The prepared solutions were applied to the leaves of the plants by spray method twice, on April 26 and May 9, to cover the whole plant. At the same time, stress issues were initiated on May 9 and ten-day flooding stress was created. Irrigation was made so that 2–3 cm of water remained on the soil surface and the decreasing water was added every day. For full irrigation issues, the calculated amount of water continued to be given. During the experimental period, aeration of the potting soil, weed control, disease and pest control was done on time and regularly as needed. During the experimental period, no factor limiting plant growth was encountered. After completing ten days under flooding stress, the plants were harvested, and necessary measurements were made.
Analyses of Growth Parameters
The leaves on the onion plants (NL) were counted and recorded just before the harvest. Then, the fresh weights (WPF) of five plants cut from the soil surface were taken and the root weights (WRF) of the plants whose roots were cleaned were determined. The samples, whose fresh weights were determined, were first dried in a certain amount in the shade, then dried in an oven at 65 °C until they reached an equal weight, and the weights were determined with the help of precision scales. After calculating the dry weights taken, the weight of plant dry (WPD) and weight of root dry (WRD) were determined.
Determination of Physiological Parameters
With the LI-COR brand LI-600 fluorimeter device, leaf temperature (°C) before the harvest from the old leaves between 9:00 and 11:00 a.m. on a sunny day stomatal conductance (GSW) and actual photosynthetic efficiency (PhiPSII) measurements were made (Liang et al. 2019). Leaf relative water content (RWC) and membrane permeability (MD) were determined using the method of Lutts et al. (1996).
Analyses of Color Pigments, Proline, and Protein Contents
Measurements of chlorophyll a and b from fully expanded leaves were determined for each replicate by taking readings at 663 and 470 nm wavelengths in a spectrophotometer (Shimadzu-UV-6300PC) using the method of Lichtenthaler and Buschmann (2001). The carotenoid content in the samples was analyzed according to Jaspar’s formula (Witham et al. 1971). On the determination of proline content in onion leaves, Bates et al. (1973) followed, while the method of Bradford (1976) was used to determine the protein content.
Analyses of Malondialdehyde, Hydrogen Peroxidase, and Antioxidant Enzyme Activity
Samples taken from onion leaves were prepared according to the method of Heath and Packer (1968), and their malondialdehyde (MDA) contents were determined by measuring at wavelengths of 532 nm and 600 nm with a spectrophotometer. Hydrogen peroxide concentration was determined by spectrophotometer according to the method explained by Velikova et al. (2000). Samples were prepared according to Angelini and Federico (1989) for the determination of enzyme activities in onion leaves. SOD activity according to Agarwal and Pandey (2004), POD activity according to Chance (1955), and CAT activity according to Havir and McHale (1987) were determined in the prepared samples by spectrophotometer.
Statistical Analysis
Morphological, physiological, and biochemical measurements were subjected to the JMP-13 statistical program to determine the effects of flooding stress and applied Pro and GB applications. Significant differences between the applications were tested at the 5% significance level according to LSMeans Student’s t. Principal component analysis (PCA) was used to evaluate all the data obtained with the same statistical program. Important parameters and important applications are interpreted with the loading plot and score plot graphs drawn from the resulting PC1 and PC2.
Results
Effects of pro and GB on Growth Parameters Under Flooding Stress
It was observed that flooding (Flo) stress applied in green onions had statistically significant effects on leaf number (NL), plant fresh weight (WPF), plant dry weight (WPD), and root dry weight (WRD) (Table 1). Flooding stress showed decreases in NL, WPF, and WPD, but increased in WRD. Root fresh weight (WRF) did not show any change with the applied flooding stress. The applied proline (Pro) and glycine betaine (GB) doses showed significant changes in growth parameters. Pro3 application has shown a significant increase in all growth parameters. When the interactions were examined, the I100-Pro3 application showed a significant increase in NL and WPF. WRF was highest in I100-Pro1, I100-Pro3, and Flo-Pro3 applications. The highest WPD values were obtained from I100-Pro2 and I100-Pro3 applications. The highest WRD was obtained from I100-GB100, Flo-control, Flo-Pro1, Flo-Pro2, Flo-Pro3 and Flo-GB100 applications.
Effects of pro and GB on Physiological Parameters Under Flooding Stress
It was observed that increasing Pro and GB doses had significant effects on the physiological activities of green onions under flooding stress conditions (Table 2). Flooding stress caused an increase in leaf temperature (T leaf) and a significant decrease in leaf relative water content (RWC). Flooding stress did not show any effect on stomatal conductance (GSW), actual photosynthetic efficiency (PhiPSII), and membrane damage (MD). When the applied Pro and GB doses were examined, the highest T leaf GB doses were found in the subjects applied. The highest GSW was obtained from Pro2, Pro3, and GB50 doses. Pro3 application decreased PhiPSII compared to other application doses and control. MD was obtained from the highest control application, while the highest RWC was obtained from Pro3 and GB50 doses. When the interactions were examined, the highest T leaf was obtained from I100-GB100, I100-GB150, Flo-control, Flo-Pro1, Flo-Pro2, Flo-GB100, and Flo-GB150 applications. The highest GSW was found in I100-Pro2 and Flo-Pro3 applications. The highest MD was obtained from the controls of I100 and Flo applications. Although the highest RWC was obtained from some applications under full irrigation conditions, Pro3 and GB50 applications applied under stress conditions increased the RWC content.
Effects of pro and GB on Color Pigments, Proline, and Protein Contents Under Flooding Stress
It was observed that Pro and GB doses applied in green onions under flooding stress conditions had statistically significant effects on color pigments, proline, and protein content (Table 3). Flooding stress significantly reduced the color pigments and protein content. The administered Pro2 dose provided a significant increase in Chl a, Chl b, and carotenoid (CT) content. Proline (PL) and protein (PT) contents decreased as a result of the applied Pro and GB doses, and the highest values were obtained from the control application. When the interactions were examined, the highest Chl a, Chl b, and CT contents were obtained from the I100-Pro2 aplication. In addition In addition to the PL content was the highest from I100-control, I100-Pro1, I100-Pro2 and Flo-control applications. The highest PT content was found in the I100-GB150 interaction.
Effects of pro and GB on Malondialdehyde, Hydrogen Peroxidase, and Antioxidant Enzyme Activity Under Flooding Stress
It has been observed that Pro and GB doses applied to green onions under flooding stress conditions have significant effects on malondialdehyde (MDA), hydrogen peroxidase (H2O2), and antioxidant enzyme activity (Table 4). When the table is examined, flooding stress caused a significant increase in MDA and H2O2 content, while a decrease in antioxidants. While the GB50 dose increased MDA content, GB50 and Pro1 doses also increased H2O2 content. Pro3 and GB100 applications provided a reduction in H2O2 content compared to the control. In terms of antioxidant activity, the applied Pro3 dose increased the CAT activity, and the GB100 dose increased the SOD activity compared to the control. When the interactions were examined, the GB50 application had the highest MDA content in flooding stress. On the other hand, the highest H2O2 content was obtained from control and Pro2 application under flooding stress conditions. The highest CAT content was found in I100-Pro3, I100-GB150, and Flo-control interactions. SOD contents were obtained from GB50 and GB100 applications under full irrigation conditions, and from GB100 and GB150 applications under flooding stress conditions.
PCA Analysis
Growth, physiological and biochemical parameters obtained as a result of Pro and GB applications applied to green onions under flooding stress conditions were subjected to principal component analysis (PCA) (Table 5). As a result of PCA, the study was explained as high as 80.85% in five components. The first component explained 27.09% of the study, NL, WPF, WPD, and Chl a parameters were the most powerful explanatory parameters in the positive direction and H2O2 in the negative direction. When the second component is examined, GSW and CT are strong parameters that explain in the positive direction and SOD in the negative direction. In the third component, WRF was positive, MD, Chl b and PL were the parameters that explained it in the negative direction. While GSW, PT, and POD explained the fourth component in the positive direction, CAT explained it in the negative direction. In the last component, WRD and PhiPSII were the strongest explanatory parameters in the positive direction, MDA and SOD in the negative direction.
A loading plot was drawn from PC1 and PC2 to examine the strong correlations between the parameters (Fig. 2). When the graph is examined, a strong positive correlation was observed between the properties that show a significant increase in abiotic stress conditions such as MDA, H2O2, and T leaf. It is seen that these parameters exhibit a strong negative correlation with growth parameters such as WPD, WPF, and NL. On the other hand, significant positive correlations were found between parameters such as GSW, CAT, CT, RWC, Chl a, and Chl b. These parameters are defined as the parameters that are located in the positive region of both components and define the applications that give the best results. When the drawn score plot graph is examined, the first component is the one that defines the effect of flooding stress (Fig. 3). On the right side of PC1, there are full watering topics, while in the negative area there are flooding stress subjects. MDA, H2O2, and T leaf flooding stress are the parameters that provide the most significant increase and define the stress. I100-Pro2, I100-Pro3, and I100-GB150 interactions have been applications showing significant increases in terms of GSW, CAT, CT, RWC, Chl a, and Chl b. In other words, in addition to Pro2 and Pro3 applications applied under full irrigation conditions, GB50 application will contribute to plant growth. Although all Pro and GB doses remain in the negative region under flooding stress conditions, the fact that Pro3 and GB50 doses are close to the positive region shows that it is effective. It is seen that especially the dose of Pro3 will make a significant contribution to avoiding the stress of green onions under flooding stress conditions.
Score plot from PC1 and PC2 based on PCA for growth, physiological, biochemical contents, and chlorophyll fluorescence measurement of increasing doses of Pro and GB applied to green onion under flooding and non-stress conditions. FIo (Flooding irrigation), S100 (Non-stress), Cont (No proline and glycine betaine), Pro1 (1 µM proline), Pro2 (2 µM proline), Pro1 (3 µM proline), GB50 (50 µM glycine betaine), GB100 (100 µM glycine betaine), GB150 (150 µM glycine betaine)
Discussion
Flooding stress applied in green onions revealed significant changes in growth, physiological and biochemical contents. There was a 28% loss in leaf number, 43% in plant fresh weight, and 32% in dry weight of the plant, which was limited under flooding stress conditions. The applied Pro3 dose showed significant increases in growth parameters (Table 1). It has been reported that there are significant decreases in the number of leaves in onions (Gedam et al. 2021), tomatoes (Ezin et al. 2010), and spinach (Seymen 2021) under flooding stress conditions. On the other hand, it has been reported by various researchers that plant fresh and dry weight and root fresh and dry weight decrease under flooding stress conditions (Yiu et al. 2009; Ghodke et al. 2018; Rasheed et al. 2018). It has been revealed that Pro and GB applications applied in many stress studies increase the growth parameters (Heuer 2003; Osman 2015; Kayak et al. 2022). Known as an osmotic regulator, proline stabilizes cell membranes and proteins helps scavenge free radicals, and contributes to the buffering of cellular redox potential, helping to protect against the negative effects of stress (Ashraf and Foolad 2007). At the same time, Pro, which is a protein-compatible hydro trope, helps to maintain NADP+/NADPH ratios compatible with metabolism (Srinivas and Balasubramanian 1995). Pro applications acted as an important preservative in plant growth under flooding stress conditions in green onions.
Flooding stress caused a significant increase in leaf temperature and decreased RWC content (Table 2). In many studies, it has been reported that there are significant increases in leaf temperature under stress conditions (Puengsungwan and Jirasereeamornkul 2020) and decreases in RWC content (Yiu et al. 2009; Kayak et al. 2022). The administered Pro3 and GB50 doses contributed to the GSW and RWC content. A decrease in stomatal conductivity occurs when plants are exposed to flooding stress (Folzer et al. 2006). In addition to increased stomatal resistance in plants under flooding stress conditions, water deficit occurs in the cells because of limited water intake (Parent et al. 2008). On the other hand, the decrease in the O2 level negatively affects hydraulic conductivity by preventing root permeability (Else et al. 2001). The lack of oxygen usually causes a decrease in the net rate of photosynthesis (Ashraf et al. 2011). This decrease in sweating and photosynthesis results in the closure of stomata (Ashraf and Arfan 2005). Sometimes, the decrease in chlorophyll content causes decreases in photosynthesis and stomatal conductivity. Yordanova et al. (2005) reported that barley plants rapidly closed their stomata under flooding stress conditions and a decrease in stomatal conductivity occurred. Similarly, in a study conducted on peas, it was reported that flooding stress caused stomata to close (Zhang and Zhang, 1994).
Photosynthesis is an important event for plant development and the realization of biochemical events in the plant. The increase in leaf temperature under flooding stress conditions causes the closure of stomata. This results in a decrease in the amount of photosynthesis (Barickman et al. 2019). The stress of flooding causes a decrease in photosynthesis efficiency by limiting CO2 entry, and oxidative damage on photosynthesis II with an accumulation of ROS and accumulation of ethylene (Rao and Li 2003; Bansal and Srivastava 2015). Castro-Duque et al. (2020) reported that the flooding stress they applied in gooseberry affected PSII negatively, and the GB they applied made a significant contribution to the PSII activity. In a study, drought stress applied in rice caused a decrease in PhiPSII, but the applied GB dose did not make any contribution (Chaum et al. 2013). In our study, although flooding stress did not adversely affect photosynthesis inputs, it was observed that the applied Pro and GB doses contributed to net photosynthesis.
In this study, flooding stress decreased the content of Chl a (22%), Chl b (20%), CT (6%), and PT (11%) in green onions, while increasing the content of PL (2%). The applied Pro2 dose contributed significantly to the color pigments content (Table 3). Chlorophyll and carotenoids are two important types of photosynthetic pigments found as pigment-protein complexes in the thylakoid membrane of chloroplasts (Bartley and Scolnik 1995). With the aging of the leaves, the chloroplasts are fragmented, there is a decrease in Chl content, and photosynthetic activity decreases (Cardinif and Bonzi 2005). The negative effect of photosynthesis in flooding stress causes significant decreases in chlorophyll content. In a study conducted on 12 different onion genotypes, it was reported that flooding stress caused significant losses in color pigments (Gedam et al. 2021). Similarly, it has been reported that there is a decrease in color pigments in cowpea under flooding stress conditions (El-Enany et al. 2013). According to Kayak et al. (2022) reported that Pro and GB applications applied under drought stress conditions contributed to the color pigments.
In this study, flooding stress caused an increase in MDA and H2O2 content, while it caused a decrease in antioxidant enzymes. The administered Pro3 dose helped to increase CAT activity while decreasing the H2O2 content. Similarly, the GB100 application caused decreases in H2O2 content and contributed to SOD activity (Table 4). Many researchers emphasize that H2O2 and MDA increase under flooding stress conditions (Cao et al. 2017; Radmann et al. 2018; Seymen, 2021). Enzymes such as SOD, CAT, and POT have an important contribution to the scavenging of ROS formed under flooding stress conditions. They can catalyze SOD superoxide to molecular oxygen (O2) and hydrogen peroxide (H2O2). At the same time, CAT and POD enzymes can decompose H2O2 by converting it to oxygen and water (Qamer et al. 2021). As a matter of fact, in our study, it was seen that Pro and GB applications contributed to the antioxidant defense system against flooding stress.
PCA is an analysis method that guides researchers in determining important parameters, tolerant genotypes, and important applications under flooding stress conditions (Seymen 2021; Zhou et al. 2021). As a result of PCA analysis, it has been reported that the first two components are explained above 25%, which will give important results for the study and its usability (Mohammadi and Prasanna 2003; Yavuz et al. 2021; Yavuz et al. 2022). In our study, the first two components explained 44% of the study. In stress studies, there are many studies in which the first component shows the effect of stress (Seymen et al. 2019; Seymen 2021; Yavuz et al. 2022). In the present study, the first component’s positive region included the exact irrigation subject and explanatory parameters, while the negative region included the flooding stress issues and parameters (Figs. 2 and 3). MDA, H2O2, and T leaf were the parameters that showed the highest increase under flooding stress conditions and explained the stress. It has been explained by many researchers that these parameters increase under stress conditions (Cao et al. 2017; Radmann et al. 2018; Puengsungwan and Jirasereeamornkul 2020; Seymen 2021). It has been revealed that Pro2, Pro3, and GB50 applications applied under full irrigation conditions, Pro3 and GB50 doses under flooding stress conditions will contribute to plant growth.
Conclusion
It has been observed that the doses of proline and glycine betaine applied in green onions under flooding stress conditions have significant effects on growth, physiological and biochemical contents. The ten-day flooding stress applied to green onions significantly limited the growth of the plant, resulting in 28% loss in leaf number, 43% in plant fresh weight, and 32% loss in plant dry weight. MDA, H2O2, and T leaf flooding stress conditions were found to be the parameters that provide the most significant increase and define the stress. It was concluded that, in addition to Pro2 and Pro3 applications applied under full irrigation conditions, GB50 application will contribute to plant growth. In flooding stress conditions, it was observed that the doses of Pro3 and GB50 applied reduced the negative effect of flooding stress. It is recommended that these practices are important in terms of sustainable agriculture and that they can be applied as protective practices in terms of yield and quality in the lands where green onion cultivation is made because of increasing floods as a result of global climate change.
References
Agarwal S, Pandey V (2004) Antioxidant enzyme responses to NaCl stress in Cassia angustifolia. Biol Plant 48(4):555–560. https://doi.org/10.1023/B:BIOP.0000047152.07878.e7
Angelini R, Federico R (1989) Histochemical evidence of polyamine oxidation and generation of hydrogen peroxide in the cell wall. J Plant Physiol 135(2):212–217. https://doi.org/10.1016/S0176-1617(89)80179-8
Anjum NA, Sofo A, Scopa A, Roychoudhury A, Gill SS, Iqbal M, Lukatkin AS, Pereira E, Duarte AC, Ahmad I (2015) Lipids and proteins—major targets of oxidative modifications in abiotic stressed plants. Environ Sci Pollut Res 22(6):4099–4121. https://doi.org/10.1007/s11356-014-3917-1
Ashraf M, Arfan M (2005) Gas exchange characteristics and water relations in two cultivars of Hibiscus esculentus under waterlogging. Biol Plant 49(3):459–462
Ashraf MFMR, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59(2):206–216. https://doi.org/10.1016/j.envexpbot.2005.12.006
Ashraf MA, Ahmad MSA, Ashraf M, Al-Qurainy F, Ashraf MY (2011) Alleviation of waterlogging stress in upland cotton (Gossypium hirsutum L.) by exogenous application of potassium in soil and as a foliar spray. Crop Pasture Sci 62(1):25–38. https://doi.org/10.1071/CP09225
Bansal R, Srivastava J (2015) Effect of waterlogging on photosynthetic and biochemical parameters in pigeonpea. Russ J Plant Physiol 62(3):322–327
Barickman TC, Simpson CR, Sams CE (2019) Waterlogging causes early modification in the physiological performance, carotenoids, chlorophylls, proline, and soluble sugars of cucumber plants. Plants 8(6):160. https://doi.org/10.3390/plants8060160
Bartley GE, Scolnik PA (1995) Plant carotenoids: pigments for photoprotection, visual attraction, and human health. Plant Cell 7:1027–1038. https://doi.org/10.1105/tpc.7.7.1027
Bates LS, Waldren RP, Teare I (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39(1):205–207. https://doi.org/10.1007/BF00018060
Boulange J, Hanasaki N, Yamazaki D, Pokhrel Y (2021) Role of dams in reducing global flood exposure under climate change. Nat Commun 12(1):1–7. https://doi.org/10.1038/s41467-020-20704-0
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1016/0003–2697(76)90527–3
Cao Y, Ma C, Chen G, Zhang J, Xing B (2017) Physiological and biochemical responses of Salix integra Thunb. under copper stress as affected by soil flooding. Environ Pollut 225:644–653. https://doi.org/10.1016/j.envpol.2017.03.040
Cardinif F, Bonzi LM (2005) Carotenoid composition and its chemotaxonomic significance in leaves of ten species of the genus Ceratozamia (Cycads). J Plant Physiol 162:517–528. https://doi.org/10.1016/j.jplph.2004.06.007
Castro-Duque NE, Chávez-Arias CC, Restrepo-Díaz H (2020) Foliar glycine betaine or hydrogen peroxide sprays ameliorate waterlogging stress in cape gooseberry. Plants 9(5):644. https://doi.org/10.3390/plants9050644
Chance B (1955) Assay of catalase and peroxidase. Meth Enzymol 2:765–775. https://doi.org/10.1016/S0076-6879(55)02300-8
Chaum S, Samphumphuang T, Kirdmanee C (2013) Glycinebetaine alleviates water deficit stress in indica rice using proline accumulation, photosynthetic efficiencies, growth performances and yield attributes. Aust J Crop Sci 7(2):213–218
Chen TH, Murata N (2008) Glycinebetaine: an effective protectant against abiotic stress in plants. Trends Plant Sci 13(9):499–505. https://doi.org/10.1016/j.tplants.2008.06.007
Choi BH, Bhusal N, Jeong WT, Park IH, Han SG, Yoon TM (2020) Waterlogging tolerance in apple trees grafted on rootstocks from G, CG, and M series. Hortic Environ Biotechnol 61(4):685–692. https://doi.org/10.1007/s13580-020-00258-2
Das K, Roychoudhury A (2014) Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front Environ Sci 2:53. https://doi.org/10.3389/fenvs.2014.00053
Dikilitas M, Simsek E, Roychoudhury A (2020) Role of proline and glycine betaine in overcoming abiotic stresses. In: Protective chemical agents in the amelioration of plant abiotic stress: biochemical and molecular perspectives, pp 1–23
El-Enany AE, Al-Anazi AD, Dief N, Al-Taisan WA (2013) Role of antioxidant enzymes in amelioration of water deficit and waterlogging stresses on Vigna sinensis plants. J Biol Earth Sci 3(1):B144–B153
Else MA, Coupland D, Dutton L, Jackson MB (2001) Decreased root hydraulic conductivity reduces leaf water potential, initiates stomatal closure and slows leaf expansion in flooded plants of castor oil (Riccinus communis) despite diminished delivery of ABA from the roots to shoots in the xylemsap. Physiol Plant 111:46–54. https://doi.org/10.1034/j.1399-3054.2001.1110107.x
Ezin V, Pena RDL, Ahanchede A (2010) Flooding tolerance of tomato genotypes during vegetative and reproductive stages. Braz J Plant Physiol 22:131–142
FAO (2022) Food and agriculture organization, crop production statistics. http://www.fao.org/faostat/en/#data/QC. Accessed 26 Aug 2022
Folzer H, Dat J, Capelli N, Rieffel D, Badot PM (2006) Response to flooding of sessile oak: an integrative study. Tree Physiol 26:759–766
Gedam PA, Thangasamy A, Shirsat DV, Ghosh S, Bhagat K, Sogam OA, Gupta A, Mahajan V, Soumia P, Salunkhe VN (2021) Screening of onion (Allium cepa L.) genotypes for drought tolerance using physiological and yield based indices through multivariate analysis. Front Plant Sci 12:600371. https://doi.org/10.3389/fpls.2021.600371
Ghanbari M, Arabi M, Kao SC, Obeysekera J, Sweet W (2021) Climate change and changes in compound coastal-riverine flooding hazard along the US coasts. Earths Future 9(5):e2021EF002055. https://doi.org/10.1029/2021EF002055
Ghodke P, Shirsat D, Thangasamy A, Mahajan V, Salunkhe V, Khade Y, Singh M (2018) Effect of water logging stress at specific growth stages in onion crop. Int J Curr Microbiol Applied Sci 7:3438–3448. https://doi.org/10.20546/ijcmas.2018.701.405
Gray SB, Brady SM (2016) Plant developmental responses to climate change. Dev Biol 419(1):64–77. https://doi.org/10.1016/j.ydbio.2016.07.023
Greenway H, Armstrong W, Colmer TD (2006) Conditions leading to high CO2 (〉 5 kPa) in waterlogged–flooded soils and possible effects on root growth and metabolism. Ann Bot 98(1):9–32. https://doi.org/10.1093/aob/mcl076
Haghighi M (2022) Effects of grafting on cucumber growth under flooding stress during 15 days in vegetative stage. J Agric Sci Technol 24(4):873–883
Havir EA, McHale NA (1987) Biochemical and developmental characterization of multiple forms of catalase in tobacco leaves. Plant Physiol 84(2):450–455. https://doi.org/10.1104/pp.84.2.450
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125(1):189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Heuer B (2003) Influence of exogenous application of proline and glycinebetaine on growth of salt-stressed tomato plants. Plant Sci 165(4):693–699. https://doi.org/10.1016/S0168-9452(03)00222-X
Jackson MB, Ishizawa K, Ito O (2009) Evolution and mechanisms of plant tolerance to flooding stress. Ann Bot 103(2):137–142. https://doi.org/10.1093/aob/mcn242
Kayak N, Kal Ü, Dal Y, Yavuz D, Seymen M (2022) Do proline and glycine betaine mitigate the adverse effects of water stress in spinach? Gesunde Pflanz. https://doi.org/10.1007/s10343-022-00675-6
Kishor PBK, Sangam S, Amrutha RN, Laxmi PS, Naidu KR, Rao KRSS, Rao S, Reddy KJ, Theriappan P, Sreenivasulu N (2005) Regulation of proline biosynthesis, degradation, uptakeand transport in higher plants: its implications in plant growth and abiotic stress tolerance. Curr Sci 88(3):424–438
Liang D, Ni Z, Xia H, Xie Y, Lv X, Wang J, Lin L, Deng Q, Luo X (2019) Exogenous melatonin promotes biomass accumulation and photosynthesis of kiwifruit seedlings under drought stress. Sci Hortic 246:34–43. https://doi.org/10.1016/j.scienta.2018.10.058
Lichtenthaler H, Buschmann C (2001) Chlorophylls and Carotenoids: measurement and characterization by UV-VIS spectroscopy. Curr Protoc Food Anal Chem. https://doi.org/10.1002/0471142913.faf0403s01
Lutts S, Kinet J, Bouharmont J (1996) NaCl-induced senescence in leaves of rice (Oryza sativa L.) cultivars differing in salinity resistance. Ann Bot 78(3):389–398. https://doi.org/10.1006/anbo.1996.0134
Ma J, Rukh G, Ruan Z, Xie X, Ye Z, Liu D (2022) Effects of hypoxia stress on growth, root respiration, and metabolism of phyllostachys praecox. Life 12(6):808. https://doi.org/10.3390/life12060808
Martín-Rodríguez AJ (2022) Respiration-induced biofilm formation as a driver for bacterial niche colonization. Trends Microbiol. https://doi.org/10.1016/j.tim.2022.08.007
Martínez-Arias C, Witzell J, Solla A, Martin JA, Rodríguez-Calcerrada J (2022) Beneficial and pathogenic plant-microbe interactions during flooding stress. Plant Cell Environ 45:2875–2897. https://doi.org/10.1111/pce.14403
Mirza M (2011) Climate change, flooding in South Asia and implications. Reg Environ Change 11(1):95–107. https://doi.org/10.1007/s10113-010-0184-7
Mohammadi SA, Prasanna BM (2003) Analysis of genetic diversity in crop plants salient statistical tools and considerations. Crop Sci 43(4):1235–1248. https://doi.org/10.2135/cropsci2003.1235
Osman HS (2015) Enhancing antioxidant–yield relationship of pea plant under drought at different growth stages by exogenously applied glycine betaine and proline. Ann Agric Sci 60(2):389–402. https://doi.org/10.1016/j.aoas.2015.10.004
Parent C, Berger A, Folzer H, Dat J, Crevecoeur M, Badot P‑M, Capelli N (2008) A novel nonsymbiotic hemoglobin from oak: Cellular and tissue specificity of gene expression. New Phytol 177:142–154. https://doi.org/10.1111/j.1469-8137.2007.02250.x
Patel PK, Singh A, Tripathi N, Yadav D, Hemantaranjan A (2014) Flooding: abiotic constraint limiting vegetable productivity. Adv Plants Agric Res 1(3):96–103. https://doi.org/10.15406/apar.2014.01.00016
Ponting J, Kelly TJ, Verhoef A, Watts MJ, Sizmur T (2021) The impact of increased flooding occurrence on the mobility of potentially toxic elements in floodplain soil—A review. Sci Total Environ 754:142040. https://doi.org/10.1016/j.scitotenv.2020.142040
Puengsungwan S, Jirasereeamornkul K (2020) IoT based root stress detection for lettuce culture using infrared leaf temperature sensor and light intensity sensor. Wirel Pers Commun 115(4):3215–3233
Qamer Z, Chaudhary MT, Du X, Hınze L, Azhar MT (2021) Review of oxidative stress and antioxidative defense mechanisms in Gossypium hirsutum L. in response to extreme abiotic conditions. J Cotton Res 4(1):1–9. https://doi.org/10.1186/s42397-021-00086-4
Radmann EB, Klumb EK, Deuner S, Bianchi VJ (2018) Antioxidant capacity in leaf and root tissues of prunus spp under flooding. J Exp Agric Int. https://doi.org/10.9734/JEAI/2018/43650
Rao R, Li Y (2003) Management of flooding effects on growth of vegetable and selected field crops. HortTechnology 13(4):610. https://doi.org/10.21273/HORTTECH.13.4.0610
Rasheed R, Iqbal M, Ashraf MA, Hussain I, Shafiq F, Yousaf A, Zaheer A (2018) Glycine betaine counteracts the inhibitory effects of waterlogging on growth, photosynthetic pigments, oxidative defence system, nutrient composition, and fruit quality in tomato. J Hortic Sci Biotechnol 93(4):385–391. https://doi.org/10.1080/14620316.2017.1373037
Rockström J, Williams J, Daily G, Noble A, Matthews N, Gordon L, Wetterstrand H, DeClerck F, Shah M, Steduto P (2017) Sustainable intensification of agriculture for human prosperity and global sustainability. Ambio 46(1):4–17. https://doi.org/10.1007/s13280-016-0793-6
Seymen M (2021) How does the flooding stress occurring in different harvest times affect the morpho-physiological and biochemical characteristics of spinach? Sci Hortic 275:109713. https://doi.org/10.1016/j.scienta.2020.109713
Seymen M, Yavuz D, Dursun A, Kurtar ES, Türkmen Ö (2019) Identification of drought-tolerant pumpkin (Cucurbita pepo L.) genotypes associated with certain fruit characteristics, seed yield, and quality. Agric Water Manag 221:150–159. https://doi.org/10.1016/j.agwat.2019.05.009
Sharma S, Villamor JG, Verslues PE (2011) Essential role of tissue-specific proline synthesis and catabolism in growth and redox balance at low water potential. Plant Physiol 157(1):292–304. https://doi.org/10.1104/pp.111.183210
Srinivas V, Balasubramanian D (1995) Proline is a protein-compatible hydrotrope. Langmuir 11:2830–2833. https://doi.org/10.1021/la00007a077
Teklić T, Parađiković N, Špoljarević M, Zeljković S, Lončarić Z, Lisjak M (2021) Linking abiotic stress, plant metabolites, biostimulants and functional food. Ann Appl Biol 178(2):169–191. https://doi.org/10.1111/aab.12651
Valenzuela-Soto EM, Figueroa-Soto CG (2019) Biosynthesis and degradation of glycine betaine and its potential to control plant growth and development. In: Osmoprotectant-mediated abiotic stress tolerance in plants. Springer, Cham, pp 123–140 https://doi.org/10.1007/978-3-030-27423-8_5
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants protective role of exogenous polyamines. Plant Sci 151:59–66. https://doi.org/10.1016/S0168–9452(99)00197–1
Witham FH, Blaydes DF, Devlin RM (1971) Experiments in plant physiology. Van Nostrand Reinhold Compan, New York, pp 55–56
Yavuz D, Seymen M, Yavuz N, Çoklar H, Ercan M (2021) Effects of water stress applied at various phenological stages on yield, quality, and water use efficiency of melon. Agric Water Manag 246:106673. https://doi.org/10.1016/j.agwat.2020.106673
Yavuz D, Kılıç E, Seymen M, Dal Y, Kayak N, Kal Ü, Yavuz N (2022) The effect of irrigation water salinity on the morph-physiological and biochemical properties of spinach under deficit irrigation conditions. Sci Hortic 304:111272. https://doi.org/10.1016/j.scienta.2022.111272
Yiu JC, Juang LD, Fang DYT, Liu CW, Wu SJ (2009) Exogenous putrescine reduces flooding-induced oxidative damage by increasing the antioxidant properties of Welsh onion. Sci Hortic 120(3):306–314. https://doi.org/10.1016/j.scienta.2008.11.020
Yordanova R, Uzunova A, Popova L (2005) Effects of short-term soil flooding on stomata behaviour and leaf gas exchange in barley plants. Biol Plant 49(2):317–319
Zhang J, Zhang X (1994) Can early wilting of old leaves account for much of the ABA accumulation in flooded pea plants? J Exp Bot 45(9):1335–1342
Zhou P, Qian J, Yuan W, Yang X, Di B, Meng Y, Shao J (2021) Effects of interval flooding stress on physiological characteristics of apple leaves. Horticulturae 7(10):331. https://doi.org/10.3390/horticulturae7100331
Zouari M, Elloumi N, Labrousse P, Rouina BB, Abdallah FB, Ahmed CB (2018) Olive trees response to lead stress: exogenous proline provided better tolerance than glycine betaine. S Afr J Bot 118:158–165. https://doi.org/10.1016/j.sajb.2018.07.008
Acknowledgements
This research was funded by the SÜ. BAP (Project No. 22201006) and is part of the Master’s thesis written by Abdullah Şamil Şahin.
Author information
Authors and Affiliations
Contributions
M. Seymen writing-original draft, data curation, investigation, methodology, validation, A. Ş. Şahin formal analysis, data curation, Ö. B. Tanrıverdi data curation.
Corresponding author
Ethics declarations
Conflict of interest
M. Seymen, A. Ş. Şahin and Ö. B. Tanrıverdi declare that they have no competing interests.
Rights and permissions
Springer Nature oder sein Lizenzgeber (z.B. eine Gesellschaft oder ein*e andere*r Vertragspartner*in) hält die ausschließlichen Nutzungsrechte an diesem Artikel kraft eines Verlagsvertrags mit dem/den Autor*in(nen) oder anderen Rechteinhaber*in(nen); die Selbstarchivierung der akzeptierten Manuskriptversion dieses Artikels durch Autor*in(nen) unterliegt ausschließlich den Bedingungen dieses Verlagsvertrags und dem geltenden Recht.
About this article
Cite this article
Seymen, M., Şahin, A.Ş. & Tanrıverdi, Ö.B. Mitigation Effects of Proline and Glycine Betaine to Green Onion Under Flooding Stress. Gesunde Pflanzen 75, 1639–1652 (2023). https://doi.org/10.1007/s10343-023-00854-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10343-023-00854-z