Abstract
Chiari malformation (CM) and basilar invagination (BI) are mesodermal malformations with disproportion between the content and volume of posterior fossa capacity and overcrowding of neural structures at the level of foramen magnum. Several alternatives for posterior approaches are available, including extradural (ED), extra-arachnoidal, and intradural (ID) approaches. The objectives are to evaluate the effect of several surgical techniques for posterior fossa decompression (PFD) in the outcomes of patients with CCJM and to evaluate complications in the techniques reported. A systematic review of the literature on the effects of PFD surgery was performed using the MEDLINE (via PubMed) database and the Cochrane Central Register of Controlled Trials. The PRISMA statement and MOOSE recommendations were followed. Five hundred and thirty-nine (539) articles were initially selected by publication title. After abstract analysis, 70 articles were selected for full-text analysis, and 43 were excluded. Ultimately, 27 studies were evaluated. The success rate (SR) with ED techniques was 0.76 versus 0.81 in EA technique and 0.83 in IA technique. All posterior fossa decompression techniques were very successful. Results from observational studies were similar to that of the randomized trial. The main complications were CSF fistulas, most common in patients with IA approach. The overall mortality rate was 1%.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Chiari malformation (CM) and basilar invagination (BI), also known as adult craniocervical junction malformations (CCJMs), are mesodermic malformations characterized by disproportion between the content and capacity of the posterior fossa, consequently leading to overcrowding of neural structures at the foramen magnum. In BI, horizontal clivus, flattening of the clivus-canal angle, and anterior brainstem compression also occur [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20].
There are several controversies regarding patient selection, outcomes, risks, and complications of surgical approaches to CCJMs [21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40]. Some authors advocate the posterior approach as the route of choice, while others state that an exclusively posterior approach may be associated with neurological deterioration, and propose that the anterior approach should be preferred for first-line treatment [41].
Several alternatives for posterior approaches are available, including extradural (ED), extra-arachnoidal, and intradural (ID) approaches, with and without tonsillectomy and duraplasty [42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69].
The aim of this study is to evaluate the effects and complications of several surgical techniques for posterior fossa decompression (PFD) on the outcomes of patients with CCJM.
Methods
A systematic review of the literature on the effects of PFD surgery was performed using MEDLINE (via PubMed) and the Cochrane Central Register of Controlled Trials.
This review was done under PRISMA statement and MOOSE recommendations.
Structure of the search strategy
The literature search was based on structured questions, which defined the criteria for inclusion of papers, based on types of participants (patients), intervention and control, follow-up, and types of studies, following the PICOT strategy.
Types of participants
Only adult patients, of both genders, with CCJM (basilar invagination and/or Chiari malformation) who underwent posterior fossa decompression (PFD) were evaluated. Acquired CCJM and unstable BI patients who underwent only craniocervical fusion were excluded, as were those who received any kind of shunt.
Types of intervention/exposure
Any kind of posterior fossa decompression in CCJM was evaluated in this study.
Control
Results of different techniques were compared.
Outcomes
The outcomes of interest were the rates of surgical success, complications, and mortality.
Types of study
We aimed to include clinical trials of all designs, from the highest level of evidence from randomized trials (if available) to cohorts. Series of fewer than 10 cases, technical reports, discussions, points of view, and reviews were excluded.
Language
Articles published in English, Portuguese, French, and Spanish from 1964 to 2016 were considered for analysis.
Analysis of risk of bias
The risk of bias was analyzed in accordance with the MOOSE recommendations for observational studies and according to the Cochrane Collaboration recommendations for randomized studies.
Search strategies
Electronic database searches were performed by two authors independently, and disagreements were resolved by discussion.
The used search strategy is detailed below:
-
1.
(“Arnold-Chiari Malformation” [Mesh] OR “Chiari Malformation” [All Fields] OR “Platybasia” [Mesh]) OR “basilar invagination” [All Fields] AND “Decompression, Surgical” [Mesh]
-
2.
(“Chiari Malformation” [All Fields] OR “basilar invagination” [All Fields]) AND (“posterior fossa decompression” [All Fields] OR (“decompression” [MeSH Terms] OR “decompression” [All Fields]) OR (“surgery” [Subheading] OR “surgery” [All Fields] OR “surgical procedures, operative” [MeSH Terms] OR (“surgical” [All Fields] AND “procedures” [All Fields] AND “operative” [All Fields]) OR “operative surgical procedures” [All Fields] OR “surgery” [All Fields] OR “general surgery” [MeSH Terms] OR (“general” [All Fields] AND “surgery” [All Fields]) OR “general surgery” [All Fields])).
Statistics
Results were described as event rates (success rate, SR), confidence interval (95% CI), and Z and p values. Meta-analysis was performed in R (www.r-project.org). Heterogeneity was described by the I-squared (I 2) and tau-squared (τ 2) statistics, using a random effects model.
When applicable, chi-square tests were used to compare complication rates among different groups.
Data extraction
The following surgical techniques were evaluated: extradural, extra-arachnoidal, and intra-arachnoidal techniques.
Unconventional techniques such as tonsillectomy without duraplasty, expansive cranioplasty, obex obstruction, shunting, endoscopic posterior fossa decompression, durotomy according to intraoperative ultrasound findings, and others were described separately, according to their relevance to the scope of this review [15, 35, 52, 55, 57, 60].
Complications were described stratified by surgical technique. All studies describing complications were evaluated.
Definition of success and failure rate
Patients who had clinical improvement (partial or complete) at late follow-up, according to the original author’s discretion, were defined as successes. Failure was defined as loss of clinical improvement or deterioration of symptoms.
Reoperations were not taken into account, and only patients with primary surgeries were analyzed.
Results
Search
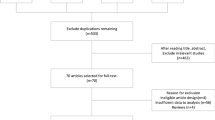
The initial electronic search identified 908 papers. A further 12 articles were obtained by cross-reference search, and 3 additional studies were obtained by the authors’ knowledge. Three hundred and eighty-four (384) duplicate entries were excluded. Five hundred and thirty-nine titles (539) were selected by title and their abstracts were analyzed. Seventy papers were in full evaluated. Of these, 43 papers were excluded and 27 papers were ultimately analyzed (Fig. 1).
We contacted the authors of five studies to clarify technical details and outcome data. Three responded [6, 21, 55], while contact attempts with the other two [28, 53] were unsuccessful.
Description of included studies
Nine of the included papers were described as comparative studies [7, 21, 22, 26, 29, 40, 52, 67]. We added the results of each study to the arm corresponding to its respective surgical technique, for pooled analysis of the results of cohort papers describing the same methods. A study by El-Ghandour [21] was excluded from the pooled analysis because results were not stratified by specific technique. Only one of the included studies was a randomized trial [22].
Randomized study
Erdogan et al. [22] compared the ED approach versus an intra-arachnoid (IA) approach with duraplasty in 27 patients. The success rate was 83.3% (N = 12) in the ED group and 73.3% (N = 15) in the IA group. There were two CSF fistulas. Of the 12 patients in the ED group, four exhibited syringomyelia; all experienced improvement in syringomyelia score on imaging and clinically. Of the 15 patients in the IA group, 11 had syringomyelia. The proportion of syrinx regression was 28 ± 10% in the ED group and 36 ± 33% in the IA group. Statistical significance was found for both groups in the postoperative period (p < 0.05) [22].
Studies by surgical technique
Extradural approach
Randomized study
The success rate in the extradural group of the Erdogan study was 83.3% (10 of 12), 95% CI = 0.52–0.95 [22].
Observational studies
Six studies evaluated ED techniques in patients both with and without syringomyelia. Two studies used the extradural technique with dural splitting (DS) [50, 69]. Four performed extradural decompression without DS. The results of Khan et al. [30] and El-Ghandour [21] were excluded from pooled analysis because both were restricted to patients without syringomyelia. Data from these studies are described under the “Excluded studies” heading.
Quon et al. [49] performed a retrospective analysis of the ED approach in 18 patients. The success rate was 88.8%, and there was no mortality. One patient experienced dysphagia and dysphonia after surgery. Two patients with syringomyelia improved clinically and on imaging [50].
Zhang et al. [69] retrospectively studied 316 patients with CM, of whom 236 had syringomyelia. The surgical technique was DS with possibility of extension of laminectomy to C4 if necessary to treat syringomyelia.
Munshi et al. [40] evaluated 34 patients. The ED technique was used in 11. Of seven who had syringomyelia, five improved, including three with a decrease in cavity size; in the remaining two, there was no improvement, and the cavity size actually increased. Of the four patients who did not have syringomyelia, three improved. Overall, the success rate was 73% (8 of 11 patients), and there was no mortality. Regarding complications, superficial wound infection occurred in 9% of patients operated extradurally.
Yilmaz et al. [67] evaluated 82 patients, of whom 24 were operated extradurally. The success rate in this group was 79.1% (19 of 24).
Guyotat et al. [26] evaluated 75 patients who underwent PFD. Forty-two patients were treated with ED technique alone, with a success rate of 64.8%.
Effect summary (Fig. 2): The ED-only group included 256 patients, with a success rate of 75% (0.6–0.86) (heterogeneity: I 2 = 75.8%, τ 2 = 0.4174, p = 0.0024).
Extra-arachnoid approach with duraplasty
Fourteen studies evaluated patients who underwent extra-arachnoidal decompression.
Noudel et al. [44] studied 11 adult patients who were operated via the extra-arachnoid (EA) technique. All improved (100%), with complete improvement in four cases and partial improvement in seven. No complications were reported over a follow-up time of 45 months.
Wetjen et al. [65] prospectively studied 29 cases treated via an EA approach. Twenty-eight patients (96.5%) had improved after 1 year, with no complications reported.
Di Lorenzo et al. [17] evaluated 20 cases treated with duraplasty over 2.4 years of follow-up. Mortality and morbidity were absent. Eight patients improved, 11 achieved stable disease, and one deteriorated.
Salunke et al. [55] evaluated 15 adult patients with CM and high-intensity ventral brainstem compression in the absence of BI. All underwent posterior decompression via an EA technique. All patients improved after surgery, although one worsened 6 months after initial improvement. The success rate was 93.33%.
Saceda-Gutiérrez et al. [51] evaluated 36 cases using EA techniques. Outcomes were classified as excellent in 55%, good in 29%, and poor (worse than baseline) in 16%. The overall success rate was 84%. There were six cases of postoperative CSF fistula.
Deng et al. [15] evaluated 38 patients with CM and ventricular dilatation who underwent PFD with EA techniques. Symptoms improved in 33 patients (86.8%).
Spena et al. [59] retrospectively studied 36 patients treated via the EA route with duraplasty. The success rate was 80.5%, and the complication rate was 8.1%.
Munshi et al. [40] evaluated 34 patients, of whom 23 were treated via EA techniques. The success rate was 87%, and there was no mortality; 4.1% experienced minor complications.
Yilmaz et al. [67] evaluated 82 patients, of whom 58 were in the EA group. In this arm, 52 patients improved (89.1%), of whom 45 (77.5%) had syringomyelia. On follow-up, 41 patients (91.1%) had a reduction in size of syringomyelia and 37 (90.2%) had improved clinically.
Kunert et al. [35] studied 38 patients divided into two groups. In the EA group (N = 17), 82% experienced improvement. There were nine complications in 21 patients in the IA group (five cases of CSF fistula and one of meningitis) and no complications in the EA group.
Lee et al. [36] studied 25 patients treated via EA approaches. In this group, the improvement rate was 80%. Overall, 24% experienced complications (three cases of transient headaches, one of transient weakness in the lower limbs, and two infections).
Parker et al. [46] analyzed 50 cases in a retrospective manner and reported 100% improvement, 18% complications, and no mortality.
Sahuquillo et al. [52] evaluated 20 subjects. In the EA group, the success rate was 80%. In the IA group (historic series), success was only 20%. There was one case of meningitis in the EA group. Due to the historic design of the IA group, we excluded it from analysis.
Kalb et al. [29] evaluated 104 patients. The dura was opened in 101; only three were treated via ED approaches. The success rate was 66.7% in the ED group, 77% in the EA group (N = 38), and 79% in the IA group (N = 63), for an overall success rate of 69.6%. All complications occurred in patients who had undergone duraplasty (six infections, one CSF fistula, one aseptic meningitis, and one pseudomeningocele). Overall, 32 patients in the sample had syringomyelia. Of the three patients in the ED group, two improved and one deteriorated and was subsequently operated by the IA technique, with clinical and radiological improvement.
Effect summary (Fig. 2): The pooled data analysis evaluated 14 studies (407 patients) using extra-arachnoidal techniques. The overall success rate was 84% (0.76–0.89) (heterogeneity: I 2 = 59.4%, τ 2 = 0.4561, p = 0.0024).
Intra-arachnoid technique with duraplasty
Randomized study
The success rate in the Erdogan [22] IA group was 0.73% (95% CI = 0.46–0.9).
Nonrandomized studies
Twelve papers evaluated 594 patients who underwent IA approaches with duraplasty.
Alfieri et al. [1] prospectively and retrospectively evaluated 109 cases of IA duraplasty with tonsillectomy, with a 90% success rate and an 11% complication rate (CSF fistula was the most common, occurring in 2.7% of patients). There was no mortality.
Prat and Galeano [47] analyzed 13 cases in a prospective manner. Overall, 84.61% of patients experienced improvement (85.71% in syringomyelia). Complications and mortality were not described.
da Silva et al. [13, 14] prospectively analyzed 10 cases treated via IA with tonsillectomy. Improvement in all cases was reported, with no description of complications or mortality.
Depreitere et al. [16] retrospectively analyzed 22 cases operated via IA with tonsillectomy. On initial follow-up, 16 patients (76%) were improved. In late follow-up, the success rate was 68%.
Mueller and Oro [39] evaluated quality of life (QoL) before and after surgery in 172 patients with CM with and without syringomyelia, all treated via an IA technique with duraplasty. Improvement was observed in 84% of patients. Among 16 participants who had deterioration of QoL, external factors not related to surgery were implicated. The complications were occipital neuralgia in 2.6%, superficial infection in 1.8%, and one case of arachnoid cyst.
Badie et al. [4] retrospectively evaluated 20 cases treated via an IA technique with duraplasty and tonsillectomy. The success rate was 85%, with no mortality. Complications were not described.
Zhang et al. [68] analyzed 132 patients in a comparison of small and large craniectomy using the IA technique. For pooled analysis, we used data from both groups in the same technique. Of the 132 patients, 106 improved, 21 achieved stable disease, and 5 deteriorated. Stratified by groups, 54 patients in the large craniotomy group improved, 10 became stable, and 5 worsened, versus 52 improved and 11 stabilized in the small craniotomy group. Only three patients (4.76%) experienced complications in the small craniotomy group (one CSF fistula, one aseptic meningitis, and one case of craniocervical instability). In the large craniotomy, 49.27% of patients experienced complications, with CSF fistula being most frequent.
Alzate et al. [2] analyzed 66 patients who underwent IA, with tonsillectomy performed in most cases. Of 34 patients with syringomyelia, 22 received shunts and all improved clinically and on imaging, with only one reoperation due to shunt migration. All 12 patients with syringomyelia who did not receive shunts improved. Long-term results were excellent in 82% and good in 18%. There was no mortality, and complications were not described.
Liang et al. [37] evaluated 21 patients, of whom 15 had syringomyelia. The success rate was 90.4%, and follow-up ranged from 6 months to 3 years. The authors described no mortality. The complication rate was 9.5% (two cases of systemic infection treated successfully with intravenous antibiotics). The infection site was not specified.
Pritz [48] evaluated 14 patients (eight with syringomyelia) operated via the IA technique. The author used pericranial grafts and observed improvement in 100% of patients, without complications. Follow-up time ranged from 4 months to 3 years.
Effect summary (Fig. 2): 12 studies were evaluated in 689 patients and the success rate was 85% (0.80–0.89) (heterogeneity: I 2 = 47.1%, τ 2 = 0.1613, p = 0.0356).
Evaluation of craniotomy size
Only one study compared outcomes according to craniotomy size.
Zhang et al. [68] retrospectively analyzed 132 patients in a comparison of small and large craniectomy with IA technique. As noted above, the success rate was 78.2% in the large craniotomy group, with nearly half of patients developing complications, the most common of which was CSF fistula (11.5%), whereas in the small craniotomy group, the success rate was 82.5% with only three cases of complications. This study demonstrated the superiority of smaller craniotomies. While the success rate was not significantly different between sizes (χ 2 = 32.346, p = 0.53), the complication rate was significantly lower in the small craniotomy group (p < 0.0001, [68]).
Other techniques
Studies which applied unique or otherwise distinct techniques are described separately below.
Takayasu et al. [60] analyzed 16 patients who underwent cranioplasty (with titanium miniplates) and duraplasty, without arachnoid opening. The success rate was 93.75%.
Kunert et al. [35] described the retrospective results of 38 patients divided into two groups: one received an IA technique and tonsillectomy without duraplasty (N = 21) and the other group an EA technique (N = 17). In the IA group, 58% of patients improved and 33.3% experienced symptom progression. As duraplasty was not performed in the IA group, the results were not pooled for analysis.
Sakamoto et al. [53] developed a method of expansive suboccipital cranioplasty to enlarge the posterior fossa and improve CSF flow and tested it in 20 patients. All experienced improved CSF flow. Another 20 patients were treated with obex occlusion, with an 80% success rate, one case of temporary improvement with late arachnoid adhesions, and three patients with stable disease, although syringomyelia decreased on postoperative MRI.
Sindou et al. [57] studied 44 patients (15 with syringomyelia) treated with an EA technique via a far lateral approach. Improvement was observed in 37 patients (84.1%), 13 of whom had syringomyelia. The mean follow-up was 4 years. Some minor complications were reported.
Risk of bias
Analysis of the risk of bias was conducted separately for each type of publication.
Risk of bias in the randomized trial
Erdogan et al. [22] compared the extradural approach versus the intra-arachnoid technique and duraplasty in a sample of 27 patients. The authors mention that the choice of technique was done randomly, but do not describe the randomization process. However, their patient histories were collected through a chart review, which is a common practice in retrospective studies. The differences between baseline characteristics of the groups were described in tables but not statistically compared. There were no attempts at allocation concealment, and the authors do not state who collected the results. This suggests that there was no blinding for outcome assessment. There was no reported loss of patient data. There was no evidence of selective outcome reporting [22].
Risk of bias of observational studies (MOOSE recommendation)
All publications included above, except for the Erdogan trial [22], were noncomparative observational studies of heterogeneous patient cohorts. For these publications, we analyzed the results of each group stratified by the surgical technique employed.
In the EA technique group, Noudel, Wetjen, Di Lorenzo, Salunke, Saceda-Gutiérrez, Deng, and Spena described cohorts of patients who underwent the same surgical procedure, although there was substantial heterogeneity in clinical pictures and imaging findings, including different percentages of patients with syringomyelia. Munshi, Yilmaz, Kunert, and Lee described cohorts of patients who underwent different surgical procedures; only those in whom EA techniques were performed were taken into account for analysis.
In the IA with duraplasty group, all authors described the same surgical procedure. In the ED technique group, only one study (Quon) involved the ED approach with DS exclusively. The other authors performed both ED and other techniques; therefore, only the ED arms of these studies were considered in the pooled analysis.
All studies selected for pooled analysis described the type of intervention and the outcome measure evaluated in this study (success rate). Studies which did not describe the above data were excluded from evaluation.
Length of follow-up varied widely, from 0.5 to 152 months (mean, 31.3 months). None of the studies reported significant loss to follow-up.
ED technique and syringomyelia
In studies of the ED technique, we evaluated the outcomes of patients with syringomyelia and, when possible, in comparative studies, compared the results of cases with and without duraplasty. There were five comparative studies of extradural techniques (Erdogan, Munshi, Bond, Yilmaz, and Zhang) and one noncomparative study (Quon).
The Zhang et al. [69] study was excluded from analysis because it pooled results of patients with and without syringomyelia, while the El-Ghandour study [21] reported the results of 14 cases operated via the ED route, all without syringomyelia.
Overall, 67 patients underwent ED techniques (34 with syringomyelia), with a success rate of 76.47%. Among the 112 patients operated with duraplasty (EA and IA), an improvement rate of 85.9% was observed. However, only 64 of these patients were considered, as 12 were lost to imaging follow-up.
Studies excluded from pooled analysis and the respective reasons for exclusion are described in detail in Table 1.
Complications
Among the 64 studies evaluated, 14 did not describe complications at all (Badie, Sakamoto, Ono, Yilmaz, Kumar, Hekman, Sakushima, Alzate, Wetjen, Cui, Tisell, Prat, da Silva, Heiss). Three studies described complications, but either did not provide detailed analyses of complications with different techniques or described complications in a pooled fashion (Ramnarayam, Klekamp, Zhang), while four reported absence of complications among their patient cohorts (Noudel, Deng, Chauvet, Dilorenzo).
Some papers, although not used for pooled outcomes evaluation, did report analyses of complications (Botelho, da Silva, Takayasu, Batzdorf, Khan, da Silva, Arruda, Bond).
Overall, there were 195 complications among 1451 patients (13.5%). The most frequent complications were CSF fistula (N = 63, 4.3%), meningitis (N = 17, 1.2%), and infection (N = 13, 0.92%). Other complications are reported in Table 2. There were 15 deaths (overall mortality 1%), 13 of which were reported in a single cohort (da Silva) [13].
The complication rate did not differ significantly across the three techniques (15.9 vs. 9.4 vs. 7.9%; χ 2 = 3.25; p = 0.19) (Table 2). The majority of deaths were reported in a single study (13 of 15); in all other cohorts, the mortality rate was <1% (Table 2).
Postoperative neurological deterioration
Zileli and Cagli [71] described nine patients who underwent transoral odontoidectomy. One of these had deteriorated rapidly after previous PFD. Klekamp [33] reported late postoperative neurological deterioration in three patients, two cases due to hardware failure and one due to craniocervical instability.
Discussion
Treatment of patients with CCJM aims to restore the normal cerebrospinal fluid (CSF) dynamics and decompress neural structures in the craniocervical junction [1,2,3,4,5,6,7,8,9,10].
Novel methods for meta-analysis of observational studies have been developed recently. Meta-analysis has the ability to incorporate the heterogeneity of primary studies on the effect summary, and is often the only way to reveal the effect size and effect direction of an intervention [22].
Although randomized trials are the gold-standard design for meta-analysis, when such data are lacking, observational studies can provide the best available evidence.
There are several proposed techniques for PFD, but three are most often described and employed, from extradural bone-only decompression to intra-arachnoid tonsillar resection. There is still controversy regarding the safety of PFD for all cases of BI and CM, and some authors propose an anterior approach for selected cases [20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43]. Until recently, the diagnostic criteria for BI were very poorly defined, and authors did not distinguish clearly between CM and BI.
Our review identified only one paper described as a randomized trial, but with several methodological weaknesses compared to current high-quality randomized designs [22]. Thus, we included randomized and cohort observational studies.
The success rates of the three decompression techniques were not statistically different. Additionally, the success rate of the sole randomized trial was similar to those reported in the cohort studies.
Patients with CCJM may also have syringomyelia, which is associated with poorer outcomes and signals a more aggressive disease. Among patients with syringomyelia who underwent surgery with ED techniques, 76% exhibited improvement in MRI findings, a rate lower than in the group that underwent duraplasty (84%), perhaps reflecting greater change in CSF dynamics in patients undergoing duraplasty at the level of the craniocervical junction.
The choice of approach in patients with significant ventral compression is controversial, as is the need for fusion. Salunke evaluated 15 adult patients with CM and significant cervical spinal cord ventral compression in the absence of BI [54]. Fifteen patients underwent posterior decompression. All patients experienced improvement of cervicomedullary compression symptoms after surgery. Only one patient later deteriorated, 6 months after initial improvement.
There were 195 complications among 1451 patients in the studies included in the present review (13.5%). The most frequent complications were CSF fistula and infection, especially in patients who underwent IA techniques.
Although there were no significant differences in overall complication rate across the three techniques, CSF fistula only occurred when the dura was opened. Fistula occurred in 4.1% of patients who underwent the EA technique, versus 4.9% with the IA technique. CSF leakage predisposes to meningitis and pseudomeningocele, which are severe and potentially harmful complications.
Although there were 15 deaths, most were reported in only one study. Overall analysis revealed a low mortality rate (0.1%) (Table 2).
When analyzing acute postoperative decompensations after PDF, Dyste et al. [20] studied a sample of 72 patients, nine of whom were treated by the anterior approach after acute decompensation (in four patients) or late decompensation (in five patients) post-PFD. Patient age ranged from 6 to 82 years, and 29 were children. The sample was too heterogeneous to allow any conclusions to be drawn.
Zileli and Cagli [71] described nine patients who underwent transoral odontoidectomy. One of these had deteriorated rapidly after previous posterior decompressive surgery.
Only one paper addressed the role of craniotomy size [68]. The success rate was 78.2% in the large craniotomy group, with 49.27% of patients experiencing complications (most often CSF fistulae, which occurred in 11.5%). In patients undergoing small craniotomy, the success rate was 82.5%, with only 4.76% experiencing complications [68]. Thus, we could not draw any conclusion regarding the size of craniotomy and the effect of decompression.
In a recent systematic review, Zhao et al. [70] presented data regarding PFD in adult and pediatric populations. The authors also included manuscripts which used imaging modalities other than MRI. Eighteen papers served as the basis for their pooled analysis [70]. Conversely, we considered that adult and pediatric patients with CM and BI are very different and, thus, should ideally be analyzed separately. Additionally, we only included patients with an MRI-based diagnosis. We were able to perform a pooled analysis of 27 studies.
Limitations of the study
There was only one randomized trial and several observational (cohort) studies. The randomized trial had several methodological weaknesses. Thus, the review did not reach a sufficient level of evidence to recommend one surgical technique over another, but only to describe them as options and guide future work. The most significant weakness of this manuscript reflects the weakness of the level of evidence of the published trials. However, malformations of the craniocervical transition are relatively rare diseases, and the literature has not yet reached the level of organization sufficient to conduct multicenter studies.
Conclusions
PFD was efficient to alleviate symptoms in patients with CCJM. No significant difference in overall success rate among evaluated techniques could be disclosed by this review, but the extradural technique was not frequently suited. Postoperative neurological deterioration after PFD was described in only two patients.
The main complication was CSF leakage (4.3%). Overall mortality was low, at 1% overall (15 patients), and largely accounted by a single cohort. Randomized trials are needed to confirm observational data.
References
Alfieri A, Pinna G (2012) Long-term results after posterior fossa decompression in syringomyelia with adult Chiari type I malformation. J Neurosurg Spine 17:381–387
Alzate J, Kothbauer K, Jallo G, Epstein F (2001) Treatment of Chiari type I malformation in patients with and without syringomyelia: a consecutive series of 66 cases. Neurosurg Focus 11(1):E3
Arruda JA, Costa CM, Tella OI Jr (2004) Results of the treatment of syruingomyelia associated with Chiari malformation: analysis of 60 cases. Arq Neuropsiquiatr 62(2):237–244
Badie B, Mendoza D, Batzdorf U (1995) Posterior fossa volume and response to suboccipital decompression in patients with Chiari malformation. Neurosurgery 37(2):214–218
Batzdorf U, McArthur DL, Bentson JR (2013) Surgical treatment of Chari malformation with and without syringomyelia: experience with 177 adult patients. J Neurosurg 118(2):232–242
Blagodatsky MD, Larionov SN, Alexandrov YA, Velm AI (1999) Surgical treatment of Chiari I malformation with or without syringomyelia. Acta Neurochir 141(9):963–968
Bond A, Jane J, Liu K, Oldfield E (2015) Changes in cerebrospinal fluid flow assessed using intraoperative MRI during posterior fossa decompression for Chiari malformation. J Neurosurg 122:1068–1075
Botelho RV, Bittencourt LR, Rotta JM, Tufik S (2010) The effects of posterior fossa decompressive surgery in adult patients with Chiari malformation and sleep apnea. J Neurosurg 112:800–807
Chauvet D, Carpentier A, George B (2009) Dura splitting decompression in Chiari type 1 malformation: clinical experience and radiological findings. Neurosurg Rev 32(4):465–470
Chotai S, Kshettry VR, Lamki T, Ammirati M (2014) Surgical outcomes using wide suboccipital decompression for adult Chiari I malformation with and without syringomyelia. Clin Neurol Neurosurg 120:129–135
Coumans JV, Walcott BP, Butler WE, Nahed BV, Kahle KT (2011) Volumetric analysis of syringomyelia following hindbrain decompression for Chiarimalformation type I: syringomyelia resolution follows exponential kinetics. Neurosurg Focus 31(3):E4
Cui LG, Jiang L, Zhang HB, Liu B, Wang JR, Jia JW, Chen W (2011) Monitoring of cerebrospinal fluid flow by intraoperative ultrasound in patientswith Chiari I malformation. Clin Neurol Neurosurg 113(3):173–176
da Silva JA, dos Santos AA Jr, Melo LR, de Araújo AF, Regueira GP (2011) Posterior fossa decompression with tonsillectomy in 104 cases of basilar impression, Chiari malformation and/or syringomyelia. Arq Neuropsiquiatr 69(5):817–823
da Silva JA, Holanda MM (2003) Basilar impression, Chiari malformation and syringomyelia: a retrospective study of 53 surgically treated patients. Arq Neuropsiquiatr 61(2B):368–375
Deng X, Wu L, Yang C, Tong X, Xu Y (2013) Surgical treatment of Chiari I malformation with ventricular dilation. Neurol Med Chir (Tokyo) 53(12):847–852
Depreitere B, Van Calenbergh F, van Loon J, Goffin J, Plets C (2000) Posterior fossa decompression in syringomyelia associated with a Chiari malformation: a retrospective analysis of 22 patients. Clin Neurol Neurosurg 102(2):91–96
Di Lorenzo N, Palma L, Palatinsky E, Fortuna A (1995) “Conservative” craniocervical decompression in the treatment of syringomyelia-Chiari I complex. A prospective study of 20 adult cases. Spine (Phila Pa 1976) 1 20(23):2479–2483
Dones J, De Jesús O, Colen CB (2003) Clinical outcomes in patients with Chiari I malformation: a review of twenty-seven cases. Surg Neurol 60:142–147
Duddy JC, Allcutt D, Crimmins D, O’Brien D, O’Brien DF, Rawluk D, Sattar MT, Young S, Caird J (2014) Foramen magnum decompression for Chiari I malformation: a procedure not to be underestimated. Br J Neurosurg 28(3):330–334
Dyste GN, Menezes AH, VanGilder JC (1989) Symptomatic Chiari malformations. An analysis of presentation, management, and long-term outcome. J Neurosurg 71:159–168
El-Ghandour NM (2012) Long-term outcome of surgical management of adult Chiari I malformation. Neurosurg Rev 35(4):537–546; discussion 546-7
Erdogan E, Cansever T, Secer HI, Temiz C, Sirin S, Kabatas S, Gonul E (2010) The evaluation of surgical treatment options in the Chiari malformation type I. Turk Neurosurg 20(3):303–313
Fenoy AJ, Menezes AH, Fenoy KA (2008) Craniocervical junction fusions in patients with hindbrain herniation and syringohydromyelia. J Neurosurg Spine 9(1):1–9
Gambardella G, Caruso G, Caffo M, Germanò A, La Rosa G, Tomasello F (1998) Transversemicroincisions of the outer layer of the dura mater combined with foramen magnum decompression as treatment for syringomyelia with Chiari I malformation. Acta Neurochir 140(2):134–139
Goel A (2009) Basilar invagination, Chiari malformation, syringomyelia: a review. Neurol India 57(3):235–246
Guyotat J, Bret P, Jouanneau E, Ricci A-C, Lapras C (1998) Syringomyelia associated with type I Chiari malformation. A 21-year retrospective study on 75 cases treated by foramen magnum decompression with a special emphasis on the value of tonsils resection. Acta Neurochir 140:745–754
Heiss JD, Suffredini G, Bakhtian KD, Sarntinoranont M, Oldfield EH (2012) Normalization of hindbrain morphology after decompression of Chiari malformation type I. J Neurosurg 117(5):942–946
Hekman KE, Aliaga L, Straus D, Luther A, Chen J, Sampat A, Frim D (2012) Positive and negative predictors for good outcome after decompressive surgery for Chiari malformation type 1 as scored on the Chicago Chiari Outcome Scale. Neurol Res 34(7):694–700
Kalb S, Perez-Orribo L, Mahan M, Theodore N, Nakaji P, Bristol RE (2012) Evaluation of operative procedures for symptomatic outcome after decompression surgery for Chiari type I malformation. J Clin Neurosci 19(9):1268–1272
Khan MB, Bashir MU, Kumar MBBSR, Bari ME (2015) Surgical treatment of symptomatic Chiari I malformation: a consecutive case series from a developing country. Neurosurg Q 25(1):56–60
Klekamp J (2011) Treatment of basilar invagination. Eur Spine J 23(8):1656–1665
Klekamp J (2015) Chiari I malformation with and without basilar invagination: a comparative study. Neurosurg Focus 38(4):E12
Klekamp J (2012) Neurological deterioration after foramen magnum decompression for Chiari malformation type I: old or new pathology? J Neurosurg Pediatr 10(6):538–547
Kumar R, Kalra SK, Vaid VK, Mahapatra AK (2008) Chiari I malformation: surgical experience over a decade of management. Br J Neurosurg 22(3):409–414
Kunert P, Janowski M, Zakrzewska A, Marchel A (2009) Comparision of results between two different techniques of craniocervical decompression in patients with Chiari I malformation. Neurol Neurochir Pol 43(4):337–345
Lee HS, Lee SH, Kim ES, Kim JS, Lee JI, Shin HJ, Eoh W (2012) Surgical results of arachnoid-preserving posterior fossa decompression for Chiari I malformation with associated syringomyelia. J Clin Neurosci 19(4):557–560
Liang CJ, Dong QJ, Xing YH, Shan M, Wen LX, Qiang ZY, Ping ZQ, Tao PZ, Ping HX (2014) Posterior fossa decompression combined with resection of the cerebellomedullary fissure membrane and expansile duraplasty: a radical and rational surgical treatment for Arnold-Chiari type I malformation. Cell Biochem Biophys 70(3):1817–1821
Matsumoto T, Symon L (1989) Surgical management of syringomyeliae—current results. Surg Neurol 32:258–265
Mueller D, Oro JJ (2005) Prospective analysis of self-perceived quality of life before and after posterior fossa decompression in 112 patients with Chiari malformation with or without syringomyelia. Neurosurg Focus 18(2):ECP2
Munshi I, Frim D, Stine-Reyes R, Weir BK, Hekmatpanah J, Brown F (2000) Effects of posterior fossa decompression with and without duraplasty on Chiari malformation-associated hydromyelia. Neurosurgery 46(6):1384–1389;discussion 1389-90
Silva JA, Santos AA Jr, Costa Mdo D, Almeida EB (2013) Suboccipitalcraniectomy with opening of the fourth ventricle and duraplasty: study of 192 cases of craniovertebral malformations. Arq Neuropsiquiatr 71(9A):609–614
Neyeloff JL, Fuchs SC, Moreira LB (2012) Meta-analyses and Forest plots using a microsoft excel spreadsheet: step-by-step guidefocusing on descriptive data analysis. BMC Res Notes 20(5):52
Nohria V, Oakes WJ (1990–1991) Chiari I malformation: a review of 43 patients. Pediatr Neurosurg 16:222–227
Noudel R, Gomis P, Sotoares G, Bazin A, Pierot L, Pruvo JP, Bordet R, Roche PH (2011) Posterior fossa volume increase after surgery for Chiari malformation type I: a quantitative assessment using magnetic resonance imaging and correlations with the treatment response. J Neurosurg 115(3):647–658
Ono A, Numasawa T, Wada K, Yokoyama T, Takeuchi K, Suetsuna F, Ueyama K, Toh S (2010) Surgical outcomes of foramen magnum decompression forsyringomyelia associated with Chiari I malformation: relation between the location of the syrinx and body pain. J Orthop Sci 15(3):299–304
Parker SL, Godil SS, Zuckerman SL, Mendenhall SK, Wells JA, Shau DN, McGirt MJ (2013) Comprehensive assessment of 1-year outcomes and determination of minimum clinically important difference in pain, disability, and quality of life after suboccipital decompression for Chiari malformation I in adults. Neurosurgery 73(4):569–581; discussion 581
Prat R, Galeano I (2009) Pain improvement in patients with syringomyelia and Chiari I malformation treated with suboccipital decompression and tonsillar coagulation. J Clin Neurosci 16(4):531–534
Pritz MB (2003) Surgical treatment of Chiari I malformation: simplified technique and clinical results. Skull Base 13:173–177
Quon JL, Grant RA, DiLuna ML (2015) Multimodal evaluation of CSF dynamics following extradural decompression for Chiari malformation type I. J Neurosurg Spine 22(6):622–630
Ramnarayan R, Praharaj MS, Jayakumar PN (2008) Chiari 1 malformations: an Indian hospital experience. Singapore Med J49(12):1029–1034
Saceda-Gutiérrez JM, Isla-Guerrero A, Álvarez-Ruíz F, Odene-Cantero C, Hernández-García B, Márquez Pérez TM (2011) Postoperative complications in Chiari I malformation: duraplasty and cerebrospinal fluid leak. Neurocirugia (Astur) 22(1):36–42; discussion 42-3
Sahuquillo J, Rubio E, Poca MA, Rovira A, Rodriguez-Baeza A, Cervera C (1994) Posterior fossa reconstruction: a surgical technique for the treatment of Chiari I malformationand Chiari I/syringomyelia complex—preliminary results and magnetic resonance imaging quantitative assessment of hindbrain migration. Neurosurgery 35(5):874–884; discussion 884-5
Sakamoto H, Nishikawa M, Hakuba A, Yasui T, Kitano S, Nakanishi N, Inoue Y (1999) Expansive suboccipitalcranioplasty for the treatment of syringomyelia associated with Chiari malformation. Acta Neurochir 141(9):949–960 discussion 960-1
Sakushima K, Hida K, Yabe I, Tsuboi S, Uehara R, Sasaki H (2013) Different surgical treatment techniques used by neurosurgeons and orthopedists for syringomyelia caused by Chiari I malformation in Japan. J Neurosurg Spine 18(6):588–592
Salunke P, Sura S, Futane S, Aggarwal A, Khandelwal NK, Chhabra R, Mukherjee KK, Gupta SK (2012) Ventral compression in adult patients with Chiari 1 malformation sans basilar invagination: cause and management. Acta Neurochir 154(1):147–152
Shamji MF, Ventureyra EC, Baronia B, Nzau M, Vassilyadi M (2010) Classification of symptomatic of Chiari I malformation to guide surgical strategy. Can J Neurol Sci 37(4):482–487
Sindou M, Chávez-Machuca J, Hashish H (2002) Craniocervical decompression for Chiari type I-malformation, adding extreme lateral foramen magnum opening and expansileduraplasty with arachnoid preservation. Technique and long-term functional results in 44 consecutive adult cases—comparison with literature data. Acta Neurochir 144(10):1005–1019
Smith JS, Shaffrey CI, Abel MF, Menezes AH (2010) Basilar invagination. Neurosurgery 66(3Suppl):39–47
Spena G, Bernucci C, Garbossa D, Valfrè W, Versari P (2010) Clinical and radiological outcome of craniocervicalosteo-dural decompression for Chiari I-associated syringomyelia. Neurosurg Rev 33(3):297–303;discussion 303-4
Takayasu M, Takagi T, Hara M, Anzai M (2004) A simple technique for expansive suboccipitalcranioplasty following foramen magnum decompression for the treatment of syringomyelia associated with Chiari I malformation. Neurosurg Rev 27(3):173–177
Taricco MA, Melo LR (2008) Retrospective study of patients with Chiari: malformation submitted to surgical treatment. Arq Neuropsiquiatr 66(2A):184–188
Tisell M, Wallskog J, Linde M (2009) Long-term outcome after surgery for Chiari I malformation. Acta Neurol Scand 120(5):295–299
Tubbs RS, McGirt MJ, Oakes WJ (2003) Surgical experience in 130 pediatric patients with Chiari I malformations. J Neurosurg 99:291–296
Vakharia VN, Guilfoyle MR, Laing RJ (2012) Prospective study of outcome of foramen magnum decompressions in patients with syrinx and non-syrinx associated Chiari malformations. Br J Neurosurg 26(1):7–11
Wetjen NM, Heiss JD, Oldfield EH (2008) Time course of syringomielya resolution followin following decompression of Chiari malformation type I. J Neurosurg Pediatr 1(2):118–123
Wu T, Zhu Z, Jiang J, Zheng X, Sun X, Qian B, Zhu F, Qiu Y (2012) Syrinx resolution after posterior fossa decompression in patients with scoliosis secondary toChiari malformation type I. Eur Spine J21(6):1143–1150
Yilmaz A, Kanat A, Musluman AM, Colak I, Terzi Y, Kayacı S, Aydin Y (2011) When is duraplasty required in the surgical treatment of Chiari malformation type I based on tonsillar descending grading scale? World Neurosurg 75(2):307–313
Zhang Y, Zhang N, Qiu H, Zhou J, Li P, Ren M, Shen G, Chen L, Zhou C, Yang D, Liu Y, Mao Y, Gu X, Zhao Y (2011) An efficacy analysis of posterior fossa decompression techniques in the treatment of Chiari malformation with associated syringomyelia. J Clin Neurosci 18(10):1346–1349
Zhang ZQ, Chen YQ, Chen YA, Wu X, Wang YB, Li XG (2008) Chiari I malformation associated with syringomyelia: a retrospective study of 316 surgically treated patients. Spinal Cord 46(5):358–363
Zhao JL, Li MH, Wang CL, Meng W (2016) A systematic review of Chiari I malformation: techniques and outcomes. World Neurosurg 88:7–14
Zileli M, Cagli S (2002) Combined anterior and posterior approach for managing basilar invagination associated with type I Chiari malformation. J Spinal Disord Tech 15(4):284–289
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The present research project was approved by Ethics and Research Committee of HSPE (Hospital do Servidor Público de São Paulo).
Informed consent
Once it is a review of other published data, we did not apply any informed consent to participants.
Funding statement
There was not any funding received to support this work.
Rights and permissions
About this article
Cite this article
de Oliveira Sousa, U., de Oliveira, M.F., Heringer, L.C. et al. The effect of posterior fossa decompression in adult Chiari malformation and basilar invagination: a systematic review and meta-analysis. Neurosurg Rev 41, 311–321 (2018). https://doi.org/10.1007/s10143-017-0857-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-017-0857-5