Abstract
A subset of patients with crescentic glomerulonephritis (CGN) is characterized serologically by the presence of antineutrophil cytoplasmic antibody (ANCA) and anti-glomerular basement membrane antibody (anti-GBM) called “double positive” disease. The clinical significance of the occurrence of both antibodies is not clear. This study aims to describe the clinical and histologic characteristics and outcomes of CGN in a US cohort of double positive (DP) patients and compare them to patients with anti-GBM disease only or ANCA only (ANCA-associated vasculitis (AAV)). Renal biopsies with a diagnosis of CGN with either pauci-immune or linear immunofluorescence were selected and classified as AAV, anti-GBM disease, or DP based on serologic testing at the time of biopsy. Data on demographics, clinical presentation, treatment, and outcome were obtained by chart review. Six patients with anti-GBM disease, 9 with DP disease, and 18 AAV patients matched for year of diagnosis with DP were identified. Extrarenal disease manifestations were more prominent in the DP patients. The DP patients had severe renal dysfunction at presentation with eight of nine patients requiring dialysis at presentation. Renal biopsy findings of DP patients were similar to anti-GBM disease with majority of glomeruli showing cellular crescents. Eighty-nine percent of patients were treated with immunosuppressive therapy and 78 % with plasmapheresis. At 1 year, all nine DP patients reached end-stage renal disease. We conclude that the DP patients share extrarenal manifestations similar to AAV patients while the renal manifestations resemble anti-GBM patients clinically and histologically. The renal prognosis of DP patients remains poor despite treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) is a group of systemic small vessel vasculitis, including granulomatosis with polyangiitis, microscopic polyangiitis, and eosinophilic granulomatosis with polyangiitis. AAV are characterized serologically by the presence of circulating antineutrophil cytoplasmic antibody. Anti-glomerular basement membrane disease (anti-GBM) is a disease characterized by antibodies directed against the glomerular basement membrane. AAV typically presents in older age group while anti-GBM disease presents typically in younger patients. Both ANCA and anti-GBM antibody cause crescentic glomerulonephritis and rapidly progressive renal failure. However, differences exist in response to immunosuppressive therapy particularly in dialysis-dependant patients at presentation. In pure ANCA-related renal disease, 75 % of such patients recover renal function with appropriate immunosuppressive therapy [1] while renal recovery is rare in isolated anti-GBM disease patients who are dialysis dependant on presentation [2].
In a subset of patients with rapidly progressive glomerulonephritis, both antibodies coexist at the time of presentation, also called as “double positive” disease and first reported by O’Donoghue et al. in 1989 [3]. Between 10 and 40 % of patients with anti-GBM disease tested positive for ANCA, mostly directed against MPO antigen [4–7] and 5 to 14 % of patients with AAV tested positive for anti-GBM antibody [8–10] in previous studies. The mechanism for the coexistence of these is not clear. It has been hypothesized that one autoantibody may expose an epitope and lead to the development of a second autoantibody [5]. No differences in antigen specificities were found when comparing sera from those with double positive disease with sera from anti-GBM disease alone [9]. One group suggested that low levels of ANCA antibodies preceded the development of anti-GBM antibodies by years or decades. It is hypothesized that disruption of glomerular basement membrane by glomerular damage caused by ANCA could expose the target antigen and trigger the production of anti-GBM antibody [7]. The clinical significance of the occurrence of both antibodies is not clear.
More controversial, however, is the prognosis of these double positive patients. While earlier reports suggest that these patients fare better than those with anti-GBM autoantibodies alone [11] but worse than those with ANCA autoantibodies, more recent analyses of cohorts of European and Chinese patients have shown that the clinical features and outcomes of double positive patients resemble those of patients with anti-GBM disease [10, 12, 13]. However, no such study has been done with a cohort of North American patients. This may be related to the rare occurrence of these diseases. It is also possible that double positive disease is under recognized since all patients presenting with crescentic glomerulonephritis may not be uniformly subjected to testing for the simultaneous presence of both types of antibodies and this may relate to low prevalence of this disease.
The current study aims to describe the clinical and histologic characteristics and outcomes of crescentic glomerulonephritis in a US cohort of double positive patients, and compare them to patients with anti-GBM disease only or AAV only.
Methods
For this study, biopsy entries in the Johns Hopkins Hospital renal biopsy database with a diagnosis of crescentic glomerulonephritis (CGN) between 1999 and 2011 were reviewed. CGN biopsies with either pauci-immune or linear immunofluorescence were then selected, and these patients were classified as either ANCA positive (n = 18), anti-GBM positive (n = 6), or double positive (n = 9) based on serologic testing conducted at the time of biopsy. The study protocol was approved by the institutional review board.
ANCA testing was done by standard indirect immunofluorescence assay on ethanol-fixed neutrophils (INOVA Diagnostics, San Diego, CA). PR3 and MPO testing was done by direct enzyme-linked immunosorbent assay according to manufacturer’s instructions (INOVA Diagnostics, San Diego, CA). ANCA titer of less than 10 was considered to be negative for c-ANCA and p-ANCA. PR 3 and MPO levels of less than 10 units were negative. Anti-GBM antibody testing was done by ELISA from 1999 to early 2010 (QUANTA Lite™ GBM ELISA) and from 2010 to 2011 by immunoassay (BioPlex 2200 System; Bio-Rad Laboratories Clinical Diagnostics Group, Hercules, CA). Patients were characterized as double positive if assays for both ANCA and anti-GBM antibody were positive.
The following data were extracted from review of patient records: demographics; details of clinical presentation; ANCA and anti-GBM serologies; serum creatinine at diagnosis, 12 months, and 24 months when available and at last follow-up when available; and details of immunosuppressive therapy. Glomerular filtration rate (GFR) was estimated using modification of diet in renal disease (MDRD) formula [14]. End-stage renal disease (ESRD) was defined as need for dialysis or transplant. Finally, patients were said to have recovered renal function if they were no longer dialysis dependent following treatment. Extrarenal disease was defined as follows: constitutional symptoms, presence of subjective fever, weight loss and fatigue, sinus symptoms (bloody nasal discharge, nasal crusting, nasal septal perforation, nasal obstruction and sinusitis), ear-related symptoms (conductive or sensorineural hearing loss), eye-related symptoms (conjunctivitis, episcleritis, scleritis, uveitis, retinal hemorrhage or exudates, retro-orbital mass or proptosis), lung-related symptoms (pleurisy, endobronchial involvement, nodules, cavities or infiltrates on chest imaging, alveolar hemorrhage, respiratory failure), peripheral nerve-related symptoms (peripheral sensory neuropathy, motor mononeuritis multiplex), and joints-related symptoms (arthralgias, arthritis).
Histological characteristics of renal biopsy samples were reviewed and the percent of normal glomeruli, percent glomeruli with cellular crescents, percent glomeruli with global sclerosis, and the type of immunofluorescence were recorded. The renal biopsies were either read by four different pathologists at Johns Hopkins during the study period or by pathologists at referring institution. The biopsies were not read centrally and hence we used the pathology report to obtain data on the different histologic variables described above.
Statistical analyses were performed using the Graph Pad Prism 5 program. Kruskal–Wallis was used to assess differences between quantitative data while Fischer Test of exact Probabilities was used to assess differences between qualitative data. Cross-tabulated data were tested using chi-square analysis. Kaplan–Meier estimates for survival distribution were used to calculate posttreatment survival curves. The end point for renal survival analysis was the start of renal replacement therapy in form of dialysis or transplant. Differences were considered statistically significant if the two-tailed p value was less than 0.05.
Results
Two hundred and seventy-two cases of isolated AAV, 6 cases of isolated anti-GBM disease, and 9 cases of double positive disease were identified. For every double positive patient, two patients with isolated AAV were selected matched for the year of diagnosis. Among the double positive patients, all nine cases were positive for anti-GBM antibodies and ANCA (five c-ANCA/PR3 ELISA positive and four p-ANCA/MPO ELISA positive). Among 18 patients that were ANCA positive only, 12 were p-ANCA/MPO ELISA positive and the rest were c-ANCA/PR3 ELISA positive. The demographics and clinical presentation at the time of biopsy are listed in Table 1. There was no significant difference in mean age at presentation between the groups. There were a higher proportion of males in the ANCA group compared to double positive and anti-GBM group. The frequency of involvement of the ears, eyes, peripheral nerves, and joints in patients in the double positive group was similar to patients in the ANCA group. The frequency of alveolar hemorrhage was similar between the double positive and anti-GBM disease groups. The serum creatinine at presentation and the number of patients that were dialysis dependant at presentation were significantly higher in the double positive patients when compared to ANCA-positive patients but similar to patients with anti-GBM disease.
All patients underwent a renal biopsy. The data on information of time to renal biopsy from symptom onset were available in six patients with double positive group, all patients with anti-GBM group, and in 15 patients in the ANCA group. The median time from symptom onset to renal biopsy was 3 months in double positive group, 1 month in the anti-GBM group, and 2 months in the ANCA group. The percent of normal glomeruli was significantly less in patients in the double positive group and anti-GBM disease compared to patients in the ANCA group (Table 2). The percent glomeruli with cellular crescents were significantly higher in patients in the double positive group and anti-GBM disease compared to patients in the ANCA group. Among the patients in the double positive group, 89 % of patients had linear staining for IgG on immunofluorescence.
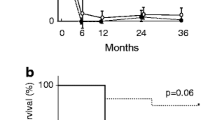
The treatment details and clinical outcome are shown in Table 3. The majority of patients in the double positive group were treated with a combination of glucocorticoids and cyclophosphamide therapy similar to those with anti-GBM disease and ANCA disease. The majority of patients with double positive disease were treated with plasmapheresis similar to patients in the anti-GBM disease group. Among patients in the double positive group, eight were dialysis dependant at presentation and none recovered renal function with treatment. The remaining one patient suffered a relapse and became dialysis dependant after 6 years. Among patients with anti-GBM disease, all were dialysis dependant on presentation and all received plasmapheresis and immunosuppressive treatment with cyclophosphamide and glucocorticoids. Two of these six patients recovered renal function and were able to discontinue dialysis after 2 and 5 months of dialysis initiation. Among those with ANCA disease, there were three patients who were dialysis dependant at presentation and were treated with cyclophosphamide and glucocorticoids. One patient recovered renal function after 10 weeks to discontinue dialysis while the remaining two remained on long-term dialysis. The 1-year renal survival was worse in the double positive patients with eight patients reaching ESRD compared to those with anti-GBM disease and AAV (Fig. 1). There was one death in the AAV group due to aspiration pneumonia at 1 year and none in the double positive or the anti-GBM group.
Kaplan–Meier curve of overall renal survival. Combined log rank analysis shows a significant difference among the three groups (p ≤ 0.0001). Pairwise comparison per group showed a significant difference between the double positive group and ANCA group and not between double positive group and anti-GBM group (p ≤ 0.001 and p = 0.14), respectively. There were 1, 2, and 16 patients still at risk at 1 year in the double positive, anti-GBM, and ANCA groups, respectively
The median follow-up time differed among the groups. The nine patients in the double positive group had a median follow-up of 66 months. At the time of last follow-up, there were no deaths in this group. Six patients underwent a renal transplant for end-stage renal disease and the remaining three patients were on dialysis. The six patients in the anti-GBM group had a median follow-up of 41 months. At the time of last follow-up, four patients in this group were on dialysis and the remaining two patients had a median serum creatinine of 1.9 mg/dl and an estimated GFR of 36 ml/min at a median follow-up of 29 months. The 18 patients in the ANCA group had a median follow-up of 61 months. At the time of last follow-up, there was one death and four patients were on dialysis in this group. The remaining 13 patients had a median serum creatinine of 1.5 mg/dl and an estimated GFR of 50 ml/min at a median follow-up of 61 months.
Discussion
This is the first descriptive study of a US cohort of double positive patients describing the demographics, clinical and histological features at presentation, and treatment response in comparison to patients with anti-GBM disease and AAV alone. Our study shows that double positive patients share extrarenal disease manifestations similar to patients with ANCA disease. The serum creatinine at presentation and number of patients requiring dialysis at presentation were similar to those patients with anti-GBM disease but worse compared to patients with ANCA disease. The renal histology was similar to those with anti-GBM disease with a large proportion of glomeruli revealing cellular crescents. The double positive patients had worse renal outcomes compared to those with anti-GBM disease or AAV.
The mechanism for the coexistence of these two autoantibodies is not clear. Recent studies have suggested that low level ANCA may be detectable years before the detection of anti-GBM antibody and onset of clinical disease [7]. ANCA may disrupt the glomerular basement membrane exposing the cryptic _α 3(IV) NC1 antigen and triggering anti-GBM production. It is also hypothesized that ANCA may inhibit the intracellular proteolytic enzymes involved in the disruptive process allowing surface expression of _α 3(IV)NC1 and trigger anti-GBM production. This sequence of occurrence of the antibodies likely explains the extrarenal features of patients with double positive disease being similar to the ANCA-positive patients and the poor renal outcome of double positive patients due to the more extensive glomerular injury mediated by anti-GBM antibodies.
The double positive patients comprised 3 % of our patients with crescentic glomerulonephritis that were either pauci-immune or had linear IgG staining and 57 % of renal biopsies that had linear IgG staining on immunofluorescence. Previous studies on double positive patients report older age at presentation. The mean age in our cohort of double positive patients was similar to those with anti-GBM disease and ANCA disease. A male predominance has been reported in previous series [8–12, 15], though the majority of our patients were females. Five of our nine patients were PR3 ANCA positive in comparison to MPO ANCA predominant patients reported by other groups [8–12, 15].
Multi-system involvement was common in double positive patients in our cohort similar to AAV patients. This is similar to the Chinese cohort reported by Zhao et al. [13]. Our patients with double positive disease had severe renal disease at presentation both clinically and histologically similar to anti-GBM patients and in agreement with the observations in the Chinese cohort of double positive patients [13].
Treatment with immunosuppression and plasmapheresis was given to the majority of our patients in contrast to only 60 to 80 % patients reported by others [10, 12, 13]. Despite treatment, the renal outcomes of double positive patients in our cohort were worse than those with anti-GBM disease and AAV. This is similar to outcomes reported by other series [10, 12, 13]. In contrast, other groups have reported better renal outcome in double positive patients [8, 11, 16]. The 1-year patient survival was 100 % in our cohort of double positive patients compared to higher mortality reported by other groups. The majority of patients in the latter two studies were p-ANCA/MPO ELISA positive.
Our study has limitations because of the retrospective nature of the study and reliance on clinical notes for data collection. We are unable to estimate the true prevalence of double positive disease due to the lack of stored sera. The renal biopsies were not read centrally by pathologists blinded to clinical data. Our study, like others, is limited by the small patient numbers and we lack the statistical power to make solid conclusions based on multivariate analysis. Given the rarity of this condition, multicenter collaboration is required to pool the numbers required to make concrete association and implications of the coexistence of anti-GBM antibody and ANCA. Despite these limitations, our study provides new data for this rare but devastating condition, in a USA-based cohort.
Conclusions
Our cohort of double positive patients differs from previously reported cohorts in having a predominance of PR3 ANCA-positive patients. The double positive patients in our cohort share extrarenal manifestations similar to ANCA-positive patients while the renal manifestations resemble anti-GBM patients clinically and histologically. The 1-year renal survival is worse compared to anti-GBM disease patients and AAV patients despite use of immunosuppressive therapy and plasmapheresis.
References
Jayne D, Rasmussen N, Andrassy K, Bacon P, Tervaert JW, Dadoniene J, Ekstrand A, Gaskin G, Gregorini G, de Groot K, Gross W, Hagen EC, Mirapeix E, Pettersson E, Siegert C, Sinico A, Tesar V, Westman K, Pusey C (2003) A randomized trial of maintenance therapy for vasculitis associated with antineutrophil cytoplasmic autoantibodies. N Engl J Med 349:36–44
Levy JB, Turner AN, Rees AJ, Pusey CD (2001) Long-term outcome of anti-glomerular basement membrane antibody disease treated with plasma exchange and immunosuppression. Ann Intern Med 134:1033–1042
O'Donoghue DJ, Short CD, Brenchley PE, Lawler W, Ballardie FW (1989) Sequential development of systemic vasculitis with anti-neutrophil cytoplasmic antibodies complicating anti-glomerular basement membrane disease. Clin Nephrol 32:251–255
Jia XY, Cui Z, Yang R, Hu SY, Zhao MH (2012) Antibodies against linear epitopes on the Goodpasture autoantigen and kidney injury. Clin J Am Soc Nephrol 7:926–933
Pedchenko V, Bondar O, Fogo AB, Vanacore R, Voziyan P, Kitching AR, Wieslander J, Kashtan C, Borza DB, Neilson EG, Wilson CB, Hudson BG (2010) Molecular architecture of the Goodpasture autoantigen in anti-GBM nephritis. N Engl J Med 363:343–354
Yang R, Hellmark T, Zhao J, Cui Z, Segelmark M, Zhao MH, Wang HY (2007) Antigen and epitope specificity of anti-glomerular basement membrane antibodies in patients with goodpasture disease with or without anti-neutrophil cytoplasmic antibodies. J Am Soc Nephrol 18:1338–1343
Olson SW, Arbogast CB, Baker TP, Owshalimpur D, Oliver DK, Abbott KC, Yuan CM (2011) Asymptomatic autoantibodies associate with future anti-glomerular basement membrane disease. J Am Soc Nephrol 22:1946–1952
Jayne DR, Marshall PD, Jones SJ, Lockwood CM (1990) Autoantibodies to GBM and neutrophil cytoplasm in rapidly progressive glomerulonephritis. Kidney Int 37:965–970
Hellmark T, Niles JL, Collins AB, McCluskey RT, Brunmark C (1997) Comparison of anti-GBM antibodies in sera with or without ANCA. J Am Soc Nephrol 8:376–385
Levy JB, Hammad T, Coulthart A, Dougan T, Pusey CD (2004) Clinical features and outcome of patients with both ANCA and anti-GBM antibodies. Kidney Int 66:1535–1540
Bosch X, Mirapeix E, Font J, Borrellas X, Rodriguez R, Lopez-Soto A, Ingelmo M, Revert L (1991) Prognostic implication of anti-neutrophil cytoplasmic autoantibodies with myeloperoxidase specificity in anti-glomerular basement membrane disease. Clin Nephrol 36:107–113
Rutgers A, Slot M, van Paassen P, van Breda Vriesman P, Heeringa P, Tervaert JW (2005) Coexistence of anti-glomerular basement membrane antibodies and myeloperoxidase-ANCAs in crescentic glomerulonephritis. Am J Kidney Dis 46:253–262
Zhao J, Yang R, Cui Z, Chen M, Zhao MH, Wang HY (2007) Characteristics and outcome of Chinese patients with both antineutrophil cytoplasmic antibody and antiglomerular basement membrane antibodies. Nephron Clin Pract 107:c56–c62
Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D (1999) A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 130:461–470
Cui Z, Zhao MH, Xin G, Wang HY (2005) Characteristics and prognosis of Chinese patients with anti-glomerular basement membrane disease. Nephron Clin Pract 99:c49–c55
Segelmark M, Hellmark T, Wieslander J (2003) The prognostic significance in Goodpasture's disease of specificity, titre and affinity of anti-glomerular-basement-membrane antibodies. Nephron Clin Pract 94:c59–c68
Disclosures
Amar Srivastava, Gautam K Rao, Paul E Segal, and Megha Shah have nothing to disclose while Duvuru Geetha, M.D. served as a consultant and received honoraria from Genentech for educational lectures for sales force.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Srivastava, A., Rao, G.K., Segal, P.E. et al. Characteristics and outcome of crescentic glomerulonephritis in patients with both antineutrophil cytoplasmic antibody and anti-glomerular basement membrane antibody. Clin Rheumatol 32, 1317–1322 (2013). https://doi.org/10.1007/s10067-013-2268-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-013-2268-5