Abstract
We report on a novel strategy for the electrochemical detection of cocaine. It is based on the use of a supramolecular aptamer, rolling circle amplification (RCA), and multiplex binding of a biotin-strepavidin system. The aptamer fragments were assembled to a supramolecular aptamer which, in the presence of cocaine, conjugates to streptavidin for anchoring of biotinylated circular DNA. This initiates RCA and enables sensitive electrochemical-enzymatic readout. A significant signal amplification was obtained by using streptavidin linked to alkaline phosphatase that binds to the remaining biotinylated detection probes and catalyzes the hydrolysis of the synthetic enzyme substrate α-naphthylphosphate. This dual amplification strategy tremendously increases the detection limit of the aptasensor. Under optimal conditions and using differential pulse voltammetry, cocaine can be detected in the concentration range between 2 and 500 nM with a detection limit as low as 1.3 nM (at S/N = 3). The method is specific and acceptably reproducible. It was successfully applied to the detection of cocaine in (spiked) urine samples. The data were in good agreement with those obtained by the GC-MS reference method.

Cascade signal amplification strategy for cocaine electrochemical detection by supramolecular aptamer and rolling circle amplification.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cocaine, a small molecule, is one of the most illegally abused drugs available all over the world because of its instantaneous and overwhelming effects on the central nervous system [1–3]. Quantification of cocaine plays an important role in forensic science, pharmacy, therapy, and metabolomics. Traditional methods for cocaine analyses are mainly based on GC-MS, and HPLC-coupled fluorescence [4–6]. These approaches are powerful and sensitive for cocaine determination, but their high cost and complicated operation limit their extensive application. Recently, the aptamer-based detection methods for cocaine and some small molecules have gained conspicuous attention.
Aptamers, in vitro selected by systematic evolution ligands by exponential enrichment (SELEX), can bind respective targets such as small inorganic or organic substances [7], proteins [8], and cells [9]. Due to their easy modification, high affinity, and good stability, aptamers have been increasingly used as recognition biomaterials in biosensors [10, 11]. Based on different technologies, miscellaneous aptasensors have been developed, such as colorimetry [12], fluorescence [13], surface plasmon resonance [14], and electrochemistry [15]. Unlike the double-antibody sandwich assays, it is challengeable not only to bind selectively with the same target molecules but also to possess effective labeling strategies for sensitive detection of small molecules. Recently, Szostak [16] and Landry [17] groups, respectively, have pioneered the development of aptamer-based sandwich assays for the detection of ATP and cocaine. In their experiment, one single stranded ATP or cocaine binding aptamer was split into two fragments, which could combine with the target molecules at specific region. In addition, an electroactive species was used to label one of the aptamers to achieve sensitive detection of ATP and cocaine. Inspired by previous advances, a great deal of supramolecular aptasensors have been developed, such as electrochemical aptasensors [18–21], fluorescence aptasensors [2, 22, 23], and colorimetric aptasensors [24]. However, it remains difficult to realize a low detection limit for cocaine detection, partially because of the surface-induced perturbation of the aptamer structure and the reduced accessibility of the target molecule to probes at a heterogeneous and locally crowded surface [25, 26].
In recent years, RCA, an isothermal DNA amplification technique [27], is widely used as an effective amplification technique in the field of biosensors. RCA can generate a linear concatenated DNA molecule containing up to 1000 complementary copies of the circular DNA [28]. Compared with polymerase chain reaction (PCR), the RCA possesses a great deal of merits, such as in situ capturing target molecules, the isothermal amplification procedure, and the linear kinetic mode [29]. Therefore, it has been obtained considerable attention as a novel tool to amplify the recognition events for ultrasensitive detections of DNA [30], RNA [31], and proteins [32].
In the present work, to increase methodological sensitivity, a novel supramolecular aptasensor was established based on supramolecular aptamer, rolling circle amplification combined with multiplex binding of the biotin-strepavidin system for cocaine detection. The established aptasensor demonstrated excellent analytical performance towards cocaine, which might be a novel detection strategy for cocaine in forensic toxicological analysis and drug abuse control.
Materials and methods
Reagents
6-Mercapto-1-hexanol (MCH), streptavidin-alkaline phosphatase (ST-AP), α-naphthyl phosphate (α-NP), bovine serum albumin (BSA), salmon sperm DNA, and Tris (2-carboxyethyl) phosphine hydrochloride (TCEP) were purchased from Sigma-Aldrich (St. Louis, MO, USA, http://www.sigmaaldrich.com). Cocaine hydrochloride, morphine hydrochloride, methamphetamine hydrochloride, and ketamine hydrochloride were obtained from the Second Research Institute of the Ministry of Public Security (Beijing, China, http://www.b2b168.com/c168-686332.html). Phi29 DNA polymerase and dNTP mixture were obtained from Sangon Inc. (Shanghai, China, http://www.sangon.com). T4 DNA ligase was purchased from Takara (Dalian, China, http://www.takara.com.cn). All other reagents were of analytical grade. All aqueous solutions were prepared using Millipore-Q water (≥18 МΩ, http://www.mollipore.com). Twenty millimolar Tris–HCl buffer (pH 7.4) containing 0.10 M NaCl, 5.0 mM MgCl2, and 0.005 % Tween-20 was used as washing buffer. Hybridization buffer (pH 7.4) contained 0.3 M NaCl and 0.03 M sodium citrate. Diethanolamine (DEA) buffer (pH 9.6) contained 0.1 M DEA, 1 mM MgCl2, and 100 mM KCl.
DNA oligonucleotides were designed according to the literatures [33, 34]. All oligonucleotides were synthesized and purified by Sangon Inc. (Shanghai, China, http://www.sangon.com), and the base sequences were showed in Table 1. All oligonucleotides were dissolved in tris-ethylenediaminetetraacetic acid (TE) buffer (pH 8.0, 10 mM Tris–HCl, 1 mM EDTA) and stored at −20 °C, which were diluted in appropriate buffer prior to use.
Apparatus
The electrochemical measurements were performed on a CHI 660D electrochemical analyzer (Shanghai Chenhua Instruments Co. Ltd., China, http://www.chinstruments.com) with a three-electrode system composed of platinum wire as auxiliary, Ag/AgCl electrode as reference, and a 3-mm-diameter gold electrode as working electrode. Differential pulse voltammetry (DPV), electrochemical impedance spectroscopic (EIS), and square wave voltammetry (SWV) were carried out at room temperature.
Preparation of electrochemical aptasensor
The bare gold electrode was polished with 0.05 μm alumina slurries and ultrasonically treated in deionized water for a few minutes. To remove all contaminants, the electrode was soaked in piranha solution (H2SO4:H2O2 = 3:1) for 10 min, followed by rinsing with deionized water and allowing to dry at room temperature. Ten micro liter of fifty nanomole thiolated aptamer probe (Co3S) in TE buffer was dropped onto the pretreated gold electrode surface carefully and incubated overnight at 4 °C. After rinsing with the washing buffer, the electrode was immersed into 10 μL of 1 mM MCH for 1 h to obtain well-aligned DNA monolayer and occupy the left bare sites. The electrode was further washed with the washing buffer and treated in 125 μg mL−1 salmon sperm DNA and 2 % BSA for 2 h to avoid nonspecific adsorption of DNA and enzyme on the electrode surface.
Cocaine detection protocol
To detect cocaine, 10 μL of a sample in the phosphate buffer (PB) (pH 7.4, 0.1 M phosphate, 5 mM KCl, 5 mM MgCl2, and 40 μM TCEP) containing 60 nM Co3B and different concentrations of cocaine were droped onto the Au/Co3S sensing interface for 40 min. After the formation of supramolecular aptamer, the aptasensor was incubated with 10 μL of 15 nM streptavidin in PB at 37 °C for 30 min. Following rinsing with the washing buffer, 10 μL of biotinylated circular DNA containing 15 nM circular template DNA and 15 nM biotinylated primer DNA in PB were added to each electrode and incubated at 37 °C for 30 min. The electrode was further rinsed with the washing buffer; RCA reaction was initiated by addition of 0.4 units of phi29 DNA polymerase in 10 μL of reaction buffer (33 mM, pH 7.9 Tris-acetate buffer, 10 mM Mg-acetate, 66 mM K-acetate, 1 mM dithiothreitol, 0.5 mM dNTP, and 0.1 % Tween 20) and continued for 1 h at 37 °C. After washing with the washing buffer, 10 μL of 1 μM biotinylated detection probe was dropped onto the biosensor surface and hybridized for 1 h at 37 °C. Following washed by DEA buffer, the aptasensor reacted with 10 μL of 1.25 μg mL−1 ST-AP at 37 °C for 30 min, and washed thoroughly with DEA buffer. The DPV measurement was performed in DEA buffer containing 0.75 mg mL−1 of α-NP substrate with modulation time of 0.05 s, interval time of 0.017 s, step potential of 5 mV, modulation amplitude of 70 mV and potential scan from 0.0 to +0.6 V.
Circularization of DNA Template
One hundred nanomole of circular template oligonucleotide and 100 nM of biotinylated primer oligonucleotide were mixed in 100 μL of ligation buffer (66 mM, pH 7.6 Tris–HCl buffer, 6.6 mM MgCl2, 10 mM dithiothreitol, and 0.1 mM ATP) and vortexed for 1 min. Then, 1 unit of T4 DNA ligase was added and incubated at 37 °C for 1 h. After ligation, T4 DNA ligase was inactivated by heating the reaction mixture at 65 °C for 10 min. The resulting mixture could be used directly or stored at −20 °C.
Results and discussion
Design of the aptasensor for cocaine detection
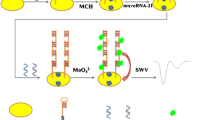
The biosensing process of aptasensor for the cocaine detection was conceptually depicted in Scheme 1. Anti-cocaine aptamer was cleaved into two fragments, Co3S and Co3B. Co3S was immobilized on gold electrode via Au-S bond, while Co3B was labeled with biotin at its 3′-end before being conjugated streptavidin. In the presence of cocaine, two fragments could self-assemble into a supramolecular aptamer. The streptavidin was then bound to the biotinylated supramolecular aptamer to act as a bridge for anchoring of biotinylated circular DNA. In the presence of nucleotides and phi29 DNA polymerase, the RCA was initiated to produce micrometer-long single-strand DNA which contained hundreds of tandem-repeat sequences for linear periodic assembly of many biotinylated detection probes. Then a significant amplification for the detection of cocaine was obtained by using ST-AP, which bound to the remaining biotinylated detection probes and catalyzed α-NP substrate to produce enzymatic electrochemical signal for quantitative detection of cocaine detection.
Characterization of the electrochemical aptasensor
Electrochemical impedance spectroscopy (EIS) and square wave voltammetry (SWV) were employed to investigate stepwise modification of the electrode. Figure 1a showed the Nyquist plots of 5 mM [Fe(CN)6]3-/4- containing 1 M KCl at the different electrodes and its surface electron transfer resistance (Ret) was assessed for the modified electrodes. The bare gold electrode exhibited a nearly straight line in the Nyquist plots of impedance spectroscopy (curve a), which was characteristic of a mass diffusion limiting step of the electron-transfer process. After the immobilization of thiol-modified aptamer fragment, an obvious semicircle up to the mid-frequencies region was exhibited and the Ret value increased (curve b). This could be attributed to the negatively charged phosphate backbone of the oligonucleotides immobilized on the gold electrode, which prevented the negative charged redox probe [Fe(CN)6]3−/4− from reaching the gold electrode and inhibited interfacial charge transfer. When biotinylated supramolecular aptamer was formed by target cocaine and two aptamer fragments, Ret further enhanced to a much larger value (curve c). Target complex layer on the electrode surface acted to block charge exchange and mass transfer, further insulted the conductive support and significantly hindered the access of redox probe towards the electrode surface. After the streptavidin bound with the biotinylated supramolecular aptamer, the semicircle and the value of Ret became larger (curve d). The increase of Ret in the presence of streptavidin was ascribed to the fact that streptavidin was successfully bound to biotinylated supramolecular aptamer. When the RCA was successfully initiated and produced a lot of tandem-repeat sequences. The semicircle and the value of Ret became greater (curve e). The increase of Ret in the presence of many tandem-repeat sequences was ascribed to the fact that the RCA was successfully initiated to produce micrometer-long single-strand DNA, which contained hundreds of tandem-repeat sequences. The impedance results were quite consistent with the conclusions obtained from square wave voltammetry (SWV) (Fig. 1b), in which the peak currents varied upon the assembly, binding, anchoring, and amplification processes.
Optimization of experimental conditions
To obtain excellent analytical performance, different experimental conditions were optimized (Fig. 2). At first, the effect of various Co3B concentrations was investigated. Miscellaneous concentrations of Co3B in PB (without cocaine) were incubated with Co3S at 37 °C. As shown in Fig. 2a, when the concentration of Co3B was over 80 nM, a significant increase of the peak current was observed, which may be implicated by the designed aptamer. It is known that the aptamer, split into two fragments, could equilibrate by cooperative aptamer-target and base-pairing interaction. When the concentrations of the two strands were high enough, they could hybridize by themselves without cocaine. Therefore, 60 nM Co3B was used in the following experiments. Since streptavidin concentration was another important parameter affecting the signal readout. With the increasing concentration of streptavidin, the DPV response decreased gradually, this was attributed to that the residual and biotinylated supramolecular complex immediately bond ST-AP, which could not initiate the RCA. Finally the electrochemical signal tended to be a steady value at 15 nM (Fig. 2b) which was chosen as the optimized streptavidin concentration. As shown in Fig. 2c, the peak current increased gradually with the increase of the incubation time of RCA reaction and then kept constant after 60 min, thus, the incubation time of 60 min was adopted. At the ST-AP concentration of 1.25 μg mL−1, the signal increased gradually with the increasing concentration of α-NP and reached the stable value at 0.75 mg mL−1 (Fig. 2d). Therefore, 0.75 mg mL−1 was decided as the optimal α-NP concentration.
Analytical performance of the electrochemical aptasensor
Sensitivity of aptasensor for cocaine detection
To evaluate the analytical performance of the established electrochemical aptasensor, DPV was exploited to characterize aptasensor. The DPV responses of the aptasensor to different concentrations of cocaine in PB were obtained under optimal conditions (Fig. 3a). It was observed that the DPV signal increased with the increment of cocaine concentration. Figure 3b showed the good linear relationship between the DPV responses and cocaine concentrations in the range of 2 to 500 nM. The resulting linear equation was ip (μA) = 0.02 CCocaine + 2.73 with a correlation coefficient of 0.9969. Additionally, the limit of detection (LOD) was estimated to be 1.3 nM at a signal to noise of 3. To further highlight the merits of the designed aptasensor, the analytical properties were compared with those of other supramolecular aptasensors in Table 2. It proved that the cocaine supramolecular aptasensor in this work hold the feature of the lowest detection limit among the reported supramolecular aptasensors because of the implement of different amplification strategies. Thus, this aptasensor could be applied to quantification of cocaine with low detection concentration.
Specificity and reproducibility of the aptasensor
Four drugs including Cocaine hydrochloride, morphine hydrochloride, methamphetamine hydrochloride, and ketamine hydrochloride were chosen to investigate the selectivity of the established aptasensor. A significant increase, induced by the effect of the aptasensor with 500 nM cocaine was observed, but the DPV responses of the other three drugs (5 μM) were of almost the same signal as the blank background signal. In addition, for further evaluating the reproducibility of the aptasensor, six electrodes were prepared for the detection of 2 and 300 nM cocaine. The relative standard deviations (RSD) of six replicates measurements were 7.58 and 1.07 % at cocaine concentrations of 2 and 300 nM, respectively. Herein, the established aptasensor displayed good specificity and acceptable reproducibility for the cocaine detection.
Detection cocaine in urine samples
To further evaluate the analytical reliability and potential application, the aptasensor was used to detect cocaine with a wide range of concentration in 25 % diluted urine samples, and compared with the reference GC-MS method. As shown in Table 3, the results obtained using the developed aptasensor were in good agreement with the reference GC-MS method with relative errors from 1.0 to 7.8 %. Moreover, other components in diluted urine samples had little interference. Thus, the aptasensor could quantitatively detect cocaine in diluted urine samples, and showed higher sensitivity than reference GC-MS method.
Conclusions
In summary, we have developed a novel and sensitive aptasensor for cocaine detection based on supramolecular aptamer, RCA, and multiplex binding of the biotin-strepavidin system with enzymatic electrochemical readout. The RCA and the multiplex binding of the biotin-strepavidin systems can powerfully enhance aptasensor’s sensitivity. The integration of these signal amplification strategies enables a low detection limit of 1.3 nM for cocaine. In addition, the designed aptasensor possesses the numerous advantages of good specificity, a wide linear range, and acceptable reproducibility, which was successfully applied to cocaine detection in urine samples. This novel biosensing strategy provided an innovative, fast and sensitive platform for the detection of cocaine, which may become a powerful tool for the detection of drug abuse in the future.
References
Lu Y, O’Donnell RM, Harrington PB (2009) Detection of cocaine and its metabolites in urine using solid phase extraction-ion mobility spectrometry with alternating least squares. Forensic Sci Int 189(1):54–59
Zhang CY, Johnson LW (2009) Single quantum-dot-based aptameric nanosensor for cocaine. Anal Chem 81(8):3051–3055
Du Y, Chen CG, Zhou M, Dong SJ, Wang EK (2011) Microfluidic electrochemical aptameric assay integrated on-chip: a potentially convenient sensing platform for the amplified and multiplex analysis of small molecules. Anal Chem 83(5):1523–1529
Jiang BY, Wang M, Chen Y, Xie JQ, Xiang Y (2012) Highly sensitive electrochemical detection of cocaine on graphene/AuNP modified electrode via catalytic redox-recycling amplification. Biosens Bioelectron 32(1):305–308
Strano-Rossi S, Colamonici C, Botrè F (2008) Parallel analysis of stimulants in saliva and urine by gas chromatography/mass spectrometry: perspectives for “in competition” anti-doping analysis. Anal Chim Acta 606(2):217–222
Wu AHB, Bristol B, Sexton K, Cassella-Mclane G, Holtman V, Hill DW (1999) Adulteration of Urine by “Urine Luck”. Clin Chem 45(7):1051–1057
Lee J, Jo M, Kim TH, Ahn JY, Lee D, Kim S, Hong S (2011) Aptamer sandwich-based carbon nanotube sensors for single-carbon-atomicresolution detection of non-polar small molecular species. Lab Chip 11(1):52–56
Cheng W, Ding SJ, Li Q, Yu TX, Yin YB, Ju HX, Ren GS (2012) A simple electrochemical aptasensor for ultrasensitive protein detection using cyclic target-induced primer extension. Biosens Bioelectron 36(1):12–17
Ray P, Rialon-Guevara KL, Veras E, Sullenger BA, White RR (2012) Comparing human pancreatic cell secretomes by in vitro aptamer selection identifies cyclophilin B as a candidate pancreatic cancer biomarker. J Clin Invest 122(5):1734–1741
Liu F, Wang SM, Zhang M, Wang YH, Ge SG, Yu JH, Yan M (2014) Aptamer based test stripe for ultrasensitive detection of mercury(II) using a phenylene-ethynylene reagent on nanoporous silver as a chemiluminescence reagent. Microchim Acta 181(5–6):663–670
Hai H, Yang F, Li JP (2014) Highly sensitive electrochemiluminescence “turn-on” aptamer sensor for lead (II) ion based on the formation of a G-quadruplex on a graphene and gold nanoparticles modified electrode. Microchim Acta 181(9–10):893–901
Yuan JL, Yu Y, Li C, Ma XY, Xia Y, Chen J, Wang ZP (2014) Visual detection and microplate assay for Staphylococcus aureus based on aptamer recognition coupled to tyramine signal amplification. Microchim Acta 181(3–4):321–327
Duan YF, Ning Y, Song Y, Deng L (2014) Fluorescent aptasensor for the determination of Salmonella typhimurium based on a graphene oxide platform. Microchim Acta 181(5–6):647–653
Chang CC, Lin S, Lee CH, Chuang TL, Hsueh PR, Lai HC, Lin CW (2012) Amplified surface plasmon resonance immunosensor for interferon-Gamma based on a streptavidin-incorporated aptamer. Biosens Bioelectron 37(1):68–74
Jia F, Duan N, Wu SJ, Ma XY, Xia Y, Wang ZP, Wei XL (2014) Impedimetric aptasensor for Staphylococcus aureus based on nanocomposite prepared from reduced graphene oxide and gold nanoparticles. Microchim Acta. 181(9–10):967–974
Huizenga DE, Szostak JW (1995) A DNA aptamer that binds adenosine and ATP. Biochemistry 34(2):656–665
Stojanovic MN, de Prada P, Landry DW (2000) Fluorescent sensors based on aptamer self-assembly. J Am Chem Soc 122(46):11547–11548
Zhang DW, Zhang FT, Cui YR, Deng QP, Krause S, Zhou YL, Zhang XX (2012) A label-free aptasensor for the sensitive and specific detection of cocaine using supramolecular aptamer fragments/target complex by electrochemical impedance spectroscopy. Talanta 92:65–71
Zhang DW, Sun CJ, Zhang FT, Xu L, Zhou YL, Zhang XX (2012) An electrochemical aptasensor based on enzyme linked aptamer assay. Biosens Bioelectron 31(1):363–368
Wen YL, Pei H, Wan Y, Su Y, Huang Q, Song SP, Fan CH (2011) DNA nanostructure-decorated surfaces for enhanced aptamer-target binding and electrochemical cocaine sensors. Anal Chem 83(19):7418–7423
Zuo XL, Xiao Y, Plaxco KW (2009) High specificity, electrochemical sandwich assays based on single aptamer sequences and suitable for the direct detection of small-molecule targets in blood and other complex matrices. J Am Chem Soc 131(20):6944–6945
Wu CC, Yan L, Wang CM, Lin HX, Wang C, Chen X, James Yang CY (2010) A general excimer signaling approach for aptamer sensors. Biosens Bioelectron 25(10):2232–2237
He JL, Wu ZS, Zhou H, Wang HQ, Jiang JH, Shen GL, Yu RQ (2010) Fluorescence aptameric sensor for strand displacement amplification detection of cocaine. Anal Chem 82(4):1358–1364
Du Y, Li BL, Guo SJ, Zhou ZX, Zhou M, Wang EK, Dong SJ (2011) G-Quadruplex-based DNAzyme for colorimetric detection of cocaine: using magnetic nanoparticles as the separation and amplification element. Analyst 136(3):493–497
Wong ELS, Chow E, Gooding JJ (2005) DNA recognition interfaces: the influence of interfacial design on the efficiency and kinetics of hybridization. Langmuir 21(15):6957–6965
Levicky R, Herne TM, Tarlov MJ, Satija SK (1998) Using self-assembly to control the structure of DNA monolayers on gold: a neutron reflectivity study. J Am Chem Soc 120(38):9787–9792
Liu DY, Daubendiek SL, Zillman MA, Ryan K, Kool ET (1996) Rolling circle DNA synthesis: small circular oligonucleotides as efficient templates for DNA polymerases. J Am Chem Soc 118(7):1587–1594
Lizardi PM, Huang XH, Zhu ZR, Bray-Ward P, Thomas DC, Ward DC (1998) Mutation detection and single-molecule counting using isothermal rolling-circle amplification. Nat Genet 19(3):225–232
Cheng W, Yan F, Ding L, Ju HX, Yin YB (2010) Cascade signal amplification strategy for subattomolar protein detection by rolling circle amplification and quantum dots tagging. Anal Chem 82(8):3337–3342
Ding CF, Wang NN, Zhang J, Wang ZF (2013) Rolling circle amplification combined with nanoparticle aggregates for highly sensitive identification of DNA and cancer cells. Biosens Bioelectron 42:486–491
Cheng YQ, Zhang X, Li ZP, Jiao XX, Wang YC, Zhang YL (2009) Highly sensitive determination of microRNA using target-primed and branched rolling-circle amplification. Angew Chem Int Ed 121(18):3318–3322
Cheng W, Ding L, Chen YL, Yan F, Ju HX, Yin YB (2010) A facile scanometric strategy for ultrasensitive detection of protein using aptamer-initiated rolling circle amplification. Chem Commun 46(36):6720–6722
Neves MAD, Reinstein O, Saad M, Johnson PE (2010) Defining the secondary structural requirements of a cocaine-binding aptamer by a thermodynamic and mutation study. Biophys Chem 153(1):9–16
Schweitzer B, Wiltshire S, Lambert J, O’Malley S, Kukanskis K, Zhu ZR, Kingsmore SF, Lizardi PM, Ward DC (2000) Immunoassays with rolling circle DNA amplification: a versatile platform for ultrasensitive antigen detection. Proc Natl Acad Sci U S A 97(18):10113–10119
Acknowledgments
This work was funded by the National Natural Science Foundation of China (21075141) and (81101638), the Science and Technology Plan Project of Yuzhong District of Chongqing (20120212) and Natural Science Foundation Project of CQ (CSTC2013jjB10019).
Author information
Authors and Affiliations
Corresponding author
Additional information
Bo Shen and Jianbo Li contributed equally to this work.
Rights and permissions
About this article
Cite this article
Shen, B., Li, J., Cheng, W. et al. Electrochemical aptasensor for highly sensitive determination of cocaine using a supramolecular aptamer and rolling circle amplification. Microchim Acta 182, 361–367 (2015). https://doi.org/10.1007/s00604-014-1333-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-014-1333-3