Abstract
Purpose
Aggressive giant cell tumor (GCT) is an important subtype of GCT and is relatively rare in the spine. There is little published information regarding this subject. The objective of our study is to discuss prognostic factors for primary aggressive GCT in the spine.
Methods
A retrospective study of patients who had primary aggressive GCT in the spine and underwent surgical treatment at our center between 2000 and 2012 was conducted. Univariate and multivariate analyses were performed to identify the factors that might affect recurrence. T test, Chi-square test and rank sum test were used to analyze a single factor for recurrence, and factors with P ≤ 0.1 were subjected to multivariate analyses by binary logistic regression analyses. P values of ≤0.05 were considered statistically significant.
Results
A total of 71 patients with primary aggressive GCT in the spine were included in the study. The mean follow-up period was 73.9 (range 23–167) months. Recurrence was detected in 24 patients after the initial surgery in our center with a recurrence rate of 33.8 %. The statistical analyses suggested that age more than 40 years, pathology grade III, total en bloc spondylectomy, and bisphosphonate treatment were independent prognostic factors for recurrence of primary aggressive GCT in the spine.
Conclusion
Total en bloc spondylectomy together with bisphosphonate treatment could significantly decrease recurrence risk of primary aggressive GCT in the spine. Jaffe grade III was an adverse prognostic factor for recurrence, while age less than 40 years was a favorable prognostic factor.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Giant cell tumor (GCT) of bone is a common benign tumor and typically arises in the metaphyseal regions of long bones [1, 2]. It usually occurs between the ages of 20–40 years with female gender predominance [2, 3]. GCT is composed of three major cell types and spindle-like stromal cell is the neoplastic component [4]. Spine is a relatively rare site for GCT which accounts for 1.4–9.4 % of primary spine tumors [2, 3, 5]. Although classified as benign, GCT has the characteristic of local aggressiveness and malignant potential with lung metastasis occasionally reported in the literature [3, 6–8]. Complete resection is advocated for spinal GCT, but anatomic constraints hinder such efforts and overall recurrence rate ranges from 22.4 to 41.7 % [5, 9].
GCT can be classified as nonaggressive, intermediate, or aggressive based on their biological behavior [10, 11]. Aggressive GCT exhibits an extraosseous extension, articular involvement, or extensive metaphyseal destruction [12, 13]. As to spinal GCT, aggressive GCT refers to those with extraosseous involvement (layer A by WBB system) [14]. There were some case series of spinal GCT focusing on surgical options and recurrence [2, 3, 5, 9, 11, 15–17], but the information about aggressive GCT was quite rare. Aggressive GCT in the extremity was considered to be more likely to recur and should be treated with radical resection [12, 13]. For aggressive GCT in the spine, extraosseous involvement makes the tumor hard to deal with and complete resection more difficult to achieve. Therefore, a systematic analysis of a large case series to analyze treatment and outcome of primary aggressive GCT in the spine is very essential. The aim of this study is to identify the prognostic factors of primary aggressive GCT in the spine by focusing specifically on the postoperative recurrence (Fig. 1).
Materials and methods
Patients
The study was performed at the bone tumor center of Changzheng Hospital (Shanghai, China). In order to identify diagnostic and prognostic criteria based on the study of a homogeneous series, patient selection was restricted to fulfill the following inclusion criteria: (1) patient presented with a Campanacci grade III giant cell tumor in the spine [10]. (2) Patients had not received surgical intervention and any other treatment before admission into our institution. (3) Patients were consecutive and received treatment by surgical team led by Doctor Xiao. (4) Patients were admitted to our center and treated between January 2000 and May 2012. (5) Patients completed a minimum follow-up of 24 months or died before (most of recurrences occur within 2 years after surgery [2, 9]).
A total of 71 were selected according to the inclusion criteria. The study was approved by the hospital ethics committee, and written informed consent was obtained from all patients.
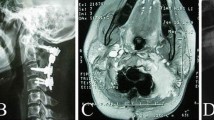
The clinical and pathologic data of all patients were retrieved from the maintained database of our center. The diagnosis of aggressive GCT was confirmed by pathology and preoperative imaging data in all patients (Fig. 2). The preoperative neurologic status was classified according to the Frankel score [18], and the pathologic features were evaluated by Jaffe grading system [19]. The individualized surgical strategy was decided for each patient according to Weinstein–Boriani–Biagini (WBB) system [14]. All patients were surgically treated by subtotal resection, piecemeal total spondylectomy, or total en bloc spondylectomy, which were performed by posterior approach, anterior approach, or a combined way [20, 21].
The study tried to identify prognostic factors for patients with aggressive GCT in the spine by focusing on recurrence after the initial surgery in our center. The recurrence status was confirmed on the basis of clinical manifestations and imaging findings in outpatient follow-up or pathologic evaluation of second surgery. All patients were followed up at 3, 6 and 12 months after surgery, every 6 months for the next 2 years, and then annually for life [22, 23]. Follow-up data were obtained from follow-up visits and telephone interviews.
Statistical method
Quantitative data are described by median (range), and qualitative data are described as counts and percentages. The univariate and multivariate analyses of various clinical factors were performed to identify independent variables that could predict recurrence. The patient factors were age, gender, duration of symptoms and preoperative Frankel score. The tumor factors were location, number of involved segments, aneurysmal bone cyst (ABC), and pathologic features. The treatment factors were preoperative selective artery embolism (PAE), surgical approach, resection mode, local treatment, intraoperative blood loss, bisphosphonate treatment, and adjuvant radiotherapy. Data were analyzed using SPSS version 17.0. (SPSS Inc.). T test, Chi-square test and rank sum test were used to analyze the single factors for recurrence. Factors with P ≤ 0.1 were subjected to multivariate analysis by binary logistic regression analysis. P values of ≤0.05 were considered statistically significant.
Results
Patient features
The series comprised 28 men and 43 women, with a mean age of 33 (median 32, range 13–69) years. Of these patients, 45 (63.4 %) patients ranged from 20 to 40 years old. Lesions were detected in the cervical spine (n = 21), thoracic spine (n = 21), lumbar spine (n = 8), and the sacrum (n = 21). Tumor with one segmental involvement occurred in 35 cases, while the other 36 cases had tumors with multisegmental involvement. Localized pain in the spine was the most consistent complaint, and duration of symptoms ranged from 1 to 108 (mean 8, median 4) months. Fifty-six patients (78.9 %) had varying degrees of cord compression at diagnosis, and aneurysmal bone cyst (ABC) was found in ten patients.
The mean follow-up period was 73.9 (median 68.0, range 23–167) months. Recurrence was detected in 24 patients (33.8 %) after the initial surgery in our center. Two patients died in the follow-up period, with postoperative survival time of 23 and 29 months, respectively. The mean time from surgery to recurrence was 16.75 (median 12.5, range 2–67) months. Of these patients, 19 (79.2 %) developed recurrence within 24 months.
Univariate analysis of prognostic factors for recurrence
Postoperative recurrence is not uncommon for spinal GCT and incomplete resection is considered to be the main reason [2, 3, 9]. Aggressive GCT in the spine is considered to be more likely to recur, but detailed recurrence rate and related prognostic factors are rarely reported in the literature. The overall recurrence rate was 33.8 % in our series, and the univariate analysis of clinical factors is shown in Table 1. In this study, recurrence rate was significantly higher in patients more than 40 years old (P < 0.0005). Patients with multisegmental involvement had higher recurrence rate than those with one segmental involvement (P = 0.015). Recurrence rate was significantly different between patients with Jaffe grade I–III (P < 0.0005).
Subtotal resection was applied in 31 patients, piecemeal total spondylectomy in 27 patients, and total en bloc spondylectomy in 13 cases. Recurrence rate was obviously different between patients with three different resection modes (P < 0.0005). Cisplatin and methotrexate were used as local treatment to soak the surgery field after tumor resection in our center [2, 23]. But no significant difference of recurrence rate was found in patients who received local treatment (P = 0.773). The mean intraoperative blood loss was 2,420 (median 2,500, range 100–6,500) ml. There was no significant difference of recurrence rate in patients with intraoperative blood loss >2,000 ml and those ≤2,000 ml (P = 0.948). PAE was used in 33 patients to reduce intraoperative blood loss, but no significant difference in recurrence rate was observed (P = 0.353).
Bisphosphonate is confirmed to control human GCT cells and relieve cancer pain in clinical treatment [24–27]. Zoledronic acid and incadronate disodium which are bisphosphonate are used to control osteolytic lesions of bone tumors in our center [2, 23]. Thirty patients received bisphosphonate treatment in our series and significantly lower recurrence rate was found in them (P = 0.002). Adjuvant radiotherapy (30–55 Gy) was administered postoperatively in 40 patients, but no significant difference in recurrence rate was found (P = 0.442). There was no significant difference in other factors of gender, duration of symptoms, ABC, location, preoperative Frankel score, follow-up period and surgical approach.
Multivariate analysis of prognostic factors
Potential prognostic factors extracted by univariate analysis were age group II, number of segmental involvement, Jaffe pathology grade, resection mode, and bisphosphonate treatment. They were submitted to multivariate analysis by binary logistic regression.
The risk of recurrence was significantly increased in patients more than 40 years (P = 0.003, HR = 17.718), and meanwhile patients with Jaffe grade III had significantly higher recurrence rate (P = 0.008, HR = 1,747.9). Total en bloc spondylectomy and bisphosphonate treatment significantly decreased the risk of recurrence. The hazard ratio (HR) for bisphosphonate treatment was 0.019 (P = 0.007). Total en bloc spondylectomy was significantly associated with lower recurrence rate (P = 0.021, HR = 0.015).
The above results showed that age group II, Jaffe grade III, total en bloc spondylectomy, and bisphosphonate treatment were independent prognostic factors for primary aggressive GCT in the spine. Details are listed in Table 2.
Discussion
GCT is a common primary bone tumor with local aggressiveness [1, 2]. Spine is a relatively rare site, but poses great challenge for the treatment of GCT [2, 3, 9]. As an important subtype, aggressive GCT in the spine should be carefully treated but the detailed information about it is still unclear. In this study, we analyzed the clinical data of 71 patients with primary aggressive GCT in the spine and performed statistical analyses to investigate the prognostic factors affecting postoperative recurrence. The results suggested that age group II, Jaffe grade III, total en bloc spondylectomy, and bisphosphonate treatment were independent prognostic factors.
In our series, 63.4 % of patients with primary aggressive GCT were between 20 and 40 years with female gender predominance, which was similar to the findings of previous reports about spinal GCT [1–3, 9, 15]. Localized pain in the spine was the most consistent complaint and 74.6 % of cases endured less than 6 months prior to admission. In our study, primary aggressive GCT was less likely to infringe upon lumbar spine and there is no difference in the distribution of cervical spine, thoracic spine and sacrum. Multisegmental involvement was found in more than half of cases and 78.9 % of cases endured neurologic disorders. Aneurysmal bone cyst is an expansile cystic lesion commonly secondary to GCT and was found in ten cases in our series. The results of our study showed that patient age was found to be a prognostic factor for primary aggressive GCT in the spine and patients more than 40 years had significantly higher recurrence rate. But gender, duration of symptoms, location, number of segmental involvement, preoperative Frankel score, and ABC were not independent prognostic factor.
Histopathologically, aggressive GCT can be classified by Jaffe grade system into three categories: grade I without appreciable atypia of stromal cells, few mitoses, none abnormal; grade II with stromal cells showing only slight or more marked atypia, but not enough to justify a diagnosis of malignancy; and grade III with obvious features of malignancy [7, 19]. Sanerkin [7] considered that this grading system was unable to predict the clinical behavior and prognosis of GCT. But in our series, we found that the Jaffe grade III was closely associated with higher recurrence risk for primary aggressive GCT in the spine.
Surgery with the aim at preserving functionality, relieving pain, controlling local recurrence and promising a prolonged survival is the foundational treatment strategy for aggressive GCT in the spine. The surgical procedures applicable to spinal column include the simplest subtotal resection (curettage), piecemeal total spondylectomy, and the most complex total en bloc spondylectomy [14]. Some authors suggested that aggressive GCT in long bones should be best treated with total en bloc spondylectomy to minimize the risks of local recurrence [14, 28–30]. The findings in our study suggested that total en bloc spondylectomy could significantly decrease recurrence rate of aggressive GCT in the spine.
Piecemeal total spondylectomy is associated with a possibility of tumor cell contamination in the surgical field and is turned out to be superior to subtotal resection for spinal GCT in our previous research [2]. But we found it could not significantly reduce recurrence rate of primary aggressive GCT in the spine when compared with subtotal resection. The possible reason might be that extraosseous involvement makes tumor cell contamination more likely to happen and hence causes recurrence. So total en bloc spondylectomy which is hard to achieve because of anatomical complexity of the spine is confirmed to have excellent prognosis for aggressive GCT in the spine.
Total en bloc spondylectomy is a procedure aimed at surgically removing a tumor in a single, intact piece, with fully tumor-free margins [14]. However, it is not always feasible because of anatomical complexity of the spine, especially in the challenging region such as upper cervical and lower lumbar spine. Careful surgical planning according to Tomita classification, Enneking stage, and WBB systems is of great importance for total en bloc spondylectomy [23]. Total en bloc spondylectomy is considered to cause more complications than the other two surgical procedures and the complications in the spine can be divided into major and minor according to McDonnell [31–33].
Bisphosphonate is confirmed to control GCT cells in vitro studies [24, 25], and it could significantly relieve cancer pain and the progression of GCT in clinical treatment [26, 27]. Tse et al. [34] found that bisphosphonate could be used as an adjuvant therapy to reduce the local recurrence rate of GCT of the extremity after surgery. We further confirmed in previous study that long-term use of bisphosphonate-assisted surgery to reduce recurrence rate of GCT in the mobile spine [2]. In this study, we found that bisphosphonate treatment could significantly reduce recurrence risk of primary aggressive GCT in the spine and served as an effective adjuvant therapy after surgery. Furthermore, pain control effect of bisphosphonate was also verified in the treatment of spinal aggressive GCT in our center.
Radiotherapy and chemotherapy were used as adjuvant therapies for spinal GCT. Postoperative radiotherapy was considered to provide excellent local control and even reduce recurrence risk of GCT in the extremity [35–37]. The intraoperative local treatments were widely used and their positive effect was confirmed in extremity GCTs [38–40]. But in our study, both postoperative radiotherapy and intraoperative local treatment were not independent prognostic factors for primary aggressive GCT in the spine.
PAE could reduce intraoperative blood loss and improve the excision rate and the safety of surgery [41, 42], but it could not improve the prognosis of aggressive GCT in the spine in our series. The same results were achieved for intraoperative blood loss and surgical approach.
The nature of a retrospective study is the limitation of this research. But aggressive GCT in the spine is a topic rarely reported and as far as we know, our series is the largest series to date until now. Furthermore, the overall follow-up time is quite long with a 2-year minimum limit, which increases the credibility of the results.
In conclusion, total en bloc spondylectomy together with bisphosphonate treatment could significantly decrease the recurrence risk of primary aggressive GCT in the spine. Pathology grade III was an adverse prognostic factor for recurrence, while age less than 40 years is a favorable prognostic factor for aggressive GCT in the spine.
References
Gupta R, Seethalakshmi V, Jambhekar NA et al (2008) Clinicopathologic profile of 470 giant cell tumors of bone from a cancer hospital in western India. Ann Diagn Pathol. 12(4):239–248
Xu W, Li X, Huang W et al (2013) Factors affecting prognosis of patients with giant cell tumors of the mobile spine: retrospective analysis of 102 patients in a single center. Ann Surg Oncol 20(3):804–810
Wu Z, Yang X, Xiao J et al (2011) Aneurysmal bone cyst secondary to giant cell tumor of the mobile spine: a report of 11 cases. Spine (Phila Pa 1976) 36(21):E1385–E1390
Wu Z, Yin H, Liu T et al (2014) MiR-126-5p regulates osteoclast differentiation and bone resorption in giant cell tumor through inhibition of MMP-13. Biochem Biophys Res Commun. 443(3):944–949
Sanjay BK, Sim FH, Unni KK et al (1993) Giant-cell tumours of the spine. J Bone Joint Surg Br 75(1):148–154
Dahlin DC (1985) Caldwell lecture. Giant cell tumor of bone: highlights of 407 cases. AJR Am J Roentgenol 144(5):955–960
Sanerkin NG (1980) Malignancy, aggressiveness, and recurrence in giant cell tumor of bone. Cancer 46(7):1641–1649
Rock MG, Pritchard DJ, Unni KK (1984) Metastases from histologically benign giant-cell tumor of bone. J Bone Joint Surg Am 66(2):269–274
Boriani S, Bandiera S, Casadei R et al (2012) Giant cell tumor of the mobile spine: a review of 49 cases. Spine (Phila Pa 1976) 37(1):E37–E45
Campanacci M, Baldini N, Boriani S et al (1987) Giant-cell tumor of bone. J Bone Joint Surg Am 69(1):106–114
Enneking WF (1986) A system of staging musculoskeletal neoplasms. Clin Orthop Relat Res 204:9–24
Puloski SK, Griffin A, Ferguson PC et al (2007) Functional outcomes after treatment of aggressive tumors in the distal radius. Clin Orthop Relat Res 459:154–160
Flouzat-Lachaniette CH, Babinet A, Kahwaji A et al (2013) Limited arthrodesis of the wrist for treatment of giant cell tumor of the distal radius. J Hand Surg Am 38(8):1505–1512
Boriani S, Weinstein JN, Biagini R (1997) Primary bone tumors of the spine. Terminology and surgical staging. Spine (Phila Pa 1976) 22(9):1036–1044
Martin C, McCarthy EF (2010) Giant cell tumor of the sacrum and spine: series of 23 cases and a review of the literature. Iowa Orthop J 30:69–75
Junming M, Cheng Y, Dong C et al (2008) Giant cell tumor of the cervical spine: a series of 22 cases and outcomes. Spine (Phila Pa 1976) 33(3):280–288
Savini R, Gherlinzoni F, Morandi M et al (1983) Surgical treatment of giant-cell tumor of the spine. The experience at the Istituto Ortopedico Rizzoli. J Bone Joint Surg Am 65(9):1283–1289
Frankel HL, Hancock DO, Hyslop G et al (1969) The value of postural reduction in the initial management of closed injuries of the spine with paraplegia and tetraplegia. I. Paraplegia 7(3):179–192
Jaffe HL, Lichtenstein L, Portis RB (1940) Giant cell tumor of bone. Its pathologic appearance, grading, supposed variants and treatment. Arch Pathol 30:993–1031
Boriani S, Biagini R, De Iure F et al (1996) En bloc resections of bone tumors of the thoracolumbar spine. A preliminary report on 29 patients. Spine (Phila Pa 1976) 21(16):1927–1931
Tomita K, Kawahara N, Baba H et al (1997) Total en bloc spondylectomy. A new surgical technique for primary malignant vertebral tumors. Spine (Phila Pa 1976) 22(3):324–333
Yin H, Zhang D, Wu Z et al (2014) Desmoplastic fibroma of the spine: a series of 12 cases and outcomes. Spine J 14(8):1622–1628
Yin H, Zhou W, Meng J et al (2014) Prognostic factors of patients with spinal chondrosarcoma: a retrospective analysis of 98 consecutive patients in a single center. Ann Surg Oncol 21(11):3572–3578
Cheng YY, Huang L, Lee KM et al (2004) Bisphosphonates induce apoptosis of stromal tumor cells in giant cell tumor of bone. Calcif Tissue Int 75(1):71–77
Chang SS, Suratwala SJ, Jung KM et al (2004) Bisphosphonates may reduce recurrence in giant cell tumor by inducing apoptosis. Clin Orthop Relat Res 426:103–109
Balke M, Campanacci L, Gebert C et al (2010) Bisphosphonate treatment of aggressive primary, recurrent and metastatic Giant Cell Tumour of Bone. BMC Cancer 29(10):462
Zhang W, Zhang Y, Li P et al (2011) Administration of sodium ibandronate in the treatment of complicated giant cell tumor of the spine. Spine (Phila Pa 1976) 36(17):E1166–E1172
Vander Griend RA, Funderburk CH (1993) The treatment of giant-cell tumors of the distal part of the radius. J Bone Joint Surg Am 75(6):899–908
Noellert RC, Louis DS (1985) Long-term follow-up of nonvascularized fibular autografts for distal radial reconstruction. J Hand Surg Am 10(3):335–340
Murray JA, Schlafly B (1986) Giant-cell tumors in the distal end of the radius. Treatment by resection and fibular autograft interpositional arthrodesis. J Bone Joint Surg Am 68(5):687–694
Boriani S, Bandiera S, Donthineni R et al (2010) Morbidity of en bloc resections in the spine. Eur Spine J 19(2):231–241
Bandiera S, Boriani S, Donthineni R et al (2009) Complications of en bloc resections in the spine. Orthop Clin North Am 40(1):125–131
McDonnell MF, Glassman SD, Dimar JR 2nd et al (1996) Perioperative complications of anterior procedures on the spine. J Bone Joint Surg Am 78(6):839–847
Tse LF, Wong KC, Kumta SM et al (2008) Bisphosphonates reduce local recurrence in extremity giant cell tumor of bone: a case-control study. Bone 42(1):68–73
Raskin KA, Schwab JH, Mankin HJ et al (2013) Giant cell tumor of bone. J Am Acad Orthop Surg 21(2):118–126
Shi W, Indelicato DJ, Reith J et al (2013) Radiotherapy in the management of giant cell tumor of bone. Am J Clin Oncol 36(5):505–508
Griffin AM, Ferguson PC, Catton CN et al (2012) Long-term outcome of the treatment of high-risk tenosynovial giant cell tumor/pigmented villonodular synovitis with radiotherapy and surgery. Cancer 118(19):4901–4909
Zhen W, Yaotian H, Songjian L et al (2004) Giant-cell tumour of bone. The long-term results of treatment by curettage and bone graft. J Bone Joint Surg Br 86(2):212–216
Dürr HR, Maier M, Jansson V et al (1999) Phenol as an adjuvant for local control in the treatment of giant cell tumour of the bone. Eur J Surg Oncol 25(6):610–618
Malawer MM, Bickels J, Meller I et al (1999) Cryosurgery in the treatment of giant cell tumor. A long-term followup study. Clin Orthop Relat Res 359:176–188
Ozkan E, Gupta S (2011) Embolization of spinal tumors: vascular anatomy, indications, and technique. Tech Vasc Interv Radiol 14(3):129–140
Vetter SC, Strecker EP, Ackermann LW et al (1997) Preoperative embolization of cervical spine tumors. Cardiovasc Intervent Radiol 20(5):343–347
Acknowledgments
This work was supported by the Shanghai Science Foundation of China (12DZ2295103) and National Natural Science Foundation of China (Grants 81402222, 81402223).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
H. Yin, X. Yang, and W. Xu contributed equally to this work, and all should be considered first authors.
Rights and permissions
About this article
Cite this article
Yin, H., Yang, X., Xu, W. et al. Treatment and outcome of primary aggressive giant cell tumor in the spine. Eur Spine J 24, 1747–1753 (2015). https://doi.org/10.1007/s00586-015-3777-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-015-3777-5