Abstract
Background
The surgical approach to the colonic splenic flexure cancer (SFC) is yet to be technically standardized. The aim of this study has been to retrospectively evaluate the oncologic long-term results of our cases comparing our data with other authors’ experiences.
Methods
Clinical data of patients with SFC operated on at our institute were retrospectively analyzed. The laparoscopic approach was used in the whole series, with limited resection distally and proximally to splenic flexure and the origin ligation of left colic artery and left branch of middle colic artery. Data on the oncological long-term safety were compared to our laparoscopic series of extended right colectomy for proximal two-third transverse colon cancer and high anterior resections for sigmoid-high rectal cancer and to the main evidences in the literature, found after a comprehensive review.
Results
From March 2008 to May 2018, we completed 53 laparoscopic splenic flexure resections (26 female and 27 male, age 71.5 ± 15.4 years). The conversion rate was 3.8%. Most of the cases were stage II (pT3 73.5%, the mean number of lymph nodes harvested was 19.1, with positivity for malignancy in 45.3%). During the FU (43.5 months), 2 patients dropped out. Out of the 51 residual cases, 37 were alive (72.5%) and 14 are deceased (27.5%).
Discussion
Compared to the literature, our survival rate does not show significant differences. The other oncological outcomes seem to be comparable with the data evaluated.
Conclusions
More extended resections seem not to confer an increase of the overall survival rate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Colorectal cancer (CRC) is one of the most common gastrointestinal malignancy (the fourth cancer for incidence worldwide), with more than 80% of the left section cancer occurs in the sigmoid. Cancers located between distal transverse and proximal descending colon, usually known as “splenic flexure cancers” (SFC), are on the other hand much less common [1]. According with most of the Authors, the SF is precisely defined as the “junction between the distal third of the transverse colon and the first part of the descending colon within 10 cm from the anatomic flexure” [2].
Due to its rarity, the clinical-pathological features of SFC are poorly defined and surgical treatment is yet to be technically standardized. In particular, there are many controversies on the appropriate extent of the colic resection and lymph node dissection; the risk and consequences of inadvertent splenectomy; and the type of anastomosis, whether terminal–terminal or lateral–lateral and, in case of laparoscopy, intra- or extracorporeal [3,4,5]. Such controversies have their roots in the anatomical and embryological complexity of this region.
From the embryological point of view, the midgut, which is supplied by the superior mesenteric artery (SMA), gives rise to the intestine comprised between the duodenum and the distal transverse colon at the junction between the proximal two-third and the distal one-third. The hindgut becomes the distal transverse colon, descending colon, and rectum and it is served by the inferior mesenteric artery (IMA). Venous and lymphatic ducts mirror their arterial counterparts and follow the same embryological division [6].
From the anatomical point of view, regarding the blood supply, the middle colic artery (MCA), arising from the SMA, anatomizes with its left branch the ascending branch of the left colic artery (LCA) which arises from the IMA. However, it is not uncommon to find another artery supplying the left colic flexure, which arises directly from the Aorta or from the SMA, with a reported incidence of about 14% [7, 8].
Considering all these aspects, it is clear that the splenic flexure is located in a sort of border area and is supplied both the by SMA and IMA. Therefore, the authors according to their interpretation of the oncological consequences of these particularities have addressed their management of such tumor in different ways. Some have preferred a more extended colectomy, such as right/transverse resection with ileocolic anastomosis or left hemicolectomy with colorectal anastomosis and division of IMA at the origin. Others have preferred a “segmental” resection defined as an excision of the anatomical SF with division of the LCA and the left branch of MCA, with a colocolic anastomosis [9].
The aim of this study has thus been to retrospectively evaluate the surgical and oncologic long-term results on a series of patients with SFC, treated exclusively with the single technique of laparoscopic segmental resection. Data have been subsequently compared to the results of the main studies in the literature where different techniques were chosen, and to different cases of the proximal two-third of transverse colon cancer (TCC) and sigmoid and high rectal cancer (SHRC) operated on at our hospital through the same selected timeframe.
Materials and methods
We have retrospectively selected from the electronic database all patients operated on for CRC in our Surgical Unit from March 2008 to May 2018. The diagnosis was made by colonoscopy and biopsy in all patients. Cancer staging was performed by CT scan of the chest and abdomen with and without contrast media. In case of partial colonoscopy due to the presence of blocked obstruction, the colon study was completed with a virtual colonoscopy. During traditional colonoscopy, tattooing with ink was made in all patients. After discharge, the patients were examined at least two times in the outpatient clinic for a period up to a month or more if necessary. The oncological follow-up (FU) was assessed by periodical physical examination, blood test analysis, ultrasonography, CT scan, colonoscopy, and positron emission tomography CT (PET-CT) according to the NCCN guidelines. These data were recorded in a computerized database which has been widely consulted in order to establish the oncological outcomes.
Out of these series, we firstly excluded the following categories: patients with right sided cancer, those with more than one CRC, those who underwent previous colic surgery, and those operated on in emergency setting. Among the remnants, we then excluded patients with cancer located in the first two-third of the transverse colon and those with left sided cancer in which the IMA had to be ligated at the origin due to the anatomical position. From the final group of patients, we considered solely those operated on with a segmental resection of the splenic flexure, with the ligature of the LCA and the left branch of MCA only. Finally, the few patients on whom an open operation was directly performed were considered not eligible for this study, due to its intent of single technique analysis (Fig. 1).
This series was examined for all personal, pathological, and clinical features. The main end-point of our work has been on oncological safety parameters such as the number of harvested lymph nodes, the overall survival (OS), and disease-free survival (DFS). Secondly, attention was paid to the outcome parameters of surgery such as operative time, major and minor complications (according to the Clavien–Dindo Scale). Data on the oncological safety and outcome parameters were compared to the main results in the literature which were found after a comprehensive review of PubMed, Cochrane Database and EMBASE, particularly focusing on studies that have reported more than 20 laparoscopic splenic flexure resection, either adopting the same technique presented in this series (named as Segmental Left Colectomy, SLC) or a technique with IMA ligation at the origin (named as Left Colectomy, LC).
Furthermore, the data were compared, only considering laparoscopic operations, to cases of TCC undergoing extended right colectomy (ERC) and to cases of SHRC undergoing high anterior resection (HAR) operated on from March 2008 to May 2018 at our institution.
All the patients belonging to the studied series were treated according to the protocols outlined, particularly in light of the provisions of the 1964 Helsinki declaration and its later amendments. A written informed consent was obtained from all the patients for the publication of their clinical data. The research was supervised at all stages by our local Ethics Committee, whose purpose is to protect the rights and welfare of human subjects during their participation to clinical studies. This article does not contain any studies on animals performed by any of the Authors.
Surgical technique
All patients received perioperative antimicrobial and antithrombotic prophylaxis. All procedures were performed by experienced surgeons in laparoscopic colorectal surgery.
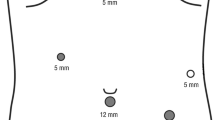
For the laparoscopic approach, the operator was placed on the right side of the patient and the first assistant on his right. If a second assistant was present, he positioned by the main operator’s left side. The operation begins using five ports. The first one (Hasson) was placed on the midline through an open access via the umbilicus. A 10–12-mm trocar was positioned in the right lower quadrant for the operator’s right hand. A 5 mm for his left hand was inserted in the right hypocondrium. A 5-mm trocars was finally placed in the epigastrium (midline or immediately on the left) and another 5 mm in the left flank (Fig. 2). Anti-Trendelenburg position was used as much as possible and the patient was rotated on his/her right flank in order to let the small bowel loops slide in the opposite side of the operation field. The inferior mesenteric vein was identified at the inferior border of the pancreas and the peritoneum was incised just below the vein to create the plane between the Toldt’s fascia and Gerota’s fascia.
Going down with the peritoneum dissection, the IMA and left colic artery (LCA) were exposed. The LCA was isolated and divided at its origin. The dissection continued medial-to-lateral using the inferior mesenteric vessels as landmarks, trying to identify and preserve the ureter at its origin and the gonadic vessels. A gauze was placed in order to protect these retroperitoneal structures. The descending colon was then extensively mobilized from the left abdominal wall.
Afterwards, the omental bursa was opened by dissecting the greater omentum while preserving the gastroepiploic arcade. The middle colic artery was then identified and its left branch dissected at its origin. The transverse mesocolon was divided at the inferior border of the pancreas after clipping the inferior mesenteric vein. The mobilization of the splenic flexure was then realized by a lateral-to-medial approach. Sometimes it was possible to find an accessory artery for the splenic flexure. This artery originates either directly from the aorta or from the SMA via MCA. If it was present, this vessel was divided at the inferior border of the pancreas. Finally, a small mini-laparotomy was performed in the left hypocondrium and the mobilized colon was pulled out after a placement of a skin protector. The SF of the externalized colon was resected and an end-to-end anastomosis was manually performed. Usually a 19fr drain was placed in the left parietocolic gutter.
Results
Since March 2008 up to May 2018, we have completed 53 laparoscopic partial splenic flexure resections for SFC.
Personal and pathological features of patients are summarized in Table 1. As evident, most of the patients were in the eight decades, there was not any sex or tumor location prevalence. Moreover, the most frequently found stage was an advanced one (T3).
From the oncological point of view, it is to be noted that as much as 16.9% of patients had a number of harvested lymph nodes less than the recommended quantity of 12 (Table 2). In one case, we found a positive proximal margin at the pathological examination of the specimen (R1 resection). The patient was reoperated and an extended right hemicolectomy was performed.
Out of the 53 patients included in the study, 2 (3.7%) dropped out during the FU period, 37 (69.8%) were alive with no disease recurrence, and 14 (26.4%) deceased at a mean FU of 43.5 months. However, only 10 of the latter patients (18.9%) died for the disease progression with a median disease-free survival (DFS) of 13.7 months. Out of those, 3 had peritoneal carcinomatosis and 7 had liver metastases, of whom 1 was positive for lung metastases too.
Outcomes of mortality related to demographics and oncological features are summarized in Table 3. It could be noticed that there is a statistically significant difference between the survival related to nodal positivity. It is to be noted that 6 patients with less than 12 lymph nodes harvested have an AJCC stage of I or II.
The Kaplan–Meier survival probability curve is shown in Fig. 3, with time of FU expressed in months.
The operatory and post-operative outcome data are summarized in the Table 4 and compared with the outcomes of other authors.
The conversion rate was 3.8% (2 procedures converted for anatomical complexity).
Complication rate was of about 20%, but only in 5.6% of those cases an invasive maneuver including a reintervention was required. The indication of surgery was an anastomotic leakage in two patients which required the creation of a stoma in both cases. In another case, reoperation was due to an early small bowel occlusion.
In one case during the mobilization of the splenic flexure, an invasion of the tail of the pancreas was suspected and an intraoperative histological examination was performed. This confirmed the adenocarcinoma invasion and a distal spleno-pancreatectomy was executed alongside to the colic resection. In total, 3 splenectomies were performed, the other 2 for bleeding.
There was no 30-day post-operative mortality.
In Table 5, the comparison between our oncologic outcomes and those of the literature is summarized, from which it emerges that our series is one of the most numerous and also that the mean FU is one of the shortest. Our data demonstrate a DFS of 69.8% and an OS of 69.8%.
Table 6 shows the comparison between cases of SF resection versus ERC for cancers of the proximal two-third of the transverse colon, and versus HAR for cancers of sigmoid colon and high rectum. As it is shown, the only statistically significant difference can be noticed for the number of patients with less than twelve lymph nodes harvested compared to ERC.
Discussion
Compared to the literature, which includes different surgical techniques for the treatment of cancers of this site, our survival rate does not show significant differences.
In our survival analysis, the only statistically significant difference is shown between patients with positive lymph nodes (N+) and negative lymph nodes (N−). Interestingly, there is no difference in survival between patients with a number of harvested lymph nodes more or less than 12. This could be due to the earlier stage of the patients with less than 12 lymph nodes harvested.
The series was subsequently compared to cases of TCC and SHRC operated on only with laparoscopic technique in the same period of time in order to evaluate the oncological outcomes of more standardized operations. A higher percentage of OS and DSF are present in the group of SHRC. Thus a lower percentage of OS and DFS can be noticed for TCC with a higher mean number of harvested lymph nodes. The latter is obviously due to the more extended resection performed in ERC where the ligation of the ileocolic trunk, the gastroepiploic arcade, and the MCA allows collection of a higher number of lymph nodes. Anyway, the only statistically significant difference can be highlighted for the number of patients with less than 12 lymph nodes harvested.
It is worth to note that there is a lack of standardization in the definition of the resection. We use the definition of Nakagoe et al. [9] of SLC that is defined as LC by other authors [12]. Even the resection extended at the right colon is lacking of definitional clarity, considering that is defined by some authors as extended right colectomy and as subtotal colectomy by others. De’ Angelis et al. [10] reported in a comparative match study two different laparoscopic techniques: ERC and left colectomy (LC) (dividing the IMA at the origin). They reported an OS and a DFS throughout 5-year FU, respectively, of 72.8–75.1% and 67.1–66.7%, comparing these two techniques. No statistically significant differences were found between the two techniques regarding OS and DFS at a mean FU time of 70.9 months. Our series is one of the largest reporting outcomes of OS and DSF. In some cases such as Okuda et al. [11], a 5-year OS and DFS is divided for pT stage and for laparoscopic or open approach with a mean FU time of 84 months. They used the LC technique. OS reported for stage II is 94.6% for laparoscopy, 85.6% for open approach. In stage III they reported a 73.5% and 66.2%, respectively. DFS reported for stage II is 91.1% for laparoscopy and 85.6% for open. In stage III, they reported a DFS of 60.3% and 56.7%, respectively. In other cases, the reported technique was not the same for the entire series. Kim et al. [13] use the LC technique dividing the IMA at the origin only when suspected lymph nodes metastasis at this site are present, otherwise they divided the LCA at its origin. They reported an OS of 84.3% and 76% comparing laparoscopic and open technique, respectively, and DFS of 93.8% and 74.5%. No statistically significant differences were found at a median FU of 59 months.
For these reasons, it is not simple to carefully review and analyze the literature, and to make comparison between the series.
Regarding the lymph node harvesting, the mean number reported in this series is appropriate. It is worth to note that in 16.9% of cases, there is a number of lymph nodes lower than the 12 requested for adequate N staging. Considering that the mean number is higher than 12 and, as previously seen, literature reports do not show an inadequate number of lymph node harvesting in LC technique, this could be due to the impact of the pathologist in lymph node harvesting rather than to the surgical technique, given that the former has been shown to be more important than the specialty of the surgeon [16]. Nakashima et al. [14] performed a technique with the ligation of the LCA and the left branch of MCA at their origin only for stage II/III and the ligation of only one of these vessels at the origin for stage 0/I, reporting a number of harvested lymph nodes of 16.
The rate of tumor recurrence is also hardly comparable because of the differences between the mean FU. Pisani Cerretti et al. [3] reported one distant recurrence and one local (33 months of FU). Carlini et al. [12] also reported two recurrence but at a long-term FU of 58 months. Han et al. [15] reported three recurrence, one liver metastasis, one lung metastasis, and one liver metastasis with peritoneal carcinomatosis during a relatively short FU period of 21 months.
The number of surgeons performing this technique of segmental resection is rapidly increasing. There is a solid embryological and anatomical background that allows surgeons performing this technique [6, 9]. Performing this limited resection has the advantage of preserving large segments of the colon, such as the right colon and the ileocecal valve, lowering the risk of post-operatory diarrhea and reducing negative effects on nutrients’ absorption, and the rectosigmoid junction, allowing to preserve an important part of the colon that contributes to the defecatory mechanism. Other advantages are the lower or even absent risk of injury to the left ureter and the autonomic para-aortic nerves. Moreover, the main lymphatic flow is directed towards the LCA; consequently, the involvement of nodes along the MCA is neglectable so as to making it seemingly unnecessary to extend the resection on this side [9, 17, 18]. Furthermore, no statistically significant differences were found when the studies compared LC or SLC with ERC in OS [10, 19].
On the other hand, the use of indocyanine green for intraoperative sentinel lymph node mapping has been proposed for lymph node harvesting in early stages [20] to allow for the detection of the lymphatic flow of the segment of the colon. This kind of approach could be useful in the surgery of SF where the lymphatic flow is not always standardized.
Laparoscopic technique is technically demanding but as shown in this series no significant differences can be highlighted in the oncological outcomes vis à vis the techniques reported in the principal data of the literature and the series of our laparoscopic cases of ERC and HAR. We performed more than 50 procedures over 10 years by laparoscopy, making this as one of the largest series presented to date.
Undoubtedly, this study has some limitations because it is a single-institution retrospective study; however, the data reported, thanks to the large series, seem to be satisfactory. In our opinion, it is necessary to standardize the definition of the resections in relation to vessel ligation and the extent of the resection in order to set up multicenter prospective studies on this emerging topic.
Conclusions
Since the resection that we applied has a minimal extension compared to other literature series, our data shows that such surgical approach could be oncologically safe. More extended resections, possibly associated to splenectomy or distal pancreatectomy, seem not to confer an increase of the overall survival rate, even though further studies are mandatory.
References
Boyle P, Langman JS (2000) ABC of colorectal cancer: epidemiology. BMJ 321:805–808
Steffen C, Bokey EL, Chapuis PH (1987) Carcinoma of the splenic flexure. Dis Colon Rectum 30:872–874
Pisani Ceretti A, Maroni N, Sacchi M, Bona S, Angiolini MR, Bianchi P, Opocher E, Montorsi M (2015) Laparoscopic colonic resection for splenic flexure cancer: our experience. BMC Gastroenterol 15:76
McGory ML, Zingmond DS, Sekeris E, Ko CY (2007) The significance of inadvertent splenectomy during colorectal cancer resection. Arch Surg 142:668–674
Rao TUM, Reddy BS, Chilukoti S (2017) Splenic flexure cancer optimum level of vessel ligation. Int Surg J 4:1355–1359
Matsuda T, Iwasaki T, Sumi Y, Yamashita K, Hasegawa H, Yamamoto M, Matsuda Y, Kanaji S, Oshikiri T, Nakamura T, Suzuki S, Kakeji Y (2017) Laparoscopic complete mesocolic excision for right-sided colon cancer using a cranial approach: anatomical and embryological consideration. Int J Colorectal Dis 32:139–141
Rusu MC, Vlad M, Voinea LM, Curca GC, Sisu AM (2008) Detailed anatomy of a left accessory aberrant colic artery. Surg Radiol Anat 30:595–599
Hamabe A, Park S, Morita S, Tanida T, Tomimaru Y, Imamura H, Dono K (2018) Analysis of the vascular interrelationships among the first jejunal vein, the superior mesenteric artery, and the middle colic artery. Ann Surg Oncol 25:1661–1667
Nakagoe T, Sawai T, Tsuji T, Jibiki M, Ohbatake M, Nanashima A, Yamaguchi H, Yasutake T, Kurosaki N, Ayabe H, Ishikawa H (2001) Surgical treatment and subsequent outcome of patients with carcinoma of the splenic flexure. Surg Today 31:204–209
de’Angelis N, Hain E, Disabato M, Cordun C, Carra MC, Azoulay D, Brunetti F (2016) Laparoscopic extended right colectomy versus laparoscopic left colectomy for carcinoma of the splenic flexure: a matched case-control study. Int J Colorectal Dis 31:623–630
Okuda J, Yamamoto M, Tanaka K, Masubuchi S, Uchiyama K (2016) Laparoscopic resection of transverse colon cancer at splenic flexure: technical aspects and results. Updates Surg 68:71–75
Carlini M, Spoletini D, Castaldi F, Giovannini C, Passaro U (2016) Laparoscopic resection of splenic flexure tumors. Updates Surg 68:77–83
Kim MK, Lee IK, Kang WK, Cho HM, Kye BH, Jalloun HE, Kim JG (2017) Long-term oncologic outcomes of laparoscopic surgery for splenic flexure colon cancer are comparable to conventional open surgery. Ann Surg Treat Res 93:35–42
Nakashima M, Akiyoshi T, Ueno M, Fukunaga Y, Nagayama S, Fujimoto Y, Konishi T, Noaki R, Yamakawa K, Nagasue Y, Kuroyanagi H, Yamaguchi T (2011) Colon cancer in the splenic flexure: comparison of short-term outcomes of laparoscopic and open colectomy. Surg Laparosc Endosc Percutan Tech 21:415–418
Han KS, Choi GS, Park JS, Kim HJ, Park SY, Jun SH (2010) Short-term outcomes of a laparoscopic left hemicolectomy for descending colon cancer: retrospective comparison with an open left hemicolectomy. J Korean Soc Coloproctol 26:347–353
Evans MD, Barton K, Rees A, Stamatakis JD, Karandikar SS (2008) The impact of surgeon and pathologist on lymph node retrieval in colorectal cancer and its impact on survival for patients with Dukes’ stage B disease. Colorectal Dis 10:157–164
Watanabe J, Ota M, Suwa Y, Ishibe A, Masui H, Nagahori K (2017) Evaluation of lymph flow patterns in splenic flexural colon cancers using laparoscopic real-time indocyanine green fluorescence imaging. Int J Colorectal Dis 32:201–207
Vasey CE, Rajaratnam S, O’Grady G, Hulme-Moir M (2018) Lymphatic drainage of the splenic flexure defined by intraoperative scintigraphic mapping. Dis Colon Rectum 61:441–446
Odermatt M, Siddiqi N, Johns R, Miskovic D, Khan O, Khan J, Parvaiz A (2014) Short- and long-term outcomes for patients with splenic flexure tumours treated by left versus extended right colectomy are comparable: a retrospective analysis. Surg Today 44:2045–2051
Currie AC, Brigic A, Thomas-Gibson S, Suzuki N, Moorghen M, Jenkins JT, Faiz OD, Kennedy RH (2017) A pilot study to assess near infrared laparoscopy with indocyanine green (ICG) for intraoperative sentinel lymph node mapping in early colon cancer. Eur J Surg Oncol 43:2044–2051
Acknowledgements
The authors would like to thank Dr. Valeria Alessandra Testori for polishing the English content of the paper.
Funding
The authors received no specific funding for this work.
Author information
Authors and Affiliations
Contributions
MA conceived, designed and drafted the work, collecting data. CB, JM, PP, AV, contributed to the analysis, interpretation of data and revised it critically. All authors approved the final version to be published. All authors agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Disclosures
Ardu Massimiliano, Bergamini Carlo, Martellucci Jacopo, Prosperi Paolo, and Valeri Andrea have no conflict of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ardu, M., Bergamini, C., Martellucci, J. et al. Colonic splenic flexure carcinoma: is laparoscopic segmental resection a safe enough oncological approach?. Surg Endosc 34, 4436–4443 (2020). https://doi.org/10.1007/s00464-019-07221-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-019-07221-y