Abstract
The Amazon represents one of the most complex biomes in the world; however, the temporal variations in parasite community structure of fishes inhabiting this region remain poorly understood. Therefore, processes generating such variations are still unknown. The present study evaluated the long-term temporal variation of community structure of metazoan parasites of Pimelodus blochii collected in Iaco River, State of Acre (Southwestern Brazilian Amazon). A total of 196 parasites were collected over a 6-year period (2012–2017). Twenty-four different taxa of parasites were found, of which 5 Monogenea, 11 Nematoda, 3 Digenea, 1 Acanthocephala, 1 Cestoda, and 3 Crustacea. The overall species richness ranged from 4 in 2012 to 17 in 2016, in which nematodes (larvae and adults) showed higher numerical dominance, diversity, and species richness. However, the annual species richness was similar between the study years, except in 2016, where it showed a distinctly higher value. The overall parasite diversity was also different in 2012 and 2016, whereas the overall abundance differed in 2013 and 2017. The prevalence and abundance of some infracommunities of parasites varied over time. The temporal changes in the parasite community structure of P. blochii are probably related to variations in host-related features, i.e., body size and shift in diet composition as well as to the occurrence of parasites with distinct life history and biology (mainly monogeneans, digeneans, and nematodes). This is the first evaluation of a long-term temporal variation in the structure of the parasite community in fish from the Amazon.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Several tributaries form the extensive floodplain area delimited by the Amazon basin; this large area exhibits different geomorphology and phytophysiognomy that vary according to regional rainfall patterns, resulting in a variety of rich ecosystems supporting complex trophic webs. These characteristics exert strong influence on the communities of invertebrates and fish inhabiting these areas (Garcez et al. 2017). In this sense, zooplankton has a key role in this ecosystem, linking primary producers to higher trophic levels (planktivorous and omnivorous fish) of the food web (Nakajima et al. 2017). The numerous tributary rivers of the Amazon support a rich native fish fauna, providing food, reproduction, and nursery grounds. Moreover, the region and its ecosystems are very important to the survival of riparian populations. In recent years, with the growth of agriculture, livestock, and deforestation, together with poor sanitation, has threatened the water quality of the Amazonian ecosystems (Ríos-Villamizar et al. 2017). This scenario is very worrisome given the great importance of this region.
The Bloch’s catfish, Pimelodus blochii (Siluriformes: Pimelodidae), is a medium-sized species with considerable economic importance, widely distributed in the Northern part of South America, feeding on invertebrates, seeds, fruit, debris, and leaves (Lundberg and Littmann 2003; Froese and Pauly 2019). Therefore, P. blochii is a generalist and opportunistic fish with broad feeding plasticity (Soares et al. 2011; Froese and Pauly 2019). This fish is not in IUCN list. Regarding its parasite fauna, a recent study evaluated the influence of seasonality and environment on the parasite community structure of P. blochii, from the same area of the present study (Negreiros et al. 2018), but this study was a short-term approach. However, most of the parasitological surveys regarding P. blochii are limited to taxonomic descriptions or species checklist (Gil de Pertierra 2004; Luque and Tavares 2007; Kohn et al. 2007; Luque et al. 2011; Mendoza-Palmero and Scholz 2011; Orélis-Ribeiro and Bullard 2015; Cavalcante et al. 2017, 2018; Negreiros et al. 2019).
Long-term approaches regarding the parasite community dynamics, in regions of both temperate and tropical climates, have been well addressed (e.g., Kennedy et al. 2001; Kennedy and Moriarty 2002; Zander 2005; Vidal-Martínez et al. 2014; Villalba-Vasquez et al. 2018; Gallegos-Navarro et al. 2018; Hoshino and Tavares-Dias 2019). These dynamics have been related to seasonality (thermal, rainfall, and other environmental fluctuations) and to biological features of hosts (general behavior, phenotype, and migratory aspects) (e.g., Kennedy et al. 2001; Kennedy and Moriarty 2002; Zander 2005; Vidal-Martínez et al. 2014; Villalba-Vasquez et al. 2018; Gallegos-Navarro et al. 2018; Hoshino and Tavares-Dias 2019). However, a variation on parasite community structure can be expected over time. Information on long-term dynamics of parasite community structure of fish in the Neotropical region is restricted to marine environments (e.g., Vidal-Martínez et al. 2014; Villalba-Vasquez et al. 2018; Gallegos-Navarro et al. 2018). Recently, Hoshino and Tavares-Dias (2019) investigated short-term dynamics in the parasite community structure of Hemibrycon surinamensis from the eastern Amazon. Therefore, little is known about how parasites can be influenced by long-term variations in Amazonian fish, because this topic has been poorly addressed. Thus, the present study evaluated the community structure of metazoan parasites in P. blochii, collected in Iaco River, State of Acre, Brazil (western Amazon), in a long-term temporal scale, identifying possible factor causing changes on this structure.
Materials and methods
Study area, sampling of hosts, and parasites
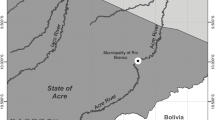
From June 2012 to March 2017 (June 2012, June 2013, June 2014, June to November 2015, May to September 2016, and March 2017), 196 individuals of P. blochii were collected with the help of local fisherman in the Iaco River, Municipality of Sena Madureira, State of Acre, Brazil (Fig. 1). Fish were kept in small, oxygenated water tanks prior to parasitological procedures in the Laboratório de Aquicultura do Instituto Federal de Educação, Ciência e Tecnologia do Acre (acronym IFAC), then euthanized by spinal cord section, measured, weighed, and analyzed for the presence of parasites. Gills, body surface, and digestive tract (esophagus, stomach, caeca, and intestine) were observed using a stereomicroscope. The organs were placed on individual Petri dishes containing saline (0.9% NaCl); body surface was also washed with saline and the resulting rinse analyzed. Parasites were collected, fixed, preserved, quantified, and processed for identification according to Eiras et al. (2006). Voucher specimens were deposited in the Coleção Zoológica de Referência da Universidade Federal do Mato Grosso do Sul (ZUFMS).
Data and statistical analysis
The Shapiro–Wilk test was applied to determine whether data to be tested fitted to a normal distribution. Differences in fish length and weight were tested between the years (2012–2017) using the Kruskal–Wallis test, followed by the Tukey’s post hoc test (Zar 2010).
A non-parametric species accumulation curve was generated with 100 replications, in order to evaluate if host sampling was enough for recovering all or most of the parasite species over time.
Parasitological terminology used herein follows Bush et al. (1997). The frequency of dominance (percentage of infracommunities in which a parasite species is numerically dominant) was estimated according to Rohde et al. (1995). The following descriptors for the parasite community were calculated using the Diversity software (Pisces Conservation Ltd., UK): species richness, Shannon–Wiener diversity index (H′), and evenness (E) (Magurran 2004). Annual differences in the Shannon–Wiener diversity index, evenness, and species richness were tested using Kruskall–Wallis test, followed by Tukey’s post hoc test (Zar 2010).
For component community of parasites, the asymptotic richness was examined based on the Clench’s model equation (Soberon and Llorente 1993), as well as the final slope of the randomized species accumulation curve.
Differences in similarity of the parasite component communities were tested using the analysis of similarities (ANOSIM), between the years (2012–2017). ANOSIM was based on ranked matrixes, generated from Jaccard index and Bray–Curtis distance (Magurran 2004), with 10,000 permutations. This analysis was performed using PAST software (Hammer et al. 2001). In order to evaluate the differences in the Bray–Curtis distances between the years, the dissimilarity matrices were ranked and subjected to a Permutational Multivariate Analysis of Variance (PERMANOVA) using 9999 permutations and TukeyHSD post hoc test (Anderson et al. 2006). The test was based on the distances from each year to the group centroid in the full dimensional space calculated in a Principal Coordinates Analysis (PCoA). The Bray–Curtis dissimilarity matrix was also used to perform this test on 9999 permutations. As the use of non-metric dissimilarity coefficients produced principal coordinate axes with negative eigenvalues, we computed the square root of distances prior to the analyses (Legendre and Legendre 1998). Additionally, the data were ordered using non-metric multidimensional scaling (NMDS) Bray–Curtis index. All these statistical procedures were performed in R version 3.5.0 (R Core Team 2018) with the vegan package (Oksanen et al. 2017).
Results
The length of P. blochii collected in 2014 was lower than individuals collected in 2012, 2013, and 2017. The weight of P. blochii collected in 2013 was higher than those collected in 2014, 2015, and 2016 (Fig. 2).
A total of 24 parasite species were collected, but only Demidospermus peruvianus, Demidospermus striatus, Demidospermus leptosynophallus, Ameloblastella amazonica, Procamallanus (S.) pimelodus, Cucullanus pinnai, Anisakidae gen. sp., and Dadaytrema oxycephala were frequent over years. In all years, the dominance was of helminth species. In 2013, it was collected the highest number of parasites and in 2012 and 2014, it was collected the lowest number of parasites (Table 1). The NMDS presented a good ordination (stress = 0.0568, r2 = 0.5129) and showed that the parasite community of 2013 was formed by a differentiated group due to the presence of Orientatractis moraveci, Scleroductus yuncens, and Anisakidae gen. sp. (Fig. 3). Most hosts were infected with no parasite species (Fig. 4). The species accumulation curve showed that most of the parasite species from component community were sampled (Fig. 5).
Non-metric multidimensional scaling (NMDS) on Bray–Curtis index of Pimelodus blochii collectected during 6 years in the Iaco River, in Brazilian Amazon region. Am, Ameloblastella amazonica; Dp, Demidospermus peruvianus; Ds, Demidospermus striatus; Dl, Demidospermus leptosynophallus; Sy, Scleroductus yuncens; Er, Ergasilus sp.; Mf, Minilernaea floricapitella; Md, Metacercariae type-Diplostomulum; Cp, Cucullanus pinnai; Om, Orientatractis moraveci; Pp, Procamallanus (S.) pimelodus; Pr, Procamallanus (S.) rarus; Pa, Philometroides acreanensis; Rr, Rondonia rondoni; Na, Anisakidae gen. sp.; Sp, Spiruroidea gen. sp.; Di, Dioctophymatidae gen. sp.; Ra, Raphidascaris sp.; Po, Prosthenhystera obesa; Da, Dadaytrema oxycephala; Mo, Monticellia magna; Ec, Echinorhynchus sp.; Di, Dipteropeltis sp.
In P. blochii gills, the abundance of D. peruvianus was higher in 2013, while abundance of D. leptosynophallus was higher in 2013 and 2014. In intestine, the abundance of O. moraveci was higher in 2013 and P. (S.) pimelodus was higher in 2016 and 2017, but the abundance of R. rondoni was higher in 2016. The abundance of Anisakidae gen. sp. was higher in 2012 and abundance of D. oxycephala was higher in 2016 (Table 2).
Shannon–Wiener diversity and evenness in hosts collected in 2012 were lower than in the other years, while Shannon–Wiener diversity and species richness of parasites were higher in 2016 (Fig. 6). In host populations, component communities of parasites exhibited, in general, similarity qualitative between 2017 with 2012 and 2016 according to Jaccard index (Table 3). ANOSIM detected differences in the composition and abundance of parasite species between host populations of 6 years (RJaccard = 1.067, p = 1.00). The pattern of abundance of the parasites varied significantly among years (PERMANOVA F[2.7] = 2.33, p < 0.05) and Tukey’s HSD test indicated differences (p < 0.05) between parasite communities of 2013 and 2015, indicating that the fish assemblages have distinct composition of parasite species in these years (Fig. 7).
Discussion
Parasites play an intrinsic but hidden role in freshwater ecosystems. Hence, the understanding of many aspects of parasite ecology in freshwater fish from many tropical regions is hindered by a paucity of long-term datasets and studies on almost all aspects of environment–host–parasite interactions (Hoshino and Tavares-Dias 2019). Therefore, there is an urgent need for long-term dataset to our understanding on the aspects of parasite ecology in Amazon fish, a large biome from South America. Here, we focused our efforts on a long-term study on parasites of P. blochii from the Iaco River, in western Amazon region, northern Brazil, in which we used quantitative analyses that are baseline, besides multivariate analyses. In long-term studies, it is seldom practicable to collect samples in many months, especially in the Amazon region. Preferably, it should be collected samples in the same months for such studies. The assumption is that this minimizes seasonal effects on community diversity, and the samples are therefore representative of the community diversity in that year (Kennedy 1997). Therefore, it seems reasonable to accept that our samples collected over 6 years are representative of the parasite community diversity in P. blochii. In addition, the species accumulation curve supported the hypothesis that the majority of parasite species of the component community in P. blochii were collected.
One of the most important results of this study is related to the species richness of parasites that was found during the 6 years in the assemblages of P. blochii from the Iaco River. In this host, collected predominately in the rainy seasons during 6 years, the component community was composed of 24 species, mainly helminth species 87.5% and crustacean species 12.5%, which had low parasitic infection levels and with sporadic occurrence. There was a variation in the number of parasites between the years sampled (4 to 17 species), and the highest species richness was found in hosts in 2016, suggesting that the component community was far richer at the end of the period investigated. The parasite community of hosts collected in 2013 was different when compared with other years due to the presence of O. moraveci, S. yuncens, and Anisakidae gen. sp. Similarly, for this same host species from the rivers Iaco and Acre, the metazoan parasite community consisted of 22 taxa, with predominance of 86.4% helminth species and 13.6% crustacean species (Negreiros et al. 2018). In addition, we observed that P. blochii had a dominance of nematode species in adult stages (Anisakidae gen. sp., O. moraveci, P. (S) pimelodus, and R. rondoni), which varied between the years, while nematodes in larval stages were only sporadic over the years. This result was not expected, since digeneans are the most abundant and diverse group of helminth parasites in fish in tropical regions (Villalba-Vasquez et al. 2018). Monogeneans were the numerically the second most diverse taxon of parasites, but D. peruvianus, D. striatus, and D. leptosynophallus were the most frequent over the years. Previous studies in P. blochii from the Iaco River reported that the species composition in the metazoan parasite community and species richness do not vary between the rainy and dry seasons, since only D. peruvianus increased during the dry season (Negreiros et al. 2018). Therefore, the communities and infracommunities of metazoan parasites in P. blochii do not change in response to rainfall levels in the Amazon region. In contrast, other studies on parasites from the tropical region showed that changes in rainfall patterns lead to changes in the proportion of hosts infected with a number of parasite species (Violante-González et al. 2009; Pech et al. 2010; Vidal-Martínez et al. 2014).
Studies on communities of parasites in fish populations are useful for the knowledge they provide on community structure and dynamics (Violante-González et al. 2009; Pech et al. 2010; Gallegos-Navarro et al. 2018). For parasitological comparisons between fish populations, the body size of hosts (age) is an important factor that can affect the infection levels and species richness of parasites (Zander 2004; Violante-González et al. 2009; Gallegos-Navarro et al. 2018; Villalba-Vasquez et al. 2018). In P. blochii from the Iaco River, the hosts were larger and lighter across years, probably because they were preparing for the breeding season. Therefore, the size of hosts can be a secondary factor influencing the parasite communities and should not be disregarded.
Our results indicated that the infracommunities, species richness, and diversity of parasites in P. blochii depended heavily on the occurrence of a distinctive set of component species such as monogeneans D. peruvianus, D. striatus, and D. leptosynophallus; nematodes P. (S.) pimelodus, C. pinai, and Anisakidae gen. sp.; and digenean D. oxycephala; all parasites whose frequency varied between the years. Considering that endohelminths have indirect life cycle that requires a variety of invertebrate and vertebrate hosts (Violante-González et al. 2009; Villalba-Vasquez et al. 2018; Gallegos-Navarro et al. 2018; Hoshino and Tavares-Dias 2019), the communities of these parasites on the host of this study reflect the presence of other organisms in the Iaco River ecosystem. Recent studies also reported that variations in infracommunity composition of parasites over time could be associated with variations in the prevalence and/or abundance of dominant taxa (Villalba-Vasquez et al. 2018; Gallegos-Navarro et al. 2018; Hoshino and Tavares-Dias 2019). In general, we also observed that for P. blochii from the Iaco River, values of the Jaccard index similarity were high between most of the years sampled. However, the multivariate analyses (PCoA) indicated a dissimilarity in the parasite community composition between 2013 and 2015.
The general characteristics of component communities of endohelminths in P. blochii from the Iaco River, especially in 2016, were very similar to those described by Negreiros et al. (2018), for this same host from the rivers Iaco and Acre, both tributaries of the Purus River, western Amazon. In addition, for P. blochii, temporal differences in species richness, component communities, and infracommunities of parasites revealed less variation than expected, with a low species richness of parasites, almost stable over the years, except in 2016, but the Shannon–Wiener diversity and evenness increased over the years. Similarly, findings unexpected were also reported for the helminth Anguilla anguilla from a small stream from the Clyst River (Kennedy 1993) and from an isolated river in England (Kennedy 1997) and for metazoan parasites of Parapseuttus panamensis from the Mexican Pacific coast (Villalba-Vasquez et al. 2018). For H. surinamensis from the eastern Amazon, it was also reported low short-term variation in the parasite community structure dynamics (Hoshino and Tavares-Dias 2019). In contrast, large temporal variations were reported in studies on the component communities, infracommunities, and species richness of parasites in other wild fish populations, which were linked to the feeding behavior and body size of hosts, and occurrence of a set of parasite species (Villalba-Vasquez et al. 2018; Gallegos-Navarro et al. 2018). The Iaco River basin, environment of this study, is characterized as a river system with extensive floodplains that constitute physical systems connected via a silted-up river that forms a drain for freshwater. During the rainy season, the waters are rich in nutrients because of the fast decomposition of grass and animal remains, and because the layer of humus that the forest produces is spread across the floodplain. This leads to a large growth of vegetation and invertebrate biomass (e.g., crustaceans and mollusks), which are used as food by fish species. This basin is not subjected to intense human activities, such as sewer and garbage disposal, urbanization, agriculture, and livestock, which results in suspended organic and inorganic debris. The vegetation is formed by species that form the floodplain forest along the waterways, along with species of macrophytes (Pereira and Morais 2014; Negreiros et al. 2018).
Trophic niche is another important factor that may influence the composition of component and infracommunities of parasites and may explain the question as to which parasite species can exist together in host populations (Zander 2003). The complexity of the food web structure in Iaco River (Silva et al. 2012; Nakajima et al. 2017; Negreiros et al. 2018) should be also a factor influencing the species richness of parasites in P. blochii, as well as the variation in diversity of parasites over the years, since food variations are known to affect species richness and diversity of endoparasites in host fish (Zander 2003, 2004; Villalba-Vasquez et al. 2018; Gallegos-Navarro et al. 2018; Hoshino and Tavares-Dias 2019). The assemblages of invertebrates, which are intermediate hosts for diverse endohelminths (Villalba-Vasquez et al. 2018; Gallegos-Navarro et al. 2018) from the Amazon region, are highly diverse and have a high density and biomass (Silva et al. 2012; Nakajima et al. 2017), but the density of mollusks should be low in Iaco River, because few digenean species (3 species) were found in P. blochii. Given the conditions of aquatic environments in Amazon, naturally dynamic, and as consequence, rivers, floodplains, and lakes suffer profound changes over the years (Silva et al. 2012; Nakajima et al. 2017; Garcez et al. 2017; Hoshino and Tavares-Dias 2019); thus, shifts in host–parasite interactions also can occur. These changes could affect the parasites in a direct or indirect way by influencing their free-living infective stages or their host range. Hence, environmental changes can result in gain or loss of parasite species as well as in increased or decreased host susceptibility. Therefore, the variations, over the years, in diversity of helminth parasites in P. blochii, a generalist and opportunistic fish that feeds on invertebrates, seeds, fruits, debris, and leaves (Soares et al. 2011; Froese and Pauly 2019), could be also related to fluctuations related to changes in diet of the host populations, which increase or decrease the opportunity for transmission of the parasite species.
A comparison of the component communities of parasites in P. blochii during 6 years showed a low number of individuals and consequently a low parasite prevalence and abundance levels. In addition, the prevalence and abundance of helminth species varied over the years, and such fluctuations seem to be also related to variations in the availability of infective stages of parasites in environments from the Iaco River. For Sciades guatemalensis from Mexico, long-term studies reported that the infection parameters of the four most frequent and abundant species of helminths suggest that the parasite transmission process was attributed to temporal fluctuations related to changes in the availability of intermediate hosts in the environment. The main factors structuring the parasite infracommunities and component communities were the feeding behavior and seasonal variation in the availability of infective stages, both influenced by the seasonal dry/rainy cycle (Violante-González et al. 2009).
In summary, as nematode helminth larvae are transmitted to their definitive hosts by predation, P. blochii is an intermediate host for Raphidascaris sp., Dioctophymatidae gen. sp., Anisakidae gen. sp., Pharyngodonidae gen. sp., and Spiruroidea gen. sp., and a definitive host for the other species of endoparasites was found. The occurrence of many species of metazoan parasites in P. blochii was not constant and the dominant species was little regular over the years, except the nematode and monogenean species, which were numerically dominant species. At the component community level, the parasite species richness increased from 2012 to 2017, suggesting that this was far richer at the end of the period. The infracommunities of parasites exhibited low infection levels, i.e., low prevalence and abundance. Furthermore, the structure and species composition of the parasite communities little varied between the sampling years. Such variations seem to be the result of factors linked to host features, such as body size, besides the occurrence of a set of distinctive parasite species in the component communities, and possible variations in the availability of infective stages over the years.
References
Anderson MJ, Ellingsen KE, McArdle BH (2006) Multivariate dispersion as a measure of beta diversity. Ecol Lett 9:683–693. https://doi.org/10.1111/j.1461-0248.2006.00926.x
Bush AO, Lafferty KD, Lotz JM, Shostak AW (1997) Parasitology meets ecology on its own terms: Margolis et al. Revisited. J Parasitol 83:575
Cavalcante PHO, Silva MT, Santos EGN, Chagas-Moutinho VA, Santos CP (2017) Orientatractis moraveci n. sp. and Rondonia rondoni Travassos, 1920 (Nematoda: Atractidae), parasites of Pimelodus blochii (Osteichthyes, Pimelodidae) from the Acre and Xapuri Rivers, western Amazon, Brazil. Parasitology 144:226–236. https://doi.org/10.1017/S0031182016001736
Cavalcante PHO, Moravec F, Santos CP (2018) The philometrid nematode Philometroides acreanensis n. sp. from the stomach wall of the catfish Pimelodus blochii in north-western Brazil. J Helminthol 92:109–115. https://doi.org/10.1017/S0022149X1700013X
Eiras JC, Takemoto RM, Pavanelli GC (2006) Métodos de estudo e técnicas laboratoriais em parasitologia de peixes. Eduem, Maringá
Froese R, Pauly D. Editors. (2019) FishBase. World Wide Web electronic publication. www.fishbase.org, version (02/2019)
Gallegos-Navarro Y, Violante-González J, Monks S, García-Ibáñez S, Rojas-Herrera AA, Pulido-Flores G, Rosas-Acevedo JL (2018) Factors linked to temporal and spatial variation in the metazoan parasite communities of green jack Caranx caballus (Günther 1868) (Pisces: Carangidae) from the Pacific coast of Mexico. J Nat Hist 52(39-40):2573–2590. https://doi.org/10.1080/00222933.2018.1546914
Garcez RCS, Souza LA, Frutuoso ME, Freitas CEC (2017) Seasonal dynamic of Amazonian small-scale fisheries is dictated by the hydrologic pulse. Bol Inst Pesca 43:207–221
Gil de Pertierra AA (2004) Redescription of Monticellia magna (Rego, dos Santos & Silva, 1974) (Eucestoda: Monticelliidae) parasite of Pimelodus spp. (Pisces: Siluriformes) from Argentina, and morphological study of microtriches. Rev Suisse Zool 11:11–20
Hammer O, Harper DAT, Ryan PD (2001) PAST: Paleontological statistics software package for education and data analyses. Pelaeont Eletron 4:1–9
Hoshino EM, Tavares-Dias M (2019) Interannual and seasonal variation in protozoan and metazoan parasite communities of Hemibrycon surinamensis, a characid fish inhabiting the Brazilian Amazon. Acta Parasitol. https://doi.org/10.2478/s11686-019-00057-5
Kennedy CR (1993) The dynamics of intestinal helminth communities in eels Anguilla anguilla in a small stream: long-term changes in richness and structure. Parasitology 107:71–78
Kennedy CR (1997) Long-term and seasonal changes in composition and richness of intestinal helminth communities in eels Anguilla anguilla of an isolated English river. Folia Parasitol 44:267–273
Kennedy CR, Moriarty C (2002) Long-term stability in the richness and structure of helminth communities in eels, Anguilla anguilla, in Lough Derg, River Shannon, Ireland. J Helminthol 76:315–322
Kennedy CR, Shears PC, Shears JA (2001) Long-term dynamics of Ligula intestinalis and roach Rutilus rutilus: a study of three epizootic cycles over thirty-one years. Parasitology 123:257–269
Kohn A, Fernandes BMM, Cohen SC (2007) South American Trematodes parasites of fishes. Editora Imprinta Express Ltda, Rio de Janeiro
Legendre P, Legendre L (1998) Numerical ecology, 2nd edn. Elsevier, Canada
Lundberg JG, Littmann MW (2003) Family Pimelodidae (Long-whiskered catfishes). In: Reis RE, Kullander SO, Ferraris JRCJ (eds) Check list of the freshwater fishes of South and Central America. EDIPUCRS, Porto Alegre, pp 432–446
Luque JL, Tavares LER (2007) Checklist of Copepoda associated with fishes from Brazil. Zootaxa 1579:1–39
Luque JL, Aguiar JC, Vieira FM, Gibson D (2011) Santos CP (2011) Checklist of Nematoda associated with the fishes of Brazil. Zootaxa 3082:1–88
Magurran AE (2004) Measuring biological diversity. Blackwell Science, Oxford
Mendoza-Palmero CA, Scholz T (2011) New species of Demidospermus (Monogenea: Dactylogyridae) of pimelodid catfish (Siluriformes) from Peruvian Amazonia and the reassignment of Urocleidoides lebedevi Kritsky and Thatcher, 1976. J Parasitol 97:586–592. https://doi.org/10.1645/GE-2655.1
Nakajima R, Rimachi EV, Santos-Silva EN, Calixto LSF, Leite RG, Khen A, Yamane T, Mazeroll AI, Inuma JC, Utumi EYK, Akira Tanaka A (2017) The density and biomass of mesozooplankton and ichthyoplankton in the Negro and the Amazon Rivers during the rainy season: the ecological importance of the confluence boundary. PeerJ 5:e3308. https://doi.org/10.7717/peerj.3308
Negreiros LP, Pereira FB, Tavares-Dias M, Tavares LER (2018) Community structure of metazoan parasites from Pimelodus blochii in two rivers of the western Brazilian Amazon: same seasonal traits, but different anthropogenic impacts. Parasitol Res 117:3791–3798. https://doi.org/10.1007/s00436-018-6082-5
Negreiros LP, Tavares-Dias M, Elisei C, Tavares LER, Felipe B, Pereira FB (2019) First description of the male of Philometroides acreanensis and phylogenetic assessment of Philometridae (Nematoda: Dracunculoidea) suggest instability of some taxa. Parasitol Inter 69:30–38. https://doi.org/10.1016/j.parint.2018.10.010
Oksanen JF, Blanchet G, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2017) Vegan: Community ecology package. R Package version 2.4-3. https://CRAN.R-project.org/package=vegan. Accessed 18 May 2019
Orélis-Ribeiro R, Bullard SA (2015) Blood flukes (Digenea: Aporocotylidae) infecting body cavity of South American catfishes (Siluriformes: Pimelodidae): two new species from rivers in Bolivia, Guyana and Peru with a re-assessment of Plehniella Szidat, 1951. Folia Parasitol 62:1–17. https://doi.org/10.14411/fp.2015.050
Pech D, Aguirre-Macedo ML, Lewis JW, Vidal-Martínez VM (2010) Rainfall induces time-lagged changes in the proportion of tropical aquatic hosts infected with metazoan parasites. Inter J Parasitol 40:937–944
Pereira TKK, Morais JF (2014) Técnicas de geoprocessamento aplicadas aos problemas ambientais que afetam o Rio Iaco dentro do limite do município de Sena Madureira-AC. Rev Brasil Gestão Amb 8:1–6
R Core Team (2018) R: A language and environment for statistical computing. R Foundation for statistical computing, Vienna, Austria. URL https://www.R-project.org/. Accessed 18 May 2019
Ríos-Villamizar EA, Piedade MTF, Junk WJ, Waichman AV (2017) Surface water quality and deforestation of the Purus river basin, Brazilian Amazon. Int Aquat Res 9:81–88. https://doi.org/10.1007/s40071-016-0150-1
Rohde K, Hayward C, Heap M (1995) Aspects of the ecology of metazoan ectoparasites of marine fishes. Int J Parasitol 25:945–970. https://doi.org/10.1016/0020-7519(95)00015-T
Silva ES, Keppeler EC, Silvério JF (2012) Composition of zooplankton of the small river Jesumira, located in a cleared in area at the Park National Serra do Divisor, State of Acre, Brazil. Semina: Ciên Biol Saúde 33:201–210. https://doi.org/10.5433/1679-0367.2012v33n2p201
Soares MGM, Costa EL, Siqueira-Souza FK, Anjos HDB, Yamamoto KC, Freitas CEC (2011) Peixes de lagos do médio Rio Solimões, 2nd edn. Instituto Piatam, Manaus
Soberon JM, Llorente BJ (1993) The use of species accumulation functions for the prediction of species richness. Conserv Biol 7:480–488. https://doi.org/10.1046/j.1523-1739.1993.07030480.x
Vidal-Martínez VM, Pal P, Aguirre-Macedo ML, May-Tec AL, Lewis JW (2014) Temporal variation in the dispersion patterns of metazoan parasites of a coastal fish species from the Gulf of Mexico. J Helminthol 88:112–122
Villalba-Vasquez PJ, Violante-González J, Monks S, Marino-Romero JU, Ibáñez SG, Rojas-Herrera AA, FloresGarza R, Rosas Guerrero V (2018) Temporal and spatial variations in the metazoan parasite communities of the Panama spadefish, Parapsettus panamensis (Pisces: Ephippidae), from the Pacific coast of Mexico. Invert Biol 137:339–354
Violante-González J, Aguirre-Macedo ML, Rojas-Herrera A, Guerrero SG (2009) Metazoan parasite community of blue sea catfish, Sciades guatemalensis (Ariidae), from Tres Palos Lagoon, Guerrero, Mexico. Parasitol Res 105:997–1005
Zander CD (2003) Four-year monitoring of parasite communities in gobiid fishes of the south-western Baltic: II. Infracommunity. Parasitol Res 93:17–29
Zander CD (2004) Four-year monitoring of parasite communities in gobiid fishes of the southwestern Baltic II. Infracommunity. Parasitol Res 93:17–29. https://doi.org/10.1007/s00436-004-1087-7
Zander CD (2005) Four-year monitoring of parasite communities in gobiid fishes of the southwest Baltic. III. Parasite species diversity and applicability of monitoring. Parasitol Res 95:136–144. https://doi.org/10.1007/s00436-004-1252-z
Zar JH (2010) Biostatistical analysis. PrenticeHall, New Jersey
Funding
The authors thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil) for the productivity research grant awarded to Dr. M. Tavares-Dias (# 303013/2015-0).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Disclosures
All procedures involving animals were authorized by the Instituto Chico Mendes para Conservação da Biodiversidade (acronym SISBIO, No. 60899-1) and were strictly according to the protocols and rules of the Committee on Ethics of Animal Use of the Embrapa
Amapá (Protocol No. 002-CEUA-CPAFAP).
Additional information
Section Editor: Stephen A. Bullard
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Negreiros, L.P., Florentino, A.C., Pereira, F.B. et al. Long-term temporal variation in the parasite community structure of metazoans of Pimelodus blochii (Pimelodidae), a catfish from the Brazilian Amazon. Parasitol Res 118, 3337–3347 (2019). https://doi.org/10.1007/s00436-019-06480-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-019-06480-x