Abstract
The present investigation evaluated the influence of seasonality and locality on the structure of the parasite community of the catfish Pimelodus blochii. A total of 160 fish were collected from two rivers in the State of Acre, western Brazilian Amazon: 80 fish in River Acre and 80 in River Iaco, with 40 in each season (rainy and drought). The overall prevalence was 78.7% and 1461 parasite specimens (adults and larvae) were allocated in 22 taxa: 5 of Monogenea, 10 of Nematoda, 3 of Digenea, 1 of Cestoda and 3 of Crustacea. In drought season, Procamallanus (Spirocamallanus) pimelodus and Demidospermus peruvianus were more prevalent in River Acre and Iaco, respectively. The parasite diversity (Brillouin index) as well as the prevalence and abundance of the monogeneans D. peruvianus, D. striatus, Demidospermus sp. and Ameloblastella sp. were higher in River Acre. The parasite community structure were dissimilar among rivers, and seasonally in River Iaco. These results suggest that environmental traits may overshadow seasonal influences on the parasite community structure, which may be related to the higher anthropization in River Acre. Furthermore, seasonality exerted less influence on the parasite community than expected, probably because the two rivers have different hydrological traits than those of other rivers in the Brazilian Amazon. New host and locality records expanded our knowledge of parasite biodiversity of P. blochii.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Several factors are known to play some role on the structure of the parasite communities in fish. These factors may be related to the environment (e.g. organic and inorganic conditions) and/or to the host (e.g. feeding, behaviour, physiology, sex) (Blasco-Costa et al. 2014; Oliveira et al. 2017). Furthermore, the abundance of parasites with complex life cycles may be strongly dependent of the population density of intermediate and definitive hosts, superposing time and space (Blasco-Costa et al. 2014).
The Amazon represents one of the greatest flood plain areas associated with large rivers. This region supports the most diverse ichthyofauna in the world, which is composed of species from almost all orders of freshwater fishes (Reis et al. 2001). Through the evolutionary process, the Amazon river basin has been passing through several environmental changes, influencing the ichthyofauna to develop unique physiological, behavioural and biochemical adaptations (Lowe-McConnell 1999). Moreover, the Amazon includes complex environments that, together with the marked flooding oscillations, provide rich resources for fishes (e.g. habitat, shelter and food) influencing their population dynamics directly (Junk 1997).
The parasite community structures of fishes in Amazon have been largely reported oscillating according to seasonal changes, which is annually based on two well-defined periods of rain and drought (Neves et al. 2013; Tavares-Dias et al. 2014; Carvalho and Tavares-Dias 2017). This marked seasonality causes drastic changes in the landscape and is expected to influence all the associated organic and inorganic environmental conditions.
Currently, there are no studies regarding the ecology of parasite communities from the catfish Pimelodus blochii Valenciennes (Siluriformes: Pimelodidae); studies have been focused solely on taxonomy of parasites and species catalogue (Gil de Pertierra 2004; Luque and Tavares 2007; Kohn et al. 2007; Luque et al. 2011; Mendoza-Palmero and Scholz 2011; Orélis-Ribeiro and Bullard 2015; Cavalcante et al. 2016, 2018). Pimelodus blochii has economic importance and is widely distributed in South America (Lundberg and Littmann 2003; Froese and Pauly 2018); therefore, the community dynamics of parasites from this fish should be better understood. In this sense, the present study evaluated the influences of locality and seasonality of the community structure of the metazoan parasites of P. blochii from two different rivers in the State of Acre, western Brazilian Amazon. The main motivation was that the studied areas are under the same seasonal conditions, but show different anthropogenic impacts.
Materials and methods
Study area, sampling of hosts and parasites
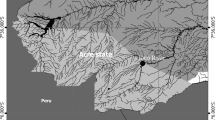
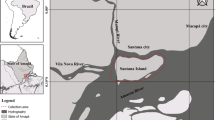
Fish were collected with the help of local fisherman, from June 2015 to April 2017, from two different rivers in the State of Acre, Brazil. One collection locality was in the River Acre, Municipality of Rio Branco and the other was in River Iaco, Municipality of Sena Madureira (Fig. 1). According to Pereira and Morais (2015) and Duarte (2017), River Acre is subjected to higher intensity of human activities (i.e. sewer and garbage disposal, agriculture), which results in higher organic and inorganic debris in suspension than in River Iaco. A total of 160 fish were collected: 80 in each river, being 40 during the rainy and 40 during the drought season; the collections were proportionally distributed within each month of the study with respect to the seasonal periods (i.e. 6 to 8 fish collected monthly during 22 months, of which 3 to 4 fish in each locality). Definition of each seasonal period was according to Silva et al. (2008) and Duarte (2017), based on rainfall indices and other climatic variables, in which the rainy season is from November to May and the drought season from June to October.
Fish were kept in small oxygenated water tanks prior to parasitological procedures in the Laboratório de Aquicultura do Instituto Federal de Educação, Ciência e Tecnologia do Acre (acronym IFAC). Hosts were euthanized by spinal cord transection, measured, weighed and analysed for parasites. Gills, skin and digestive tract (oesophagus, stomach, caeca and intestine) were individually observed in Petri dishes containing saline (0.9% NaCl), using a stereomicroscope. Parasites were collected, fixed, stored, quantified and processed for identification according to Eiras et al. (2006).
Voucher specimens were deposited in the Coleção Zoológica de Referência da Universidade Federal do Mato Grosso do Sul (acronym ZUFMS).
Data and statistical analysis
Parasitological terminology used herein follows that of Bush et al. (1997). Species with prevalence < 10% were not included in the statistical analyses. The Shapiro-Wilk test was used to determine whether data of abundance and prevalence of parasites fitted a curve of normal distribution.
A series of preliminary tests were performed to evaluate the homogeneity of host sampling. Differences in the number of individuals from each sex, and length and weight of males and females were tested by locality and seasonality (with overall data and separated by locality), using the Mann-Whitney (U) followed by the post hoc of Dunn when pertinent (Zar 2010). A linear regression was performed to confirm the expected positive correlation between host length and weight. Overall differences in the prevalence and in the mean abundance of parasites from male and female hosts were verified using generalized linear models (GLM), i.e. Poisson regression in the case of abundance and logistic regression in the case of prevalence (see next paragraphs for details). These preliminary tests were performed to avoid biased and/or wrong interpretation of the results, because the analyses were focused on comparing the structure of the parasite community by locality and season.
Differences in prevalence and abundance of parasites were tested between the two sampling areas, and seasonally by each locality, separately. These comparisons were chosen after testing the adequateness of GLMs, based on the Akaike information criteria (Burnham and Anderson 2002) followed by a one-way ANOVA, in which the mixed models with interaction of season and locality showed responses statistically similar to the models with season and locality tested separately. These tests were performed using the package lme4 (Bates et al. 2015), implemented in the software R (R Development Core Team 2008). Therefore, the differences in the prevalence were tested using the logistic regression and those in abundance using the Poisson regression (Dohoo et al. 2003). The odds ratio (OR) for each GLM was calculated in order to evaluate the presence and the degree of association between the dependent and the independent variable, where 0 < OR < 1 indicates negative association, OR = 1 indicates lack of association and OR > 1 indicates positive association (variables of reference were River Iaco and drought season) (Dohoo et al. 2003). The confidence interval (CI) of 95% for all the GLM was estimated, and if 1 was included in the interval, the model was considered null.
The diversity of parasite infracommunities was estimated using the Brillouin index (HB), and its differences between the two localities and the two seasons within each river separately were evaluated using the U test (Zar 2010). The Spearman’s rank correlation (rs) was used to verify correlations between host length and parasite diversity (HB), species richness and abundance (overall and of each species) according to the two localities and seasonally by locality, separately (Zar 2010).
Differences in similarity of the parasite component communities were tested using the analysis of similarities (ANOSIM), between the two rivers and seasonally by locality. ANOSIM was based on ranked matrixes, generated from Jaccard index and Bray-Curtis distance (Magurran 2004), with 10,000 permutations.
All the statistical analyses were performed in R (R Development Core Team 2008) and in PAST software (Hammer et al. 2001) with significance level of p < 0.05.
Results
Of the 160 Pimelodus blochii examined, 117 were females and 43 males. In River Acre, 30 female and 10 male specimens were collected during the rainy season and the same quantity during the drought, totalizing 80 hosts. In River Iaco, 30 females and 10 males were collected in the rainy season and 27 females and 13 males in the drought, totalizing 80 hosts as well. The number of male and female fish were not statistically different between locality and season (Online Resource 1), as well as length and weight were similar between sexes (Online Resources 2, 3); however, fishes were slightly heavier in the rainy period (Online Resource 3). Fish length and weight were positively correlated (p < 0.001, r2 = 0.86). Prevalence and abundance of each parasite taxa were not statistically different between male and female hosts (Online Resource 4).
From all of the P. blochii examined, 78.7% harboured at least one species of parasite; the overall abundance was 1461, of which 972 specimens parasitizing fish from River Acre (overall prevalence 88.7%) and 489 parasitizing those from River Iaco (68.7% overall prevalence). Twenty-two different taxa were found; the most prevalent species was the nematode Procamallanus (Spirocamallanus) pimelodus Pinto, Fábio, Noronha & Rolas, 1974 (48.8%) followed by the monogenean Demidospermus peruvianus Mendoza-Palmero & Scholz, 2011 (32.5%); 4 taxa were found as larvae, three nematodes and one digenetic (Table 1). Seventeen taxa co-occurred in both component communities from River Acre and River Iaco (Table 1).
Differences in prevalence and abundance of parasites were more evident comparing the two localities rather than seasonally, in which monogeneans showed higher prevalence and abundances in River Acre (Table 1). Locally, during the drought season, P. (S.) pimelodus and D. peruvianus were more prevalent in River Acre and River Iaco, respectively (Table 2).
The mean diversity was higher in River Acre than in River Iaco (H = 0.7 ± 0.6 vs. 0.4 ± 0.4, respectively; U = 2035.5, p < 0.001), as well as during the drought in River Iaco (H = 0.49 ± 0.48 vs. 0.24 ± 0.36 in drought and rainy season respectively; U = 565, p = 0.014), but was not different among seasons in the River Acre (H = 0.75 ± 0.63 vs. 0.74 ± 0.54 in rainy and drought season respectively; U = 776, p = 0.83). There was no correlation between host length and parasite diversity (H), species richness and abundance in any of the comparisons performed (see Online Resource 5 for all the related results).
The ANOSIM results indicated no qualitative and quantitative similarities comparing the component communities from the two rivers (Jaccard index matrix R = 0.075, p = 0.0001; Bray-Curtis distance matrix R = 0.090, p = 0.001), as well as comparing the component communities between seasons in the River Iaco (Jaccard index matrix R = 0.026, p = 0.045; Bray-Curtis distance matrix R = 0.027, p = 0.045). However, the ANOSIM indicated similarity among rainy and drought periods in River Acre (Jaccard index matrix R = 0.008, p = 0.063; Bray-Curtis distance matrix R = 0.009, p = 0.089).
Discussion
Almost 60% of the parasite species found in the present study have been reported in P. blochii in different regions of South America (Gil de Pertierra 2004; Mendoza-Palmero and Scholz 2011; Orélis-Ribeiro and Bullard 2015). However, the monogeneans D. peruvianus, D. striatus Mendoza-Palmero and Scholz, 2011, and Scleroductus sp., the nematode Raphidascaris sp. and the crustaceans Dipteropeltis sp., Ergasilus sp. and Minilernea floricapitella Thatcher & Huergo, 2005, represent new host records for P. blochii in Brazil. Furthermore, the nematodes Cucullanus pinnai pinnai Travassos, Artigas & Pereira, 1928, P. (S.) pimelodus and P. (S.) rarus Travassos, Artigas & Pereira, 1928, and the cestode Monticellia magna (Rego, dos Santos & Silva, 1974) infecting P. blochii in the State of Acre expand the geographic range of these parasites to the western Amazon.
The parasite community of P. blochii showed a richer fauna of endoparasites, but most of the species of monogeneans were highly prevalent (see Table 1). Moreover, almost 18% of all parasite specimens were found as larval stages. These results indicate that P. blochii has important and different roles on the transmission of the present parasite species, acting as intermediate, paratenic and/or definitive host, and the transmission patterns are largely linked to the omnivory of the fish (Sánchez-Botero and Araújo-Lima 2001). In this sense, infections by endoparasites that exhibit diverse life cycles and different transmission mechanisms may be favoured (Moravec 1998; Tavares-Dias et al. 2014; Oliveira et al. 2017), superposing the richness of ectoparasites.
The Amazon has a well-defined seasonality, which is based on periods of intense rainfall followed by those of drought, and these characteristics are known to commonly influence the structure of parasite communities in the biome (Neves et al. 2013; Tavares-Dias et al. 2014; Carvalho and Tavares-Dias 2017). However, the present results showed some different and interesting patterns rather than the expected regarding the River Acre, where variations in the parasite community were very slight.
In the River Acre, the only seasonal variation observed was in the prevalence of P. (S.) pimelodus that was higher during the drought season. This result may be related to the host dietary habits, because fish are predominantly herbivorous in the rainy season, but generalists in the drought (López-Casas and Jiménez-Segura 2007; Soares et al. 2011) which increases the probability of ingesting intermediate hosts harbouring P. (S.) pimelodus infective larvae.
In River Iaco, parasite communities were seasonally dissimilar, with higher prevalence of D. peruvianus in the drought resulting in higher diversity in this season. During the drought period, flooded areas are reduced, consequently favouring the fish aggregation and the transmission of ectoparasites with direct life cycle (e.g. monogeneans) as has been observed in the Brazilian Amazon (Neves et al. 2013; Amarante et al. 2015).
Several factors determine differences in the structure of parasite communities of hosts from different populations and localities (Oliveira et al. 2017; Yamada et al. 2017). The present results showed a more diverse parasite fauna in River Acre, as same as the ANOSIM indicated no similarity among the two rivers. Even though P. blochii migrates during the breeding season and may overlap the two sampling areas (López-Casas and Jiménez-Segura 2007; Soares et al. 2011), the local conditions seem to be important on structuring the studied component community. An example is that monogeneans were more prevalent and abundant in River Acre as they are monoxenic ectoparasites in unremitting contact with the external environment. Moreover, the nearly complete lack of seasonal variations in the parasite community structure in River Acre compared with those in River Iaco may be related to the different anthropogenic impacts that are higher in River Acre, drastically increasing the amount of organic and inorganic sediments in suspension (Pereira and Morais 2015; Duarte 2017). Therefore, even though both rivers are in the same basin and exhibit similar limnology and hydrology (Silva et al. 2008), the impact of human activities may cause significant environmental changes on a local scale (close to cities and/or areas of agriculture). These issues support the idea that the local environment has influence on the parasite component community of the present study and, in River Acre, it seems to be stronger than the seasonal variations.
It should be mentioned that fishes were heavier in the rainy period, probably because it represents the breeding season and flooded areas for foraging are increased (López-Casas and Jiménez-Segura 2007; Soares et al. 2011; Froese and Pauly 2018). However, as the fish weight showed strong and positive correlation with fish length, which had no influence on the parasite fauna (see Online Resource 3), it is clear that fish weight is not determinant in the structure of this parasite community.
Based on the present results, the seasonal influence on the component community structure of parasites was less intense than expected, contrasting with similar approaches in the Amazon (Neves et al. 2013; Tavares-Dias et al. 2014; Carvalho and Tavares-Dias 2017). Therefore, it should be highlighted that River Acre and Iaco, both tributaries of River Purus, have their headwaters in Peru, at the base of the Andes mountain range, resulting in different hydrological and fluvial level fluctuations when compared with those from most of the other rivers in the Brazilian Amazon (Bardales et al. 2010; Wadt et al. 2010). Most likely, this resulted in the less intense seasonal influence observed here, compared with studies in other Amazonian sub basins (see Neves et al. 2013; Tavares-Dias et al. 2014; Carvalho and Tavares-Dias 2017).
These results were generated from a 2-year sampling, and despite that sampling data was robust, in a longer temporal scale, data may exhibit different behaviour as the low R values of ANOSIM suggested that other factors than locality and seasonality are influencing the homogeneity of the parasite communities. Nevertheless, the higher values of prevalence and abundance of monogeneans in River Acre (more anthropized) contrasting with those in Rive Iaco (less anthropized) suggest a possible biological indicator role for these parasites as asserted by other authors (Sanchez-Ramires et al. 2007; Palm 2011). Finally, these results expanded our knowledge of parasite biodiversity in P. blochii, an economically important species with farming potential.
References
Amarante CF, Tassinari WS, Luque JL, Pereira MJS (2015) Factors associated with parasite aggregation levels in fishes from Brazil. Rev Bras Parasitol Vet 24:174–182. https://doi.org/10.1590/S1984-29612015031
Bardales NG, Rodrigues TE, Oliveira H, Amaral EF, Araújo EA, Lani JL (2010) Formação, classificação e distribuição geográfica dos solos do Acre. In: Souza CM, Araújo EA, Medeiros MFST, Magalhãe AA (eds) Recursos naturais: geologia, geomorfologia e solos do Acre. SEMA, Rio Branco, pp 48–60
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–51. https://doi.org/10.18637/jss.v067.i01
Blasco-Costa I, Rouco C, Poulin R (2014) Biogeography of parasitism in freshwater fish: spatial patterns in hot spots of infection. Ecography 37:1–10. https://doi.org/10.1111/ecog.01020
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2 end edn. Springer, New York
Bush AO, Lafferty KD, Lotz JM, Shostak AW (1997) Parasitology meets ecology on its own terms: Margolis et al. revisited. J Parasitol 83:575–583. https://doi.org/10.2307/3284227
Carvalho AA, Tavares-Dias M (2017) Diversity of parasites in Cichlasoma amazonarum Kullander, 1983 during rainy and dry seasons in eastern Amazon (Brazil). J Appl Ichthyol 33:1178–1183. https://doi.org/10.1111/jai.13451
Cavalcante HO, Silva MT, Santos EGN, Chagas-Moutinho VA, Santos CP (2016) Orientatractis moraveci n. sp. and Rondonia rondoni Travassos, 1920 (Nematoda: Atractidae), parasites of Pimelodus blochii (Osteichthyes, Pimelodidae) from the Acre and Xapuri Rivers, Western Amazon, Brazil. Parasitology 144:226–236. https://doi.org/10.1017/S0031182016001736
Cavalcante PHO, Moravec F, Santos CP (2018) The philometrid nematode Philometroides acreanensis n. sp. from the stomach wall of the catfish Pimelodus blochii in north-western Brazil. J Helminthol 92:109–115. https://doi.org/10.1017/S0022149X1700013X
Dohoo I, Martin W, Stryhn H (2003) Veterinary epidemiologic research. AVC Inc., Charlottetown
Duarte AF (2017) Climatologia das chuvas e efeitos antrópicos da urbanização na Bacia do Rio Acre, Amazônia Ocidental. Bol Observ Amb Alberto Ribeiro Lamego 1:199–213. https://doi.org/10.19180/2177-4560.v11n12017p199-213
Eiras JC, Takemoto RM, Pavanelli GC (2006) Métodos de estudo e técnicas laboratoriais em parasitologia de peixes, 2nd edn. Editora da Universidade Estadual de Maringá, Maringá
Froese R, Pauly D (2018) FishBase. World Wide Web electronic publication. www.fishbase.org, version (02/2018)
Gil de Pertierra AA (2004) Redescription of Monticellia magna (Rego, Santos & Silva, 1974) (Eucestoda: Monticelliidae) parasite of Pimelodus spp. (Pisces: Siluriformes) from Argentina, and morphological study of microtriches. Rev Suisse Zool 111:11–20. https://doi.org/10.5962/BHL.part.80222
Hammer O, Harper DAT, Ryan PD (2001) PAST: Paleontological statistics software package for education and data analyses. Pelaeontologia Eletronica 4:1–9
Junk WJ (1997) The central Amazon floodplain: ecology of a pulsing system. Springer, New York
Kohn A, Fernandes BMM, Cohen SC (2007) South American trematodes parasites of fishes. Ed. Imprinta Express Ltda, Rio de Janeiro
López-Casas S, Jiménez-Segura LF (2007) Reproducción y hábitos alimenticios del nicuro, Pimelodus blochii (Valenciennes, 1840) (Pisces: Pimelodidae), en la ciénaga de cachimbero, río Magdalena, Colômbia. Actualidade Biológica 29:199–207
Lowe-McConnell RH (1999) Estudos ecológicos de comunidades de peixes tropicais. EDUPS, São Paulo
Lundberg JG, Littmann MW (2003) Family Pimelodidae (Long-whiskered catfishes). In: Reis RE, Kullander SO, Ferraris JRCJ (eds) Check list of the freshwater fishes of South and Central America. EDIPUCRS, Porto Alegre, pp 432–446
Luque JL, Tavares LER (2007) Checklist of Copepoda associated with fishes from Brazil. Zootaxa 1579:1–39. https://doi.org/10.11646/zootaxa.1579.1.1
Luque JL, Aguiar JC, Vieira FM, Gibson DI, Santos CP (2011) Checklist of Nematoda associated with the fishes of Brazil. Zootaxa 3082:1–88
Magurran AE (2004) Measuring biological diversity. Blackwell Publishing, Oxford
Mendoza-Palmero CA, Scholz T (2011) New species of Demidospermus (Monogenea: Dactylogyridae) of pimelodid catfish (Siluriformes) from Peruvian Amazonia and the reassignment of Urocleidoides lebedevi Kritsky and Thatcher, 1976. J Parasitol 97:586–592. https://doi.org/10.1645/GE-2655.1
Moravec F (1998) Nematodes of freshwater fishes of the Neotropical region. Academia, Praha
Neves LR, Pereira FB, Tavares-Dias M, Luque JL (2013) Seasonal influence on the parasite fauna of a wild population of Astronotus ocellatus (Perciformes: Cichlidae) from the Brazilian Amazon. J Parasitol 99:718–721. https://doi.org/10.1645/12-84.1
Oliveira MSB, Gonçalves RA, Ferreira DO, Pinheiro DA, Neves LR, Dias MKR, Tavares-Dias M (2017) Metazoan parasite communities of wild Leporinus friderici (Characiformes: Anostomidae) from Amazon River system in Brazil. Stud Neotropical Fauna Environ 52:146–156. https://doi.org/10.1080/01650521.2017.1312776
Orélis-Ribeiro R, Bullard SA (2015) Blood flukes (Digenea: Aporocotylidae) infecting body cavity of South American catfishes (Siluriformes: Pimelodidae): two new species from rivers in Bolivia, Guyana and Peru with a re-assessment of Plehniella Szidat, 1951. Folia Parasit 62:1–17. https://doi.org/10.14411/fp.2015.050
Palm HW (2011) Fish parasites as biological indicators in a changing world: can we monitor environmental impact and climate change? In: Mehlhorn H (ed) Progress in parasitology. Parasitology Research Monographs, vol 2. Springer, Berlin, pp 223–250. https://doi.org/10.1007/978-3-642-21396-0_12
Pereira TKK, Morais FJ (2015) Técnicas de geoprocessamento aplicadas aos problemas ambientais que afetam o rio Iaco dentro do limite do município de Sena Madureira-AC. Rev Eletron Gestão, Educação e Tecnol Amb 19:11–20. https://doi.org/10.5902/2236117013867
R Development Core Team (2008) R: a language and environment for statistical computing. Vienna, Austria
Reis RE, Kullander SO, Ferraris JRCJ (2001) Check list of the freshwater fishes of South and Central America. EDIPUCRS, Porto Alegre
Sánchez-Botero JI, Araújo-Lima CARM (2001) As macrófitas aquáticas como berçário para a ictiofauna da várzea do Rio Amazonas. Acta Amazon 31(3):437–447. https://doi.org/10.1590/1809-43922001313447
Sanchez-Ramires C, Vidal-Martinez VM, Aguirre-Macedo ML, Rodriguez-Canul RP, Gold-Bouchot G, Sures B (2007) Cichlidogyrus sclerosus (Monogenea: Ancyrocephalinae) and its host, the Nile tilapia (Oreochromis niloticus), as bioindicators of chemical pollution. J Parasitol 93:1097–1106. https://doi.org/10.1645/GE-1162R.1
Silva AEP, Angelis CF, Machado LAT, Waichaman AV (2008) Influência da precipitação na qualidade da água do Rio Purus. Acta Amazon 38:733–742. https://doi.org/10.1590/S0044-59672008000400017
Soares MGM, Costa EL, Siqueira-Souza FK, Anjos HDB, Yamamoto KC, Freitas CEC (2011) Peixes de lagos do médio Rio Solimões, 2nd edn. Instituto Piatam, Manaus
Tavares-Dias M, Oliveira MSB, Gonçalves R, Silva LMA (2014) Ecology and seasonal variation of parasites in wild Aequidens tetramerus, a Cichlidae from the Amazon. Acta Parasitol 54:158–164. https://doi.org/10.2478/s11686-014-0225-3
Wadt PGS, Araújo EA, Costa FS (2010) Manejo de fertilizantes e resíduos na Amazônia sul-ocidental. In: Prado RB, Turetta APD, Andrade AG (eds) Manejo e conservação do solo e da água no contexto das mudanças ambientais. Embrapa Solos, Rio de Janeiro, pp 141–170
Yamada FH, Bongiovani MF, Yamada POF, Silva FJ (2017) Parasite infracommunities of Leporinus friderici: a comparison of three tributaries of the Jurumirim Reservoir in southeastern Brazil. An Acad Bras Cienc 89:953–963. https://doi.org/10.1590/0001-3765201720160554
Zar JH (2010) Biostatistical analysis, 5th edn. Prentice Hall, New Jersey
Acknowledgements
Authors would like to thank Dr. Tomaš Scholz and Dr. Philippe Vieira Alves for the identification of cestodes as well as the staff of the Laboratory of Animal Parasitology from the Universidade Federal de Mato Grosso do Sul for support and providing research facilities.
Funding
M. T.-D. was supported by a researcher fellowship of the National Council for Research and Technological Development (Conselho Nacional de Pesquisa e Desenvolvimento Tecnologico, CNPq, Brazil) (# 303013/2015-0). F. B. P. was supported by a Post-doctoral fellowship PNPD-CAPES (Programa Nacional de Pós-Doutorado-Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-CAPES, Brazil).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Disclosures
All procedures involving animals were authorised by the Instituto Chico Mendes para Conservação da Biodiversidade (acronym SISBIO, N° 60899-1) and were strictly according to the protocols and rules of the Committee on Ethics of Animal Use of the Embrapa Amapá (Protocol No. 002-CEUA-CPAFAP).
Additional information
Section Editor: Guillermo Salgado-Maldonado
Luciano P. Negreiros and Felipe B. Pereira gave equal intellectual contribution for this publication.
Rights and permissions
About this article
Cite this article
Negreiros, L.P., Pereira, F.B., Tavares-Dias, M. et al. Community structure of metazoan parasites from Pimelodus blochii in two rivers of the Western Brazilian Amazon: same seasonal traits, but different anthropogenic impacts. Parasitol Res 117, 3791–3798 (2018). https://doi.org/10.1007/s00436-018-6082-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-018-6082-5