Abstract
Main conclusion
FaMAPK5 and FaMAPK10 genes were involved in ABA-mediated strawberry fruit ripening and could enhance the antioxidant capacity by increasing non-enzymatic components and enzymatic antioxidants.
Abstract
Mitogen-activated protein kinases (MAPKs) are the key proteins involved in plant stress response by activating an antioxidant defense system, which cooperates with plant hormones. However, the involvement of MAPKs in the regulation of strawberry fruit ripening and resistance is unclear. In this study, two genes, FaMAPK5 and FaMAPK10, were isolated, and their expression pattern and function analysis were conducted. The results showed FaMAPK5 and FaMAPK10 were expressed in all tested tissue/organ types and reached the highest expression level at the white stage during strawberry fruit development and ripening. Transient overexpression of FaMAPK5 and FaMAPK10 increased the fruit anthocyanin, abscisic acid (ABA), total sugar, and glucose contents. ABA and especially hydrogen peroxide (H2O2) treatment induced the production of large amounts of H2O2 and noticeably increased the expression levels of FaMAPK5 and FaMAPK10 in strawberry fruit, while the reduced glutathione (GSH) had the opposite effect. The level of total phenol and activities of superoxide dismutase (SOD), catalase (CAT) and peroxidase (POD) significantly increased in FaMAPK5 overexpression fruit, and increased activities of SOD and CAT were observed in FaMAPK10 overexpression fruit. In addition, Botrytis cinerea treatment showed that overexpression of FaMAPK5 conferred retarded disease symptom development and enhanced fruit disease resistance. Our research revealed that FaMAPK5 and FaMAPK10 might participate in ABA-mediated H2O2 signaling in regulating strawberry fruit ripening and resistance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mitogen-activated protein kinases (MAPKs) are identified when cells are stimulated by mitogen. The basic components of MAPK signaling cascades are well conserved from yeast to human and plant, including MAPK kinase kinase (MAPKKK, MAP3K or MEKK), MAPK kinases (MAPKK, MAP2K, MKK or MEK) and MAPK (MPK) (Pitzschke et al. 2009). Each level of the three-kinase cascades has multiple members in a cell, which helps to transmit signal specificity. In recent years, screening and identification of MAPK gene family have been studied in several plants. For instance, about 110 genes encoding MAPK pathway components are identified in the Arabidopsis genome, including 20 MAPKs, 10 MAPKKs and more than 80 MAPKKKs (Colcombet and Hirt 2008). A total of 16 MAPKs, 5 MAPKKs and 89 MAPKKKs are present in tomato (Kong et al. 2012; Wu et al. 2014). Fourteen MAPKs, 5 MAPKKs and 62 MAPKKKs can be found in grapevine (Cakir and Kılıçkaya 2015). Recently, 12 FvMAPKs, 7 FvMAPKKs, 73 FvMAPKKKs and 1 FvMAPKKKK genes are identified in the strawberry (Fragaria vesca) genome (Zhou et al. 2017). Therefore, the interlinked MAPK cascades have thousands of different MAPKKK–MAPKK–MAPK combinations. However, only a limited number of entire MAPK cascades have been deeply investigated in model plants. For example, AtMEKK1–AtMKK4/5–AtMPK3/6 involves the flagellin-mediated innate immune response (Asai et al. 2002), and AtMEKK1–AtMKK2–AtMPK4/6 regulates tolerance to freezing and other abiotic stresses in Arabidopsis thaliana (Kovtun et al. 2000; Teige et al. 2004).
The MAPK controls plant development and various stress responses through effects on phosphorylation of transcription factors and other signaling components (Meng and Zhang 2013). It has been reported that MAPKs are involved in these biological processes by cooperating with plant hormones and reactive oxygen species (ROS). ROS, especially hydrogen peroxide (H2O2) initiates the MAPK cascade to respond to oxidative stresses, a highly conserved pathway in plant (Liu and He 2017). In Arabidopsis thaliana, AtMPK6 and AtMPK3 can be activated by various concentration of H2O2 (Kovtun et al. 2000). In tomato, H2O2 application might enhance chilling tolerance by activating gene expressions of SlMAPK1/2/3 and SlCBF1, and by increasing activities of antioxidant enzymes including superoxide dismutase (SOD), catalase (CAT), ascorbate peroxidase (APX), and peroxidase (POD), and by regulating the concentrations of phytohormones (Wang et al. 2017). H2O2 production is critical for ABA-induced stress tolerance in plants (Zhou et al. 2014). ABA-induced H2O2 generation activated calcium channels of guard cells and promoted the stomatal closure (Pei et al. 2000). Recently, MAPK cascades have been shown to participate in ABA signaling in response to biotic and abiotic stress (Jammes et al. 2011; Liu 2012; Danquah et al. 2014; de Zelicourt et al. 2016). Several lines of evidence indicate that there is a crosstalk among ABA, H2O2 and MAPKs (Smékalová et al. 2014). In Arabidopsis, AtMKK1–AtMPK6 could regulate CAT1 expression in ABA-induced H2O2 production (Xing et al. 2008).
In our previous study, FaMAPK5 and FaMAPK10 expression levels were increased as a result of ABA application and ABA-mediated H2O2 production accelerated strawberry ripening (Luo et al. 2020). Given that MAPKs have been implicated in plant development and defense by mediating ABA and H2O2 signaling, we sought to investigate the role of FaMAPK5 and FaMAPK10 in strawberry fruit ripening and resistance by gene transient overexpression and Botrytis cinerea inoculation experiments, which provide promising candidate genes for strawberry breeding.
Materials and methods
Plant materials
Strawberry (Fragaria × ananassa cv. Benihoppe) was grown in a plastic greenhouse under natural conditions in Chengdu, China. Fruits were harvested at different stages: small green (SG), large green (LG), de-greening (DG), white (WT), initial red (IR) and full red (FR). In addition, roots, shoot, leaves and flowers of strawberry were collected. All plant tissues were harvested 100 days after strawberry planting. These materials were quickly frozen in liquid nitrogen and stored at − 80 °C until use.
Cloning of the FaMAPK5 and FaMAPK10 genes
Total RNA was extracted using a modified CTAB protocol (Jia et al. 2008). Approximately, 1 μg of total RNA was reverse transcribed to cDNA using a SMART™ RACE cDNA Synthesis Kit (Clontech, Mountain View, CA, USA). The primers used for FaMAPK5 and FaMAPK10 gene cloning are listed in Table S1. The PCR reaction protocol was as follows: 94 °C for 5 min, followed by 35 cycles at 94 °C for 30 s, 58 ºC for 30 s and 72 ºC for 2 min, with a final extension at 72 °C for 10 min.
Plasmid construction
The primers used for plasmid construction are listed in Table S2. For overexpression, the open reading frames (ORF) of FaMAPK5 and FaMAPK10 were, respectively, inserted into the modified pCAMBIA1301 (Lin et al. 2018) under the control of the cauliflower mosaic virus (CaMV) 35S promoter using the restriction enzymes BamH I and Xba I, referred to as 35S::MAPK5 and 35S::MAPK10. These constructs and empty vector pC1301-35s-Nos were transformed into Agrobacterium tumefaciens strain GV3101 by the freeze–thaw method (Jia et al. 2011).
Transient gene expression in strawberry fruit
Transient gene expression assays in strawberry fruit were performed as described by Jia et al. (2011). A single Agrobacterium colony carrying the plasmid of interest was inoculated in 5-mL Luria–Bertani (LB) medium supplemented with appropriate antibiotics, and grown overnight on a rotary shaker at 28 °C. Then, 0.1 mL of overnight cultures was added into 50 mL of the same medium and incubated at 28 °C overnight again. The cells were harvested by centrifugation at 4000 g for 15 min at room temperature, and subsequently resuspended in infiltration buffer (10 mM MgCl2, 10 mM MES, 200 mM acetosyringone) to reach an OD600 of 0.8–1.0. Detached fruits at the DG stage were injected with about 800 μL of Agrobacterium suspension using a 1-mL syringe. To do this, the needle tip was inserted into the fruit center from the top, and then the Agrobacterium suspension was slowly and evenly injected into the fruits. After injection, the fruits were incubated at 23 °C with a 16 h light, 8 h dark photoperiod and 80–90% humidity. Ten individual fruits were sampled 5 days later for each infiltration. The experiment was repeated three times.
Quantitative real-time PCR
Quantitative real-time PCR (qRT-PCR) was performed on the CFX96 real-time PCR system (Bio-Rad, Hercules, CA, USA). The primer sequences used for qRT-PCR are shown in Table S3. The amplification program consisted of one cycle at 95 °C for 2 min, followed by 40 cycles of 94 °C for 20 s, 54 °C for 20 s, and 72 °C for 30 s. FaActin gene (AB116565) was used as the internal control. The qRT-PCR experiment was performed on three independent biological replicates with technical replicates. The relative expression levels of the target genes were calculated according to the 2−ΔΔCt algorithm.
Determination of total anthocyanin, ABA, total sugar, sucrose, glucose, fructose, total phenol, H2O2, and reduced glutathione (GSH)
The total anthocyanin determination was conducted using the pH differential method (Cheng and Breen 1991). Briefly, the extraction was conducted from 0.5 g strawberry fruit homogenized with 1.8 mL of cold 1% HCl-ethanol. Then, the homogenate was centrifuged at 8000 g for 25 min at 4 °C. Next, the supernatants were used for measuring the total anthocyanin content. Anthocyanin concentration was expressed as mg pelargonidin 3-glucoside equivalents/100 g of fresh weight.
Total sugar, sucrose, glucose, fructose, total phenol and GSH content were, respectively, determined by the commercial assay kits purchased from Suzhou Comin Biotechnology Co., Ltd. (Suzhou, China). The strawberry fruit (0.2 g) was extracted with the respective extract provided in the kit and centrifuged at 8000 g for 15 min, and then the supernatants were used for the determination of corresponding substances according to the manufacturer’s protocol.
The ABA content was measured by the commercial determination kit purchased from Shanghai MLBIO Biotechnology Co., Ltd. (Shanghai, China). The strawberry fruit (0.2 g) was extracted with 1.8-mL PBS (phosphate buffer saline) for 30 min and centrifuged at 5000 g for 20 min, and then the supernatants were used for measuring the ABA content according to the manufacturer’s protocol by enzyme-linked immunosorbent assay.
H2O2 was measured as described by Cao et al. (2007). Briefly, strawberry fruits (0.5 g) were extracted in 5 mL pre-cooled acetone, followed by centrifugation at 12,000 g for 20 min at 4 °C. The 1-mL supernatant was mixed with 0.1 mL of 10% TiCl4–HCl (v/v) and 0.2 mL of concentrated ammonia. The mixture was kept for 5 min and then centrifuged at 12,000 g for 15 min at 4 °C. The sediment was dissolved by 3 mL of 10% H2SO4 (v/v) and then measured spectrophotometrically at 412 nm to determine the H2O2 content.
Determination of superoxide dismutase (SOD), peroxidase (POD), and catalase (CAT) enzyme activity
The activities of antioxidant enzymes were determined according to the method of Qin et al. (2015). All operations were done at 4 °C. The strawberry fruit (0.5 g) was homogenized in a mortar with 3 mL of 50 mM phosphate buffer at pH 7. Homogenates were filtered through two layers of Miracloth and the filtrate was centrifuged at 15,000 g for 15 min, at 4 °C. The resulting supernatant was stored at −80 °C. The CAT, POD, and SOD enzyme activities were measured spectrophotometrically. The CAT activity was measured by monitoring the decrease in absorbance at 240 nm in 50 mM phosphate buffer (pH 7.5) containing 20 mM H2O2. The POD activity was measured by monitoring the increase in absorbance at 470 nm in 50 mM phosphate buffer (pH 5.5) containing 1 mM guaiacol and 0.5 mM H2O2. The SOD activity in apoplastic fractions was estimated by recording the decrease in optical density of nitroblue tetrazolium dye at 560 nm by the enzyme.
Infection experiment by Botrytis cinerea
The strawberry fruits at the initial red stage were randomly divided into 3 groups and each group had 20 fruits. The sepals were removed from the fruits, and the fruit surface was cleaned with distilled water and dried in the air. Three groups were, respectively, injected with the Agrobacterium suspension harboring the 35S::MAPK5, 35S::MAPK10 and pC1301-35s-Nos plasmids. Three days later, strawberry fruits of each group were all inoculated with Botrytis cinerea. The original Botrytis cinerea was isolated from an infected strawberry fruit. Prior to each experiment, Botrytis cinerea was cultured on PDA (potato dextrose agar) medium at 25 °C for 12 days. The spore suspension was prepared according to the method described as Zhang et al. (2020a). Next, 30 μL of spore suspension was evenly sprayed on the surface of the fruit, and then fruits were placed in artificial climate box, which environment condition was 20 °C, 16/8 h day/night cycle and 90% relative humility. The number of mildewed fruits and the infected area was recorded on the third and fourth day after inoculation.
Diaminobenzidine (DAB) staining
De-greening strawberry fruit discs were treated with H2O, 25 mg/L ABA, 100 mM sucrose, 100 mM GSH or 100 mM H2O2 by immersing discs in these solutions for 6 h, respectively. After that, the treated discs were immediately washed in distilled water and dried by absorbent paper. Subsequently, these discs were stained with DAB for detection of H2O2, according to the method described by Zhang et al. (2020b).
Results
Spatiotemporal expression profiles of FaMAPK5 and FaMAPK10
The expression profiles of FaMAPK5 and FaMAPK10 at different tissues/organs and different stages of fruit development were detected by qRT-PCR. The results showed that FaMAPK5 and FaMAPK10 constitutively expressed in all tested tissues/organs, but they displayed different expression patterns. The most abundant expression levels of FaMAPK5 were detected in leaves, followed by flower, stem, root and fruit (Fig. 1a). The highest transcripts of FaMAPK10 were detected in fruit, followed by flower, root, stem and leaves (Fig. 1b). Clearly, the expression profiles of FaMAPK5 and FaMAPK10 were similar during the fruit development. They displayed low transcript abundance at SG, LG and DG stages, and had a significant transcript accumulation at WT, IR and FR stages (Fig. 1c, 1d).
Transcription levels of FaMAPK5 and FaMAPK10 in different tissues/organs and developmental stages. a Expression patterns of FaMAPAPK5 gene in different tissues of strawberry. The fruits were sampled at full red. b Expression patterns of FaMAPK10 gene in different tissues of strawberry. The fruits were sampled at full red. c Expression patterns of FaMAPK5 gene at different developmental stages of strawberry fruit. d Expression patterns of FaMAPK10 gene at different developmental stages of strawberry fruit. Small green (SG, 7 days after anthesis), large green (LG, 14 days after anthesis), de-greening (DG, 18 days after anthesis), white (WT, 20 days after anthesis), initial red (IR, 23 days after anthesis) and full red (FR, 28 days after anthesis). Data are expressed as means ± standard error of three biological replicates. Different lower case letters indicate significant difference (P < 0.05, ANOVA followed by the Duncan test)
Overexpression of FaMAPK5 and FaMAPK10 affects the levels of ABA, sugar and anthocyanin
To test the role of FaMAPK5 and FaMAPK10 in fruit ripening, 35S::MAPK5 and 35S::MAPK10 were generated, and respectively, infiltrated into the strawberry fruits. The expression of FaMAPK5 and FaMAPK10 significantly increased in the overexpression fruits compared to the control (Fig. 2a, b). ABA is a critical signal molecule in regulating strawberry fruit development and ripening. The results showed that ABA content increased to about 1.12-fold and 1.19-fold as a result of FaMAPK5 and FaMAPK10 overexpression (Fig. 2c). Moreover, overexpression of FaMAPK5 and FaMAPK10 promoted strawberry fruit coloration and anthocyanin accumulation (Fig. 2d). The contents of sucrose and fructose did not significantly increase in the FaMAPK5 overexpression fruits, yet they significantly decreased in the FaMAPK10 overexpression fruits (Fig. 2e, f). However, the contents of glucose and total sugar increased significantly in the FaMAPK5 and FaMAPK10 overexpression fruit compared with that of the control (Fig. 2g, h). These results suggested that alteration of FaMAPK5 and FaMAPK10 transcripts could influence ABA, sugar and anthocyanin contents of strawberry fruit.
Overexpression for the FaMAPK5 and FaMAPK10 genes in strawberry fruits. a Expression levels of FaMAPK5 in the FaMAPK5 overexpression fruit and control by qRT-PCR. b Expression levels of FaMAPK10 in the FaMAPK10 overexpression fruit and control by qRT-PCR. c ABA content of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. d Anthocyanin content of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. e Sucrose content of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. f Fructose content of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. g Glucose content of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. h Total sugar content of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. Data are expressed as means ± standard error of three biological replicates. Asterisk indicates significant difference from empty vector (P < 0.05, ANOVA followed by the Duncan test)
Oxidative stress responses of FaMAPK5 and FaMAPK10
MAPK and H2O2 are involved in the stress response process; thus, the relationship between them is particularly important under oxidative stress. Diaminobenzidine (DAB) staining can visually detect the content of H2O2 in plants. The peroxidase in cells can release oxygen from hydrogen peroxide, and then oxidize DAB to form brown deposit located at the active site of peroxidase. Normally, the darker the brown deposit, the more hydrogen peroxide is accumulated. In this experiment, the strawberry fruit discs were, respectively, treated with distilled water, 25 mg/L ABA, 100 mM sucrose, 100 mM GSH and 100 mM H2O2 for 6 h and then stained with DAB. The results showed that the darker brown color was observed in ABA-, H2O2- and sucrose-treated fruits, while a lighter brown color was observed in the GSH-treated fruits compared with the control (Fig. 3a). To further verify the results, the content of H2O2 in these treated strawberry fruit discs was determined. Compared with the control, the level of H2O2 was higher in ABA-, sucrose- and H2O2-treated fruit discs, especially in H2O2-treated fruit discs, and the content of H2O2 was lower in GSH-treated fruit discs (Fig. 3b), which was consistent with the DAB staining. Taken together, these results indicated that ABA, sucrose, H2O2 and GSH treatment induced or relieved oxidative stress by regulating the H2O2 level of strawberry fruit discs.
Effect of ABA, sucrose, H2O2 and GSH on H2O2 content and expression level of FaMAPK5 and FaMAPK10. a Diaminobenzidine (DAB) staining of the control (H2O, CK), ABA-, sucrose-, H2O2- and GSH-treated strawberry fruit discs. b The corresponding H2O2 content in the control (H2O, CK), ABA-, sucrose-, H2O2- and GSH-treated strawberry fruit discs. c Transcription level of FaMAPK5 at the control (H2O, CK), ABA, sucrose, H2O2 and GSH treatments. d Transcription level of FaMAPK10 at the control (H2O, CK), ABA, sucrose, H2O2 and GSH treatments. Suc: sucrose. Data are expressed as means ± standard error of three biological replicates. Different lower-case letters indicate significant difference (P < 0.05, ANOVA followed by the Duncan test)
To explore the response characteristics of FaMAPK5 and FaMAPK10 gene to different levels of oxidative stress, the expression levels of FaMAPK5 and FaMAPK10 in strawberry fruit discs were detected under the above treatments. The qRT-PCR results showed that transcript levels of FaMAPK5 and FaMAPK10 in strawberry fruit discs were significantly upregulated after ABA and H2O2 treatments, while there was no significant change after sucrose treatment, and the expression levels were significant down-regulated after GSH treatment (Fig. 3c, d). These results indicated that FaMAPK5 and FaMAPK10 might act as an important regulator in ABA and H2O2-mediated or GSH-mediated redox signaling.
Overexpression of FaMAPK5 and FaMAPK10 affects the levels of H2O2, GSH, total phenol and activity of antioxidant enzymes
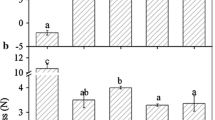
Plant maintains a delicate balance between generation and removal of ROS, which is principally accomplished by the antioxidant defense system that includes enzymatic antioxidants and reduced non-enzymatic components. The H2O2 content did not significantly change in FaMAPK5 and FaMAPK10 overexpression fruits compared with the control (Fig. 4a). The GSH content was significantly increased in overexpressed FaMAPK5 and FaMAPK10 fruit (Fig. 4b). In addition, the level of total phenol and the activities of CAT, SOD and POD increased significantly in overexpressing FaMAPK5 fruit, but increases in total phenol content and POD activity were not observed in overexpressing FaMAPK10 fruit (Fig. 5). These results indicated that overexpression of FaMAPK5 and FaMAPK10 could affect the redox state of strawberry fruit by regulating the levels of endogenous H2O2, non-enzymatic components (GSH and total phenol) and enzymatic antioxidants (CAT, SOD and POD).
Overexpression of FaMAPK5 and FaMAPK10 influences the levels of H2O2 and GSH. a H2O2 content of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. b GSH content of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. Data are expressed as means ± standard error of three biological replicates. Asterisk indicates significant difference from empty vector (P < 0.05, ANOVA followed by the Duncan test)
Effects of transient overexpression of FaMAPK5 and FaMAPK10 on the antioxidant enzyme activities and total phenol content. a CAT activity of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. b SOD activity of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. c POD activity of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. d Total phenol content of strawberry fruit after overexpression of FaMAPK5 and FaMAPK10. Data are expressed as means ± standard error of three biological replicates. Asterisk indicates significant difference from empty vector (P < 0.05, ANOVA followed by the Duncan test)
Overexpression of FaMAPK5 and FaMAPK10 affects resistance to Botrytis cinerea
Due to antioxidant and redox regulation of FaMAPK5 and FaMAPK10 genes, a pathogen infection experiment in vitro was designed to verify whether the genes were involved in strawberry fruit defense against Botrytis cinerea. The results showed that incidence of the gray mold decay was 45%, 10% and 25% in the empty plasmid, overexpressing FaMAPK5 and FaMAPK10 fruits on the third day of inoculation with Botrytis cinerea, respectively. On the 4th day, the decay incidence was 65%, 25% and 65% in above treatments, respectively (Table 1, Fig. 6). The area of disease symptom development in FaMAPK5 overexpressing fruits was smaller than that in fruits of other treatments (Fig. 6). Taken together, overexpression of the FaMAPK5 gene showed a remarkable decrease susceptibility to Botrytis cinerea infection in strawberry fruit, while overexpression of FaMAPK10 did not show an obvious effect.
Effect of overexpression of FaMAPK5 and FaMAPK10 on strawberry fruit disease resistance against Botrytis cinerea. The phenotype of strawberry fruits in the first row shows the third day after infection with Botrytis cinerea, and the phenotype of strawberry fruits in the second row shows the fourth day after infection with Botrytis cinerea
Discussion
It is well known that MAPK cascades are important components for integrating responses to intra- and extracellular signaling in plants (Taj et al. 2010). Each level of MAPK cascades has multiple members, which confers their specific functions in various signal transduction process. Spatial and temporal expression profiles of gene are used for the early analysis of gene function. However, little is known about MAPK expression profiles during fruit development. In our study, the results showed that the expressions of FaMAPK5 and FaMAPK10 were detected in all tested tissues/organs types, and both of which had a significant transcript accumulation at fruit coloration stages (Fig. 1), indicating that FaMAPK5 and FaMAPK10 may play a certain role in strawberry fruit ripening. Color is always used as the index of fruit maturity, and the degree of red coloration in strawberry is determined by the content and composition of anthocyanins (da Silva et al. 2007). Anthocyanins have antioxidant properties, which contribute to free radical scavenging, anti-aging, anti-cancer and immunity boosting in human body (Pojer et al. 2013). Xia et al. (2009) reported that anthocyanins can inhibit apoptosis of many cells by inhibiting the activation of MAPK. In the present study, transient overexpression of FaMAPK5 and FaMAPK10 increased anthocyanin content significantly, which indicated a relationship between anthocyanin accumulation and MAPK expression (Fig. 2d). In addition, fruit sweetness is another good indicator of fruit ripening and flavor that influences consumers’ purchase decision (Schwieterman et al. 2014). Sucrose, fructose and glucose are the main sources of sugar in strawberry fruit. Our results showed that overexpression of FaMAPK5 and FaMAPK10 could remarkably increase the total sugar and glucose content of strawberry fruit, suggesting these two genes increased the total sugar content mainly depending on the glucose content, especially for FaMAPK10 (Fig. 2e–h). More importantly, the enhanced expression of FaMAPK5 and FaMAPK10 caused the increase of ABA content (Fig. 2c). It has been documented that ABA is associated with non-climacteric fruit ripening and it can promote sugar, anthocyanin metabolism and accumulation in fleshy fruits (Jia et al. 2011; Luo et al. 2020). Hence, FaMAPK5 and FaMAPK10 was related to strawberry fruit ripening by regulating the content of anthocyanin, sugar and ABA.
The production of ROS and especially H2O2 is acknowledged as a signal for activation of defense mechanism under various stress conditions, in addition to the toxicity that could lead to oxidative damages in plant. It has been reported that H2O2 interacts with other signaling molecules such as plant hormones and MAPKs to respond to abiotic and biotic stresses (Xia et al. 2014; Saxena et al. 2016; Lee and Back 2017). Here, exogenous ABA and H2O2 application caused a significant increase of H2O2, which also noticeably enhanced the transcription of FaMAPK5 and FaMAPK10. However, GSH treatment decreased the H2O2 content and inhibited the expression of FaMAPK5 and FaMAPK10 (Fig. 3). These results indicated that FaMAPK5 and FaMAPK10 might function in strawberry redox signaling, which was related to the endogenous H2O2 level. As a signal molecule, the generation of H2O2 in stress response is induced by ABA. In response to osmotic stress, ABA-triggered H2O2 accumulation promotes stomatal closure in plant leaves (Li et al. 2015), whereas reducing H2O2 level inhibits ABA-induced stomatal closure (An et al. 2016). Several evidences suggest that MAPKs are involved in ABA and H2O2 signaling (Jammes et al. 2009; Saxena et al. 2016). ABA-induced H2O2 generation activates MAPK and results in upregulation of the expression and activities of antioxidant enzymes. The activation of MAPK also enhances the H2O2 production, forming a positive feedback loop (Zhang et al. 2006, 2007). The exogenous H2O2 activates MAPK cascade, which in turn is mediated by ABA and other hormones (Cristina et al. 2010; Sinha et al. 2011). In this study, increased expression of FaMAPK5 and FaMAPK10 notably upregulated the ABA content, whereas they did not significantly influence the H2O2 content, which was probably a result of a marked increase of non-enzymatic components (GSH and total phenol) and enzymatic antioxidants (CAT, SOD and POD) (Figs. 4, 5). It is speculated that overexpression of FaMAPK5 and FaMAPK10 can enhance the antioxidant capacity in strawberry fruit.
Gray mold caused by Botrytis cinerea is one of the most destructive strawberry diseases worldwide and can cause more than 50% yield losses in severe cases (Cordova et al. 2017). Increasing evidences have shown that MAPKs are indeed involved in pathogen defense (Adachi et al. 2016; Bi and Zhou 2017; Zhang et al. 2018). In citrus, CsMAPK1 plays an important role in defense response to the citrus canker pathogen by inducing defense gene expression and ROS accumulation (de Oliveira et al. 2013). In tomato, SlMPK1/2/3 are involved in nitric oxide-induced disease resistance against Botrytis cinerea. Meanwhile, plant hormones and ROS participate in the disease response of SlMPK1/2/3 (Zheng et al. 2014). In apple, MAPK cascade is closely related to ROS signaling, which together regulate disease resistance against blue mold infection (Cheng et al. 2020). Recent researches have identified that a few MAPKs in strawberry implicate in pathogen response (Wei et al. 2017; Zhang et al. 2020a). In our experiment, overexpression of FaMAPK5 and FaMAPK10 significantly upregulated the ABA content, and activated the ROS scavenging system, and thus inhibited the ROS burst, indicating that the genes may play a regulatory role in strawberry fruit resistance against Botrytis cinerea. However, only FaMAPK5 overexpression outstandingly retarded disease symptom development and conferred enhanced resistance to Botrytis cinerea, while FaMAPK10 overexpression failed in doing that (Fig. 6). Whether FaMAPK10 gene is effective in inhibiting the growth of other pathogens needs further studies.
In conclusion, it was found that FaMAPK5 and FaMAPK10 genes are involved in ABA-mediated strawberry fruit ripening. The expression level of FaMAPK5 and FaMAPK10 is jointly mediated by ABA and H2O2, and they have a feedback regulation on the content of ABA. Overexpression of FaMAPK5 and FaMAPK10 enhanced the antioxidant capacity by increasing non-enzymatic components (GSH and total phenol) and enzymatic antioxidants (CAT, SOD and POD). In addition, FaMAPK5 can improve the disease resistance of strawberry fruit against Botrytis cinerea.
Author contribution statement
YTZ, YL and HRT conceived and designed the study. YTZ wrote the paper. YL, YTL, MY, LXW and XYL conducted experiments and analyzed data. YZ, QC, MYL and YXL reviewed drafts of the paper. All the authors read and approved the final version.
Abbreviations
- CAT:
-
Catalase
- GSH:
-
Glutathione
- MAPK:
-
Mitogen-activated protein kinase
- POD:
-
Peroxidase
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
References
Adachi H, Ishihama N, Nakano T, Yoshioka M, Yoshioka H (2016) Nicotiana benthamiana MAPK-WRKY pathway confers resistance to a necrotrophic pathogen Botrytis cinerea. Plant Signal Behav 11(6):e1183085
An Y, Liu L, Chen L, Wang L (2016) ALA inhibits ABA-induced stomatal closure via reducing H2O2 and Ca2+ levels in guard cells. Front Plant Sci 7:482
Asai T, Tena G, Plotnikova J, Willmann MR, Chiu W-L, Gomez-Gomez L, Boller T, Ausubel FM, Sheen J (2002) MAP kinase signalling cascade in Arabidopsis innate immunity. Nature 415(6875):977–983
Bi G, Zhou J-M (2017) MAP kinase signaling pathways: a hub of plant-microbe interactions. Cell Host Microbe 21(3):270–273
Cakir B, Kılıçkaya O (2015) Mitogen-activated protein kinase cascades in Vitis vinifera. Front Plant Sci 6:556
Cao J, Jiang W, Zhao Y (2007) The guidance of postharvest physiology and biochemistry of fruits and vegetables. China Light Industry press
Cheng GW, Breen PJ (1991) Activity of phenylalanine ammonia-lyase (PAL) and concentrations of anthocyanins and phenolics in developing strawberry fruit. J Amer Soc Hortic Sci 116(5):865–869
Cheng Y, Li C, Hou J, Li Y, Jiang C, Ge Y (2020) Mitogen-activated protein kinase cascade and reactive oxygen species metabolism are involved in acibenzolar-S-methyl-induced disease resistance in apples. J Agric Food Chem 68(39):10928–10936
Colcombet J, Hirt H (2008) Arabidopsis MAPKs: a complex signalling network involved in multiple biological processes. Biochem J 413(2):217–226
Cordova LG, Amiri A, Peres NA (2017) Effectiveness of fungicide treatments following the strawberry advisory system for control of botrytis fruit rot in Florida. Crop Prot 100:163–167
Cristina MS, Petersen M, Mundy J (2010) Mitogen-activated protein kinase signaling in plants. Annu Rev Plant Biol 61:621–649
da Silva FL, Escribano-Bailón MT, Alonso JJP, Rivas-Gonzalo JC, Santos-Buelga C (2007) Anthocyanin pigments in strawberry. LWT-Food Sci Technol 40(2):374–382
Danquah A, de Zelicourt A, Colcombet J, Hirt H (2014) The role of ABA and MAPK signaling pathways in plant abiotic stress responses. Biotechnol Adv 32(1):40–52
de Oliveira MLP, de Lima Silva CC, Abe VY, Costa MGC, Cernadas RA, Benedetti CE (2013) Increased resistance against citrus canker mediated by a citrus mitogen-activated protein kinase. Mol Plant-Microbe Interact 26(10):1190–1199
de Zelicourt A, Colcombet J, Hirt H (2016) The role of MAPK modules and ABA during abiotic stress signaling. Trends Plant Sci 21(8):677–685
Jammes F, Song C, Shin D, Munemasa S, Takeda K, Gu D, Cho D, Lee S, Giordo R, Sritubtim S (2009) MAP kinases MPK9 and MPK12 are preferentially expressed in guard cells and positively regulate ROS-mediated ABA signaling. Proc Natl Acad Sci USA 106(48):20520–20525
Jammes F, Yang X, Xiao S, Kwak JM (2011) Two Arabidopsis guard cell-preferential MAPK genes, MPK9 and MPK12, function in biotic stress response. Plant Signal Behav 6(11):1875–1877
Jia H-F, Zhu X-Q, Jin X-L, Shen Y-Y (2008) An effective method and its modifications for isolation of high-quality total RNA from fruit pulps. J Agric Sci Technol 2(1):58–62
Jia H-F, Chai Y-M, Li C-L, Lu D, Luo J-J, Qin L, Shen Y-Y (2011) Abscisic acid plays an important role in the regulation of strawberry fruit ripening. Plant Physiol 157(1):188–199
Kong F, Wang J, Cheng L, Liu S, Wu J, Peng Z, Lu G (2012) Genome-wide analysis of the mitogen-activated protein kinase gene family in Solanum lycopersicum. Gene 499(1):108–120
Kovtun Y, Chiu W-L, Tena G, Sheen J (2000) Functional analysis of oxidative stress-activated mitogen-activated protein kinase cascade in plants. Proc Natl Acad Sci USA 97(6):2940–2945
Lee HY, Back K (2017) Melatonin is required for H2O2- and NO-mediated defense signaling through MAPKKK3 and OXI1 in Arabidopsis thaliana. J Pineal Res 62(2):e12379
Li H, Wang Y, Xiao J, Xu K (2015) Reduced photosynthetic dark reaction triggered by ABA application increases intercellular CO2 concentration, generates H2O2 and promotes closure of stomata in ginger leaves. Environ Exp Bot 113:11–17
Lin Y, Jiang L, Chen Q, Li Y, Zhang Y, Luo Y, Zhang Y, Sun B, Wang X, Tang H (2018) Comparative transcriptome profiling analysis of red-and white-fleshed strawberry (Fragaria× ananassa) provides new insight into the regulation of the anthocyanin pathway. Plant Cell Physiol 59(9):1844–1859
Liu Y (2012) Roles of mitogen-activated protein kinase cascades in ABA signaling. Plant Cell Rep 31(1):1–12
Liu Y, He C (2017) A review of redox signaling and the control of MAP kinase pathway in plants. Redox Biol 11:192–204
Luo Y, Ge C, Ling Y, Mo F, Yang M, Jiang L, Chen Q, Lin Y, Sun B, Zhang Y (2020) ABA and sucrose co-regulate strawberry fruit ripening and show inhibition of glycolysis. Mol Genet Genomics 295(2):421–438
Meng X, Zhang S (2013) MAPK cascades in plant disease resistance signaling. Annu Rev Phytopathol 51:245–266
Pei Z-M, Murata Y, Benning G, Thomine S, Klüsener B, Allen GJ, Grill E, Schroeder JI (2000) Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature 406(6797):731–734
Pitzschke A, Schikora A, Hirt H (2009) MAPK cascade signalling networks in plant defence. Curr Opin Plant Biol 12(4):421–426
Pojer E, Mattivi F, Johnson D, Stockley CS (2013) The case for anthocyanin consumption to promote human health: a review. Compr Rev Food Sci Food Saf 12(5):483–508
Qin X, Gao M, Jiang X, Si L, Yang Y, Yang R, Cai Z, Xiao H (2015) Effects of spraying antagonistic yeast on postharvest storage performance of strawberry. J Nanjing Agric Univ 38(1):152–160
Saxena I, Srikanth S, Chen Z (2016) Cross talk between H2O2 and interacting signal molecules under plant stress response. Front Plant Sci 7:570
Schwieterman ML, Colquhoun TA, Jaworski EA, Bartoshuk LM, Gilbert JL, Tieman DM, Odabasi AZ, Moskowitz HR, Folta KM, Klee HJ (2014) Strawberry flavor: diverse chemical compositions, a seasonal influence, and effects on sensory perception. PLoS ONE 9(2):e88446
Sinha AK, Jaggi M, Raghuram B, Tuteja N (2011) Mitogen-activated protein kinase signaling in plants under abiotic stress. Plant Signal Behav 6(2):196–203
Smékalová V, Doskočilová A, Komis G, Šamaj J (2014) Crosstalk between secondary messengers, hormones and MAPK modules during abiotic stress signalling in plants. Biotechnol Adv 32(1):2–11
Taj G, Agarwal P, Grant M, Kumar A (2010) MAPK machinery in plants: recognition and response to different stresses through multiple signal transduction pathways. Plant Signal Behav 5(11):1370–1378
Teige M, Scheikl E, Eulgem T, Dóczi R, Ichimura K, Shinozaki K, Dangl JL, Hirt H (2004) The MKK2 pathway mediates cold and salt stress signaling in Arabidopsis. Mol Cell 15(1):141–152
Wang L, Zhao R, Zheng Y, Chen L, Li R, Ma J, Hong X, Ma P, Sheng J, Shen L (2017) SlMAPK1/2/3 and antioxidant enzymes are associated with H2O2-induced chilling tolerance in tomato plants. J Agric Food Chem 65(32):6812–6820
Wei W, Chai Z, Xie Y, Gao K, Cui M, Jiang Y, Feng J (2017) Bioinformatics identification and transcript profile analysis of the mitogen-activated protein kinase gene family in the diploid woodland strawberry Fragaria vesca. PLoS ONE 12(5):e0178596
Wu J, Wang J, Pan C, Guan X, Wang Y, Liu S, He Y, Chen J, Chen L, Lu G (2014) Genome-wide identification of MAPKK and MAPKKK gene families in tomato and transcriptional profiling analysis during development and stress response. PLoS ONE 9(7):e103032
Xia M, Ling W, Zhu H, Ma J, Wang Q, Hou M, Tang Z, Guo H, Liu C, Ye Q (2009) Anthocyanin attenuates CD40-mediated endothelial cell activation and apoptosis by inhibiting CD40-induced MAPK activation. Atherosclerosis 202(1):41–47
Xia X, Gao C, Song L, Zhou Y, Shi K, Yu J (2014) Role of H2O2 dynamics in brassinosteroid-induced stomatal closure and opening in Solanum lycopersicum. Plant Cell Environ 37(9):2036–2050
Xing Y, Jia W, Zhang J (2008) AtMKK1 mediates ABA-induced CAT1 expression and H2O2 production via AtMPK6-coupled signaling in Arabidopsis. Plant J 54(3):440–451
Zhang A, Jiang M, Zhang J, Tan M, Hu X (2006) Mitogen-activated protein kinase is involved in abscisic acid-induced antioxidant defense and acts downstream of reactive oxygen species production in leaves of maize plants. Plant Physiol 141(2):475–487
Zhang A, Jiang M, Zhang J, Ding H, Xu S, Hu X, Tan M (2007) Nitric oxide induced by hydrogen peroxide mediates abscisic acid-induced activation of the mitogen-activated protein kinase cascade involved in antioxidant defense in maize leaves. New Phytol 175(1):36–50
Zhang M, Su J, Zhang Y, Xu J, Zhang S (2018) Conveying endogenous and exogenous signals: MAPK cascades in plant growth and defense. Curr Opin Plant Biol 45:1–10
Zhang G, Jia S, Yan Z, Wang Y, Zhao F, Sun Y (2020a) A strawberry mitogen-activated protein kinase gene, FaMAPK19, is involved in disease resistance against Botrytis cinerea. Sci Hortic 265:109259
Zhang Y, Luo M, Cheng L, Lin Y, Chen Q, Sun B, Gu X, Wang Y, Li M, Luo Y, Wang X, Zhang Y, Tang H (2020b) Identification of the cytosolic glucose-6-phosphate dehydrogenase gene from strawberry involved in cold stress response. Int J Mol Sci 21(19):7322
Zheng Y, Hong H, Chen L, Li J, Sheng J, Shen L (2014) LeMAPK1, LeMAPK2, and LeMAPK3 are associated with nitric oxide-induced defense response against Botrytis cinerea in the Lycopersicon esculentum fruit. J Agric Food Chem 62(6):1390–1396
Zhou J, Wang J, Li X, Xia X-J, Zhou Y-H, Shi K, Chen Z, Yu J-Q (2014) H2O2 mediates the crosstalk of brassinosteroid and abscisic acid in tomato responses to heat and oxidative stresses. J Exp Bot 65(15):4371–4383
Zhou H, Ren S, Han Y, Zhang Q, Qin L, Xing Y (2017) Identification and analysis of mitogen-activated protein kinase (MAPK) cascades in Fragaria vesca. Int J Mol Sci 18(8):1766
Acknowledgements
This study was supported by the National Natural Science Foundation of China (3180817); the State Education Ministry, Key projects of Sichuan Provincial Education Department (172A0319) and Key projects of Sichuan Provincial Science and Technology Department (2018NZ0126) and Service Station Projects of New Rural Development Research Institute, Sichuan Agricultural University (2018, 2020) and the Cooperation Project of Ya'an City and Sichuan Agricultural University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, Y., Long, Y., Liu, Y. et al. MAPK5 and MAPK10 overexpression influences strawberry fruit ripening, antioxidant capacity and resistance to Botrytis cinerea. Planta 255, 19 (2022). https://doi.org/10.1007/s00425-021-03804-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-021-03804-z