Abstract
Purpose
While non-noxious local cooling is widely used in physical medicine, its effect on muscle sympathetic nerve activity (MSNA) and cardiovascular regulation are not clear. The purpose of the present study was to assess the responses of MSNA, blood pressure (BP), heart rate (HR) and local blood flow during non-noxious local cooling.
Methods
The study included two protocols. Both protocols consisted of 10-min rest in supine position, followed by 15-min local cooling (15 °C) of the shin and anterior foot, and 20-min recovery. MSNA of the right common peroneal nerve, BP, HR, and shin skin temperature (TSK) were recorded in eight men in the first protocol, while leg blood flow (LBF) was measured in the same subjects by strain-gauge plethysmography in the second protocol.
Results
TSK gradually decreased from 31.5 ± 0.02 to 16.0 ± 1.01 °C (mean ± SEM) during local cooling, and gradually increased after the end of local cooling. No subject complained of pain, and BP and HR remained constant. The MSNA burst rate increased significantly (p < 0.05) to 141.1 ± 12.5 % during local cooling, but decreased significantly (p < 0.05) to 73.6 ± 5.9 % during the recovery period. Total MSNA also increased to 148.0 ± 14.2 % (p < 0.05) during local cooling, and decreased to 74.0 ± 13.9 % (p < 0.05) at recovery. LBF remained constant through the experiment.
Conclusions
The results suggest that MSNA is activated by non-noxious local cooling, and attenuated after the end of local cooling without any changes in HR and BP.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Muscle sympathetic nerve activity (MSNA) can be recorded from the muscle nerve fascicles in the limbs by the technique of microneurography (Delius et al. 1972a, b, c; Fagius et al. 1989; Hagbarth et al. 1972; Hagbarth and Vallbo 1968; Mark et al. 1985; Mitchell and Victor 1996; Seals and Victor 1991; Vallbo et al. 1979, 2004; Victor et al. 1987). MSNA has been studied during various types of physiological stimuli and induced various changes by these stimuli (Cui et al. 2007; Mano 1998; Mizushima et al. 1998; Nakamura et al. 2005; Niimi et al. 1997; Tanaka et al. 1991). There is general agreement that MSNA primarily represents the activity of the vasoconstrictor fibers throughout the body, which innervate skeletal blood vessels and thus play an important role in the regulation of systemic blood pressure (BP) (Charkoudian et al. 2005; Mitchell and Victor 1996; Saito et al. 1990). Therefore, activation of MSNA generally induces a rise in BP, while reduction of MSNA induces a fall in BP.
We reported previously that non-noxious local heat application to the leg reduced MSNA but neither changed BP nor heart rate (HR) in healthy men (Takahashi et al. 2011). We concluded that the response of MSNA during non-noxious local heat was induced by a local reflex mechanism based on the lack of change in BP, and thus the control of BP and HR was not affected by reduction in MSNA, suggesting that the effect of MSNA is limited to the local area. On the other hand, in the cold pressor test, which is often used to evaluate the sympathetic nervous system, BP, HR and MSNA increase after immersion of the hand in ice cold water, together with induction of intense pain (Kregel et al. 1992; Victor et al. 1987; Mizushima et al. 1998). To our knowledge, however, the response of BP, HR and MSNA to non-noxious local cooling of the skin has not been examined in detail.
Non-noxious local cooling, also known as cryotherapy, is one of the most generally used physical therapy in rehabilitation. Cryotherapy decreases local blood flow by causing vasoconstriction, and reduces tissue metabolism, oxygen utilization, inflammation, and muscle spasm (Nadler et al. 2004). The decrease in local blood flow is produced by the direct effect of the applied cold and by reflex mechanisms ranging from a simple monosynaptic reflex to a complex polysynaptic reflex (Lehmann and Lateur 1990). Previous studies demonstrated that application of mild non-noxious cold ice pack on the leg induced a decrease in leg blood flow (LBF) (Taber et al. 1992; Weston et al. 1994). However, Fiscus et al. (2005) reported that cold-water immersion of the lower part of the leg did not change LBF.
The present study was designed to determine the effects of non-noxious local cooling of the leg [i.e., cooling of skin temperature (TSK)] on BP, HR, MSNA and regional blood flow in normal subjects. The study was performed by measuring BP, HR, TSK, LBF and MSNA in the leg before, during, and after 15 min of non-noxious local cooling of the leg.
Methods
Subjects
Eight healthy adult men aged 21–35 years participated in this study after signing a written informed consent. All subjects were normotensive, taking no medications, and free of cardiovascular or neuromuscular diseases, based on medical history and physical examination. The study was approved by the Human Investigation Committee of the Wakayama Medical University, Japan.
Recording of sympathetic nerve activity
Multiunit recording of postganglionic MSNA was obtained from the right common peroneal nerve fascicles at the level of the fibular head in the lower extremity, according to the method described previously (Mizushima et al. 1998; Nakamura et al. 2005; Takahashi et al. 2011). For this purpose, a tungsten microelectrode with an uninsulated tip (diameter 1–5 μm) expanding to an insulated shaft (0.2 mm) was inserted into the nerve fascicles. A surface reference electrode (diameter 3 cm) was placed medially 2–3 cm from the recording electrode. The neural activity was collected through a microclipper using a shielded cord, amplified and filtered (bandwidth = 500–2000 Hz) using an electromyogram recording system (Nicolet, Viking SELECT VIASIS). For quantification of the electric signal, the amplified signal was fully rectified and integrated using a time constant of 100 ms. The mean voltage of the MSNA neurogram was continuously displayed together with the electrocardiogram (ECG) and raw MSNA neurogram on a chart recorder (NEC, Omniace II RA1300) at a paper speed of 5 mm/s. The nerve signal was also continuously routed to a loudspeaker for audio recognition of MSNA bursts. The MSNA recording was accepted when the following three criteria were met (Vallbo et al. 1979; Wallin 1983), first, when electrical stimulation (0.1 mA; 0.2 ms; 1 Hz) through the electrode inserted into the common peroneal nerve elicited an involuntary muscle contraction but not paresthesia; second, when tapping or stretching the muscle or tendon supplied by the impaled fascicle elicited afferent mechanoreceptor discharges; and third, when the neurogram displayed spontaneous, intermittent, pulse-synchronous bursts that increased during a sustained expiration and phases 2 and 3 of the Valsalva maneuver but not during an acoustic stimulus (hand clap).
Recording of leg blood flow, cardiovascular variables and skin temperature
Heart rate was obtained from the R–R interval of the ECG and arterial pressure was measured manually with a sphygmomanometer (model ES-H55D, Terumo). The mean blood pressure (MBP) was calculated from the diastolic blood pressure (DBP) and systolic blood pressure (SBP) as MBP = DBP + (SBP − DBP)/3. LBF was determined on the right side using the plethysmographic technique (plethysmography: model ECR5, Hokanson) as described previously (Whitney 1953; Gamble et al. 1993). A silicon tube filled with mercury was placed around the right calf at maximum circumference and positioned 20 cm above the heart level. The maximum circumference of the calf region was located 3 cm above a midpoint between the medical condyle of the femur and upper edge of the medical malleolus. This setup ensured that the silicone tube was not directly cooled by the application of ice packs. Changes in the calf circumference were measured by a bridge amplifier during proximal venous occlusion. In a series of preliminary experiments, we assessed the effect of local pressure on LBF by applying a weight identical to that of the ice pack to the area where the ice pack would be applied. The results showed that while the weight placement changed the baseline, it did not affect the impedance during venous occlusion. TSK of the right shin temperature was measured using a thermocouple thermometer (model LT8A, Gram Corporation).
Local cooling
Local cooling was applied to the ipsilateral anterior area from 4 cm below the midcalf region down to the toes using two ice packs (Fig. 1). The ice pack was covered with double-folded paper towels to protect the skin. The temperature on the surface part of the towels in contact with the skin was 15 °C immediately before application of the ice pack.
Experimental protocols
In the first protocol, MSNA was measured in eight subjects in a temperature-controlled room (26.0 ± 0.5 °C). Each subject reported to the laboratory at 16:00 h. After voiding, the subject rested comfortably in a supine position on the examination table wearing short pants and a T-shirt. After recording all variables for 10 min at rest (control period), local cooling was applied for 15 min (LC period) while monitoring continued. After removal of the ice packs, recovery measurements were made for 20 min (recovery period). Thus, MSNA was measured continuously for 45 min. TSK, HR and MBP were recorded every 1 min, and averaged every 2 min except during the first minute of local cooling. Furthermore, in a preliminarily study, we confirmed that TSK of the right shin was stabilized after 15 min of local cooling (the selection of this time period was based on the fact that cryotherapy is usually applied for 15 min in the clinic). For this reason, we used the 15-min duration for local cooling in present study. On the other hand, the recovery spanned over 20 min (i.e., longer than local cooling).
In the second protocol, LBF was measured in the same group of eight normal subjects without recording the MSNA, and all other parameters were recorded in the same manner as in the first protocol. The second protocol was conducted on a separate day within 1 week of the first protocol, and under conditions (room temperature and time of experiment) identical to the first protocol.
Data analysis
Records were divided into 1-min periods for analysis. Acceptable bursts of MSNA were identified by inspection of the mean voltage neurogram on the chart recorder, synchronized with the audio signal from loudspeaker and the pulse wave of the ECG. All microneurographic recordings were analyzed by the same investigator. MSNA was expressed as both burst rate (bursts/min) and total activity (total MSNA) in arbitrary units (bursts/min × average burst amplitude/min), according to the method described previously (Mark et al. 1985; Wallin 1983). MSNA was averaged each minute of the control, local cooling and recovery periods. Data from the 10-min control period were averaged to obtain single baseline values. To analyze the MSNA response, the mean values during local cooling and recovery periods were expressed as percentages of those recorded during the control period.
All data were reported as mean ± standard error of the mean. The data of each parameter showed skewed distribution. Changes in each variable due to treatment (control, LC period, and recovery) were examined for statistical significance using the Mann–Whitney U test. A p value less than 0.05 was considered significant in all statistical analyses.
Results
Protocol 1
Skin temperature
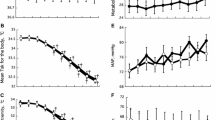
Shin TSK gradually and significantly decreased (p < 0.01) during local cooling from 31.5 ± 0.02 °C to the lowest value of 16.0 ± 1.01 °C. All subjects were questioned verbally about pain and hot sensation, and none complained of pain or soreness during local cooling. The shin TSK gradually increased after removal of the cold pack, but remained significantly lower (p < 0.01) compared with the control period until the end of the recovery period (Fig. 2).
Heart rate and mean blood pressure
HR and MBP remained constant throughout LC period and recovery (Fig. 3a, b).
Muscle sympathetic nerve activity
The mean MSNA burst rate during the control period was 14.5 ± 2.04 bursts/min, increased significantly during LC period to 141.1 ± 12.5 % (p < 0.05) and decreased significantly (p < 0.05) to become lower (73.6 ± 5.9 %) than the control value after the end of local cooling (Fig. 4a). The total MSNA also increased significantly (p < 0.05) to 148.0 ± 14.2 % during local cooling and decreased significantly (p < 0.05) to 74.0 ± 13.9 % relative to the baseline after removal of the ice pack (Fig. 4b). The percent change in MSNA burst rate during local cooling negatively correlated significantly with the shin temperature (R = −0.34, p = 0.0105, Fig. 5).
Protocol 2
Leg blood flow
LBF in the right leg remained constant throughout LC period and recovery (Fig. 6).
Discussion
The major findings of the present study were the following; (1) Local cooling (15 °C) of the skin over the shin and anterior foot increased MSNA of the ipsilateral common peroneal nerve, and removal of local cooling induced reduction of MSNA, and (2) HR, MBP and LBF remained constant throughout the study. These findings suggest that the fall in shin TSK induced activation of MSNA while increase in shin TSK induced attenuation of MSNA, and that the change in MSNA was induced by a local reflex mechanism in the absence of any response in HR and MBP.
The cold pressor test is known to induce a rapid increase in HR and MBP, as well as increase in MSNA (over 150 %) (Mizushima et al. 1998). The test usually involves the use of low-temperature water (around 0 °C) to cool the hand. On the other hand, the local cooling applied in the present study involved cooling the skin to mild temperature (15 °C) using ice packs. Such mild cooling induced a mild increase in MSNA (<50 %), relative to the cold pressor test. The present findings of changes in MSNA during and after mild local cooling without changes in HR suggest that the threshold of the MSNA response to changes in shin TSK differed from that affecting the response of cardiac sympathetic and parasympathetic nerve activities. The stable MBP and changes in MSNA during and after mild local cooling suggest that the response in MSNA may represent a local reflex response elicited in the local cooling area. Ray et al. (1997) also reported that the MBP, HR and MSNA in the lower leg did not change by 30 min mild local cooling of the forearm. Thus, the threshold change in shin TSK necessary to elicit a local MSNA response seems lower than that required to change HR and BP. The stability of HR and MBP during and after mild local cooling implies a stable cardiovascular control during and after local cooling.
In the present study, none of the participating subjects complained of pain during local cooling and HR did not increase during local cooling. Victor et al. (1987) reported that tachycardia during the cold pressor test is induced by pain sensation. In contrast, several studies that have previously addressed the effects of somatic afferent stimulation on MSNA reported increased MSNA response (Kregel et al. 1992; Mizushima et al. 1998; Nakamura et al. 2008). Somatic afferent stimulation activates the ascending tracts in the spinal cord. In particular, the spinoreticular tract projecting to the medullary reticular formation activates the vasomotor center in the medulla oblongata. Activation of the projection from the vasomotor center to the intermediolateral cell column in the thoracolumbar cord results in excitation of the sympathetic preganglionic neurons causing increased MSNA (Nakamura et al. 2008). In this regard, Kregel et al. (1992) reported that immersion of the hand in 7 °C water induced intense pain and increased MSNA in the lower leg, whereas immersion of the hand in 14 °C water that did not induce intense pain did not increase MSNA in the lower leg. Furthermore, Mizushima et al. (1998) showed that immersion of the hand in cold water increased MSNA, BP and HR and was associated with a sensation of intense pain. The significant relationship between somatic afferent stimulation and MSNA suggests that somatic stimulation during local cooling in the present study did not activate the ascending tract in the spinal cord and vasomotor center in the medulla, and that the change in MSNA in the absence of pain during local cooling was induced by a local reflex mechanism.
In the present study, MSNA of the common peroneal nerve increased rapidly during 15 min local cooling (15 °C) on the ipsilateral lower leg, and then decreased to a level lower than the control value after removal of the cold pack. Moreover, MSNA activity during local cooling correlated significantly with shin temperature. In comparison, Mizushima et al. (1998) reported that MSNA of the common peroneal nerve increased during 3 min of the cold pressor test (hand immersion in 0 °C water) and also similar to our protocol it decreased after the cold pressor test, but it remained during the 3-min recovery period at a level higher than the control value. Thus, the increase in MSNA induced by local cooling observed in the present study was similar to that seen in the cold pressor test, but a different pattern was noted after the end of local cooling. The cold pressor test induces stimulation of the MSNA throughout the entire body, where our test stimulated MSNA in only the local area of application through a local reflex mechanism. To the best of our knowledge, the present study is the first report to evaluate the response of MSNA to non-noxious local cooling.
We reported previously that non-noxious local heat application (42 °C) to the shin and anterior foot induced a significant increase in LBF (Takahashi et al. 2011). In the present study, non-noxious local cooling (15 °C) to the same area did not change LBF. Fiscus et al. (2005) reported that immersion of the lower leg in warm-water (40 °C) increased LBF whereas cold-water (13 °C) immersion did not. The LBF response observed in our study is similar to the latter. As discussed by Fiscus et al. (2005), the lack of change in LBF during cold-water immersion may represent a body reaction to preserve core body temperature or a vasomotor response. In the present and above studies, LBF was measured using the plethysmographic method. This method measures total regional blood flow of the leg, which includes mainly flow to muscles and skin. It is well known that any increase in MSNA is associated with vasoconstriction of skeletal muscle blood vessels. In the present study, the significant increase in MSNA from the common peroneal nerve recorded during local cooling of the shin and anterior foot was not associated with a decrease in LBF. We assume that the area that controls MSNA of the common peroneal nerve is located in part of the leg, and that MSNA and vasomotor response of skeletal blood vessels in other areas might show a response different to the area of the common peroneal nerve. Moreover, it is well known that the vasoconstrictor skin sympathetic nerve activity (SSNA) controls the tone of cutaneous vessels. In this regard, mild local skin cooling induced an increase in vasoconstrictor SSNA and a decrease in local skin blood flow (Sawasaki et al. 2001; Yamazaki et al. 2006). Therefore, the local skin blood flow during local cooling could have decreased in the present study, and the change in skin blood flow could also be different in the locally cooled area compared with other areas of the leg. On the other hand, several other studies (Taber et al. 1992; Weston et al. 1994) reported a decrease in LBF during application of mild cooling (10–20 °C) to parts of the leg. In these studies, the ice packs were applied for 20 min, whereas local cooling in the present study lasted 15 min. It is possible that LBF would have decreased had the local cooling been applied for longer.
Cryotherapy is widely used clinically and our finding showed that cryotherapy is a harmless treatment in improving patient care based on lack of both hypo-perfusion and changes in cardiovascular responses. Such lack of change in LBF suggests that the reduction in local blood flow observed in cryotherapy is localized in a limited area and that the sympathetic nerve response during cryotherapy seems to be limited to the local area.
Technical considerations
Our study was performed by simultaneously measuring LBF and MSNA in the lower leg before, during, and after 15 min of local cool application. In the first protocol, MSNA was measured in eight subjects while in the second protocol; LBF was measured in the same group of eight normal subjects without recording MSNA. Such design (two separate protocols) was necessary because the calf of the plethysmograph made contact with the tungsten microelectrode inserted into the common peroneal nerve. We acknowledged that such design was not ideal (i.e., no simultaneous measurements), but nevertheless necessary.
Conclusions
The present study demonstrated suppression of activation of change in MSNA during non-noxious local cooling of the skin after the end of local cooling, without changes in BP and HR. The findings indicate different MSNA control mechanisms between non-noxious local cooling and noxious local cooling, and suggest that the MSNA response during non-noxious local cooling was induced by a local reflex mechanism.
Abbreviations
- BP:
-
Blood pressure
- DBP:
-
Diastolic blood pressure
- ECG:
-
Electrocardiogram
- HR:
-
Heart rate
- LBF:
-
Leg blood flow
- MBP:
-
Mean blood pressure
- MSNA:
-
Muscle sympathetic nerve activity
- SBP:
-
Systolic blood pressure
- TSK:
-
Skin temperature
References
Charkoudian N, Joyner MJ, Johnson CP, Eisenach JH, Dietz NM, Wallin BG (2005) Balance between cardiac output and sympathetic nerve activity in resting humans: role in arterial pressure regulation. J Physiol (Lond) 568:315–321
Cui J, Durand S, Crandall CG (2007) Baroreflex control of muscle sympathetic nerve activity during skin surface cooling. J Appl Physiol 103:1284–1289
Delius W, Hagbarth KE, Hongell A, Wallin BG (1972a) General characteristics of sympathetic activity in human muscle nerves. Acta Physiol Scand 84:164–176
Delius W, Hagbarth KE, Hongell A, Wallin BG (1972b) Manoeuvres affecting sympathetic outflow in human muscle nerves. Acta Physiol Scand 84:82–94
Delius W, Hagbarth KE, Hongell A, Wallin BG (1972c) Manoeuvres affecting sympathetic outflow in human skin nerves. Acta Physiol Scand 84:177–186
Fagius J, Karhuvaara S, Sundlof G (1989) The cold pressor test: effects on sympathetic nerve activity in human muscle and skin nerve fascicles. Acta Physiol Scand 137:325–334
Fiscus KA, Kaminski TW, Powers ME (2005) Changes in lower-leg blood flow during warm-, cold-, and contrast-water therapy. Arch Phys Med Rehabil 86:1404–1410
Gamble BJ, Gartside IB, Christ F (1993) A reassessment of mercury in silastic strain gauge plethysmography for microvascular permeability assessment in man. J Physiol (Lond) 464:407–422
Hagbarth KE, Vallbo AB (1968) Pulse and respiratory grouping of sympathetic impulses in human muscle–nerves. Acta Physiol Scand 74:96–108
Hagbarth KE, Hallin RG, Hongell A, Torebjork HE, Wallin BG (1972) General characteristics of sympathetic activity in human skin nerves. Acta Physiol Scand 84:164–176
Kregel KC, Seals DR, Callister R (1992) Sympathetic nervous system activity during skin cooling in humans: relationship to stimulus intensity and pain sensation. J Physiol (Lond) 454:359–371
Lehmann JF, Lateur BJ (1990) Diathermy and superficial heat, laser, and cold therapy. In: Kottke FJ, Lehmann JF (eds) Krusen’s handbook of physical medicine and rehabilitation, 4th edn. W. B. Saunders, Philadelphia, pp 283–367
Mano T (1998) Microneurographic research on sympathetic nerve responses to environmental stimuli in humans. Jpn J Physiol 48:99–114
Mark AL, Victor RG, Nerhed C, Wallin BG (1985) Microneurographic studies of the mechanisms of sympathetic nerve responses to static exercise in humans. Circ Res 57:461–469
Mitchell JH, Victor RG (1996) Neural control of the cardiovascular system: insights from muscle sympathetic nerve recordings in humans. Med Sci Sports Exerc 28:S60–S69
Mizushima T, Tajima F, Nakamura T, Yamamoto M, Lee KH, Ogata H (1998) Muscle sympathetic nerve activity during cold pressor test in patients with cerebrovascular accidents. Stroke 29:607–612
Nadler SF, Weingand K, Kruse RJ (2004) The physiologic basis and clinical applications of cryotherapy and thermotherapy for the pain practitioner. Pain Physician 7:395–399
Nakamura T, Mizushima T, Yamato M, Kawazu T, Umezu Y, Tajima F (2005) Muscle sympathetic nerve activity during isometric exercise in patients with cerebrovascular accidents. Arch Phys Med Rehabil 86:436–441
Nakamura T, Kawabe K, Sapru HN (2008) Cold pressor test in the rat: medullary and spinal pathways and neurotransmitters. Am J Physiol 295:H1780–H1787
Niimi Y, Matsukawa T, Sugiyama Y, Shamsuzzaman AS, Ito H, Sobue G, Mano T (1997) Effect of heat stress on muscle sympathetic nerve activity in humans. J Auton Nerv Syst 19:61–67
Ray CA, Keith MH, Kathryn HG, Edward TM (1997) Muscle cooling delays activation of the muscle metaboreflex in humans. Am J Physiol 273:H2436–H2441
Saito M, Naito M, Mano T (1990) Different responses in skin and muscle sympathetic nerve activity to static muscle contraction. J Appl Physiol 69:2085–2090
Sawasaki N, Iwase S, Mano T (2001) Effect of skin sympathetic response to local or systemic cold exposure on thermoregulatory function in humans. Auton Neurosci 87:274–281
Seals DR, Victor RG (1991) Regulation of muscle sympathetic nerve activity during exercise in humans. Exerc Sport Sci Rev 19:313–349
Taber C, Contryman K, Fahrenbruch J, LaCount K, Cornwall MW (1992) Measurement of reactive vasodilation during cold gel pack application to nontraumatized ankles. Phys Ther 72:294–299
Takahashi N, Nakamura T, Kanno N, Kimura K, Toge Y, Lee KH, Tajima F (2011) Local heat application to the leg reduces muscle sympathetic nerve activity in human. Eur J Appl Physiol 111:2203–2211
Tanaka H, Sagawa S, Miki K, Tajima F, Freund BJ, Claybaugh JR, Shiraki K (1991) Sympathetic nerve activity and renal responses during continuous negative-pressure breathing in humans. Am J Physiol 261:R276–R282
Vallbo AB, Hagbarth KE, Torebjork HE, Wallin BG (1979) Somatosensory, proprioceptive, and sympathetic activity in human peripheral nerves. Physiol Rev 59:919–957
Vallbo AB, Hagbarth KE, Wallin BG (2004) Microneurography: how the technique developed and its role in the investigation of the sympathetic nervous system. J Appl Physiol 96:1262–1269
Victor RG, Leimbach W Jr, Seals DR, Wallin BG, Mark AL (1987) Effects of the cold pressor test on muscle sympathetic nerve activity in humans. Hypertension 9:429–436
Wallin BG (1983) Intraneural recording and autonomic function in man. In: Bannister R (ed) Autonomic failure. Oxford University Press, Oxford, pp 36–51
Weston M, Taber Casagranda L, Cornwall M (1994) Changes in local blood volume during cold gel pack application to traumatized ankles. J Orthop Sports Phys Ther 19:197–199
Whitney RJ (1953) The measurement of volume changes in human limbs. J Physiol (Lond) 121:1–27
Yamazaki F, Sone R, Zhao K, Alvarez GE, Kosiba WA, Johnson JM (2006) Rate dependency and role of nitric oxide in vascular response to direct cooling in human skin. J Appl Physiol 100:42–50
Acknowledgments
We thank Dr. Faiq G. Issa (www.word-medex.com.au) for the careful reading and editing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by George Havenith.
Rights and permissions
About this article
Cite this article
Ishida, K., Nakamura, T., Kimura, K. et al. Suppression of activation of muscle sympathetic nerve during non-noxious local cooling after the end of local cooling in normal adults. Eur J Appl Physiol 116, 851–858 (2016). https://doi.org/10.1007/s00421-016-3343-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-016-3343-9