Abstract
Purpose
Previous studies have shown an association between obstructive sleep apnoea syndrome (OSAS) and cardiovascular events. Whether this association is mediated by an impairment of endothelial function, which is itself a driver of elevated cardiovascular risk, has yet to be clarified, as it is the eventual protective role of several OSAS treatments. The aim of our meta-analysis is to evaluate the effect of various OSAS treatments on endothelial function calculated by means of flow-mediated dilatation (FMD).
Methods
We conducted a meta-analysis of prospective studies including patients affected by mild to severe OSAS treated with continuous positive airway pressure (CPAP), surgery, oral appliance and medical treatments. FMD was measured before and after treatment
Results
After pooling results from different treatment strategies, OSAS treatment showed a positive impact on endothelial function (Mean Difference [MD] = 2.58; 95% CI 1.95–3.20; p < 0.00001).
Conclusions
Our study supports the hypothesis that several modalities of treatment for OSAS positively impact endothelial function. Whether this effect also associates with an improvement of clinical outcomes remains to be ascertained.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Several epidemiological studies have shown an association between obstructive sleep apnoea syndrome (OSAS) and cardiovascular events, particularly stroke [1,2,3].
Endothelial dysfunction is a marker of early-stage vascular impairment and independently predicts cardiovascular morbidity and mortality in the general population and, specifically, in patients affected by OSAS [4].
OSAS treatment with continuous positive airway pressure (CPAP) appeared to be beneficial, improving endothelial function indexes [5]. Alternative medical or surgical treatments have been less extensively explored in this sense.
Ventilatory treatment is still considered the gold standard treatment for OSAS but the interest towards alternative modalities has been significantly growing, being adherence the most critical aspect of CPAP therapy. For this reason, further appropriate evidence supporting non-CPAP therapies is needed to offer patients valid alternatives.
The aim of our study was to revise and evaluate the effect of various OSAS treatment options, with a special focus on non-CPAP modalities, on endothelial function calculated by means of flow-mediated dilatation (FMD), a widely available, inexpensive, non-invasive ultrasound-based method where arterial diameter is measured in response to an increase in shear stress, which causes release of nitric oxide from the endothelium and consequent endothelium dependent dilatation.
Methods
Literature search protocol
We performed a comprehensive review of the literature until 31st March 2018 regarding the impact on endothelial function calculated by FMD in patients with OSAS. We searched PubMed, EMBASE, the Cochrane Library and CENTRAL electronic databases for keywords such as (1) Endothelial; (2) Sleep Apnoea; (3) OSAS; (4) OSA; (5) OSAHS.
The study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement and Meta-analysis of Observational Studies in Epidemiology (MOOSE) guidelines [6, 7]. The following inclusion criteria were applied to each article: (1) prospective studies; (2) diagnosis of mild to severe OSAS defined according to the American Academy of Sleep Medicine criteria [8]; (3) patients treated with CPAP or surgery or oral appliances or medications; (4) data regarding FMD collected before and after treatment. To further reduce the risk of incomplete literature search, a manual search through the references of the included papers was also performed.
The methodological quality of all included studies was assessed according to the Newcastle–Ottawa Scale. The Newcastle–Ottawa Scale guideline was carried out as follows. A study was awarded a maximum of one star for each numbered item within the patient selection and outcome categories. A maximum of four stars were given for patient selection, three stars for outcome, and two stars for comparability. A higher number of stars indicated higher quality of the eligible study. One score represented one star. One reviewer extracted all data and a second reviewer verified the accuracy of data extraction.
Outcome measurement
Endothelial function was evaluated using FMD performed before treatment initiation and after treatment completion during a follow-up visit. FMD evaluation was carried out in all selected studies according to the principles described by Celermajer et al. [9]. The diameter of the target artery was measured from two-dimensional ultrasound images. In each study, scans were taken at rest, during reactive hyperaemia, again at rest, and after sublingual glyceryl trinitrate (GTN). The target artery (superficial femoral just distal to the bifurcation of the common femoral artery, or the brachial 2–15 cm above the elbow) was scanned in longitudinal section.
The subject lay at rest for at least 10 min before a first resting scan was recorded, and arterial flow velocity was measured, by means of a pulsed Doppler signal. Increased flow was then induced by inflation of a pneumatic tourniquet to a pressure of 300 mm Hg for 45 min. A second scan was taken for 30 s before and 90 s after cuff deflation, including a repeat flow velocity recording for the first 15 s after cuff release. 15 min was allowed for vessel recovery, and then a further resting scan was taken. Sublingual GTN spray (400 ug) was then administered, and 3–4 min later the last scan was done.
The arterial diameter was measured at a fixed distance from an anatomical marker, such as a bifurcation, with ultrasonic calipers. For the reactive hyperaemia scan, diameter, measurements were taken 45–60 s after cuff deflation. Four cardiac cycles were analyzed for each scan and the measurements averaged. Vessel diameters in scans after reactive hyperaemia, 15-min rest, and GTN were expressed as percentages of the first control scan. Flow was calculated from Doppler flow velocity and vessel diameter. Reactive hyperaemia was calculated as the maximum flow recorded in the first 15 s after cuff deflation divided by the flow during the resting (baseline) scan. The mean vessel diameter and percent dilatation for each patient were obtained by averaging the measurements taken over all occasions on which that patient was studied. The post-treatment improvement of FMD was evaluated in this study.
Statistical analysis
The pooled estimate of the weighted mean difference and 95% confidence interval for continuous data were calculated. The observed treatment effects were pooled across studies applying DerSimonian–Laird random-effect model to account for different lengths of follow-up among studies. At sensitivity analysis, we first excluded individual studies to establish the impact of each trial on the pooled effect size, and then excluded studies with a follow-up < 2 weeks. A two-tailed alpha of 5% was used for hypothesis testing. Heterogeneity was assessed with Cochran Q via a chi-square test and quantified with the I2 test. Potential publication bias was assessed by visual inspection of funnel plot. Analyses were conducted using Review Manager 5.3 (Cochrane Collaboration, Oxford, United Kingdom) and Stata 13.0 (Version 13, StataCorp, Station, TX, USA).
Results
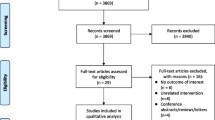
We screened 748 potentially relevant reports. After excluding reviews and studies not reporting the direct effect of OSAS treatment on endothelial function, a total of 43 studies were assessed for eligibility. Process of study selection is shown in Fig. 1. Twenty-five studies were excluded due to a non-prospective design or a lack of information regarding FMD. Finally, 18 studies could be included in the quantitative meta-analysis (Fig. 1) [10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27]. Study design and baseline clinical characteristics are summarized in Table 1. Treatment strategies for OSAS were divided in four groups: (1) ventilation therapy (i.e. CPAP and nasal CPAP); (2) ear–nose–throat (ENT) surgery (i.e. adenotonsillectomy and uvulopalatopharyngoplasty); (3) medical treatment (i.e. oral administration of allopurinol or vitamin C); (4) device therapy with oral appliances (i.e. mandibular advancement device [MAD]).
Twelve studies evaluated the effect on endothelial function of a ventilation treatment with CPAP or nCPAP. The mean-difference FMD change from baseline to follow-up ranged from 0.29 to 6.50%. The pooled effect showed a significant improvement of FMD among patients treated with CPAP or nCPAP (mean difference [MD] = 2.54; 95% CI 1.75–3.32; p < 0.00001) (Fig. 2). This result remained consistent when only studies with a longer follow-up were included (MD = 2.50; 95% CI 1.70–3.31; p < 0.00001). Within-study heterogeneity was high (I2 = 88%) and was not strictly related to length of follow-up when studies with shorter follow-up were excluded (I2 = 89%).
Three studies evaluated the impact on endothelial function of ENT surgery, namely adenotonsillectomy and uvulopalatopharyngoplasty. ENT surgery significantly improved FMD at study follow-up (MD = 2.05; 95% CI 0.20–3.89; p = 0.03) (Fig. 2). Heterogeneity between studies was high (I2 = 88%) and mostly due to the different surgical treatments and to the different mean age between the studies cohorts. At sensitivity analysis, excluding patients treated with adenotonsillectomy (Chan et al. 2015), overall treatment effect remained similar (MD = 3.07; 95% CI 2.18–3.96; p < 0.00001) with no heterogeneity (I2 = 0%). Study results remained directionally consistent when we restricted our analysis to > 2 weeks follow-up studies (MD = 1.09; 95% CI − 0.06–2.25; p = 0.06).
Two observational studies evaluated endothelial function after medical treatment in patients with OSAS with the use of allopurinol and vitamin C, respectively. Medical treatment consistently improved FMD at follow-up (MD = 3.12; 95% CI 2.57–3.68; p < 0.00001) with no between-study heterogeneity (I2 = 0%) (Fig. 2).
Finally, one single study evaluated the effect of oral appliance therapy on FMD at 8-week follow-up in patients with OSAS. As for other treatment groups, oral appliance significantly improved FMD in a similar magnitude (MD = 4.60; 95% CI 1.61–7.59; p = 0.003) (Fig. 2).
After pooling results from different treatment strategies, OSAS therapies showed a positive impact on endothelial function (MD = 2.58; 95% CI 1.95–3.20; p < 0.00001) (Fig. 2). Heterogeneity for the treatment effect within the four explored treatment strategies was low (I2 = 14%), reflecting a comparable magnitude of benefit on endothelial function among treatments.
Discussion
OSAS is widely prevalent in western countries, and its impact on public health is considerable given its association with an increased cardiovascular risk [1,2,3,4]. OSAS has also a negative impact on endothelial function, which might mediate the higher incidence of cardiovascular events observed in this population [5]. Our analysis shows that the treatment of OSAS, by means of four different strategies, produces a significant improvement of endothelial function. Hence, our results might support the hypothesis that a sustained treatment of OSAS reduces cardiovascular risk by improving vascular fitness.
Four randomized clinical trials (RCT) explored the effect of CPAP on endothelial function demonstrating a significant improvement of FMD in patients affected by moderate to severe OSAS [14, 16,17,18].
Our analysis also suggests a possible benefit, of a similar extent, from a surgical treatment of this patient population. The impact of surgery on endothelial function was explored by Yang et al. in an observational study [24]. In this study, the authors highlighted the role of uvulopalatopharyngoplasty, a common surgical technique used for OSAS treatment, in improving several endothelial function indexes [24].
We observed a consistent benefit also from a treatment with medical therapy in patients with OSAS. However, whether the positive effect of these medications (i.e. allopurinol and vitamin C) on endothelial function is mediated by an actual improvement of OSAS remains controversial, as these treatments are likely ineffective on upper airways collapse. Hence, their positive effect on endothelial function is most likely due to their direct effect on this system, rather than secondary to an improvement of OSAS.
Finally, whether the positive effect on endothelial function of OSAS treatment converts in a similar reduction of cardiovascular events remains to be ascertained. In fact, a big randomized controlled trial ultimately failed to show a reduction of the composite ischemic endpoint in OSAS patients with concomitant cardiovascular disease treated with CPAP [28]. The overall evidence in this field remains controversial, and a lack of CPAP adherence was proposed as a plausible justification for the negative results [29]. Importantly, CPAP was associated with an improvement in endothelial function only in patients adhering to the treatment [5]. Since CPAP is often not well tolerated, other treatment strategies not necessitating long-term patient adherence (e.g. surgery) are highly attractive and necessitate further study [30].
The following limitation of our analysis should be accounted: most of the studies evaluating the impact of OSAS treatment on endothelial function were observational, therefore, tending to overestimate treatment effects, and only few randomly allocated patients to treatment. High between-study heterogeneity was observed. This could be due to several differences in population selection and study design, especially in the ventilation treatment group. Nevertheless, effect direction by visual inspection invariably showed an improvement of endothelial function after treatment. Finally, taking into account that endothelial function measurements are generally applied only for scientific purposes, being time-consuming, their application on large scale appears to be improbable in sleep labs. For this reason, very few scientific works evaluating endothelial function in OSAS patients are focused on not-ventilatory modalities (e.g. MAD and surgery) and a satisfactory comparison between different therapeutic alternatives seems hard to be achieved.
In conclusion, further trials, especially focused on not-ventilatory treatments, are needed to compare the effectiveness of several therapeutic modalities, by means of polysomnography outcome evaluation (e.g. Apnea–Hypopnea Index), and their effect on endothelial function indexes. However, the clinical value of these indexes still needs to be better clarified.
Conclusions
Our study supports the hypothesis that treatment for OSAS positively impacts endothelial function. Whether this effect also associates with an improvement of clinical outcomes remains to be ascertained.
Abbreviations
- OSAS:
-
Obstructive sleep apnoea syndrome
- CPAP:
-
Continuous positive airway pressure
- FMD:
-
Flow-mediated dilatation
- PRISMA:
-
Systematic Reviews and Meta-analyses (PRISMA) Statement
- MOOSE:
-
Meta-analysis of Observational Studies in Epidemiology
- ENT:
-
Ear–nose–throat
- MAD:
-
Mandibular advancement device
- RCT:
-
Randomized clinical trials
References
Hung J, Whitford EG, Parsons RW et al (1990) Association of sleep apnoea with myocardial infarction in men. Lancet 336:261–264
Marin JM, Carrizo SJ, Vicente E et al (2005) Long-term cardiovascular outcomes in men with obstructive sleep apnoea-hypopnoea with or without treatment with continuous positive airway pressure: an observational study. Lancet 365:1046–1053
Peppard PE, Young T, Palta M et al (2000) Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med 342:1378–1384
Sanchez-de-la-Torre M, Campos-Rodriguez F, Barbe F (2013) Obstructive sleep apnoea and cardiovascular disease. Lancet Respir Med 1(1):61–72
E.I. Schwarz, M.A. Puhan, C. Schlatzer, J.R., et al. Effect of CPAP therapy on endothelial function in obstructive sleep apnoea: a systematic review and meta-analysis. Respirology 2015;20(6):889–95.
Urrutia G, Bonfill X (2010) Declaración PRISMA: Una propuesta para mejorar la publicación de revisiones sistemàticas y metaanálisis. Med Clin (Barc) 135(11):507–511
Stroup DF, Berlin JA, Morton SC, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000;283:2008–2012.
Ruehland WR, Rochford PD, O'Donoghue FJ et al (2009) The new AASM criteria for scoring hypopneas: impact on the apnea hypopnea index. Sleep 32(2):150–157
Celermajer DS, Sorensen KE, Gooch VM et al (1992) Non-invasive detection of endothelial dysfunction in children and adults at risk of atherosclerosis. Lancet 340:1111–1115
Bayram NA, Ciftci B, Keles T et al (2009) Endothelial function in normotensive men with obstructive sleep apnea before and 6 months after CPAP treatment. Sleep 32(10):1257–1263
Butt M, Khair OA, Dwivedi G, Shantsila A et al (2011) Myocardial perfusion by myocardial contrast echocardiography and endothelial dysfunction in obstructive sleep apnea. Hypertension 58(3):417–424
Chung S, Yoon IY, Lee CH et al (2010) The association of nocturnal hypoxemia with arterial stiffness and endothelial dysfunction in male patients with obstructive sleep apnea syndrome. Respiration 79(5):363–369
Del Ben M, Fabiani M, Loffredo L et al (2012) Oxidative stress mediated arterial dysfunction in patients with obstructive sleep apnoea and the effect of continuous positive airway pressure treatment. BMC Pulm Med 23:12–36
Ip MS, Tse HF, Lam B et al (2004) Endothelial function in obstructive sleep apnea and response to treatment. Am J Respir Crit Care Med 169(3):348–353
Kallianos A, Panoutsopoulos A, Mermigkis C et al (2015) Sex differences of continuous positive airway pressure treatment on flow-mediated dilation in patients with obstructive sleep apnea syndrome. Clin Interv Aging 10:1361–1366
Kohler M, Stoewhas AC, Ayers L et al (2011) Effects of continuous positive airway pressure therapy withdrawal in patients with obstructive sleep apnea: a randomized controlled trial. Am J Respir Crit Care Med 184(10):1192–1199
Kohler M, Craig S, Pepperell JC et al (2013) CPAP improves endothelial function in patients with minimally symptomatic OSA: results from a subset study of the MOSAIC trial. Chest 144(3):896–902
Nguyen PK, Katikireddy CK, McConnell MV et al (2010) Nasal continuous positive airway pressure improves myocardial perfusion reserve and endothelial-dependent vasodilation in patients with obstructive sleep apnea. J Cardiovasc Magn Reson 12:50
Ohike Y, Kozaki K, Iijima K et al (2005) Amelioration of vascular endothelial dysfunction in obstructive sleep apnea syndrome by nasal continuous positive airway pressure–possible involvement of nitric oxide and asymmetric NG NG-dimethylarginine. Circ J 69(2):221–226
Panoutsopoulos A, Kallianos A, Kostopoulos K et al (2012) Effect of CPAP treatment on endothelial function and plasma CRP levels in patients with sleep apnea. Med Sci Monit 18(12):747–751
Tulmaç M, Tireli E, Ebinç H et al (2012) Effect of overnight nasal continuous positive airway pressure treatment on the endothelial function in patients with obstructive sleep apnea. Anadolu Kardiyol Derg 12(7):560–565
Chan KC, Au CT, Chook P et al (2015) Endothelial function in children with OSA and the effects of adenotonsillectomy. Chest 147(1):132–139
Lee MY, Lin CC, Lee KS et al (2009) Effect of uvulopalatopharyngoplasty on endothelial function in obstructive sleep apnea. Otolaryngol Head Neck Surg 140(3):369–374
Yang HB, Wang Y, Dong MM (2012) Effect of Han-uvulopalatopharyngoplasty on flow-mediated dilatation in patients with moderate or severe obstructive sleep apnea syndrome. Acta Otolaryngol 132(7):769–772
El Solh AA, Saliba R, Bosinski T et al (2006) Allopurinol improves endothelial function in sleep apnoea: a randomised controlled study. Eur Respir J 27(5):997–1002
Grebe M, Eisele HJ, Weissmann N et al (2006) Antioxidant vitamin C improves endothelial function in obstructive sleep apnea. Am J Respir Crit Care Med 173(8):897–901
Lin CC, Wang HY, Chiu CH, Liaw SF (2015) Effect of oral appliance on endothelial function in sleep apnea. Clin Oral Investig 19(2):437–444
McEvoy RD, Antic NA, Heeley E et al (2016) CPAP for Prevention of Cardiovascular Events in Obstructive Sleep Apnea. N Engl J Med 375(10):919–931
Mokhlesi B, Ayas NT (2016) Cardiovascular Events in Obstructive Sleep Apnea - Can CPAP Therapy SAVE Lives? N Engl J Med 375(10):994–996
Lin HC, Friedman M, Chang HW et al (2008) The efficacy of multilevel surgery of the upper airway in adults with obstructive sleep apnea/hypopnea syndrome. Laryngoscope 118(5):902–908
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
This article does not contain any studies with human participants or animal performed by any of the authors.
Informed consent
This article does not contain any studies with human participants or animal performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on sleep apnea syndrome.
Guest Editors: Manuele Casale, Rinaldi Vittorio.
Rights and permissions
About this article
Cite this article
Cammaroto, G., Costa, F., Ruiz, M.V.G. et al. Obstructive sleep apnoea syndrome and endothelial function: potential impact of different treatment strategies—meta-analysis of prospective studies. Eur Arch Otorhinolaryngol 276, 2331–2338 (2019). https://doi.org/10.1007/s00405-019-05486-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-019-05486-6