Abstract
Purpose
A meta-analysis of prospective cohort studies was conducted to clarify the association between obstructive sleep apnoea (OSA) and future risk of all-cause mortality.
Methods
Eligible studies were identified by searching the PubMed and EMBASE databases up to July 2015. Pooled hazard ratios (HRs) and their corresponding 95 % confidence intervals (CIs) were calculated to estimate the association between OSA and risk of all-cause mortality. Sources of heterogeneity were identified by subgroup and meta-regression analyses.
Results
Twelve prospective cohort studies involving 34,382 participants were included in this meta-analysis. The pooled HR of all-cause mortality was 1.262 (95 % CI 1.093–1.431) with significant heterogeneity. Subgroup analyses indicated that the pooled HRs of all-cause mortality in patients with mild, moderate and severe OSA were 0.945 (95 % CI 0.810–1.081), 1.178 (95 % CI 0.978–1.378) and 1.601 (95 % CI 1.298–1.902), respectively. OSA severity could be a possible sources of heterogeneity. Existing publication bias produced a minor contribution to effect size.
Conclusion
Severe, but not mild to moderate, OSA is significantly associated with increased risk of all-cause mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obstructive sleep apnoea (OSA), the most common sleep disorder in clinical practice, is characterised by upper airway obstruction, which leads to repetitive apnoeas and hypopnoeas during sleep. This condition is associated with sleep fragmentation and intermittent hypoxia. The estimated prevalence of OSA is approximately 4 % in men and 2 % in women [1]. Such prevalence is increasing at an alarming rate [2].
Emerging evidence suggests that OSA is associated with the future risk of mortality. The major of published epidemiologic studies have yielded conflicting results. Recent meta-analyses of all-cause mortality in OSA have concluded that severe OSA significantly increases all-cause mortality [3, 4]. However, two analyses [3, 4] included fewer studies, did not involve patients with mild OSA and exhibited higher prevalence than patients with moderate or severe OSA. The study by Wang et.al [4] only conducted a pooled analysis. Neither studies [3, 4] explored the robustness of pooled results and the influence of several variables on effect size limited by the number of studies. The presence of heterogeneity and publication bias in previous meta-analyses can also influence the reliability of pooled results. Therefore, a meta-analysis of cohort study is crucial to verify previous conclusions and to reassess the association between OSA and all-cause mortality. In particular, all-cause mortality in patients’ mild OSA and the potential effects of relevant variables on all-cause mortality should be determined.
Methods and materials
Literature search and selection
Eligible studies were identified by systematically searching the PubMed and EMBASE databases up to July 2015. The search was limited to human, but no language restrictions were implemented. The searches combined free-text and subject terms, and the following search terms were used: ‘mortality’ or ‘death’, ‘apnoea’ or ‘apnea’, and ‘cohort’ or ‘prospective cohort studies’. The reference lists of relevant publications were also manually searched for additional studies.
Two researchers independently identified the eligible studies. Conflicting decisions were resolved through a consensus with a third researcher. Studies were considered eligible if the following criteria were met: (1) the design was prospective cohort study; (2) the studied population was adult; and (3) the outcome of relative risk (hazard ratios (HRs)) with their 95 % confidence intervals (CIs) for all-cause mortality was included, or sufficient data were provided to calculate them.
The fully adjusted HR was used to compute the pooled HR for the associations of OSA with risk of all-cause mortality; otherwise, non-adjusted HR was used. When multiple studies reported outcomes using the same patient group, the study with the largest population was included. Abstracts, case reports, editorials, expert opinions, letters, animal studies and reviews without original data were excluded.
In most studies included in this meta-analysis, the diagnosis of OSA was based on the apnoea–hypopnoea index (AHI). Therefore, the severity of OSA in this study was quantified using AHI; normal (AHI < 5), mild (AHI 5-15), moderate (AHI 15-30), and severe (AHI > 30) respectively [5].
Data extraction
Data extracted from each study included the name of the first author, publication date, country, patient recruitment, sample size (the number of participants and mortality cases), follow-up duration, the diagnosis and severity of OSA, mortality ascertainment, statistical adjustments for confounding factors and participant characteristics (age and body mass index (BMI)).
Statistical analysis
Pooled HR was used to estimate the association between OSA and risk of all-cause mortality across studies. Q and I 2 statistics were used to determine statistical heterogeneity amongst individual studies. P heterogeneity < 0.10 or I 2 > 60 % was considered to indicate significant heterogeneity. The random effects model was used when significant heterogeneity was observed; otherwise, the fixed effects model was utilised. In papers that reported HRs from a separate subgroup, the combined HR was computed using the fixed effects model prior to pooling.
Sensitivity analyses using the random effects model were conducted to explore the robustness of pooled results. Planned subgroup analyses using the random effects model were conducted to evaluate the effects of several relevant variables on the effect size and to explore possible sources of heterogeneity. Meta-regressions were also conducted to explore possible sources of heterogeneity.
Publication bias was evaluated visually with funnel plot and statistically with the Begg’s and Egger’s tests. The trim and fill method was used to identify and correct for funnel plot asymmetry arising from publication bias. Cumulative meta-analyses were conducted to detect the presence of publication bias.
A two-tailed P < 0.05 was considered to indicate statistical significance. All statistical analyses were conducted using software Stata 10.0 (StataCorp, College Station, TX, USA).
Results
Literature search and characteristics
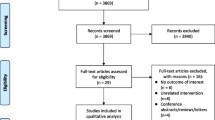
Figure 1 shows the selection process for studies included in this meta-analysis. The initial online search on the databases resulted in the identification of 546 potentially related articles. After reviewing the titles and abstracts, 19 articles were selected and further reviewed. Seven articles were excluded for the following reasons: one study duplicated the participants with the study of Marshall 2014 [6], one study did not present original data [3], three studies lacked data for the necessary computations [7–9] and two studies involved participants who were not OSA patients [10, 11]. Twelve cohort studies which involved 34,382 participants finally met the inclusion criteria and were included in this meta-analysis [12–23]. The characteristics of the included studies and the corresponding patient information are summarised in Table 1.
Pooled analysis
The heterogeneity test indicates significant heterogeneity amongst individual studies (P heterogeneity = 0.000, I 2 = 70.4 %). Hence, the random effects model was used for the pooled analysis. The result demonstrated a significant decrease in all-cause mortality (HR, 1.262, 95 % CI 1.093–1.431), whereas the fixed model yielded a non-significant result (HR, 1.024, 95 % CI 0.988 to 1.061) (Fig. 2).
Sensitivity analyses
Sensitivity analyses by omitting one study at a time indicated that the HR ranged from 1.169 (95 % CI 1.021 to 1.318) to 1.379 (95 % CI 1.134 to 1.624) after omitting the studies of Lavie 2005 [14] and Mant 1995 [16]. A borderline result was obtained after excluding two studies that diagnosed OSA on the basis of self-reporting [19, 20] (HR, 1.204, 95 % CI 1.039 to 1.368; P heterogeneity = 0.002, I 2 = 65.6 %). To decrease the effect of gender, the study of Lavie 2005 [14] which involved male participants only, was excluded, a heterogeneous (P heterogeneity = 0.009, I 2 = 57.7 %) and non-significant increase in mortality was observed (HR, 1.169, 95 % CI 1.021 to 1.318). Additional sensitivity analysis by restricting the analysis to studies which diagnosing OSA on the basis of AHI revealed a heterogeneous and significant increase in mortality (P heterogeneity = 0.045, I 2 = 49.5 %; HR, 1.280; 95 % CI 1.059 to 1.5027).
Subgroup analysis
Differences in sample size (≤1000 and >1000), severity of OSA (mild, moderate, severe), patient age (≤60 and >60 years) and BMI (≤30 and >30 kg/m2) significantly influenced all-cause mortality in patients with OSA, but not significantly in study location (Europe, USA, Asia-Pacific), recruitment-based (community and hospital) and follow-up duration (≤10 and >10 years). Heterogeneity was abolished, when grouped by disease severity. Table 2 presents the detailed results of subgroup analyses.
Specific subgroup analyses were performed by restricting the analysis to studies that directly compared mortality rate with varying OSA severity. Results of OSA severity from four studies [12, 13, 17, 22] indicated that the pooled HR were 0.945 (95 % CI 0.810 to 1.085), 1.180 (95 % CI 0.964 to 1.396) and 1.503 (95 % CI 1.154 to 1.851) for patients with mild, moderate and severe OSA, respectively, without significant heterogeneity (P heterogeneity = 0.206, I 2 = 24.2 %). Compared with patients with mild and moderate OSA, those with severe OSA exhibited higher mortality.
Meta-regression analysis
Univariate meta-regression analysis showed that the change in all-cause mortality was not statistically associated with publication year (P = 0.149), study location (P = 0.261), patients recruitment (P = 0.146), follow-up duration (P = 0.304), participant number (P = 0.266), as patient age (P = 0.637) and BMI (P = 0.223). Unfortunately, the analysis does not perform in severity of disease based on limited data available.
Publication bias
The funnel plot indicates the potential presence of publication bias. The results of Begg’s (P = 0.732) and Egger’s test (P = 0.003) further supported the occurrence of publication bias in this meta-analysis. The trim and fill method indicated the need for five additional studies to correct the funnel plot asymmetry (Fig. 3). The corrected HRs after using the fixed and random effects models were 1.015 (95 % CI 0.978 to 1.051) and 1.117 (95 % CI 0.941 to 1.293), indicating a minor contribution of publication bias to the pooled results.
The pooled results showed that the random effects estimate was more beneficial than the fixed effects one, indicating the presence of small-study effects. Accordingly, a cumulative meta-analysis was conducted to identify the presence of small-study effects. The results suggest the contribution of small-study effects (Fig. S1), and this result was strengthened by the outcome of subgroup analysis. Cumulative meta-analysis revealed that the republication year of study exerted no significant influence on publication bias (Fig. S2).
Discussion
This meta-analysis evaluated the association between OSA and future risk of all-cause mortality. Patients with OSA exhibited a significant increase in risk of all-cause mortality. Subgroup analysis suggested the discrepant effect of disease severity. Unlike patients with mild to moderate OSA, those with severe OSA exhibited significantly increased mortality.
To the best of our knowledge, this study is the first meta-analysis to evaluate the risk of all-cause mortality in patients with mild, moderate and severe OSA. Different from previous meta-analyses, which included only six studies with moderate or severe OSA [3, 4], the current study included additional six studies with three degrees of OSA severity. Therefore, the current meta-analysis has high statistical power and degree of certainty to detect the association. Inconsistent with the present study, a higher effect size was reported from previous meta-analyses [3, 4], with HRs of 1.67 (95 % CI 1.25 to 2.23) and 1.92 (95 % CI 1.38 to 2.69). When the analysis was restricted to severe OSA, similar HRs were obtained in this analysis. This result may be attributed to the disparity in disease severity.
The precise mechanisms underlying the association between OSA and future risk of all-cause mortality remain unclear. The possibility may be explained by some pathophysiological characteristics associated with OSA. OSA can exert several deleterious effects including sleep fragmentation, swings in negative intrathoracic pressure, fluid redistribution, intermittent hypoxia, alterations in sympathetic activity, increased systemic inflammation, hidden or unaccounted risk factors or other factors related to OSA such as excessive daytime sleepiness, obesity and lung disease [18, 24–27]. All these factors can induce and promote directly or indirectly multi-organ or system function damage, consequently hastening patient death.
Determining whether OSA is an independent risk factor or simply a marker is essential. Current findings seemed to support the role of OSA as an independent factor. First, if OSA is an early marker, then the value of AHI or respiratory disturbance index (RDI) should be higher when patient death is near. The follow-up duration of 4–20 years in studies included in this meta-analysis suggests that OSA is an independent risk factor. Second, although no linear trend analysis was conducted on AHI or RDI change with risk of all-cause mortality because of the limited number of studies, the result indicates a high mortality rate in patients with severe OSA, which is consistent with previous findings [12, 13, 17, 21, 22, 28]. Finally, considerable convincing mechanisms should come from the pathophysiology of OSA. As described above, OSA can induce and promote a multi-organ or system function damage [26] and hasten patient death.
Subgroup analysis suggests that the differences in sample size, severity of OSA, patient age and BMI are significantly associated with the risk of all-cause mortality. At least four studies compared the all-cause mortality in patients with mild, moderate and severe OSA, the studies suggested that severe OSA tended to be associated with increased all-cause mortality compared to mild or moderate OSA [6, 12, 13, 22]. In addition, a similar result was reported by previous meta-analysis [3, 4]. Differences in severity of OSA may be associated with risk of all-cause mortality because severe OSA is prone to develop organ damage, thereby increasing the risk of mortality, compared with mild to moderate OSA. The larger sample sizes have sufficient statistical power to detect the potential association between OSA and mortality compared with smaller sample sizes. Thus, the result obtained given a larger sample size and severe OSA subgroups associated with increased risk of all-cause mortality would have a higher degree of certainty. However, the results on the effect of patient age and BMI on mortality must be interpreted with caution. Consistent with the result of this analysis, most studies reported a decrease in mortality risk with increasing age [12, 14, 27, 29, 30]. However, other studies including two large studies have confirmed an association between increased age and increased risk of mortality [31–33]. Obesity has been recognised as an important risk factor for OSA development and progression. Increased BMI may theoretically increase severity of OSA and increase mortality. However, the effects of obesity based on BMI on all-cause mortality remain inconsistent. Three studies examined the effect of BMI on all-cause mortality in patients with OSA have been found [27, 29, 32]; two studies consistent with the result of this analysis indicate a higher mortality in higher BMI [27, 29], but the remaining one suggests otherwise [32]. In addition, no supportive results were found from meta-regression for age and BMI.
Several potential limitations should be noted when interpreting the findings of this meta-analysis. First, significant heterogeneity was present. Subgroup analyses showed that OSA severity and sample size may be possible sources of heterogeneity. However, no supportive results were obtained from meta-regression analyses. Secondly, the potential effect of publication bias was present, although its contribution was minor. Thirdly, the effect size was relatively small despite the large number of participants, and most sensitivity analyses indicated non-robust results. Fourthly, the subgroups were defined post hoc, and the means of subgroup and meta-regression analyses, rather than the individual patients, were used as data points. Finally, AHI or RDI reflecting the respiratory events during sleep including apnoea, hypopnoea and arousals may not accurately reflect the pathophysiological aspects and clinical consequences of OSA [34]. A limited number of studies evaluated the association between OSA-relevant variables, other than AHI and risk of mortality, and found that these variables influence risk of mortality. These variables include symptoms, sleep fragmentation, sympathetic active and hypoxic events [12, 17, 18, 35]. Some studies even recognised that OSA-relevant variables other than AHI were important predictors of all-cause mortality in OSA [35]. Given that OSA is a heterogeneous disorder and the causes are multifactorial [36], the precise effects of these variables on all-cause mortality need further investigation and verification.
Despite the limitations, this meta-analysis supports the hypothesis that OSA is an independent risk factor for all-cause mortality. Further evidence and verification in patients with mild and moderate OSA are needed. Further studies are needed to identify the precise predictors of all-cause mortality and to determine the optimal regimes and patients that would most benefit from OSA intervention.
Authors’ contributions
Y. Guo conceived and designed the study; L. Pan, X. Xie and Y. Guo selected the studies; D. Liu, D. Ren and Y. Guo extracted the data; X. Xie and Y. Guo performed the statistical analyses; L. Pan, X. Xie, D. Liu and Y. Guo wrote the manuscript. All authors read and revised the manuscript. The final manuscript was approved by all authors.
References
Lee W, Nagubadi S, Kryger MH, Mokhlesi B (2008) Epidemiology of obstructive sleep apnea: a population-based perspective. Expert Rev Respir Med 2:349–364
De Miguel-Diez J, Carrasco-Garrido P, Jimenez-Garcia R, Puente-Maestu L, Hernandez-Barrera V, Lopez DAA (2015) Obstructive sleep apnea among hospitalized patients in Spain, analysis of hospital discharge data 2008–2012. Sleep Breath 19:1–8
Ge X, Han F, Huang Y, Zhang Y, Yang T, Bai C, Guo X (2013) Is obstructive sleep apnea associated with cardiovascular and all-cause mortality? PLoS One 8:e69432
Wang X, Ouyang Y, Wang Z, Zhao G, Liu L, Bi Y (2013) Obstructive sleep apnea and risk of cardiovascular disease and all-cause mortality: a meta-analysis of prospective cohort studies. Int J Cardiol 169:207–214
Bradley TD, Floras JS (2009) Obstructive sleep apnoea and its cardiovascular consequences. Lancet 373:82–93
Marshall NS, Wong KK, Liu PY, Cullen SR, Knuiman MW, Grunstein RR (2008) Sleep apnea as an independent risk factor for all-cause mortality: the Busselton Health Study. Sleep 31:1079–1085
Partinen M, Jamieson A, Guilleminault C (1988) Long-term outcome for obstructive sleep apnea syndrome patients. Mortality Chest 94:1200–1204
Veale D, Chailleux E, Hoorelbeke-Ramon A, Reybet-Degas O, Humeau-Chapuis MP, Alluin-Aigouy F, Fleury B, Jonquet O, Michard P (2000) Mortality of sleep apnoea patients treated by nasal continuous positive airway pressure registered in the ANTADIR observatory. Association Nationale pour le Traitement A Domicile de l’Insuffisance Respiratoire chronique. Eur J Respir 15:326–331
Johansson P, Alehagen U, Svanborg E, Dahlstrom U, Brostrom A (2012) Clinical characteristics and mortality risk in relation to obstructive and central sleep apnoea in community-dwelling elderly individuals: a 7-year follow-up. Age Ageing 41:468–474
Korostovtseva LS, Sviryaev YV, Zvartau NE, Konradi AO, Kalinkin AL (2011) Prognosis and cardiovascular morbidity and mortality in prospective study of hypertensive patients with obstructive sleep apnea syndrome in St Petersburg, Russia. Med Sci Monit 17:CR146–CR153
Lindberg E, Janson C, Sv Rdsudd K, Gislason T, Hetta J, Boman G (1998) Increased mortality among sleepy snorers: a prospective population based study. Thorax 53:631–637
Punjabi NM, Caffo BS, Goodwin JL, Gottlieb DJ, Newman AB, O’Connor GT, Rapoport DM, Redline S, Resnick HE, Robbins JA, Shahar E, Unruh ML, Samet JM (2009) Sleep-disordered breathing and mortality: a prospective cohort study. PLoS Med 6:e1000132
Young T, Finn L, Peppard PE, Szklo-Coxe M, Austin D, Nieto FJ, Stubbs R, Hla KM (2008) Sleep disordered breathing and mortality: eighteen-year follow-up of the Wisconsin sleep cohort. Sleep 31:1071–1078
Lavie P, Lavie L, Herer P (2005) All-cause mortality in males with sleep apnoea syndrome: declining mortality rates with age. Eur J Respir 25:514–520
Yaggi HK, Concato J, Kernan WN, Lichtman JH, Brass LM, Mohsenin V (2005) Obstructive sleep apnea as a risk factor for stroke and death. N Engl J Med 353:2034–2041
Mant A, King M, Saunders NA, Pond CD, Goode E, Hewitt H (1995) Four-year follow-up of mortality and sleep-related respiratory disturbance in non-demented seniors. Sleep 18:433–438
Johansson P, Alehagen U, Ulander M, Svanborg E, Dahlstrom U, Brostrom A (2011) Sleep disordered breathing in community dwelling elderly: associations with cardiovascular disease, impaired systolic function, and mortality after a six-year follow-up. Sleep Med 12:748–753
Gooneratne NS, Richards KC, Joffe M, Lam RW, Pack F, Staley B, Dinges DF, Pack AI (2011) Sleep disordered breathing with excessive daytime sleepiness is a risk factor for mortality in older adults. Sleep 34:435–442
Marshall NS, Delling L, Grunstein RR, Peltonen M, Sjostrom CD, Karason K, Carlsson LM, Hedner J, Stenlof K, Sjostrom L (2011) Self-reported sleep apnoea and mortality in patients from the Swedish Obese Subjects study. Eur J Respir 38:1349–1354
Yeboah J, Redline S, Johnson C, Tracy R, Ouyang P, Blumenthal RS, Burke GL, Herrington DM (2011) Association between sleep apnea, snoring, incident cardiovascular events and all-cause mortality in an adult population: MESA. Atherosclerosis 219:963–968
Martinez-Garcia MA, Campos-Rodriguez F, Catalan-Serra P, Soler-Cataluna JJ, Almeida-Gonzalez C, De la Cruz MI, Duran-Cantolla J, Montserrat JM (2012) Cardiovascular mortality in obstructive sleep apnea in the elderly: role of long-term continuous positive airway pressure treatment: a prospective observational study. Am J Respir Crit Care Med 186:909–916
Lee JE, Lee CH, Lee SJ, Ryu Y, Lee WH, Yoon IY, Rhee CS, Kim JW (2013) Mortality of patients with obstructive sleep apnea in Korea. J Clin Sleep Med 9:997–1002
Marshall NS, Wong KK, Cullen SR, Knuiman MW, Grunstein RR (2014) Sleep apnea and 20-year follow-up for all-cause mortality, stroke, and cancer incidence and mortality in the Busselton Health Study cohort. J Clin Sleep Med 10:355–362
Mirrakhimov AE (2013) Supine fluid redistribution: should we consider this as an important risk factor for obstructive sleep apnea? Sleep Breath 17:511–523
Guo Y, Pan L, Ren D, Xie X (2013) Impact of continuous positive airway pressure on C-reactive protein in patients with obstructive sleep apnea: a meta-analysis. Sleep Breath 17:495–503
Leung RS, Comondore VR, Ryan CM, Stevens D (2012) Mechanisms of sleep-disordered breathing: causes and consequences. Pflugers Arch 463:213–230
Lavie P, Herer P, Lavie L (2007) Mortality risk factors in sleep apnoea: a matched case-control study. J Sleep Res 16:128–134
Muraja-Murro A, Eskola K, Kolari T, Tiihonen P, Hukkanen T, Tuomilehto H, Peltonen M, Mervaala E, Toyras J (2013) Mortality in middle-aged men with obstructive sleep apnea in Finland. Sleep Breath 17:1047–1053
Rich J, Raviv A, Raviv N, Brietzke SE (2012) All-cause mortality and obstructive sleep apnea severity revisited. Otolaryngol Head Neck Surg 147:583–587
Campos-Rodriguez F, Pena-Grinan N, Reyes-Nunez N, De la Cruz-Moron I, Perez-Ronchel J, De la Vega-Gallardo F, Fernandez-Palacin A (2005) Mortality in obstructive sleep apnea-hypopnea patients treated with positive airway pressure. Chest 128:624–633
Jennum P, Tonnesen P, Ibsen R, Kjellberg J (2015) All-cause mortality from obstructive sleep apnea in male and female patients with and without continuous positive airway pressure treatment: a registry study with 10 years of follow-up. Nat Sci Sleep 7:43–50
Nakamura H, Kanemura T, Takara C, Tsukayama A, Tohyama K, Matsumoto T, Iseki K (2009) A retrospective analysis of 4000 patients with obstructive sleep apnea in Okinawa, Japan. Sleep Biol Rhythms 7:103–112
Marti S, Sampol G, Munoz X, Torres F, Roca A, Lloberes P, Sagales T, Quesada P, Morell F (2002) Mortality in severe sleep apnoea/hypopnoea syndrome patients: impact of treatment. Eur Respir J 20:1511–1518
Punjabi NM (2015) Counterpoint: is the AHI the best way to quantify the severity of sleep disordered breathing? No Chest. doi:10.1378/chest.14-2261
Kendzerska T, Gershon AS, Hawker G, Leung RS, Tomlinson G (2014) Obstructive sleep apnea and risk of cardiovascular events and all-cause mortality: a decade-long historical cohort study. PLoS Med 11:e1001599
Eckert DJ, White DP, Jordan AS, Malhotra A, Wellman A (2013) Defining phenotypic causes of obstructive sleep apnea. Identification of novel therapeutic targets. Am J Respir Crit Care Med 188:996–1004
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Xuzhou Science and Technology Bureau provided financial support in the form of social development funding (XZZD1345). The sponsor had no role in the design or conduct of this research.
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Lei Pan, Xiaomei Xie and Dayue Liu contributed equally to this study.
Rights and permissions
About this article
Cite this article
Pan, L., Xie, X., Liu, D. et al. Obstructive sleep apnoea and risks of all-cause mortality: preliminary evidence from prospective cohort studies. Sleep Breath 20, 345–353 (2016). https://doi.org/10.1007/s11325-015-1295-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-015-1295-7