Abstract
Cloverleaf skull deformity or Kleeblattschadel syndrome is a severe condition where multiple cranial sutures are absent and prematurely fused, leading to a trilobate head shape. The remaining open sutures or fontanelles compensate for rapid brain expansion, while the constricted fused calvarium restricts brain growth and results in increased intracranial pressure. Recent data show that early posterior cranial and foramen magnum decompression positively affects infants with cloverleaf skulls. However, long-term sequelae are still rarely discussed. We hereby report a child who developed secondary metopic craniosynostosis after posterior cranial decompression, which required a front-orbital advancement and cranial remodelling as a definitive procedure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cloverleaf skull deformity or Kleeblattschadel syndrome is the most severe form of multisuture craniosynostosis. The condition presents as a trilobar head shape resulting from the brain expansion through the remaining open sutures or fontanelles after abnormal early fusion of multiple sutures. This results in brain growth restriction and impending increased intracranial pressure. Literature shows that patients with this particular condition consequently have delayed development and multiple issues, including hydrocephalus, Chiari I malformation, compromised airways, and eye proptosis [1,2,3,4].
Early cranial decompression has recently been shown to have a significant role in alleviating the raised of intracranial pressure (ICP) and increasing the intracranial volume. Posterior calvarial remodelling, whether reshaping, distraction [5], or decompression procedures [6], has been proposed and shown remarkable improvement. We have published a case series showing early positive outcomes after posterior cranial and foramen magnum decompression in infants with cloverleaf skulls [7].
Herein, in this report, we identified one case of cloverleaf skull deformity that developed secondary metopic craniosynostosis after early posterior cranial decompression, where the metopic suture was previously patent. The child developed secondary trigonocephaly, resulting in an early front-orbital reshaping procedure.
Case presentation
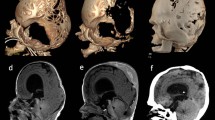
A male infant from non-consanguineous Myanmar parents with no family history of craniofacial disease was born term at 37 weeks of gestational age. He was referred to Siriraj Craniofacial Centre at the age of 1 month due to a trilobed head with tense bulging of the anterior fontanelle (Fig. 1). The overnight oxygenation monitoring showed significant desaturations, and continuous oxygen treatment was initiated. The physical examination also showed receded supraorbital rims, bilateral eye proptosis, mid-face hypoplasia, and bifid uvula. All hands and extremities were unremarkable. The fundoscopy revealed no papilloedema. The laryngotracheobronchoscopy revealed unremarkable and no tracheomalacia.
A, B, C Preoperative images demonstrating trilobar head shape and multisuture craniosynostosis. The metopic suture is patent. D Intraoperative posterior cranial decompression by wide craniectomy and foramen magnum decompression. The dotted line shows the area for foramen magnum decompression. E, F Postoperative images 4 months postoperatively show improved posterior cranial shape with remarkable bone reformation and expanded posterior cranial fossa. The fused metopic suture was observed with the upward and medial deviation of the bilateral supraorbital area
The 3D CT showed multisuture craniosynostosis involving coronal, sagittal, and lambdoid sutures. Temporal lobes were extruding through the splaying of the squamosal sutures and bulging of the brain parenchyma through the open anterior fontanelle, consistent with a cloverleaf skull deformity. There was an effacement of subarachnoid spaces and mild hydrocephalus. Further genetic testing by whole exome sequencing revealed heterogenous c.278C > G mutation with p.Ala93Gly of the SIX3 gene.
The child underwent lateral tarsorrhaphies for proptosis eye management. The posterior cranial decompression by wide craniectomy was performed along with foramen magnum decompression at the age of 2 months (Fig. 1). This involved a meticulous dissection of the bony spicules projection into the dura, resulting in a wide bony excision from just posterior to the fused coronal ring of sutures back to and including the posterior rim of the foramen magnum. The intraoperative blood salvage was used to reduce the transfusion to 35 ml. One month after surgery, the shape of the head appeared rounder and more expanded posteriorly (Fig. 2A, E). Despite this decompression, the hydrocephalus has been progressive and managed by undergoing a programmable ventriculoperitoneal (VP) shunt 2 months after the primary cranial surgery.
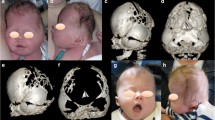
A, E One month after posterior cranial decompression. The head shape looks rounder and fully expanded posteriorly. B, F Eleven months after posterior decompression. It shows a metopic ridge and trigonal-turricephalic head shape. C, D, G, H At 14 months after primary surgery (16 months of age), the fronto-orbital advancement and cranial remodelling were performed to correct the head shape and expand the intracranial volume
Follow-up CT at postoperative 4 months after primary decompression (at 6 months of age) revealed remarkable cranial expansion in the parieto-occipital area and improved CSF space between the cerebellar and the brain stem. However, the fused metopic suture was observed with the upward medially displacement of the supraorbital area (Fig. 1F). The secondary metopic craniosynostosis with a prominent metopic ridge and trigonal-turricephalic head shape was revealed at the age of 13 months postoperatively (around 11 months after posterior decompression) (Fig. 2B, C, F, and G). The boy’s development is progressively improving and appropriate age.
Fronto-orbital advancement with remodelling was performed at the age of 16 months. The supraorbital bar was advanced 15 mm bilaterally, and the triangulated forehead was reshaped. The prominent turricephalic shape was also decompressed (Fig. 2D, H). Clinical follow-up is ongoing.
Discussion
Secondary craniosynostosis (SCS) is defined as a closure in otherwise previously patent sutures following corrective cranial vault surgery. It has been identified as between 10 and 30% in syndromic craniosynostosis children, while in non-syndromic cases, it ranges from 0 to 80% depending on the types and extension of the cranial operations [8]. Arnaud et al. reported 10% of secondary coronal synostosis after primary operation in sagittal craniosynostosis. However, only 1% required secondary decompression due to increased ICP [9].
Unander-Scharin et al. reported 38% developing secondary coronal synostosis in the operated non-syndromic sagittal craniosynostosis cases and suggested that the occurrence of secondary craniosynostosis is linked to reduced growth of the skull and increased ICP [10]. By contrast, Veca et al. identified 8.8% rate of SCS. Similar to our case report, one of the patients in their series developed pan-synostosis 1 year after posterior cranial distraction, which included the metopic suture and required a secondary FOA to correct the abnormal head shape [11]. Tahiri et al. identified 90% of SCS in previously patent lambdoid sutures after posterior cranial vault distraction. There was no association with the age at primary operation, sex, or length of distraction; however, no patient developed distortion of the head shape and required secondary operation [12].
Kim et al. published a review article that found that the idiopathic SCS was around 3%. It is most commonly developed at the bi-coronal suture, followed by the sagittal and uni-coronal sutures. Interestingly, the SCS developed in most patients who underwent primary cranial surgery before 6 months. The average interval to establish the secondary synostosis in previously patent sutures was over 18 months. However, all secondary fused sutures were adjacent to the abnormally fused ones that had been operated on [8]. In our case, the secondary fused metopic suture was far from the primary operating area leaving open the question of the pathogenesis of the SCS.
Mechanical forces over the cranial suture have been long-term proven as a factor in the cranial suture and bone development [13]. Physiological changes in the pressure and cerebrospinal fluid flow after cranial decompression might be one of the significant causes of developing SCS. In syndromic multi-suture craniosynostosis, like cloverleaf skull deformity, the constraining of the skull causes high pressure on the calvarial-and-dura interface. After the cranial decompression, the abruptly reduced tension at the dura-calvarial interface occurs. Certainly in the murine model shows that the posterior intrafrontal suture was still patent after applying tension force across the suture. The experiment showed a high proliferative and apoptotic activity with increased TGF-beta levels at the open sutures [14]. Another in vivo study in rabbits also suggested that cyclic tensile and compressive forces over the premaxillomaxillary and nasofrontal sutures induced remodelling and growth changes in cranial sutures. The tensile or compressive forces are transmitted as shear stresses and upregulate genes and gene products responsible for sutural growth [15]. Moazen et al. studied applying mechanical forces over the frontal bones of Crouzon mice. The results showed that the skull shape was morphologically normal rather than brachycephalic appearance [16]. This thus suggests that sudden changes in tension and compression force at the dura surface unbalancing the osteogenic factors of the dura-suture interface would result in osteogenic differentiation over previously patent sutures.
The cause of metopic craniosynostosis remains unknown but is thought to be multifactorial [17], including underlying brain malformation [18]. A review study proposed that the abnormally small frontal lobe volume associated with frontal stenosis and/or corpus callosum abnormalities would increase the risk of neurocognitive problems in children with metopic synostosis [19]. This suggests that the underdeveloped frontal lobe may be causal of synostosis of the metopic suture. In our case, the posterior cranial decompression and VP shunting create a large space posteriorly to allow backward expansion. This, plus VP shunting, could reduce the pressure for brain expansion anteriorly with increasing tendency for the anterior sutures, including coronal and metopic sutures, to fuse secondarily. In our current case, we noted only the metopic suture was fused. In a previous report, it was noted that secondary cranial suture fusions can occur as a result of VP shunting [20]. The report also highlighted that abnormal head shape can develop more than 7 months after the shunting procedure. However, our current case has shown an abnormal head shape at just 2 months post VP shunt procedure, indicating the possibility of secondary metopic craniosynostosis, which may not be associated with the shunting.
SIX Homeobox 3 or SIX3 gene has a role in vertebrate and insect development or has been implicated in maintaining the differentiated state of tissues (OMIM 603714). The mutation of SIX3 has been reported to be associated with holoprosencephaly, in which the forebrain does not properly develop into two hemispheres [21]. Raam et al. reported 2 cases of holoprosencephaly who had metopic craniosynostosis. However, those patients had no SIX3 mutation [22]. To date, there is no reported association of SIX3 mutation with craniosynostosis, particularly metopic synostosis, thus favouring our case presentation being secondary to loss of anterior brain drive following very early posterior cranial vault decompression.
Conclusions
Later changes in previous patent cranial sutures can happen after transcranial surgery. Early cranial vault surgery with/without VP shunting in complex multisuture craniosynostosis to idealise outcome requires close and ongoing monitoring to observe the secondary effects of the surgery—in this case, the development of secondary metopic synostosis.
Data availability
No datasets were generated or analysed during the current study.
Code availability
Not applicable.
References
Lodge ML, Moore MH, Hanieh A, Trott JA, David DJ (1993) The cloverleaf skull anomaly: managing extreme cranio-orbitofaciostenosis. Plast Reconstr Surg 91(1):1–9
Machado G, Di Rocco F, Sainte-Rose C, Meyer P, Marchac D, Macquet-Nouvion G, Arnaud E, Renier D (2011) Cloverleaf skull deformity and hydrocephalus. Childs Nerv Syst 27(10):1683–1691. https://doi.org/10.1007/s00381-011-1508-8
Tubbs RS, Sharma A, Griessenauer C, Loukas M, Shoja MM, Watanabe K, Oakes WJ (2013) Kleeblattschädel skull: a review of its history, diagnosis, associations, and treatment. Childs Nerv Syst 29(5):745–748. https://doi.org/10.1007/s00381-012-1981-8
Chaisrisawadisuk S, Hammam E, Molloy CJ, Barnett C, Anderson PJ, Moore MH (2021) Severe cloverleaf skull deformity in c.1061C>G (p.Ser354Cys) mutated fibroblast growth factor receptor 2 gene in Crouzon syndrome. J Craniofac Surg 32(1):261–264. https://doi.org/10.1097/SCS.0000000000006999
Raposo-Amaral CE, Raposo-Amaral CA, Adams JW, Ghizoni E (2022) Posterior vault distraction outcomes in patients with severe Crouzon syndrome resulting from Ser347Cys and Ser354Cys mutations. J Craniofac Surg 33(5):1545–1548. https://doi.org/10.1097/SCS.0000000000008642
Nowinski D, Di Rocco F, Renier D, SainteRose C, Leikola J, Arnaud E (2012) Posterior cranial vault expansion in the treatment of craniosynostosis. Comparison of current techniques. Childs Nerv Syst 28(9):1537–1544. https://doi.org/10.1007/s00381-012-1809-6
Moore MH, Chaisrisawadisuk S, Khampalikit I, Doorenbosch X, Jukes A, Molloy CJ (2023) Re-imagining early cloverleaf skull deformity management from front to back approach-30 years on. Childs Nerv Syst 39(12):3349–3359. https://doi.org/10.1007/s00381-023-06147-0
Kim SY, Shin HJ, Lim SY (2017) Determining the fate of cranial sutures after surgical correction of non-syndromic craniosynostosis. J Craniomaxillofac Surg 45(11):1801–1808. https://doi.org/10.1016/j.jcms.2017.08.009
Arnaud E, Capon-Degardin N, Michienzi J, Di Rocco F, Renier D (2009) Scaphocephaly part II: Secondary coronal synostosis after scaphocephalic surgical correction. J Craniofac 20(Suppl 2):1843–1850. https://doi.org/10.1097/SCS.0b013e3181b6c4c3
Unander-Scharin J, Nysjö J, Enblad P, Nowinski D (2021) Secondary coronal synostosis after early surgery for sagittal craniosynostosis: implications for cranial growth. J Craniofac Surg 32(1):113–117. https://doi.org/10.1097/SCS.0000000000007087
Vaca EE, Sheth N, Purnell CA, McGrath JL, Gosain AK (2020) Secondary suture fusion after primary correction of nonsyndromic craniosynostosis: recognition of the problem and identification of risk factors. Plast Reconstr Surg 145(2):493–503. https://doi.org/10.1097/PRS.0000000000006491
Tahiri Y, Paliga JT, Bartlett SP, Taylor JA (2015) New-onset craniosynostosis after posterior vault distraction osteogenesis. J Craniofac Surg 26(1):176–179. https://doi.org/10.1097/SCS.0000000000001186
Cohen MM Jr (1993) Sutural biology and the correlates of craniosynostosis. Am J Med Genet 47(5):581–616. https://doi.org/10.1002/ajmg.1320470507
Tholpady SS, Freyman TF, Chachra D, Ogle RC (2007) Tensional forces influence gene expression and sutural state of rat calvariae in vitro. Plast Reconstr Surg 120(3):601–611. https://doi.org/10.1097/01.prs.0000270284.69632.6b
Peptan AI, Lopez A, Kopher RA, Mao JJ (2008) Responses of intramembranous bone and sutures upon in vivo cyclic tensile and compressive loading. Bone 42(2):432–438. https://doi.org/10.1016/j.bone.2007.05.014
Moazen M, Hejazi M, Savery D, Jones D, Marghoub A, Alazmani A, Pauws E (2022) Mechanical loading of cranial joints minimizes the craniofacial phenotype in Crouzon syndrome. Sci Rep 12(1):9693. https://doi.org/10.1038/s41598-022-13807-9
Karabagli P (2013) Pathology in metopic synostosis. Childs Nerv Syst 29(12):2165–2170. https://doi.org/10.1007/s00381-013-2284-4
Teager SJ, Constantine S, Lottering N, Anderson PJ (2019) Physiologic closure time of the metopic suture in South Australian infants from 3D CT scans. Childs Nerv Syst 35(2):329–335. https://doi.org/10.1007/s00381-018-3957-9
Kapp-Simon KA, Speltz ML, Cunningham ML, Patel PK, Tomita T (2007) Neurodevelopment of children with single suture craniosynostosis: a review. Childs Nerv Syst 23(3):269–281. https://doi.org/10.1007/s00381-006-0251-z
Doorenbosch X, Molloy CJ, David DJ, Santoreneos S, Anderson PJ (2009) Management of cranial deformity following ventricular shunting. Childs Nerv Syst 25(7):871–874. https://doi.org/10.1007/s00381-009-0842-6
Ribeiro LA, El-Jaick KB, Muenke M, Richieri-Costa A (2006) SIX3 mutations with holoprosencephaly. Am J Med Genet A 140(23):2577–2583. https://doi.org/10.1002/ajmg.a.31377
Raam MS, Solomon BD, Shalev SA, Muenke M (2010) Holoprosencephaly and craniosynostosis: a report of two siblings and review of the literature. Am J Med Genet C Semin Med Genet 154C(1):176–182. https://doi.org/10.1002/ajmg.c.30234
Author information
Authors and Affiliations
Contributions
Sarut Chaisrisawadisuk: Conception and design, collection and assembly of data, manuscript writing, final approval of manuscript Inthira Khampalikit: Conception and design, collection and assembly of data, manuscript writing Ekawut Chankaew: Conception and design, manuscript writing Mark H. Moore: Conception and design, collection and assembly of data, manuscript writing.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
The patient’s parents provided written informed consent for the publication and the use of the images.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chaisrisawadisuk, S., Khampalikit, I., Chankaew, E. et al. Secondary metopic craniosynostosis after posterior cranial decompression in cloverleaf skull deformity. Childs Nerv Syst 40, 1937–1941 (2024). https://doi.org/10.1007/s00381-024-06309-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-024-06309-8