Abstract
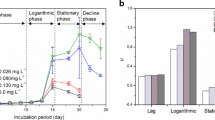
Bound extracellular polymeric substances (bEPS) play an important role in the proliferation of Microcystis. However, the understanding of bEPS characterization remains limited. In this study, three-dimensional fluorescence excitation-emission matrix (3D-EEM) spectroscopy and zeta potentiometer were used to characterize the loosely bound EPS (LB-EPS) and tightly bound EPS (TB-EPS) from two dominant Microcystis morphospecies from Taihu Lake (China) at different light intensities. Physiochemical analysis showed that the growth and TB-EPS or bEPS contents in Microcystis aeruginosa were higher than those in Microcystis flos-aquae at each light intensity. The 3D-EEM contour demonstrated that the intensities of peak B (tryptophan-like substances) in the TB-EPS from M. aeruginosa were stronger than those from M. flos-aquae when the light intensity was higher than 10 µE/(m2·s). Zeta potential analysis showed that the absolute values of the zeta potential of TB-EPS in the two species both increased with rising light intensity, except those of TB-EPS in M. aeruginosa at 105 µE/(m2·s). Moreover, the absolute values of the zeta potential of M. aeruginosa were higher than those of M. flos-aquae at each light intensity. All these results indicated that M. aeruginosa may more quickly proliferate than M. flos-aquae through increased negative charges, bEPS contents, growth, and tryptophan-like substance contents at certain light intensities.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Amaral V, Romera-Castillo C, García-Delgado M et al. 2020. Distribution of dissolved organic matter in estuaries of the southern Iberian Atlantic Basin: sources, behavior and export to the coastal zone. Marine Chemistry, 226: 103857, https://doi.org/10.1016/j.marchem.2020.103857.
Bradford M M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72(1–2): 248–254, https://doi.org/10.1016/0003-2697(76)90527-3.
Chen B F, Huang W, Ma S Z et al. 2018. Characterization of chromophoric dissolved organic matter in the littoral zones of eutrophic lakes Taihu and Hongze during the algal bloom season. Water, 10(7): 861, https://doi.org/10.3390/w10070861.
Chen M, Tian L L, Ren C Y et al. 2019. Extracellular polysaccharide synthesis in a bloom-forming strain of Microcystis aeruginosa: implications for colonization and buoyancy. Scientific Reports, 9(1): 1251, https://doi.org/10.1038/s41598-018-37398-6.
Chen W, Peng L, Wan N et al. 2009. Mechanism study on the frequent variations of cell-bound microcystins in cyanobacterial blooms in lake Taihu: implications for water quality monitoring and assessments. Chemosphere, 77(11): 1585–1593, https://doi.org/10.1016/j.chemosphere.2009.09.037.
Cruz D, Vasconcelos V, Pierre G et al. 2020. Exopolysaccharides from cyanobacteria: strategies for bioprocess development. Applied Sciences, 10(11): 3763, https://doi.org/10.3390/app10113763.
Dainard P G, Guéguen C, McDonald N et al. 2015. Photobleaching of fluorescent dissolved organic matter in Beaufort Sea and North Atlantic subtropical Gyre. Marine Chemistry, 177: 630–637, https://doi.org/10.1016/j.marchem.2015.10.004.
Dittrich M, Sibler S. 2005. Cell surface groups of two picocyanobacteria strains studied by zeta potential investigations, potentiometric titration, and infrared spectroscopy. Journal of Colloid and Interface Science, 286(2): 487–495, https://doi.org/10.1016/j.jcis.2005.01.029.
DuBois M, Gilles K A, Hamilton J K et al. 1956. Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28(3): 350–356, https://doi.org/10.1021/ac60111a017.
Fattom A, Shilo M. 1984. Hydrophobicity as an adhesion mechanism of benthic cyanobacteria. Applied and Environmental Microbiology, 47(1): 135–143, https://doi.org/10.1128/aem.47.1.135-143.1984.
Gan N Q, Xiao Y, Zhu L et al. 2012. The role of microcystins in maintaining colonies of bloom-forming Microcystis spp. Environmental Microbiology, 14(3): 730–742, https://doi.org/10.1111/j.1462-2920.2011.02624.x.
Ge H M, Zhang J, Zhou X P et al. 2014. Effects of light intensity on components and topographical structures of extracellular polymeric substances from Microcoleus vaginatus (Cyanophyceae). Phycologia, 53(2): 167–173, https://doi.org/10.2216/13-163.1.
Hadjoudja S, Deluchat V, Baudu M. 2010. Cell surface characterisation of Microcystis aeruginosa and Chlorella vulgaris. Journal of Colloid and Interface Science, 342(2): 293–299, https://doi.org/10.1016/j.jcis.2009.10.078.
Han P P, Shen S G, Wang H Y et al. 2015. Comparative metabolomic analysis of the effects of light quality on polysaccharide production of cyanobacterium Nostoc flagelliforme. Algal Research, 9: 143–150, https://doi.org/10.1016/j.algal.2015.02.019.
Han P P, Shen S G, Wang H Y et al. 2017. Applying the strategy of light environment control to improve the biomass and polysaccharide production of Nostoc flagelliforme. Journal of Applied Phycology, 29(1): 55–65, https://doi.org/10.1007/s10811-016-0963-8.
Harke M J, Steffen M M, Gobler C J et al. 2016. A review of the global ecology, genomics, and biogeography of the toxic cyanobacterium, Microcystis spp. Harmful Algae, 54: 4–20, https://doi.org/10.1016/j.hal.2015.12.007.
Huisman J, Codd G A, Paerl H W et al. 2018. Cyanobacterial blooms. Nature Reviews Microbiology, 16(8): 471–483, https://doi.org/10.1038/s41579-018-0040-1.
Huisman J, Sharples J, Stroom J M et al. 2004. Changes in turbulent mixing shift competition for light between phytoplankton species. Ecology, 85(11): 2960–2970, https://doi.org/10.1890/03-0763.
Li M, Zhu W, Gao L et al. 2013. Changes in extracellular polysaccharide content and morphology of Microcystis aeruginosa at different specific growth rates. Journal of Applied Phycology, 25(4): 1023–1030, https://doi.org/10.1007/s10811-012-9937-7.
Li M, Zhu W, Guo L L et al. 2016. To increase size or decrease density? Different Microcystis species has different choice to form blooms. Scientific Reports, 6(1): 37056, https://doi.org/10.1038/srep37056.
Li X Y, Yang S F. 2007. Influence of loosely bound extracellular polymeric substances (EPS) on the flocculation, sedimentation and dewaterability of activated sludge. Water Research, 41(5): 1022–1030, https://doi.org/10.1016/j.watres.2006.06.037.
Liu H, Fang H H P. 2002. Characterization of electrostatic binding sites of extracellular polymers by linear programming analysis of titration data. Biotechnology and Bioengineering, 80(7): 806–811, https://doi.org/10.1002/bit.10432.
Liu L Z, Huang Q, Qin B Q et al. 2016. Characterizing cell surface of blooming Microcystis in Lake Taihu, China. Water Science & Technology, 73(11): 2731–2738, https://doi.org/10.2166/wst.2016.069.
Meneghesso A, Simionato D, Gerotto C et al. 2016. Photoacclimation of photosynthesis in the Eustigmatophycean Nannochloropsis gaditana. Photosynthesis Research, 129(3): 291–305, https://doi.org/10.1007/s11120-016-0297-z.
Müller P, Li X P, Niyogi K K. 2001. Non-photochemical quenching. A response to excess light energy. Plant Physiology, 125(4): 1558–1566, https://doi.org/10.1104/pp.125.4.1558.
Ni L X, Li D Y, Rong S Y et al. 2017. Characterization of extracellular polymeric substance (EPS) fractions produced by Microcystis aeruginosa under the stress of linoleic acid sustained-release microspheres. Environmental Science and Pollution Research, 24(26): 21091–21102, https://doi.org/10.1007/s11356-017-9540-1.
Otero A, Vincenzini M. 2003. Extracellular polysaccharide synthesis by Nostoc strains as affected by N source and light intensity. Journal of Biotechnology, 102(2): 143–152, https://doi.org/10.1016/S0168-1656(03)00022-1.
Parlanti E, Wörz K, Geoffroy L et al. 2000. Dissolved organic matter fluorescence spectroscopy as a tool to estimate biological activity in a coastal zone submitted to anthropogenic inputs. Organic Geochemistry, 31(12): 1765–1781, https://doi.org/10.1016/S0146-6380(00)00124-8.
Phélippé M, Gonçalves O, Thouand G et al. 2019. Characterization of the polysaccharides chemical diversity of the cyanobacteria Arthrospira platensis. Algal Research, 38: 101426, https://doi.org/10.1016/j.algal.2019.101426.
Pokrovsky O S, Martinez R E, Golubev S V et al. 2008. Adsorption of metals and protons on Gloeocapsa sp. cyanobacteria: A surface speciation approach. Applied Geochemistry, 23(9): 2574–2588, https://doi.org/10.1016/j.apgeochem.2008.05.007.
Qu F S, Liang H, Wang Z Z et al. 2012. Ultrafiltration membrane fouling by extracellular organic matters (EOM) of Microcystis aeruginosa in stationary phase: influences of interfacial characteristics of foulants and fouling mechanisms. Water Research, 46(5): 1490–1500, https://doi.org/10.1016/j.watres.2011.11.051.
Rousso B Z, Bertone E, Stewart R A et al. 2021. Light-induced fluorescence quenching leads to errors in sensor measurements of phytoplankton chlorophyll and phycocyanin. Water Research, 198: 117133, https://doi.org/10.1016/j.watres.2021.117133.
Sheng G P, Yu H Q, Li X Y. 2010. Extracellular polymeric substances (EPS) of microbial aggregates in biological wastewater treatment systems: a review. Biotechnology Advances, 28(6): 882–894, https://doi.org/10.1016/j.biotechadv.2010.08.001.
Sirmerova M, Prochazkova G, Siristova L et al. 2013. Adhesion of Chlorella vulgaris to solid surfaces, as mediated by physicochemical interactions. Journal of Applied Phycology, 25(6): 1687–1695, https://doi.org/10.1007/s10811-013-0015-6.
Stanier R Y, Kunisawa R, Mandel M et al. 1971. Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriological Reviews, 35(2): 171–205, https://doi.org/10.1128/br.35.2.171-205.1971.
Straka L, Rittmann B E. 2017. Light attenuation changes with photo-acclimation in a culture of Synechocystis sp. PCC 6803. Algal Research, 21: 223–226, https://doi.org/10.1016/j.algal.2016.11.024.
Straka L, Rittmann B E. 2018. Light-dependent kinetic model for microalgae experiencing photoacclimation, photodamage, and photodamage repair. Algal Research, 31: 232–238, https://doi.org/10.1016/j.algal.2018.02.022.
Tan X, Shu X Q, Duan Z P et al. 2020. Two types of bound extracellular polysaccharides and their roles in shaping the size and tightness of Microcystis colonies. Journal of Applied Phycology, 32(1): 255–262, https://doi.org/10.1007/s10811-019-01937-z.
Trabelsi L, Ouada H B, Bacha H et al. 2009. Combined effect of temperature and light intensity on growth and extracellular polymeric substance production by the cyanobacterium Arthrospira platensis. Journal of Applied Phycology, 21(4): 405–412, https://doi.org/10.1007/s10811-008-9383-8.
Vogelaar J C T, De Keizer A, Spijker S et al. 2005. Bioflocculation of mesophilic and thermophilic activated sludge. Water Research, 39(1): 37–46, https://doi.org/10.1016/j.watres.2004.07.027.
Walsby A E, Hayes P K, Boje R et al. 1997. The selective advantage of buoyancy provided by gas vesicles for planktonic cyanobacteria in the Baltic Sea. New Phytologist, 136(3): 407–417, https://doi.org/10.1046/j.1469-8137.1997.00754.x.
Wang C, Kong H N, He S B et al. 2010. The inverse correlation between growth rate and cell carbohydrate content of Microcystis aeruginosa. Journal of Applied Phycology, 22(1): 105–107, https://doi.org/10.1007/s10811-009-9421-1.
Wang Q, Pang W J, Mao Y D et al. 2020. Changes of extracellular polymeric substance (EPS) during Microcystis aeruginosa blooms at different levels of nutrients in a eutrophic microcosmic simulation device. Polish Journal of Environmental Studies, 29(1): 349–360, https://doi.org/10.15244/pjoes/102367.
Xiao M, Li M, Duan P F et al. 2019. Insights into the relationship between colony formation and extracellular polymeric substances (EPS) composition of the cyanobacterium Microcystis spp. Harmful Algae, 83: 34–41, https://doi.org/10.1016/j.hal.2019.02.006.
Xiao M, Li M, Reynolds C S. 2018. Colony formation in the cyanobacterium Microcystis. Biological Reviews, 93(3): 1399–1420, https://doi.org/10.1111/brv.12401.
Xiao M, Willis A, Burford M A et al. 2017. Review: a meta-analysis comparing cell-division and cell-adhesion in Microcystis colony formation. Harmful Algae, 67: 85–91, https://doi.org/10.1016/j.hal.2017.06.007.
Xu F, Zhu W, Xiao M et al. 2016. Interspecific variation in extracellular polysaccharide content and colony formation of Microcystis spp. Cultured under different light intensities and temperatures. Journal of Applied Phycology, 28(3): 1533–1541, https://doi.org/10.1007/s10811-015-0707-1.
Xu H C, Cai H Y, Yu G H et al. 2013a. Insights into extracellular polymeric substances of cyanobacterium Microcystis aeruginosa using fractionation procedure and parallel factor analysis. Water Research, 47(6): 2005–2014, https://doi.org/10.1016/j.watres.2013.01.019.
Xu H C, He P J, Wang G Z et al. 2010. Three-dimensional excitation emission matrix fluorescence spectroscopy and gel-permeating chromatography to characterize extracellular polymeric substances in aerobic granulation. Water Science & Technology, 61(11): 2931–2942, https://doi.org/10.2166/wst.2010.197.
Xu H, Zhu G W, Qin B Q et al. 2013b. Growth response of Microcystis spp. to iron enrichment in different regions of Lake Taihu, China. Hydrobiologia, 700(1): 187–202, https://doi.org/10.1007/s10750-012-1229-3.
Yamamoto Y, Shiah F K, Chen Y L. 2011. Importance of large colony formation in bloom-forming cyanobacteria to dominate in eutrophic ponds. Annales de Limnologie-International Journal of Limnology, 47(2): 167–173, https://doi.org/10.1051/limn/2011013.
Yang H L, Cai Y F, Xia M et al. 2011. Role of cell hydrophobicity on colony formation in Microcystis (Cyanobacteria). International Review of Hydrobiology, 96(2): 141–148, https://doi.org/10.1002/iroh.201011312.
Yang Z, Geng L L, Wang W et al. 2012. Combined effects of temperature, light intensity, and nitrogen concentration on the growth and polysaccharide content of Microcystis aeruginosa in batch culture. Biochemical Systematics and Ecology, 41: 130–135, https://doi.org/10.1016/j.bse.2011.12.015.
Yang Z, Kong F X, Shi X L et al. 2008. Changes in the morphology and polysaccharide content of Microcystis aeruginosa (Cyanobacteria) during flagellate grazing. Journal of Phycology, 44(3): 716–720, https://doi.org/10.1111/j.1529-8817.2008.00502.x.
Yang Z, Kong F X. 2013. Abiotic factors in colony formation: effects of nutrition and light on extracellular polysaccharide production and cell aggregates of Microcystis aeruginosa. Chinese Journal of Oceanology and Limnology, 31(4): 796–802, https://doi.org/10.1007/s00343-013-2264-2.
Yue T, Zhang D L, Hu C X. 2014. Comparative studies on phosphate utilization of two bloom-forming Microcystis spp. (Cyanobacteria) isolated from Lake Taihu (China). Journal of Applied Phycology, 26(1): 333–339, https://doi.org/10.1007/s10811-013-0067-7.
Zhai C M, Song S, Zou S H et al. 2013. The mechanism of competition between two bloom-forming Microcystis species. Freshwater Biology, 58(9): 1831–1839, https://doi.org/10.1111/fwb.12172.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (No. 31800457) and the Natural Science Foundation of Hubei (No. 2016CFB355)
Rights and permissions
About this article
Cite this article
Wang, X., Han, X. & Ge, H. Effect of light intensity on bound EPS characteristics of two Microcystis morphospecies: the role of bEPS in the proliferation of Microcystis. J. Ocean. Limnol. 40, 1706–1719 (2022). https://doi.org/10.1007/s00343-022-1362-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00343-022-1362-4