Abstract
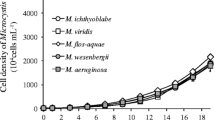
Colony morphology is important for Microcystis to sustain a competitive advantage in eutrophic lakes. The mechanism of colony formation in Microcystis is currently unclear. Extracellular polysaccharide (EPS) has been reported to play an important role in cell aggregate formation of some phytoplankton. Microcystis aeruginosa was cultivated under varied abiotic conditions, including different nutrient, light, and temperature conditions, to investigate their effects on EPS production and morphological change. The results show that nutrient concentration and light intensity have great effects on EPS productionin M. aeruginosa. There was a considerable increase in EPS production after M. aeruginosa was cultivated in adjusted culture conditions similar to those present in the field (28.9 mg C/L, 1.98 mg N/L, 0.65 mg P/L, light intensity: 100 μmol/(m2 · s)). These results indicate that abiotic factors might be one of the triggers for colony formation in Microcystis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bahat-Samet E, Castro-Sowinski S, Okon Y. 2004. Arabinose content of extracellular polysaccharide plays a role in cell aggregation of Azospirillum brasilense. FEMS Microbiol. Lett., 237: 195–203.
Bolch C J S, Blackburn S I. 1996. Isolation and purification of Australian isolates of the toxic cyanobacterium Microcystis aeruginosa Kutz. J. Appl. Phycol., 8: 5–13.
Chen L Z, Li D H, Song L R, Hu C X, Wang G H, Liu Y D. 2006. Effects of salt stress on carbohydrate metabolism in desert soil alga Microcoleus vaginatus Gom. J. Integr. Plant Biol., 48: 914–919.
De Philippis R, Vincenzini M. 1998. Exocellular polysaccharides from cyanobacteria and their possible applications. F EMS Microbiol. Rev., 22: 151–175.
De Philippis R, Margheri M C, Pelosi E, Ventura S. 1993. Exopolysaccharide production by a unicellular cyanobacterium isolated from a hypersaline habitat. J. Appl. Phycol., 5: 387–394.
De Philippis R, Sili C, Tassinato G, Vincenzini M, Materassi R. 1991. Effects of growth conditions on exopolysaccharide production by cyanospira-capsulata. Bioresource Technol., 38: 101–104.
Del Gallo M, Haegi A. 1990. Characterization and quantification of exocellular polysaccharides in Azospirillum brasilense and Azospirillum lipoferum. Symbiosis, 9: 155–161.
Di Pippo F, Ellwood N T W, Guzzon A, Siliato L, Micheletti E, De Philippis R, Albertano P B. 2012. Effect of light and temperature on biomass, photosynthesis and capsular polysaccharides in cultured phototrophic biofilms. J. Appl. Phycol., 24: 211–220.
Duan H T, Ma R H, Xu X F, Kong F X, Zhang S X, Kong W J, Hao J Y, Shang L L. 2009. Two-decade reconstruction of algal blooms in China’s lake Taihu. Environ. Sci. Technol., 43: 3 522–3 528.
Friedman C, Dubinsky Z, Arad S M. 1991. Effect of lightintensity on growth and polysaccharide production in red and blue-green Rhodophyta unicells. Bioresource Technol., 38: 105–110.
Hawes I. 1990. The effects of light and temperature on photosynthate partitioning in Antarctic freshwater phytoplankton. J. Plankton Res., 12: 513–518.
Helm R, Potts M. 2012. Extracellular Matrix (ECM). In: Whitton B ed. Ecology of Cyanobacteria II. Springer, Germany. p.461–480.
Herbert D, Phipps P J, Strange R E. 1971. Chemical analysis of microbial cells. Academic Press, London.
Humpage A R, Falconer I R. 1999. Microcystin-LR and liver tumor promotion: effects on cytokinesis, ploidy, and apoptosis in cultured hepatocytes. Environ. Toxicol., 14: 61–75.
Kroen W K, Rayburn W R. 1984. Influence of growth status and nutrients on extracellular polysaccharide synthesis by the soil alga Chlamydomonas mexicana (Chlorophyceae). J. Phycol., 20: 253–257.
Lancelot C, Mathot S, Owens N J P. 1986. Modelling protein synthesis, a step to an accurate estimate of net primary production: Phaeocystis pouchetii colonies in Belgian coastal waters. Mar. Ecol. Prog. Ser., 32: 193–202.
Li Y X, Li D H. 2012. Physiological variations of bloomforming Microcystis (Cyanophyceae) related to colony size changes during blooms. Phycologia, 51: 599–603.
Moreno J, Vargas M A, Olivares H, Rivas J, Guerrero M G. 1998. Exopolysaccharide production by the cyanobacterium Anabaena sp. ATCC 33047 in batch and continuous culture. J. Biotechnol., 60: 175–182.
Oliver R L. 1994. Floating and sinking in gas vacuolate cyanobacteria. J. Phycol., 30: 161–173.
Otero A, Vincenzini M. 2003. Extracellular polysaccharide synthesis by Nostoc strains as affected by N source and light intensity. J. Biotechnol., 102: 143–152.
Pajdak-Stos A, Fialkowska E, Fyda J. 2001. Phormidium autumnale (Cyanobacteria) defense against three ciliate grazer species. Aquat. Microb. Ecol., 23: 237–244.
Raungsomboon S, Chidthaisong A, Bunnag B, Inthorn D, Harvey N W. 2006. Production, composition and Pb2+ adsorption characteristics of capsular polysaccharides extracted from a cyanobacterium Gloeocapsa gelatinosa. Water Res., 40: 3 759–3 766.
Reynolds C S. 2007. Variability in the provision and function of mucilage in phytoplankton: facultative responses to the environment. Hydrobiologia, 578: 37–45.
Reynolds C S, Oliver R L, Walsby A E. 1987. Cyanobacterial dominance—the role of buoyancy regulation in dynamic lake environments. New Zeal. J Mar. Fresh., 21: 379–390.
Reynolds C S, Walsby A E. 1975. Water blooms. Biol. Rev., 50: 437–441.
Šejnohová L, Maršálek B. 2012. Microcystis. In: Whitton B ed. Ecology of Cyanobacteria II. Springer, Germany. p.195–228.
Staats N, Stal L J, Mur L R. 2000. Exopolysaccharide production by the epipelic diatom Cylindrotheca closterium: effects of nutrient conditions. J. Exp. Mar. Biol. Ecol., 249: 13–27.
Stanier R Y, Kunisawa R, Mandel M, Cohen-Bazire G. 1971. Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol. Rev., 35: 171–205.
van Rijssel M, Janse I, Noordkamp D J B, Gieskes W W C. 2000. An inventory of factors that affect polysaccharide production by Phaeocystis globosa. J. Sea Res., 43: 297–306.
Wang C, Kong H N, He S B, Zheng X Y, Li C J. 2010a. The inverse correlation between growth rate and cell carbohydrate content of Microcystis aeruginosa. J. Appl. Phycol., 22: 105–107.
Wang W, Liu Y, Yang Z. 2010b. Combined effects of nitrogen content in media and Ochromonas sp. grazing on colony formation of cultured Microcystis aeruginosa. J. Limnol., 69: 193–198.
Wu Z X, Song L R. 2008. Physiological comparison between colonial and unicellular forms of Microcystis aeruginosa Kutz. (Cyanobacteria). Phycologia, 47: 98–104.
Xu Y F, Ma C Z, Huo S L, Xi B D, Qian G R. 2012. Performance assessment of water quality monitoring system and identification of pollution source using pattern recognition techniques: a case study of Chaohu Lake, China. Desalin. Water Treat., 47: 182–197.
Yang Z, Kong F X, Shi X L, Zhang M, Xing P, Cao H S. 2008. Changes in the morphology and polysaccharide content of Microcystis aeruginosa (Cyanobacteria) during flagellate grazing. J. Phycol., 44: 716–720.
Yang Z, Kong F X, Yang Z, Zhang M, Yu Y, Qian S Q. 2009. Benefits and costs of the grazer-induced colony formation in Microcystis aeruginosa. Ann. Limnol. -Int. J. Lim., 45: 203–208.
Zhang X J, Chen C, Lin P F, Hou A X, Niu Z B, Wang J. 2011. Emergency drinking water treatment during source water pollution accidents in China: origin analysis, framework and technologies. Environ. Sci. Technol., 45: 161–167.
Zhang M, Kong F X, Tan X, Yang Z, Cao H S, Xing P. 2007. Biochemical, morphological, and genetic variations in Microcystis aeruginosa due to colony disaggregation. World J. Microb. Biot., 23: 663–670.
Author information
Authors and Affiliations
Additional information
Supported by the National Natural Science Foundation of China (No. 31200296) and the CAS International Partnership Creative Group (No. KZZD-EW-TZ-08-01)
Rights and permissions
About this article
Cite this article
Yang, Z., Kong, F. Abiotic factors in colony formation: effects of nutrition and light on extracellular polysaccharide production and cell aggregates of Microcystis aeruginosa . Chin. J. Ocean. Limnol. 31, 796–802 (2013). https://doi.org/10.1007/s00343-013-2264-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00343-013-2264-2