Abstract
Key message
We have defined the conditions for citrus transformations using glyphosate as selection agent. This protocol results in high transformation rate and low incidence of chimeric shoots.
Abstract
Glyphosate, the most widely used herbicide in the world, specifically inhibits 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS), an essential enzyme of the shikimate pathway. Various laboratory-generated or naturally evolved glyphosate-resistant EPSPS variants have been used to produce glyphosate-tolerant transgenic crops, enabling highly effective weed control in agriculture. In this study, we explored the potential of using a citrus EPSPS variant that mimics the previously reported Eleusine indica glyphosate-resistant TIPS (T102I + P106S) mutant for selection of transgenic citrus plants in the presence of glyphosate. We found that glyphosate did not suppress bud formation on ‘Duncan’ grapefruit seedling explants, but inhibited non-transgenic bud outgrowth to produce shoots in a concentration-dependent manner. At certain concentrations, glyphosate had dramatic effect on the transformation rate and the percentage of non-chimeric transgenic shoots in this newly developed selection system. Specifically, at 0, 10, 20, and 50 μM of glyphosate, the citrus TIPS EPSPS-based selection resulted in transformation rates of 4.02, 5.04, 14.46, and 40.78%, respectively, and 6.41, 23.96, 42.94, and 40.17% of non-chimeric transgenic shoots, respectively. These results indicate that the citrus TIPS EPSPS-glyphosate selection system is highly efficient and can be used as an alternative to antibiotic-based selection methods in citrus genetic transformation. Furthermore, the selection conditions defined in this study are expected to greatly facilitate the production of genetically modified, market-friendly citrus plants, such as cisgenic and intragenic plants.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Within the last two decades, considerable progress has been made in transgenic improvement of crop plants including tree fruit crops (Song et al. 2019). For citrus, introduction of beneficial traits into commercial cultivars through transgenic approaches is crucial, because conventional breeding is labor-intensive and time-consuming due to incompatibility, apomixis, heterozygosity, and long juvenility. Introduction of genes via Agrobacterium-mediated transformation into elite citrus cultivars significantly accelerates trait improvement and allows proof-of-concept experiments to be conducted quickly. This is particularly important at the present time, as citrus producers in many countries are suffering significant losses due to the devastating disease known as Huanglongbing (HLB). Creation of trees that are either tolerant or resistant to HLB is imperative for the survival of citrus industry in many areas, and the transgenic technology is expected to play a major role in this endeavor (NASEM 2018).
A large number of genes have been introduced into various cultivars via Agrobacterium-mediated transformation for improvement of different traits of citrus trees including resistance to diseases (Fagoaga et al. 2001, 2006; Hao et al. 2016, 2017; Mondal et al. 2012; Zhang et al. 2010; Zou et al. 2017), resistance to abiotic stress (Alvarez-Gerding et al. 2015; Orbović et al. 2017), flavor (Bachchu et al. 2011; Koca et al. 2009), and flowering time (Sinn et al. 2020). In these transgenic plants, the transfer DNA (T-DNA) regions of the binary vectors carrying the genes of interest were stably inserted into the plants’ genomes. The T-DNA regions included different selectable marker genes, which encode proteins conferring resistance to specific antibiotics, such as kanamycin, hygromycin, or the herbicide Basta.
The Escherichia coli phosphomannose isomerase encoded by the manA gene has also been used as a selectable marker in citrus genetic transformation (Dutt et al. 2010; Wu et al. 2019). This enzyme converts mannose phosphate into fructose phosphate and thereby allows the growth of plant tissue on medium supplemented only with mannose that serves as benign selective agent. Another approach is to use the green fluorescent protein (GFP) as a reporter that allows visual identification of transgenic shoots, though a large number of shoots that regenerated from the explants will need to be screened (Ballester et al. 2010). Nevertheless, the “foreign DNA” including the selectable marker genes and the GFP reporter gene introduced into the transgenic plants raises public concerns and reduces market acceptance of the resulting products. Thus, researchers are looking for alternative ways to generate beneficial changes within the genome while decreasing or completely eliminating the presence of “foreign DNA” (Yau and Stewart 2013). For instance, the Cre/loxP-mediated recombination system has been used to excise part of the T-DNA inserted into the citrus genome (Peng et al. 2015).
Another strategy to avoid “foreign DNA” is to use genetic materials derived from the species itself or from closely related sexually compatible species. This has led to the development of two new technologies, cisgenesis and intragenesis (Singh et al. 2015). Cisgenesis employs a complete copy of a natural gene with its regulatory elements, whereas intragenesis allows the use of new fusion genes via rearrangements of functional genetic elements (Singh et al. 2015). We have previously developed a T-DNA binary vector using genetic materials derived from Citrus clementina for production of intragenic citrus plants (An et al. 2013). However, identification of intragenic citrus plants was conducted through PCR under non-selective conditions, resulting in an extremely low efficiency (An et al. 2013). A selection method based on citrus DNA sequences would greatly facilitate this process.
The shikimate pathway enzyme 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS) is the target of the most widely used herbicide glyphosate (Steinrucken and Amrhein 1980). To use glyphosate in agriculture for effective weed control, various glyphosate-resistant forms of EPSPS have been identified via screening laboratory-generated or naturally evolved glyphosate-tolerant microbes and plants. Several glyphosate-resistant EPSPS variants have been successfully employed to engineer transgenic crops resistant to glyphosate (Funke et al. 2006; Padgette et al. 1995; Pollegioni et al. 2011). One such EPSPS variant was found in naturally occurring glyphosate-resistant Eleusine indica and carries a double amino acid substitution, T102I + P106S, which was named the TIPS mutation (Yu et al. 2015). The TIPS EPSPS has been used to generate the first generation commercially successful glyphosate-tolerant transgenic corn GA21 (Spencer et al. 2000).
The citrus genome encodes a single protein homolog of EPSPS, which shares high amino acid identity with the E. indica EPSPS. The two amino acids T102 and P106 of the E. indica EPSPS are conserved in the citrus EPSPS. In this study, we created a citrus TIPS EPSPS and found that it is able to confer glyphosate tolerance in the model plant Arabidopsis thaliana. We further discovered that glyphosate did not hinder bud formation on ‘Duncan’ grapefruit seedling explants, but drastically inhibited bud outgrowth. Importantly, the citrus TIPS EPSPS-based selection significantly increased the transformation rate and the percentage of non-chimeric transgenic shoots. This highly efficient citrus TIPS EPSPS-glyphosate selection system can not only serve as an alternative to antibiotic-based selection methods in citrus genetic transformation, but also facilitate the production of cisgenic and intragenic plants in citrus.
Materials and methods
Plant materials and growth conditions
The A. thaliana (L.) Heynh. ecotype Columbia (Col-0) was used for genetic transformation. Col-0 seeds were sown on autoclaved soil (Propagation Mix; Sun Gro Horticulture, Agawam, MA) and cold-treated at 4 °C for 3 days. After germination, plants were grown at ~ 23 °C under a 16-h-light/8-h-dark regime till flowering for transformation. For citrus, seeds were extracted from ‘Duncan’ grapefruit (Citrus × paradisi) fruit, surface- sterilized, and germinated in dark for 5 weeks as previously described (Orbović and Grosser 2015). Etiolated seedlings were exposed to white light for a week and the hypocotyls were then cut into 1.5 cm long segments as explants for glyphosate tolerance test and genetic transformation.
Cloning and mutagenesis of the citrus EPSPS gene
Citrus sinensis total RNA was extracted using an RNeasy Plant Mini Kit (QIAGEN Sciences Inc., Germantown, MD, USA) following the manufacturer’s instructions. Reverse transcription (RT) was performed as previously described (Zhang et al. 2010). A pair of primers SacI-CsEPSPSF (C. sinensis EPSPS) and SalI-CsEPSPS (Supplemental Table 1) was used to amplify the coding region of the citrus EPSPS from the total cDNA generated by RT. The PCR products were digested with SacI and SalI and then ligated into the corresponding sites of pBluescript II SK(+), resulting in pBluescript SK(+)-CsEPSPS. Site-directed mutagenesis of T177 and P181, which correspond to the two amino acids T102 and P106 of the E. indica EPSPS, was performed in the pBluescript SK(+)-CsEPSPS construct using a PCR-based QuickChange Site-Directed Mutagenesis Kit (Agilent Technologies, Santa Clara, CA, USA). The primers used for the site-directed mutagenesis were CsEPSPS-MuF and CsEPSPS-MuR (Supplemental Table 1). The presence of the expected mutations in the resulting construct pBluescript SK(+)-TIPS CsEPSPS was confirmed by DNA sequencing.
Glyphosate sensitivity assay of bud and shoot formation on citrus explants
Roundup Super Concentrate (active ingredient: 50.2% or 1.38 M glyphosate) was diluted with dH2O to make a 0.5 M stock solution of glyphosate. The solution was filter-sterilized and stored in aliquots at – 20 °C. Aliquots of glyphosate were thawed at room temperature and added to sterilized and cooled (50 °C) Murashige and Skoog (MS) medium (Murashige and Skoog 1962).
To determine the minimum inhibitory concentration of glyphosate for bud and/or shoot formation, the explants were incubated in liquid co-cultivation medium (Orbović and Grosser 2015), dried on sterile napkin, and placed onto MS medium supplemented with 5 mg/L of 6-benzylaminopurine (BAP). After 7 days of incubation in the dark, the ends of the explants were cut off (3-5 mm long) and placed with the cut surface down into MS medium supplemented with 1 mg/L of BAP and a series of concentrations of glyphosate ranging from 0 to 10 µM. The plates were again incubated in the dark for 7 days, and then brought into light. The buds and shoots sprouting from the explants were counted after 6–8 weeks. To estimate the biomass of the newly formed buds and shoots, the surface area of the buds and shoots on the explants exposed to different concentrations of glyphosate was measured with the ImageJ software.
Plasmid construction and plant transformation
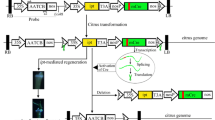
A T-DNA binary vector was built for delivery of the citrus TIPS EPSPS gene into A. thaliana and citrus. Briefly, the fragment of 2x35S promoter-polylinker-poly(A) signal was excised from pCAMBIA1300S with HindIII and EcoRI to replace the HindIII-EcoRI region in pBI101, resulting in pZM235. Meanwhile, three pairs of primers, GFP cassetteF1 + R1, F2 + R2, and F3 + R3 (Supplemental Table 1) were used to amplify the proID promoter, eGFP, and the T35S terminator, respectively, from pK7WG2D,1, resulting in three fragments with more than 20 bp overlaps. The three PCR fragments were assembled into one fragment via assembly PCR, which was then digested with HindIII and inserted into the HindIII site in pZM235, resulting in pZM332 with the proID promoter oriented in the opposite direction of the 2x35S promoter. Finally, the TIPS CsEPSPS gene was removed from pBluescript SK(+)-TIPS CsEPSPS with SacI and SalI and ligated into the corresponding sites of pZM332, resulting in pZM332-TIPS CsEPSPS. This T-DNA binary construct was mobilized into the Agrobacterium strains GV3101(pMP90) and EHA105 for transformation of A. thaliana and citrus, respectively.
Transformation of A. thaliana plants was conducted following the floral dip protocol (Clough and Bent 1998). Citrus transformation was performed as previously described with glyphosate as the selective agent (Orbović and Grosser 2015). Agrobacterium strain EHA105 carrying the pZM332-TIPS CsEPSPS plasmid was grown overnight in liquid YEP medium containing 50 µg/mL of kanamycin and 50 µg/mL of rifampicin on an orbital shaker at 28 °C and 220 rpm. Bacterial cells were pelleted by centrifugation (10 min at 5000×g) and the optical density at 620 nm was adjusted to 0.6 by resuspension in liquid co-cultivation medium (CCM) (MS salts and vitamins, 3 mg/L benzyladenine, 0.5 mg/L 2,4-D, 0.1 mg/L NAA, and 100 μM acetosyringone).
Seedling explants were incubated in the Agrobacterium suspension for 1–2 min. The infected explants were blotted dry on sterile filter paper and placed horizontally on solid CCM (same as liquid CCM with the addition of 8 g/L agar). Two days after co-incubation, the explants were transferred onto regeneration medium (MS salts and vitamins, 3 mg/L benzyladenine, 0.1 mg/L NAA, 333 μg/mL cefotaxime, 50 μg/mL kanamycin, 8 g/L agar) supplemented with indicated concentrations of glyphosate. The cultures were maintained in a 16-h photoperiod for five weeks, with 45 μE/m2s illumination and 60% relative humidity at 26 °C.
Statistical analysis
Statistical analyses were performed using the SAS software (SAS Institute, Cary, NC, USA) and the one-way ANOVA in Prism 7 (GraphPad Software, San Diego, CA, USA).
Results
The citrus TIPS EPSPS confers glyphosate tolerance in transgenic A. thaliana plants
To identify the citrus EPSPS, the E. indica EPSPS protein sequence was used as query for BLAST in Citrus Genome Database (https://www.citrusgenomedb.org/). A single homologous protein with 519 amino acids (orange1.1t00227) was obtained. The citrus EPSPS homolog shares 84% amino acid identity with the E. indica EPSPS (Supplemental Figure 1). The highly conserved region (95LFLGNAGTAMRPL107) in the E. indica EPSPS is identical with the amino acids between L170 and L182 of the citrus EPSPS (Fig. 1A). To generate a citrus TIPS EPSPS variant, the conserved amino acids T177 and P181 were substituted with isoleucine (I) and serine (S), respectively, via PCR-based site-directed mutagenesis (Fig. 1B). The resulting citrus TIPS EPSPS gene was cloned into a newly built T-DNA binary vector pZM332.
The ability of the citrus TIPS EPSPS to provide glyphosate tolerance in A. thaliana. A The highly conserved regions in the E. indica and citrus (C. sinensis) EPSPS proteins. The conserved threonine (T) and proline (P) residues are labeled in bold. B The double substitutions of the amino acids T177 and P181 isoleucine (I) and serine (S) in the citrus TIPS EPSPS. C Glyphosate tolerance of transgenic A. thaliana plants expressing the citrus TIPS EPSPS. Ten-day-old seedlings of five single insertion homozygous transgenic lines (2–4, 3–2, 7–4, 26–4, and 35–2) expressing the citrus TIPS EPSPS and the non-transgenic wild-type Col-0 were sprayed with ~ 0.27% glyphosate. The photos were taken 10 days later. Compared with the wild-type Col-0, all five transgenic lines exhibited elevated levels of tolerance to glyphosate. The experiment was repeated three times with similar results
We then tested whether the citrus TIPS EPSPS could confer glyphosate tolerance in transgenic A. thaliana plants. To this end, the T-DNA binary construct pZM332-TIPS CsEPSPS was introduced into the Agrobacterium strain GV3101(pMP90) and the resulting Agrobacteria were used to transform wild-type Col-0 plants. Single T-DNA insertion lines were isolated in the T2 generation and homozygous plants of these lines were identified in the T3 generation. A total of five single insertion homozygous lines (2–4, 3–2, 7–4, 26–4, and 35–2) were obtained and tested for glyphosate tolerance. As shown in Fig. 1C, compared with the wild-type Col-0, all five transgenic lines exhibited elevated glyphosate tolerance, though the tolerance levels varied, which might be attributed to different expression levels of TIPS CsEPSPS in the transgenic lines. Nevertheless, this result indicated that the citrus TIPS EPSPS is able to provide glyphosate tolerance when ectopically expressed in A. thaliana plants.
Glyphosate suppresses non-transgenic shoot formation on citrus explants
To use glyphosate as a selective agent in citrus genetic transformation, bud and/or shoot formation on explants must be sensitive to glyphosate. To test this possibility, slices of hypocotyl explants (Supplemental Figure 2) were placed on a series of concentrations of glyphosate ranging from 0 to 10 µM. As shown in Supplemental Figure 3 and Fig. 2A, the average number of buds and shoots that sprouted from each explant varied from 4.48 ± 0.26 to 6.83 ± 0.38 with the highest at 5 μM of glyphosate. However, the average number of shoots on each explant at 10 μM of glyphosate was 0.59 ± 0.14, which is the lowest among all the concentrations and is nearly fourfold lower than that (2.21 ± 0.19) on plates without glyphosate (Fig. 2B). As the average number of buds and shoots did not decrease in the presence of 10 μM glyphosate but the number of shoots dropped significantly, the bud/shoot ratio went up more than twelvefold from 1.08 ± 0.29 on the control plates to 13.32 ± 3.65 on plates with 10 μM of glyphosate (Fig. 2C).
The effect of glyphosate on bud and shoot formation on citrus explants. A Average number of buds and shoots formed on each explant on media with different concentrations of glyphosate. B Average number of shoots sprouting from each explant on the media with different concentrations of glyphosate. C Bud to shoot ratios in the presence of different concentrations of glyphosate. The experiment was repeated three times and data from the three times were used together for statistical analyses. In A and B, data represent the mean of 66–120 explants with standard error (SE). Different letters above the error bars indicate significant differences (P < 0.05, Duncan’s multiple range test). In C, bud to shoot ratios were calculated for each plate, and data represent the mean of three to six plates with SE. Different letters above the error bars indicate significant differences (P < 0.05, one-way ANOVA)
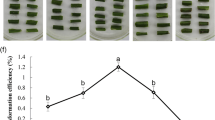
To estimate the effect of glyphosate on the biomass of the newly formed buds and shoots, the surface area corresponding to the buds and shoots that sprouted from each explant was measured. Significant inhibitory effect of glyphosate on the surface area was found at 2 μM and the inhibitory effect was further enhanced as the glyphosate concentration went up to 5 or 10 μM (Fig. 3A). At 1 μM or lower concentrations, the surface area of buds and shoots on each explant was ~ 90 mm2, whereas at 2, 5, and 10 μM of glyphosate, the surface area decreased to 60.99 ± 12.30, 50.25 ± 7.93, and 26.41 ± 3.38 mm2, respectively. This decrease could, at least partially, be attributed to the reduced number of shoots on the explant (Fig. 3B–D). Taken together, these results indicated that 10 μM of glyphosate could dramatically suppress shoot formation on the explants and suggested that 10 μM or higher concentrations of glyphosate would be needed for transgenic shoot selection.
The effect of glyphosate on the biomass of newly formed buds and shoots. A Average surface area of buds and shoots on each explant on media with different concentrations of glyphosate. The surface area of buds and shoots was used to estimate the biomass of newly formed buds and shoots on the explant. The experiment was repeated three times and data from the three times were used together for statistical analysis. Average surface area of buds and shoots per explant was calculated for each plate, and data represent the mean of three to six plates with SE. Different letters above the error bars indicate significant differences (P < 0.05, one-way ANOVA). B A representative plate with explants on medium with 0 μM of glyphosate. C A representative plate with explants on medium with 5 μM of glyphosate. D A representative plate with explants on medium with 10 μM of glyphosate. Scale bars on panels B, C, and D are 10 mm long
The citrus TIPS EPSPS confers glyphosate tolerance in transgenic citrus shoots
Since 10 μM of glyphosate is required to suppress non-transgenic shoot formation on explants, we tested the efficacy of glyphosate as a selective agent for Agrobacterium-mediated citrus transformation using 10 μM and higher concentrations of glyphosate. The T-DNA binary construct pZM332-TIPS CsEPSPS was introduced into the Agrobacterium strain EHA105 and the resulting Agrobacteria were used to transform ‘Duncan’ grapefruit seedling hypocotyl segments (Orbović and Grosser 2015). Compared with that on control plates without glyphosate, the average number of shoots on each explant was significantly reduced on the plates supplemented with glyphosate, regardless of the concentrations (Figs. 4A, 5). On the other hand, the average percentage of GFP positive (transgenic) shoots among the fully developed shoots increased significantly as the glyphosate concentration went up (Fig. 4B). The pZM332-TIPS CsEPSPS construct carries the GFP reporter cassette, proID-eGFP-T35S, in the T-DNA region, GFP fluorescence was thus used for easy identification of transgenic shoots. The percentages of GFP positive shoots at 0, 10, 20, and 50 μM of glyphosate were 4.02, 5.04, 14.46, and 40.78%, respectively (Fig. 4B). The transgenic shoots included chimeric and fully GFP fluorescent ones (Fig. 5), and the fully fluorescent shoots were considered non-chimeric transgenic shoots. In the presence of 0, 10, 20, and 50 μM of glyphosate, the percentages of non-chimeric transgenic shoots were 6.41, 23.96, 42.94, and 40.17%, respectively (Fig. 4C). Taken together, our results not only demonstrated that the citrus TIPS EPSPS-glyphosate selection system is highly efficient for identification of transgenic citrus shoots but also revealed the optimum conditions for using this newly developed selection method.
Identification of transgenic shoots using the citrus TIPS EPSPS-glyphosate selection system. A After co-incubation with Agrobacteria, average number of shoots formed on each explant on media with different concentrations of glyphosate. B Transformation rates (transgenic shoots/total shoots) in the presence of different concentrations of glyphosate. C Non-chimeric transgenic shoot rates (non-chimeric transgenic shoots/total transgenic shoots) in the presence of different concentrations of glyphosate. The experiment was repeated four times and data from the four times were used together for statistical analyses. In A, data represent the mean of 419–912 explants with standard error (SE). Different letters above the error bars indicate significant differences (P < 0.05, Duncan’s multiple range test). In B and C, transformation rates and non-chimeric transgenic shoot rates were calculated for each plate, and data represent the mean of 21–48 plates with SE. Different letters above the error bars indicate significant differences (P < 0.05, one-way ANOVA)
Representative explants and transgenic shoots on selective media. A Shoot formation on medium without glyphosate and a transgenic shoot with chimeric GFP fluorescence. B Shoot formation on medium with 10 μM glyphosate and a transgenic shoot with chimeric GFP fluorescence. C Shoot formation on medium with 20 μM glyphosate and a transgenic shoot with full GFP fluorescence. D Shoot formation on medium with 50 μM glyphosate and a transgenic shoot with full GFP fluorescence
Discussion
In this study, we generated the citrus TIPS EPSPS variant resembling the naturally evolved E. indica TIPS EPSPS mutant that conferred high-level glyphosate resistance (Yu et al. 2015). We demonstrated that the citrus TIPS EPSPS, when combined with glyphosate as the selective agent, is a valuable selectable marker in plant genetic transformation. Furthermore, we defined the optimum conditions for identification of transgenic ‘Duncan’ grapefruit shoots using the citrus TIPS EPSPS-glyphosate selection system. Additionally, the morphology of 1-year old transgenic ‘Duncan’ grapefruit plants generated with this method is similar to that of the wild-type control (Supplemental Figure 4), suggesting that the TIPS EPSPS transgene may not have adverse effects on growth and development. Thus, our newly developed system offers an alternative to antibiotic-based selection methods in citrus genetic transformation and provides a potential tool for generation of cisgenic and intragenic plants in citrus.
As the active ingredient of the herbicide Roundup, glyphosate targets EPSPS whose activity is absolutely required for the survival of plants. At micromolar concentrations, glyphosate did not negatively affect initiation of bud morphogenesis from ‘Duncan’ grapefruit explants (Fig. 2A). However, at concentrations higher than 10 μM, it strongly inhibited the development of shoots (Figs. 2B, 4A). As a result, the biomass or the surface area of buds and shoots that sprouted from the explants decreased with the increased levels of glyphosate in the medium (Fig. 3A). A previous study with poplar cell cultures showed that glyphosate fully inhibited bud morphogenesis in the millimolar range (Klimazsewska and Cheliak 1987). However, as the concentrations of glyphosate were lowered to micromolar levels, the authors observed regeneration of buds and very short shoots (Klimazsewska and Cheliak 1987). These observations are consistent with our results.
With GFP as a reporter, we could easily identify transgenic shoots that regenerated from explants exposed to various concentrations of glyphosate in the medium. Most of the large, healthy shoots with several leaves found on the plates with 20 and 50 μM of glyphosate were GFP positive and therefore tolerant to the chemical (Fig. 5). Some smaller, underdeveloped shoots were also GFP positive, but were found less frequently. Transformation rates of 14.46% and 40.78% were obtained at 20 and 50 μM of glyphosate, respectively, compared with 4.02% and 5.04% at 0 and 10 μM of the chemical, respectively (Fig. 4B). The transformation rate of 4.02% on the medium without glyphosate appeared to be acceptable. However, only 6.41% of the transgenic shoots produced in the absence of glyphosate had GFP fluorescence in all tissues, which were potentially non-chimeric transgenic shoots (Figs. 4C, 5). Consequently, the rate of producing non-chimeric transgenic shoots without glyphosate selection was ~ 0.26% (Fig. 4B, C). In contrast, with 20 and 50 μM of glyphosate in the medium, the rates of producing non-chimeric transgenic shoots were ~ 6.21 and 16.38%, respectively (Fig. 4B, C). In other words, among 100 shoots generated in the presence of 50 μM glyphosate, about 16 could be non-chimeric transgenic shoots. Thus, we recommend to use 20–50 μM of glyphosate in the selection medium if the T-DNA binary vector pZM332-TIPS EPSPS is used for generation of transgenic citrus plants in the future.
In this paper, we chose to calculate transformation rate as a percentage of transgenic shoots in relation to the total number of shoots that sprouted from explants (also used by Ballester et al. 2010 and Peng et al. 2015) based on the following justification. While shoot morphogenesis is a necessary condition for plant transformation, it is not the only one (Song et al. 2019). Other conditions, such as Agrobacterium attachment, activation of vir genes, proper movement and entry of T-DNA into plant cells and nuclei, levels of expression of transformation-related host genes, and many others, influence the transformation rate. Therefore, realized capacity of explants’ cambial tissue for shoot morphogenesis indirectly affects transformation rate. Depending on the protocol, seedling explants used for citrus transformation will yield different numbers of shoots. As a result, different amount of work would be required to identify transgenic shoots. For example, 100 explants are used for transformation. About 400 shoots are generated with protocol #1, and 100 shoots are generated with protocol #2. There are 10 transgenic shoots in both cases. If the explant numbers are used to calculate the transformation rate, there would be no difference between the two protocols (10%). However, if total shoot numbers are used, the rates for protocols #1 and #2 would be 2.5 and 10%, respectively. Protocol #2 would be a better one. Essentially, this is what happened for the selection method reported in this manuscript.
Nevertheless, it is up to the researcher who does experiments to pick the protocol and get lower or higher numbers of shoots regenerated from explants. They may also choose to express transformation efficiency/rate the way they prefer. The method developed in this study decreases the number of shoots regenerated from explants, which significantly reduces the cost for identifying the same number of transgenic shoots. We calculate the rate in relation to the total number of shoots regenerated from explants to reflect this improvement.
The citrus TIPS EPSPS is potentially useful in cisgenesis and intragenesis. In the current study, the 2x35S promoter was used to drive the citrus TIPS EPSPS gene, which is not allowed in cisgenesis and intragenesis. For cisgenesis, the TIPS mutation will need to be introduced into the native citrus EPSPS gene and the complete copy of the gene including its promoter and terminator will need to be used. As the naturally occurring TIPS mutation in the E. indica genome conferred high-level glyphosate resistance (Yu et al. 2015), it can be expected that the expression level of the citrus TIPS EPSPS gene driven by its native promoter would be sufficient for providing glyphosate tolerance. For intragenesis, on the other hand, strong citrus promoters can be used to drive the citrus TIPS EPSPS gene created in this study and the resulting fusion gene could then be cloned into the previously reported intragenic vector (An et al. 2013). It is possible that the glyphosate concentration that was required for suppression of non-transgenic shoots in this study will be suitable for other citrus varieties. However, as the strength of various promoters is different, selection conditions for specific constructs will need to be optimized. In this regard, the conditions defined for the citrus TIPS EPSPS-glyphosate selection system in this study will be valuable for optimization of conditions for future generation of cisgenic and intragenic plants in citrus.
Availability of data and material
The constructs used in this study are available upon request from the corresponding author Zhonglin Mou (zhlmou@ufl.edu).
Code availability
Not applicable.
References
Alvarez-Gerding X, Espinoza C, Inostroza-Blancheteau C, Arce-Johnson P (2015) Molecular and physiological changes in response to salt stress in Citrus macrophylla W plants overexpressing Arabidopsis CBF3/DREB1A. Plant Physiol Biochem 92:71–80. https://doi.org/10.1016/j.plaphy.2015.04.005
An C, Orbović V, Mou Z (2013) An efficient intragenic vector for generating intragenic and cisgenic plants in citrus. Am J Plant Sci 4:2131–2137
Bachchu MA, Jin SB, Park J-W, Boo K-B, Sun H-J, Kim Y-W, Lee H-Y, Riu K-Z, Kim J-H (2011) Functional expression of Miraculin, a taste-modifying protein, in transgenic Miyagawa Wase Satsuma Mandarin (Citrus unshiu Marc.). J Korean Soc Appl Biol Chem 54:24–29
Ballester A, Cervera M, Pena L (2010) Selectable marker-free transgenic orange plants recovered under non-selective conditions and through PCR analysis of all regenerants. Plant Cell Tissue Org Cult 102:329–336
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Dutt M, Lee DH, Grosser JW (2010) Bifunctional selection-reporter systems for genetic transformation of citrus: mannose and kanamycin based systems. Vitro Cell Devel Biol Plant 46:467–476
Fagoaga C, López C, de Mendoza AH, Moreno P, Navarro L, Flores R, Peña L (2006) Post- transcriptional gene silencing of the p23 silencing suppressor of Citrus tristeza virus confers resistance to the virus in transgenic Mexican lime. Plant Mol Biol 60:153–165
Fagoaga C, Rodrigo I, Conejero V, Hinarejos C, Tuset JJ, Arnau J, Pina JA, Navarro L, Peña L (2001) Increased tolerance to Phytophthora citrophthora in transgenic orange plants constitutively expressing a tomato pathogenesis related protein PR-5. Mol Breed 7:175–185
Funke T, Han H, Healy-Fried ML, Fischer M, Schonbrunn E (2006) Molecular basis for the herbicide resistance of roundup ready crops. Proc Natl Acad Sci USA 103:13010–13015
Hao G, Stover E, Gupta G (2016) Overexpression of a modified plant thionin enhances disease resistance to citrus canker and huanglongbing (HLB). Front Plant Sci 7:1078
Hao G, Zhang S, Stover E (2017) Transgenic expression of antimicrobial peptide D2A21 confers resistance to diseases incited by Pseudomonas syringae pv. tabaci and Xanthomonas citri, but not Candidatus Liberibacter asiaticus. PLoS ONE 12:e0186810
Klimazsewska K, Cheliak WM (1987) Selection for glyphosate-tolerant cell cultures in poplar. In: Proceedings of the IAE task II meeting and workshops on cell cultures and coppicing, pp17–23
Koca U, Berhow MA, Febres VJ, Champ KI, Carrillo-Mendoza O, Moore GA (2009) Decreasing unpalatable flavonoid components in Citrus: the effect of transformation construct. Phys Plant 137:101–114
Mondal S, Dutt M, Grosser J, Dewdney M (2012) Transgenic citrus expressing the antimicrobial gene Attacin E (attE) reduces the susceptibility of ‘Duncan’ grapefruit to the citrus scab caused by Elsinoë fawcettii. Eur J Plant Pathol 133:391–404
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
National Academies of Science, Engineering and Medicine (2018) A review of the citrus greening research and development efforts supported by the citrus research and development foundation: fighting a ravaging disease. The National Academies Press
Orbović V, Grosser JW (2015) Citrus transformation using juvenile tissue explants. Methods Mol Biol 1224:245–257
Orbović V, Fields J, Syvertsen J (2017) Transgenic citrus plants expressing p35 anti-apoptotic gene have altered response to abiotic stress. Hortic Environ Biotechnol 58:33–39
Padgette SR, Kolacz KH, Delannay X, Re DB, LaVallee BJ, Tinius CN, Rhodes WK, Otero YI, Barry GF, Eichholz DA, Peschke VM, Nida DL, Taylor NB, Kishore GM (1995) Development, identification, and characterization of glyphosate-tolerant soybean line. Crop Sci 35:1451–1461
Peng A, Xu L, He Y, Lei T, Yao L, Chen S, Zou X (2015) Efficient production of marker-free transgenic ‘Tarocco’ blood orange (Citrus sinensis Osbeck) with enhanced resistance to citrus canker using a Cre/loxP site-recombination system. Plant Cell Tiss Organ Cult 123:1–13
Pollegioni L, Schonbrunn E, Siehl D (2011) Molecular basis of glyphosate resistance: different approaches through protein engineering. FEBS J 278:2753–2766
Singh A, Joshi M, Devi EL (2015) Alternative to transgenesis: cisgenesis and intragenesis. In: Al-Khayri J, Jain S, Johnson D (eds) Advances in plant breeding strategies: breeding, biotechnology and molecular tools. Springer, Cham
Sinn J, Held J, Vosburg C, Klee S, Orbović V, Taylor E, Gottwald T, Stover E, Moore G, McNellis T (2020) Flowering locus T chimeric protein induces floral precocity in edible citrus. Plant Biotech J19:215–217
Song G, Prieto H, Orbović V (2019) Agrobacterium-mediated transformation of tree fruit crops: methods, progress, and challenges. Front Plant Sci 10:226
Spencer M, Mumm R, Gwyn J, inventors; DeKalb Genetics Corporation, assignee. (2000) Glyphosate resistant maize lines. U.S. patent 6040497
Steinrucken HC, Amrhein N (1980) The herbicide glyphosate is a potent inhibitor of 5-enolpyruvylshikimic acid-3-phosphate synthase. Biochem Biophys Res Commun 94:1207–1212
Wu H, Acanda Y, Canton M, Zale J (2019) Efficient biolistic transformation of immature citrus rootstocks using phosphomannose-isomerase selection. Plants 8:390. https://doi.org/10.3390/plants8100390
Yau Y, Stewart CN (2013) Less is more: strategies to remove marker genes from transgenic plants. BMC Biotech 13:36
Yu Q, Jalaludin A, Han H, Chen M, Sammons RD, Powles SB (2015) Evolution of a double amino acid substitution in the 5-enolpyruvylshikimate-3-phosphate synthase in Eleusine indica conferring high-level glyphosate resistance. Plant Physiol 167:1440–1447
Zhang X, Francis MI, Dawson WO, Graham JH, Orbović V, Triplett EW, Mou Z (2010) Overexpression of the Arabidopsis NPR1 gene in citrus increases resistance to citrus canker. Eur J Plant Pathol 128:91–100
Zou X, Jiang X, Xu L, Lei T, Peng A, He Y, Yao L, Chen S (2017) Transgenic citrus expressing synthesized cecropin B genes in the phloem exhibits decreased susceptibility to Huanglongbing. Plant Mol Biol 93:341–353
Funding
This research was supported by the USDA/NIFA Emergency Citrus Disease Research and Extension Program.
Author information
Authors and Affiliations
Contributions
ZM and VO conceived and designed research, and analyzed data. BM and XZ conducted the experiments and analyzed data. VO and ZM wrote the manuscript. ET conceived research and reviewed the manuscript. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Kan Wang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Merritt, B.A., Zhang, X., Triplett, E.W. et al. Selection of transgenic citrus plants based on glyphosate tolerance conferred by a citrus 5-enolpyruvylshikimate-3-phosphate synthase variant. Plant Cell Rep 40, 1947–1956 (2021). https://doi.org/10.1007/s00299-021-02760-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-021-02760-y