Abstract
YKL-40, also known as human cartilage glycoprotein 39, is a member of the “mammalian chitinase-like proteins” family without chitinase activity. Increased serum concentrations are associated with inflammatory processes and several types of cancer. In this study, we evaluated YKL-40 levels in serum and synovial fluid of patients with rheumatoid arthritis in comparison with the ultrasonographic findings. YKL-40 levels were measured by enzyme-linked immunosorbent assay in 25 patients with active rheumatoid arthritis and in 40 healthy subjects. B mode and power Doppler were performed to determine synovial thickening and vascularization. Serum YKL-40 level in patients was significantly higher than the concentration in healthy controls (P < 0.01). In patients with rheumatoid arthritis, the level of the glycoprotein in synovial fluid was remarkably elevated compared to the serum level (P = 0.003). The B mode and power Doppler scores correlated to YKL-40 in serum and synovial fluid (P = 0.07). Serum YKL-40 levels were related positively to serum markers of inflammation such as C-reactive protein (P = 0.004) and erythrocyte sedimentation rate (P = 0.003). This study is the first to demonstrate a relationship between YKL-40 levels and ultrasonographic examinations in Bulgarian patients with rheumatoid arthritis. The findings suggest that YKL-40 might be implicated in the pathogenesis of the disease and could indicate the level of joint inflammation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
YKL-40 is a 40-kDa glycoprotein, belonging to the family of “mammalian chitinase-like proteins” without enzymatic activity [1]. Its name is an abbreviation of the three NH2-terminal amino acids and the molecular weight. It is also known as chitinase 3-like 1 protein (CHI3L1), human cartilage-binding glycoprotein-39 (HC gp-39), and 38-kDa heparin-binding glycoprotein, chondrex.
YKL-40 is secreted by activated neutrophils and macrophages in tissues with acute or chronic inflammation, by arthritic chondrocytes [1], vascular smooth muscle cells [2], and cancer cells [3].
The normal range for YKL-40 in healthy subjects differs in various populations [4, 5]. YKL-40 was determined in 3,130 Danish people, aged 20–80 years. The mean YKL-40 level was 40 μg/l [4]. Serum YKL-40 was measured in 26 healthy people from the Turkish population, aged between 44 and 65 years. The mean values there were 114.16 ± 43.73 ng/ml [5]. It is established that the glycoprotein increases with age; thus, age-adjusted reference levels of serum or plasma YKL-40 are important when results are evaluated, along with population-based ranges.
Growing clinical evidence reveals that increased expression and secretion of YKL-40 is associated with the pathogenesis of a variety of diseases. Elevated serum levels of YKL-40 in patients with rheumatoid arthritis, osteoarthritis [5, 6], carotid atherosclerosis [7], asthma [8], and type II diabetes [9] suggest its function to be related to inflammation, extracellular remodeling, and fibrosis.
In addition, high serum levels of YKL-40 have been reported in various cancers, including breast, ovarian and colorectal, prostate carcinoma, and glioblastoma [10–14]. Several studies indicate that in rheumatoid arthritis (RA), YKL-40 might play a role as a new marker for disease activity [1, 15]. There are no data regarding a correlation between YKL-40 levels and sonographically determined joint lesions in patients with RA.
The aim of the present study was to examine YKL-40 levels in serum and synovial fluid of patients with RA and to search a correlation with the ultrasonographic findings.
Materials and methods
Patients
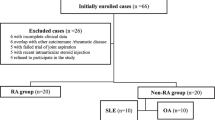
The target group consisted of 25 Bulgarian patients (13 women and 12 men) with active RA aged 24–82 years (mean age 54.43 ± 12 years). An equal number of male and female patients was included in the study due to the fact that the large joints selected for the invasive procedure were more often presented in males. Disease duration varied from several months to 22 years. Only five naïve patients were examined. All the others were treated with DMARDS and additional therapy as follows: NSAIDS (n = 10), corticosteroids (n = 5), NSAIDS and corticosteroids (n = 3), and NSAIDS and biological agents (n = 2). The patients’ clinical and biochemical characteristics are shown in Table 1.
The control group consisted of 40 age-matched healthy subjects (24 women and 14 men). Clinical and routine hematological, biochemical, and coagulation tests were completed to assess their health status. YKL-40 concentrations in serum were measured only. Despite the different number of individuals in the control and the patient group, the disparity in the gender ratio in both groups was statistically insignificant (P > 0.61) according to the performed Fischer’s exact test.
The study was approved by the university ethics committee. Informed consent was obtained from all examined individuals according to the Helsinki Declaration.
Venous blood samples were collected in the morning (4.5 mmol/l blood, Monovette, Sarstedt). Serum was obtained after centrifugation. Synovial fluid was aspirated during arthrocentesis, which was performed as a part of the diagnostic and therapeutic process. The samples were centrifuged at 2,500 rpm for 10 min. The serum and synovial fluid were stored at −70 °C before analysis.
YKL-40 detection
YKL-40 concentrations in serum and synovial fluid were determined by a two-site, sandwich-type, enzyme-linked immunosorbent assay (ELISA). The validation of the method was performed in compliance with the international standard of quality and competence of medical laboratories (BDS/EN/ISO 15189). All samples were analyzed in duplicate. The erythrocyte sedimentation rate (ESR), serum C-reactive protein (CRP), rheumatoid factor (RF), and anti-CCP antibodies were also determined.
Ultrasonographic (US) examination
A systematic clinical examination was performed at screening and baseline visits. The joints selected for assessment were 28 joints of the DAS28 (shoulder, elbow, wrist, metacarpophalangeal (MCP) 1–5, proximal interphalangeal (PIP) 1–5, knee—bilaterally).
The US examinations were performed in a darkened room. Systemic multiplanar grayscale (B mode) and power Doppler (PD) methods were carried out with commercially available real-time scanners CHISON IVIS 60 EXPERT using multifrequency linear transducers (7–13 MHz).
The PRF was set on 750 Hz. ‘Wall filter’ was fixed at the lowest possible value. Doppler frequency was adapted to the depth of the region of interest (12 MHz for small joints, 7 MHz for large joints). The EULAR–OMERACT group obtained good interobserver and intermachine reliability from the semiquantitative scoring of both greyscale synovitis and PD signal in rheumatoid joints. Semiquantitative scoring systems are used by most research groups because they are easily applicable in clinical practice, and their findings are available at the time of US examination [16]. In Bulgarian rheumatological clinical practice, these scoring systems are adopted as well.
Synovitis was defined according to the published definitions of outcome measures in RA clinical trials (OMERACT). A B mode and a PD examination were recorded for each joint. B-mode synovitis scoring was evaluated using a four-grade scale from 0 to 3 with the following subjective definitions for each category: grade 0, absence of synovial thickening; grade 1, mild synovial thickening; grade 2, moderate synovial thickening; grade 3, marked synovial thickening [17, 18].
Synovial vascularization was also evaluated using a four-grade scale from 0 to 3 with the following definitions for each category: grade 0, absence of signal, no intra-articular flow; grade 1, mild, one or two vessels signal (including one confluent vessel) for small joints and two or three signals for large joints (including two confluent vessels); grade 2, moderate confluent vessels (>grade 1) and <50 % of normal area; grade 3, marked vessels signal in more than half of the synovial area [19]. The scorer was blinded to the patient’s clinical status.
Statistical analysis
The normality of the continuous variables was examined by the Kolmogorov–Smirnov test and comparisons were completed using the t test. Pearson’s correlation coefficient was calculated to evaluate correlation between studied variables. Statistical analysis was carried out with the SPSS v 17.0 software. The level of statistical significance of the null hypothesis was P < 0.05.
Results
Concentrations of YKL-40 in serum and synovial fluid were determined in 25 Bulgarian patients with active RA and compared to 40 age-matched healthy controls. There were no significant differences in YKL-40 levels between treated and naïve patients.
The mean serum value of YKL-40 in healthy subjects was 84,19 ± 11.39 ng/ml, while the mean serum concentration of YKL-40 in RA patients was 246,17 ± 209.63 ng/ml. Serum YKL-40 level in RA patients is significantly higher compared to that of healthy controls (Fig. 1). The level of the glycoprotein in synovial fluid was 509.78 ± 375.56 ng/ml and it was significantly higher compared to the serum level (P = 0.003).
The mean of the standard indicators of disease activity was elevated toward the range (shown in Table 2). A positive correlation between serum YKL-40 concentration and conventional markers of biochemical assessment of disease activity in RA as ESR (r = 0.446, P = 0.003) and CRP (r = 0.582, P = 0.004) was determined.
A strong correlation between B mode and DAS28 was established (P = 0.006). The B-mode assessment is a method for direct visualization of synovial hyperplasia. We used the PD examination to evaluate the vascularization of synovia and the degree of inflammation. We found an association between serum and synovial YKL-40 levels and synovial thickening and vascularization in RA patients (Fig. 2).
Correlation between YKL-40 concentrations in serum and synovial fluid and sonographic parameters. a Correlation between serum YKL-40 levels and B mode. b Correlation between serum YKL-40 levels and PD. c Correlation between synovial fluid YKL-40 levels and B mode. d Correlation between synovial fluid YKL-40 levels and PD. e Correlation between serum YKL-40 levels and ESR. f Correlation between serum YKL-40 levels and CRP
Ours research is the first to reveal a correlation between serum and synovial YKL-40 levels and the sonographic examination (P = 0.07).
Discussion
RA is an autoimmune disease in which chronic inflammation is mediated by autoantigen activation of T cells. These immune cells induce an inflammatory process which results in synovial proliferation of cartilage and bone [20].
Serum CRP and ESR are suitable biochemical markers for monitoring disease activity in RA. An important problem arises from the fact that normal values are sometimes found in patients with clinically active disease [21]. Radiological progression, including bone erosion and loss of joint space, is one of the important characteristics of RA. However, the development of bone erosion can be found independently of biochemical parameters and clinical symptoms [22]. Therefore, a new biomarker of disease activity is needed.
YKL-40 is discussed as a possible novel biomarker. Our study shows that serum and synovial fluid YKL-40 levels are significantly higher in Bulgarian patients with RA compared to age-matched healthy subjects (P < 0.001). The normal range for circulating YKL-40 in healthy individuals is not established yet. Johansen [1] suggested that changes in serum YKL-40 concentration >20 % should be indicative of elevated serum range.
We determined 25 % higher concentration of YKL-40 in synovial fluid compared to serum levels in patients with RA. A positive correlation between serum and synovial fluid concentrations of YKL-40 was detected. Other scientists have observed a tenfold increased concentration of YKL-40 in synovial fluid compared to serum concentration in RA patients. A strong relationship between glycoprotein levels in serum and synovial fluid was demonstrated in different populations with RA [23, 24]. Other research groups also suggested that YKL-40 in synovial fluid and serum might reflect cartilage degradation and synovial inflammation in RA and knee osteoarthritis [1, 25].
We found a significant correlation between serum YKL-40 and standard biochemical markers of inflammation such as CRP and ESR in patients with RA (P < 0.001). Many pathological conditions influence these parameters. ESR rate depends on the aggregation and damage of erythrocytes. CRP is secreted by hepatocytes in response to high levels of proinflammatory cytokines. These laboratory tests are nonspecific indicators of synovial inflammation. It is suggested that CRP and YKL-40 reveal two different aspects of the inflammatory process in joint diseases [1]. Reliable biomarkers of joint inflammation and destruction in RA patients are proteins expressed and released by cells in synovial fluid and serum. We suppose that YKL-40 could provide a different and a more local view of the inflammatory process than conventional biomarkers such as CRP and ESR. The explanation may lay in the fact that YKL-40 is secreted by activated macrophages in the inflammed synovial membrane [26], or released by exocytosis from specific granules of neutrophils [27] and by articular chondrocytes [28]. Despite the treatment, all our patients presented with active RA, which indicates that they could be regarded as non-responders to the applied therapy. A similar phenomenon was recorded by Knudsen et al. [25] who observed unchanged YKL-40 serum levels in non-responders, while responders showed a significant decrease in YKL-40 and other biomarkers.
Serum YKL-40 is discussed as a potential biomarker for the risk of progression of joint damage in patients with RA. Some authors regard it as a serious parameter in disease diagnosis and monitoring [6, 29], and others disregard its importance in disease activity [30].
Most published studies used radiographic examination to assess the disease process in joints. Ours is the first to search a correlation between YKL-40 levels in RA patients and US characteristics. The PD sonography and B-mode ultrasound diagnosis are highly sensitive techniques for the differentiation of synovitis and joint destruction and for the quantification of the degree of inflammation. According to Weidekam et al. [31], sonography detects 20 % more abnormalities than does radiography and provides simple grading of disease activity. PD sonography is a new technique that improves the sensitivity to detect flow from small vessels and low velocity flow at the microvascular level. In our investigation, we used sonography to support the diagnosis of RA as a technique more sensitive for detecting joint inflammation than conventional radiography. A positive correlation between YKL-40 concentrations in serum and synovial fluid and sonographic parameters was demonstrated (P < 0.07). The increased concentration in serum and synovial fluid of the glycoprotein can indicate destructive changes in the cartilage. The significant relation between results from PD examination and circulating YKL-40 may lay in the fact that YKL-40 facilitates cell attachment and migration of vascular endothelial cells, which could indicate that the protein participates in angiogenesis [1]. It is well known that chronic synovitis is generally accompanied by angiogenesis. Francescone et al. [32] demonstrated that YKL-40 induced VEGF expression in the U87 glioblastoma cell line, and suggested that both molecules synergistically promoted endothelial cell angiogenesis. A study by Zivanoviz et al. [6] also suggested that YKL-40 could be used as a biomarker of joint damage in knee osteoarthritis.
In conclusion, our study is the first to show significantly elevated serum YKL-40 level in Bulgarian patients with active RA compared to healthy controls. We prove that the high YKL-40 concentration in synovial fluid and serum correlates with sonographic parameters and suppose that it might have a significant value for prediction of joint inflammation.
References
Johansen JS (2006) Studies on serum YKL-40 as a biomarker in diseases with inflammation, tissue remodelling, fibroses and cancer. Dan Med Bull 53:172–209
Rehli M, Krause SW, Andreesen R (1997) Molecular characterization of the gene for human cartilage gp-39 (CHI3L1), a member of the chitinase protein family and marker for late stages of macrophage differentiation. Genomics 43:221–225
Roslind A, Johansen JS (2009) YKL-40: a novel marker shared by chronic inflammation and oncogenic transformation. Methods Mol Biol 511:159–184
Bojesen SE, Johansen JS, Nostegaard BG (2011) Plasma YKL-40 levels in healthy subjects from the general population. Clin Chim Acta 412:709–712
Güngen G, Ardic F, Fιndıkoğlu G, Rota S (2011) The effect of mud pack therapy on serum YKL-40 and hsCRP levels in patients with knee osteoarthritis. Rheumatol Int 31:1–10
Zyvanovic S, Rackov LP, Vojvodic D, Vucetic D (2009) Human cartilage glycoprotein 39-biomarker of joint damage in knee osteoarthritis. Int Orthop 33:1165–1170
Michelsen AE, Rathcke CN, Skjelland M, Holm S, Ranheim T, Krohg-Sørensen K, Klingvall MF, Brosstad F, Oie E, Vestergaard H, Aukrust P, Halvorsen B (2010) Increased YKL-40 expression in patients with carotid atherosclerosis. Atherosclerosis 211:589–595
Letuve S, Kozhich A, Arouche N, Grandsaigne M, Reed J, Dombret MC, Kiener PA, Aubier M, Coyle AJ, Pretolani M (2008) YKL-40 is elevated in patients with chronic obstructive pulmonary disease and activates alveolar macrophages. J Immunol 181:5167–5173
Rondbjerg A, Omerovic E, Vestergaard H (2011) YKL-40 levels are independently associated with albuminuria in type 2. Cardiovasc Diabetol 10:54. doi:10.1186/1475-2840-10-54
Cintin C, Johansen JS, Christensen IJ, Price PA, Sørensen S, Nielsen HJ (2002) High serum YKL-40 level after surgery for colorectal carcinoma is related to short survival. Cancer 95:267–274
Yamac D, Ozturk B, Coskun U, Tekin E, Sancak B, Yildiz R, Atalay C (2008) Serum YKL-40 levels as a prognostic factor in patients with locally advanced breast cancer. Adv Ther 25:801–809. doi:10.1007/s12325-008-0082-2
Hogdall E, Johansen JS (2009) YKL-40 tissue expression and plasma levels in patients with ovarian cancer. Biomed Central Cancer 9:8. doi:10.1186/1471-2407-9-8
Kucur M, Isman FK, Balci C, Onal B, Hacibekiroglu M, Ozkan F, Ozkan A (2008) Serum YKL-40 levels and chitotriosidase activity as potential biomarkers in primary prostate cancer and benign prostatic hyperplasia. Urol Oncol 26:47–52
Tanwar MK, Gilbert MR, Holland EC (2002) Gene expression microarray analysis reveals YKL-40 to be a potential serum marker for malignant character in human glioma. Cancer Res 62:4364–4368
Peltomaa R, Paimela L, Harvey S, Helve T, Leirisalo-Repo M (2001) Increased level of YKL-40 in sera from patients with early rheumatoid arthritis: a new biomarker for disease activity. Rheumatol Int 20:192–196
Mandl P, Naredo E, Wakefield RG, Conaghan PG, D’agostino MA, OMERACT Ultrasound Task Force (2011) A systematic literature review analysis of ultrasound joint count and scoring systems to assess synovitis in rheumatoid arthritis according to the OMERACT filter. J Rheumatol 38:2055–2062
Schmidt WA, Schmidt H, Schicke B, Cromlica-Ichle E (2004) Standard reference values for musculoskeletal ultrasonography. Ann Rheum Dis 63:988–994
Wakefield RJ, Gibbon WW, Conaghan PG, O’Connor P, McGonagle D, Pease C, Green MJ, Veale DJ, Isaacs JD, Emery P (2000) The value of sonography in the detection of bone erosions in patients with rheumatoid arthritis: a comparison with conventional radiography. Arthritis Rheum 43:2762–2770
Naredo E, Bonilla G, Gamero F (2005) Assessment of inflammatory activity in rheumatoid arthritis: a comparative study of clinical avaluation with grey scale and power Doppler ultrasonography. Ann Rheum Dis 64:375–381
Fox DA (1997) The role of T cells in the immunopathogenesis of rheumatoid arthritis. New perspectives. Arthritis Rheum 40:598–609
Emery P, Gough A, Salmon M, Devlin J (1993) Medical management of rheumatoid arthritis. BMJ 307:425–428
Kirwan JR, Arthritis and Rheumatism Council Low-Dose Glucocorticoid Study Group (1995) The effect of glucocorticoids on joint destruction in rheumatoid arthritis. N Engl J Med 333:142–147
Johansen JS, Jensen HS, Price PA (1993) A new biochemical marker for joint injury. Analysis of YKL-40 in serum and synovial fluid. Br J Rheumatol 32:949–955
Volck B, Johansen JS, Stoltenberg M, Garbarsch C, Price PA, Østergaard M, Østergaard K, Løvgreen-Nielsen P, Sonne-Holm S, Lorenzen I (2001) Studies on YKL-40 in knee joints of patients with rheumatoid arthritis and osteoarthritis. Involvement of YKL-40 in the joint pathology. Osteoarthr Cartil 9:203–214
Knudsen LS, Hetland ML, Johansen JS, Skjodt H, Peters ND, Colic A, Grau K, Nielsen NJ, Ostergaard M (2009) Changes in plasma IL-6, plasma VEGF and serum YKL-40 during treatment with etanercept and methotrexate or etanercept alone in patients with active rheumatoid arthritis despite methotrexate therapy. Biomarker Insights 4:91–95
Kirkpatrick RB, Emery JG, Connor JR, Dodds R, Lysko PG, Rosenberg M (1997) Induction and expression of human cartilage glycoprotein 39 in rheumatoid inflammatory and peripheral blood monocyte-derived macrophages. Exp Cell Res 237:46–54
Volck B, Price PA, Johansen JS, Sørensen O, Benfield T, Calafat J, Nielsen HJ, Borregaard N (1998) YKL-40, a mammalian member of the bacterial chitinase family, is a matrix protein of specific granules in human neutrophils. Proc Assoc Am Phys 110:351–360
Hakala BE, White C, Recklies AD (1993) Human cartilage gp-39, a major secretory product of articular chondrocytes and synovial cells, is a mammalian member of a chitinase protein family. J Biol Chem 268:25803–25810
Takahashi M, Naito K, Abe M, Sawada T, Nagano A (2004) Relationship between radiographic grading of osteoarthritis and the biochemical markers for arthritis in knee osteoarthritis. Arthritis Res Ther 6:208–212
Syversen SW, Goll GL, Heijde D, Landeve R, Lie BA, Odegard S, Uhlig T, Gaarder PI, Kvien TK (2009) Prediction of radiographic progression in rheumatoid arthritis and the role of antibodies against mutated citrullinated vimentin: results from a 10-year prospective study. Ann Rheum Dis 69:345–351. doi:10.1136/ard.2009.113092
Weidekamm C, Koller M, Weber M, Kainberger F (2003) Diagnostic value of high-resolution B-mode and Doppler sonography for imaging of hand and finger joints in rheumatoid arthritis. Arthritis Rheum 48:325–333
Francescone RA, Scully S, Faibish M, Taylor SL, Oh D, Moral L, Yan W, Bentley B, Shao R (2011) Role of YKL-40 in the angiogenesis, radioresistance, and progression of glioblastoma. J Biol Chem 286:15332–15343
Acknowledgments
The study was supported by grants no. 1/2009 and 1/2010 from Medical University-Plovdiv.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kazakova, M., Batalov, A., Deneva, T. et al. Relationship between sonographic parameters and YKL-40 levels in rheumatoid arthritis. Rheumatol Int 33, 341–346 (2013). https://doi.org/10.1007/s00296-012-2387-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-012-2387-3