Abstract
Systemic sclerosis (SSc) is a rare connective tissue disease characterized by vascular, immune and fibrotic abnormalities in the skin and in many internal organs. New biomarkers with predictive value associated with target organ involvement are needed. The up-regulation of IL-6 production is associated with the disease activity and in the development of cardiopulmonary manifestations in SSc patients. The protein YKL-40 is a promising and intensively investigated biomarker related to inflammatory and tumor diseases. The objective of the study was to investigate how serum levels of YKL-40 and IL-6 correlate with articular and periarticular involvement in patients with SSc assessed by high-frequency ultrasonography. 59 SSc patients (56 women, 3 men) and 23 age-matched healthy subjects (21 women and 2 men) were investigated for serum YKL-40 and IL-6 (by ELISA). All the patients and healthy controls underwent clinically and high-frequency ultrasound assessment of articular and periarticular structures. Joint involvement was scored according to the new US10SSc score. Clinical data about the SSc patients showed significantly higher mRSS in the dcSSc patients (p = 0.015). Clinical synovitis was diagnosed in 16.9% of all patients: 22.5% of the dcSSc group and 10.7% of the lcSSc group (p = 0.306). On the other hand, US synovitis was detected in a higher percentage: 44% of all SSc patients; 54.8% of the dcSSc group and 32% of the lcSSc patients (p = 0.116). Clinical tenosynovitis was established in 6.7% of all patients: 9.7% of the dcSSc group and 3.5% of the lcSSc group (p = 0.614). US tenosynovitis was detected at a higher rate: 27% of all patients; 32.25% of the dcSSc group and 21.4% of the lcSSc group (p = 0.393). Serum level of YKL-40 was significantly higher in SSc patients (115.62 ng/ml ± 89.51, median 86.76) compared to the healthy controls (46.28 ng/ml ± 18.91, median 44.02), p < 0.001. IL-6 level was also significantly higher in the patient group (27.60 ± 48.80 pg/ml; median 8.32) vs. the healthy controls (5.79 ± 2.46 pg/ml, median 5.52). In the patient subgroups, YKL-40 and IL-6 levels were significantly elevated in dcSSc compared to lcSSc patients: YKL-40 dcSSc (159.52 ng/ml ± 102.81; median 136.20 ng/ml) vs. lcSSc patients (89.31 ng/ml ± 50.36; median 68.03 ng/ml;), p < 0.001; IL-6 dcSSc patients (49.64 pg/ml ± 46.37; median 16.36 pg/ml) vs. lcSSc patients (13.22 pg/ml ± 8.95; median 8.65 pg/ml), p = 0.048. A statistically significant correlation of high magnitude (rs = 0.884, p < 0.001) was observed between YKL-40 and the ultrasound 10 Systemic sclerosis score (US10SSc) and between IL-6 and the US10SSc score (rs = 0.808, p < 0.001). Serum YKL-40 and IL-6 in combination with US may have a potential role in defining disease activity and stratification, predicting organ involvement, and in the prognosis of SSc.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Systemic sclerosis (SSc) is a chronic autoimmune rheumatic disease characterized by vascular injury and tissue fibrosis [1]. It is a rare disease with still unclear etiology and a prevalence of between 100 and 300 cases per million of the population. Involvement of internal organs has defined the high morbidity and is associated with poor quality of life and high mortality [2,3,4]. A difficulty in treatment is caused by clinical diversity and heterogeneity and is related to specific organ involvement [5, 6].
SSc is a disease with a clinically heterogeneity and its treatment is a real challenge. According to the updated EULAR recommendations (2017) for the treatment of SSc, management of the disease such as early diagnosis of internal organ involvement and identification of patients with high risk of new organ damage are extremely important [6]. These recommendations include new treatments according to specific SSc-related organ involvement [6].
Involvement of the musculoskeletal system is reported to be observed in about 10–60% the patients with SSc at the time of diagnosis and is often the onset of the disease [7, 8]. Patients with this disease are thought to have synovial proliferation, joint effusion, tenosynovitis, calcifications, bursitis, carpal tunnel syndrome (CTS), acroosteolysis, reduction of the distance skin surface—distal phalanx, flexion contractions, digital ulcerations, fibrosing tendinitis /TFRs/ [9, 10]. Synovitis is one of the strongest predictors of skin progression and further occurrence of DUs, which directly reflects disease progression, according to the EUSTAR prospective study [11, 12]. The described pathological findings cannot always be confirmed by physical examination due to alterations in the overlying skin, which is thickened, compacted, and with low elasticity. In such cases, searching for objective methods of early diagnosis is necessary [13, 14]. Due to high-frequency ultrasonography—B-Mode (Gray scale) and Power Doppler technique, it is possible to evaluate joint, periarticular and soft tissue involvement in SSc [15,16,17].
In the context of personalized medicine, new biomarkers are sought as predictors of different organ involvement in SSc. YKL-40 (chitinase-3-like protein) is a member of the 18 glycosyl hydrolases, a mammalian chitinase family. Its biological role is still not fully understood and remains to be clarified [18]. However, it is reported that the protein takes part in many important biological processes, such as proliferation, inflammation, and angiogenesis [19, 20]. YKL-40 is expressed by mature macrophages, chondrocytes, and cancer cells. It is proposed to be involved in tissue remodeling and fibrosis [18]. There are few studies showing elevated serum YKL-40 in SSc patients [21] and none of them has focused on the gene expression of YKL-40.
Elevated serum YKL-40 is found in patients with a variety of diseases characterized by inflammation, remodeling of the extracellular matrix, and development of fibrosis, such as rheumatoid arthritis, osteoarthritis, giant cell arteritis, pneumonia, liver fibrosis, and different types of solid tumors [20]. Serum YKL-40 levels vary according to disease activity. Elevated serum YKL-40 is associated with a poor prognosis.
The link between YKL-40 and SSc has been investigated by a number of authors and an association with pulmonary involvement has been observed [21]. In previous studies related to rheumatoid arthritis, psoriatic arthritis, gout, and activated osteoarthritis, we found that YKL-40 was associated with joint involvement in these diseases [19, 22]. In SSc, dermal fibroblasts are one of the main effector cell types involved in the development of fibrotic lesions. Their biological activity is regulated by a variety of inflammatory cytokines and growth factors. YKL-40 had been implicated in other pathologies leading to excessive fibrosis [23].
Interleukin 6 (IL-6) is an interleukin that acts as both a pro- and anti-inflammatory cytokine and regulates cellular proliferation, activation and differentiation of numerous cell types. IL-6 is implicated in pathological processes including SSc [24]. The role of IL-6 is well established and there is substantial evidence that its up-regulation is associated with clinical subsets and organ involvement [25]. In comparison, little is known about YKL-40 participation in the pathogenesis of SSc.
Therefore, our aim was to assess serum levels of YKL-40 and IL-6 in SSc and to investigate their relation to ultrasound articular and periarticular involvement detected by US.
Objective
To investigate how serum levels of YKL-40 and IL-6 correlate with articular and periarticular involvement in patients with systemic sclerosis (SSc) assessed with high-frequency ultrasound examination.
Materials and methods
Patients and samples
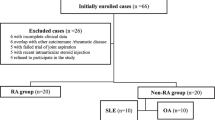
Between May 2018 and January 2019, 59 consecutive patients with SSc (31 dcSSc and 28 lcSSC) from the Rheumatology Clinic of University Hospital “Kaspela” were included in a prospective study. The study was approved by the local ethics committee (protocol № 3/31.05.2018) and all patients provided written informed consent. The diagnosis of SSc was based on the 2013 ACR/EULAR classification criteria for SSc [26]. All patients underwent clinical examination, mRSS assessment, and capillaroscopy. In the same day, US examination, blood tests as erythrocyte sedimentation rate [ESR] and C-reactive proteins [CRP] were performed. Patients were treated by NSAIDs, DMARDs, and biologic agents. Three of them who have ILD are on RTX treatment, two patients on MMF, five patients on Prednisolone, and other patients on MTX; four patients take NSAIDs. The following data were recorded for each patient: age, gender, date of diagnosis, symptoms duration, past and present therapy. Exclusion criteria for patients with SSc were: rheumatological disorders other than SSc including rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), mixed connective tissue disease (MCTD), polymyositis/dermatomyositis (PM/DM), Sjögren syndrome (SS), and eosinophilia myalgia syndrome (EMS).
Control group
The control group consisted of 23 age-matched healthy subjects (21 women and 2 men). Clinical and routine hematological and biochemical tests were completed to assess their health status. The inclusion criteria for the control group of healthy individuals were: age over 18 years; signed informed consent for participation; and lack of malignancies, inflammatory joint and autoimmune diseases.
Physical examination
All patients were examined physically for swollen and tender joints, TFRs.
Measurement of YKL-40, IL-6
Concentrations of YKL-40 and IL-6 in serum were measured by ELISA, using commercial kits from Quidel, SA (YKL-40) and R&D Systems, Minneapolis, USA (IL-6). All samples were examined in duplicate.
The US assessment
The ultrasound (US) assessment was performed using Esaote MyLab 7 and GE E9 machines with multifrequency linear probe (7–15 MHz) in a longitudinal and transversal scan. The B-mode (gray scale) and Power Doppler (PD) US settings were adjusted to optimize image resolution and sensitivity to detect the flow. Two ultrasonographers scanned all patients in a blind, independent, and consecutive manner. The new US10SSc score was applied for ultrasound assessment of SSc patients.
The new modified US10SSc score included the following joints of both hands: wrist, second and third metacarpophalangeal and proximal interphalangeal joints. Synovitis and synovial/tenosynovial vascularity were scored semi-quantitatively (grade 0–3) by gray-scale (GS) and PD ultrasound. Tenosynovitis was scored for presence (0 or 1). The scoring ranges were as follows: 0–30 for GS synovitis; 0–46 for PD synovitis; 0–14 for GS tenosynovitis/paratenonitis; and 0–42 for PD tenosynovitis/paratenonitis. Unlike previously known scoring systems, in this one erosions and joint cartilage are not included. The wrist, MCP2 and MCP3, and PIP2 and PIP3 joints of the both sides were sonographically examined in a standardized manner according to the European League Against Rheumatism (EULAR) and OMERACT guidelines [27].
Based on these results, a new modified 10‐joint US score (US10SSc score) was performed, including the sum of synovitis scores in the GSUS (0–30) and PDUS (0–46) modes, tenosynovitis/paratenonitis in the GSUS (0–14) and PDUS (0–42) modes.
Statistical analysis
Statistical analysis was performed using IBM SPSS V.25.0 package (Statistical Package for the Social Sciences, Chicago, IL, USA) (2017) and Minitab V.18.1 (2017). We examined all continuously measured data for normality through the Kolmogorov–Smirnov test. Age was normally distributed in all groups and subgroups (p > 0.05). The rest of the continuous data showed violations of normality (p < 0.05) and is presented as median, minimal and maximum values. Dichotomous data are summarized in frequencies and percentages. An independent samples t test was used for two-group comparisons on age and the Mann–Whitney U test for comparisons on the non-normally distributed continuously measured data. Associations between dichotomous variables were examined through χ2 test, and Fisher’s exact test was used for the comparison of proportions. Spearman’s rank-order correlation was employed to examine the relationship between non-normally distributed variables. Results were interpreted as statistically significant at p < 0.05.
Results
Background data
The demographic and clinical data about the SSc patients and the group of healthy controls are summarized in Table 1. There was no significant age difference between the healthy controls and the SSc patients (p = 0.68) or between the dcSSc and lcSSc patients (p = 0.349). In both samples and subsamples, the females were the dominant sex: 91% of the healthy controls and 96% of the SSc patients (p = 0.616); 93.5% of the patients with dcSSc and 94.5% of those with lcSSc (p = 0.870).
In the SSc patients, the median mRSSc was 14, with a significantly higher mRSS in the dcSSc patients (median 18) in comparison to the lcSSc group (median 12), p = 0.015. Interstitial lung disease (ILD) detected by high resolution computed tomography (HRCT) was observed in 5% of all patients: 9.6% of the dcSSc group and in none of the lcSSc group (p = 0.239). Clinical synovitis was diagnosed in 16.9% of all patients: 22.5% of the dcSSc group and 10.7% of the lcSSc group (p = 0.306). On the other hand, US synovitis was detected at a higher percentage: 44% of all SSc patients; 54.8% of the dcSSc group and 32% of the lcSSc patients (p = 0.116). Clinical tenosynovitis was established in 6.7% of all patients: 9.7% of the dcSSc group and 3.5% of the lcSSc group (p = 0.614). US tenosynovitis was detected at a higher rate: 27% of all patients; 32.25% of the dcSSc group and 21.4% of the lcSSc group (p = 0.393).
Serum YKL-40 and IL-6 in SSc patients vs. healthy controls and in dsSSc vs. lcSSc patients
Serum levels of YKL-40 and IL-6 were compared between the SSc patients and the healthy controls, and between the dcSSc and lcSSc patients through the Mann–Whitney’s U test (Table 2).
In the SSc group, we found a significantly higher level of YKL-40 (median 86.76 ng/ml) compared to the healthy controls (median 44.02 ng/ml;), p < 0.001. In the patient subgroups, a significantly higher YKL-40 level was observed in the dcSSc patients (median 136.20 ng/ml) compared to the patients with lcSSc (median 68.03 ng/ml), p < 0.001.
A similar trend was observed in relation to IL-6 levels. The SSc patients had a significantly higher IL-6 concentration (median 8.82 pg/ml) than the healthy controls (median 5.52 pg/ml), p < 0.001. IL-6 concentration in the dcSSc patients (median 16.36 pg/ml) was significantly elevated compared to the lcSSc patients (median 8.65 pg/ml), p = 0.048.
Association of YKL-40 and IL-6 with US10SSc score
As a whole, the SSc patients had a median US10SSc score of 6. In the dcSSc group, we found a significantly higher US10SSc score (median 9) compared to the lcSSc patients (median 5), p = 0.002. (Table 2).
Spearman’s correlation analysis was used to examine the relation between YKL-40 and US10SSc score and IL-6 and US10SSc score. A statistically significant correlation of high magnitude was found between YKL-40 and US10SSc score (rs = 0.884, p < 0.001). A high positive correlation was also observed between IL-6 and US10SSc score (rs = 0.808, p < 0.001) (Fig. 1).
Discussion
Many studies have evaluated biomarker levels in relation to joint involvement in SSc patients. However, this is the first study to examine the levels of YKL-40 and IL-6 in SSc patients stratified in articular and periarticular involvement by high-frequency ultrasonography. The role of ultrasound in the assessment of SSc patients has been demonstrated in other our publications, both for the evaluation of joint, skin and pulmonary involvement [28,29,30,31,32]. A significant and novel finding of this study is that both YKL-40 and IL-6 levels correlated with US joint involvement. Ultrasonography is a sensitive tool for detecting synovitis and tenosynovitis in patients with SSc. The specifically designed US10SSc score for this study is targeted to test patients with systemic sclerosis as it focuses on specific articular and periarticular changes in SSc. In our study, YKL-40 and IL-6 levels correlated with US10SSc score, providing strong evidence that YKL-40 and IL-6 are associated with the joint involvement. Several previous clinical studies identified elevated YKL-40 and IL-6 in patients with SSc. The authors associated the increased values of these biomarkers with pulmonary and cutaneous worsening of the disease. Korthagen et al. found that serum and BALF YKL-40 levels are predictors of survival in idiopathic pulmonary fibrosis (IPF) [33]. YKL-40 is increased in sera from SSc patients and correlates with obstructive ventilatory pattern, reduced DLCO and poor prognosis. The presence of single gene polymorphisms (SNPs) in the gene encoding YKL-40 (CHI3L1-329 and -321) is associated with the magnitude of serum levels of this marker [21]. De Lauretis et al. assessed the prognostic value of serum IL-6 in a large cohort of 212 patients with SSc-ILD as an independent predictor of DLCO decline in both IPF and SSc-ILD [34]. Affandi et al. described in their study that IL-6 is increased in the sera of SSc patients and has been associated with multiple organ involvement including skin, pulmonary fibrosis and increased mortality [35]. They concluded that this finding correlates with disease activity and extensive skin fibrosis. The link between increased serum level of IL-6 and skin worsening was also documented from De Lauretis et al. [34]. Ema Ball et al. [36] elucidated the role of specific cytokines in SLE arthritis stratified in terms of joint disease by US. They found that IL-6 levels correlated both with clinical and US measures of arthritis disease activity.
Montagna GL et al. in 2003 established for the first time the link between elevated serum YKL-40 in SSc patients and joint involvement [37]. But they do not find significant differences in the different SSc subgroups. There are other studies focused on YKL-40 in diseases, such as rheumatoid arthritis and osteoarthritis showing that its concentrations in both serum and synovial fluid reflected active inflammation and intensive angiogenesis in synovial membrane [38]. We reported significant relationship within CRP and ESR but in patients with RA [19]. These parameters are conventional markers that provide information about disease activity, but are not predictive separately to be used for treatment decision making [39]. High serum IL-6 levels correlate with coronary instability and have been shown to be a prognostic marker for septic shock [40]. In SSc patients, an association of IL-6 with interstitial lung disease is observed [41]. There are no studies focused on both markers (YKL-40 and IL-6) and their correlation with joint involvement. Although YKL-40 and IL-6 appear to lack sufficient specificity to be involved in diagnose of SSc, our study suggests that they may have utility as biomarkers capable of predicting other clinical manifestations of SSc. We found that the correlation between YKL-40 and articular involvement was predominant in patients with dcSSc. Our results show that the increased values of these two biomarkers are also associated with joint involvement, which is objectively proven by US—the most contemporary instrumental tool for joint assessment.
Limitations
The present study has some limitations, which have to be mentioned. The first one concerns the application of the US10SSc score which, although specially adapted to SSc patients, needs further validation. Another limitation is the absence of data for other markers of joint involvement in SSc such as ICAM-1, IL12/IL23, TNF-α, which we intend to examine in the future. Furthermore, all synovitis, joint effusion and/or tenosynovitis seen on ultrasound may not be clinically relevant.
Conclusion
The importance of our study is that, using an objective instrumental tool (high-frequency ultrasonography), we demonstrate the correlation of serum levels of YKL-40 and IL-6 with articular and periarticular involvement in SSc patients. Serum biomarkers in combination with US may have a potential role in defining disease activity and stratification, predicting organ involvement, and in the prognosis of SSc.
References
Varga J, Abraham D (2007) Systemic sclerosis: a prototypic multisystem fibrotic disorder. J Clin Invest 117:557–567. https://doi.org/10.1172/JCI31139
Campbell PM, LeRoy EC (1975) Pathogenesis of systemic sclerosis: a vascular hypothesis. Semin Arthritis Rheum 4:351–368. https://doi.org/10.1016/0049-0172(75)90017-7
Wigley FM (2002) Clinical practice. Raynaud’s phenomenon. N Engl J Med 347:1001–1008. https://doi.org/10.1056/NEJMcp013013
Avouac J, Fransen J, Walker UA, Riccieri V, Smith V, Muller C et al (2011) Preliminary criteria for the very early diagnosis of systemic sclerosis: results of a Delphi Consensus Study from EULAR Scleroderma Trials and Research Group. Ann Rheum Dis 70:476–481. https://doi.org/10.1136/ard.2010.136929
Nihtyanova SI, Ong VH, Denton CP (2014) Current management strategies for systemic sclerosis. Clin Exp Rheumatol 32:156–164
Kowal-Bielecka O, Fransen J, Avouac J, Becker M, Kulak A, Allanore Y, Distler O, Clements P, Cutolo M, Czirjak L, Damjanov N, del Galdo F, Denton CP, Distler JHW, Foeldvari I, Figelstone K, Frerix M, Furst DE, Guiducci S, Hunzelmann N, Khanna D, Matucci-Cerinic M, Herrick AL, van den Hoogen F, van Laar JM, Riemekasten G, Silver R, Smith V, Sulli A, Tarner I, Tyndall A, Welling J, Wigley F, Valentin G, Walker UA, Zulian F, Müller-Ladner U (2017) Update of EULAR recommendations for the treatment of systemic sclerosis. Ann Rheum Dis 76:1327–1339. https://doi.org/10.1136/annrheumdis-2016-209909
Avouac J, Walker U, Tyndall A, Kahan A, Matucci-Cerinic M, Allanore Y, EUSTAR, Miniati I, Muller A, Iannone F, Distler O, Becvar R, Sierakowsky S, Kowal-Bielecka O, Coelho P, Cabane J, Cutolo M, Shoenfeld Y, Valentini G, Rovensky J, Riemekasten G, Vlachoyiannopoulos P, Caporali R, Jiri S, Inanc M, Zimmermann Gorska I, Carreira P, Novak S, Czirjak L, Oliveira Ramos F, Jendro M, Chizzolini C, Kucharz EJ, Richter J, Cozzi F, Rozman B, Mallia CM, Gabrielli A, Farge D, Kiener HP, Schöffel D, Airo P, Wollheim F, Martinovic D, Trotta F, Jablonska S, Reich K, Bombardieri S, Siakka P, Pellerito R, Bambara LM, Morovic-Vergles J, Denton C, Hinrichs R, Van den Hoogen F, Damjanov N, Kötter I, Ortiz V, Heitmann S, Krasowska D, Seidel M, Hasler P, Van Laar JM, Kaltwasser JP, Foeldvari I, Juan Mas A, Bajocchi G, Wislowska M, Pereira Da Silva JA, Jacobsen S, Worm M, Graniger W, Kuhn A, Stankovic A, Cossutta R, Majdan M, Damjanovska Rajcevska L, Tikly M, Nasonov EL, Steinbrink K, Herrick A, Müller-Ladner U, Dinc A, Scorza R, Sondergaard K, Indiveri F, Nielsen H, Szekanecz Z, Silver RM, Antivalle M, Espinosa IB, de la Pena García, Lefebvre P, Midtvedt O, Launay D, Valesini F, Tuvik P, Ionescu RM, Del Papa N, Pinto S, Wigley F, Mihai C, Sinziana Capranu M, Sunderkötter C, Jun JB, Alhasani S, Distler JH, Ton E, Soukup T, Seibold J, Zeni S, Nash P, Mouthon L, De Keyser F, Duruöz MT, Cantatore FP, Strauss G, von Mülhen CA, Pozzi MR, Eyerich K, Szechinski J, Keiserman M, Houssiau FA, Román-Ivorra JA, Krummel-Lorenz B, Aringer M, Westhovens R, Bellisai F, Mayer M, Stoeckl F, Uprus M, Volpe A, Buslau M, Yavuz S, Granel B, Valderílio Feijó A, Del Galdo F, Popa S, Zenone T, Ricardo Machado X, Pileckyte M, Stebbings S, Mathieu A, Tulli A, Tourinho T, Souza R, Acayaba de Toledo R, Stamp L, Solanki K, Veale D, Francisco Marques Neto J, Bagnato GF, Loyo E, Toloza S, Li M, Ahmed Abdel Atty Mohamed W, Cobankara V, Olas J, Salsano F, Oksel F, Tanaseanu CM, Foti R, Ancuta C, Vonk M, Caramashi P, Beretta L, Balbir A, Chiàla A, Pasalic Simic K, Ghio M, Stamenkovic B, Rednic S, Host N, Pellerito R, Hachulla E, Furst DE (2010) Characteristics of joint involvement and relationships with systemic inflammation in systemic sclerosis: results from the EULAR Scleroderma Trial and Research Group (EUSTAR) database. J Rheumatol 37:1488–1501. https://doi.org/10.3899/jrheum.091165
Khanna PP, Furst DE, Clements PJ, Maranian P, Indulkar L, Khanna D (2010) Tendon friction rubs in early diffuse systemic sclerosis: prevalence, characteristics and longitudinal changes in a randomized controlled trial. Rheumatology (Oxford) 49:955–959. https://doi.org/10.1093/rheumatology/kep464
Dore A, Lucas M, Ivanco D, Medsger TA Jr, Domsic RT (2013) Significance of palpable tendon friction rubs in early diffuse cutaneous systemic sclerosis. Arthritis Care Res (Hoboken) 65:1385–1389. https://doi.org/10.1002/acr.21964
Iagnocco A, Vavala C, Vasile M, Stefanantoni K, Valesini G, Riccieri V (2013) Power Doppler ultrasound of the hand and wrist joints in systemic sclerosis. Clin Exp Rheumatol Mar-Apr 31(2 Suppl 76):89–95 [Epub 2012 Nov 28]
Cottrell TR, Wise RA, Wigley FM, Boin F (2014) The degree of skin involvement identifies distinct lung disease outcomes and survival in systemic sclerosis. Ann Rheum Dis 73:1060–1066. https://doi.org/10.1136/annrheumdis-2012-202849
Avouac J, Walker U, Hachulla E, Riemekasten G, Cuomo G, Carreira PE, Caramaschi P, Ananieva LP, Matucci-Cerinic M, Czirjak L, Denton C, Ladner UM, Allanore Y, EUSTAR collaborators (2016) Joint and tendon involvement predict disease progression in systemic sclerosis: a EUSTAR prospective study. Ann Rheum Dis 75:103–109. https://doi.org/10.1136/annrheumdis-2014-205295
Batalov A, Atanassov A (1998) Arthrosonography in the evaluation of osteoarticular and soft-tissue structures in rheumatology. Folia Med 40(3):35–43
Kane D, Pv Balint, Sturrock R, Grassi W (2004) Musculoskeletal ultrasound—a state of the art review in rheumatology. Part 2: clinical indications for musculoskeletal ultrasound in rheumatology. Rheumatology (Oxford) 43:829–838. https://doi.org/10.1093/rheumatology/keh215
Porta F, Gargani L, Kaloudi O, Schmidt WA, Picano E, Damjanov N, Matucci-Cerinic M (2012) The new frontiers of ultrasound in the complex world of vasculitides and scleroderma. Rheumatology (Oxford) 51:26–30. https://doi.org/10.1093/rheumatology/kes336
Elhai M, Guerini H, Bazeli R, Avouac J, Freire V, Drapé JL, Kahan A, Allanore Y (2012) Ultrasonographic hand features in systemic sclerosis and correlates with clinical, biologic, and radiographic findings. Arthritis Care Res (Hoboken) 64:1244–1249. https://doi.org/10.1002/acr.21668
Bandinelli F, Kaloudi O, Candelieri A, Conforti ML, Casale R, Cammarata S, Grassiri G, Miniati I, Melchiorre D, Matucci-Cerinic M (2010) Early detection of median nerve syndrome at the carpal tunnel with high-resolution 18 MHz ultrasonography in systemic sclerosis patients. Clin Exp Rheumatol 28(5 Suppl 62):S15–S18 [Epub 2010 Nov 3]
Johansen JS (2006) Studies on serum YKL-40 as a biomarker in diseases with inflammation, tissue remodelling, fibroses and cancer. Dan Med Bull 53:172–209
Kazakova M, Batalov A, Deneva T, Mateva N, Kolarov Z, Sarafian V (2013) Relationship between sonographic parameters and YKL-40 levels in rheumatoid arthritis. Rheumatol Int 33:341–346. https://doi.org/10.1007/s00296-012-2387-3
Väänänen T, Koskinen A, Paukkeri E, Hämäläinen M, Moilanen T, Moilanen E, Vuolteenaho K (2014) YKL-40 as a novel factor associated with inflammation and catabolic mechanisms in osteoarthritic joints. Mediators Inflamm. https://doi.org/10.1155/2014/215140
Nordenbæk C, Johansen JS, Halberg P, Wiik A, Garbarsch C, Ullman S, Priece PA, Jacobson S (2005) High serum levels of YKL-40 in patients with systemic sclerosis are associated with pulmonary involvement. Scand J Rheumatol 34:293–297. https://doi.org/10.1080/03009740510018598
Karalilova R, Kazakova M, Batalov A, Sarafian V (2018) Correlation between protein YKL-40 and ultrasonographic findings in active knee osteoarthritis. Med Ultrason 1:57–63. https://doi.org/10.11152/mu-1247
Lee CG, Hartl D, Lee GR, Koller B, Matsuura H, Da Silva CA, Sohn MH, Cohn L, Homer RJ, Kozhich AA, Humbles A, Kearley J, Coyle A, Chupp G, Reed J, Flavell RA, Elias JA (2009) Role of breast regression protein 39 (BRP-39)/chitinase 3-like-1 in Th2 and IL-13-induced tissue responses and apoptosis. J Exp Med 206:1149–1166
Khan K, Xu S, Nihtyanova S et al (2012) Clinical and pathological significance of interleukin 6 overexpression in systemic sclerosis. Ann Rheum Dis 71:1235–1242. https://doi.org/10.1136/annrheumdis-2011-200955
Abdel-Magied RA, Kamel SR, Said AF, Ali HM, Abdel Gawad EA, Moussa MM (2016) Serum interleukin-6 in systemic sclerosis and its correlation with disease parameters and cardiopulmonary involvement. Sarcoidosis Vasc Diffus Lung Dis 33(4):321–330
van den Hoogen F, Khanna D, Fransen J, Johnson SR, Baron M, Tyndall A, Matucci-Cerinic M, Naden RP, Medsger TA Jr, Carreira PE, Riemekasten G, Clements PJ, Denton CP, Distler O, Allanore Y, Furst DE, Gabrielli A, Mayes MD, van Laar JM, Seibold JR, Czirjak L, Steen VD, Inanc M, Kowal-Bielecka O, Müller-Ladner U, Valentini G, Veale DJ, Vonk MC, Walker UA, Chung L, Collier DH, Ellen Csuka M, Fessler BJ, Guiducci S, Herrick A, Hsu VM, Jimenez S, Kahaleh B, Merkel PA, Sierakowski S, Silver RM, Simms RW, Varga J, Pope JE (2013) 2013 classification criteria for systemic sclerosis: an American college of rheumatology/European league against rheumatism collaborative initiative. Ann Rheum Dis 72:1747–1755. https://doi.org/10.1136/annrheumdis-2013-204424
Mandl P, Naredo E, Wakefield RJ, Conaghan PG, D’Agostino MA (2011) A systematic literature review analysis of ultrasound joint count and scoring system to assess synovitis in rheumatoid arthritis according to the OMERACT filter. J Rheumatol 38:2055–2062. https://doi.org/10.3899/jrheum.110424
Karalilova R, Batalov A, Peeva Y, Kraev K, Batalov Z, Popova V (2014) Lung ultrasound for evaluation of pulmofibrosis in systemic sclerosis. Ann Rheum Dis 73(Suppl2):FRI0497,567. https://doi.org/10.1136/annrheumdis-2014-eular.5207
Karalilova R, Peeva Y, Batalov Z, Selimov P, Tsvetkova S, Batalov A (2016) Is there correlation between dermal thickness and pulmonary involvement in early diffuse systemic sclerosis?—the role of ultrasound. 4th Systemic Sclerosis World Congress. P.222. JSRD. https://doi.org/10.5301/jsrd.5000203
Karalilova R, Peeva Y, Batalov Z, Selimov P, Tsvetkova P, Batalov A (2016) Involvement of joint and tendon in systemic sclerosis. 4th Systemic Sclerosis World Congress. P.221. JSRD. https://doi.org/10.5301/jsrd.5000203
Karalilova R, Todorov P, Peeva Y, Batalov Z, Selimov P, Batalov A (2015) The correlation of high frequency ultrasound of dermal thickness and lung ultrasound for pulmonary involvement in early diffuse systemic sclerosis. Ann Rheum Dis 74(Suppl2):FRI0605, 647. https://doi.org/10.1136/annrheumdis-2015-eular.5079
Karalilova R, Kazakova M, Sarafian V, Matucci-Cerinic M, Batalov A (2018) Relationship between glycoprotein YKL-40 and musculoskeletal ultrasonography in systemic sclerosis. 5th Systemic Sclerosis World Congress. Poster presentation. JSRD. https://doi.org/10.1177/2397198317753474
Korthagen NM, van Moorsel CH, Barlo NP, Ruven HJ, Kruit A, Heron M, van den Bosch JM, Grutters JC (2011) Serum and BALF YKL-40 levels are predictors of survival in idiopathic pulmonary fibrosis. Respir Med 105:106–113. https://doi.org/10.1016/j.rmed.2010.09.012
De Lauretis A, Sestini P, Pantelidis P, Hoyles R, Hansell DM, Goh NS, Zappala CJ, Visca D, Maher TM, Denton CP, Ong VH, Abraham DJ, Kelleher P, Hector L, Wells AU, Renzoni EA (2013) Serum interleukin 6 is predictive of early functional decline and mortality in interstitial lung disease associated with systemic sclerosis. J Rheumatol 40:435–446. https://doi.org/10.3899/jrheum.120725
Affandi AJ, Radstake TR, Marut W (2015) Update on biomarkers in systemic sclerosis: tools for diagnosis and treatment. Semin Immunopathol. 37:475–487. https://doi.org/10.1007/s00281-015-0506-4
Ball E, Gibson D, Bell A, Rooney M (2014) Plasma IL-6 levels correlate with clinical and ultrasound measures of arthritis in patients with systemic lupus erythematosus. Lupus 23:46–56. https://doi.org/10.1177/0961203313512882
La Montagna G, D’Angelo S, Valentini G (2003) Cross-sectional evaluation of YKL-40 serum concentrations in patients with systemic sclerosis. Relationship with clinical and serological aspects of disease. J Rheumatol 30:2147–2215
Kawasaki M, Hasegawa Y, Kondo S, Iwata H (2001) Concentration and localization of YKL-40 in hip joint diseases. J Rheumatol 28:341–345
Atzeni F, Talotta R, Masala I, Bongiovanni S, Boccassini L, Sarzi-Puttini P (2017) Biomarkers in Rheumatoid Arthritis. Isr Med Assoc J 19:512–516
Libby P (2002) Inflammation in atherosclerosis. Nature 420(69–17):868–874
Gourh P, Arnett F, Assassi S, Tan F, Huang M, Diekman L, Mayes M, Reveille J, Agarwal S (2009) Plasma cytokine profiles in systemic sclerosis: associations with autoantibody subsets and clinical manifestations. Arthritis Res Ther 11(5):R147. https://doi.org/10.1186/ar2821
Acknowledgements
The study is supported by grant from Medical University, Plovdiv (DP06/2018) and the Bulgarian Association of Musculoskeletal Ultrasound (BAMSU).
Author information
Authors and Affiliations
Contributions
AB and VS contributed to the concept, design of the study and final approval of the manuscript. RK and MK contributed to manuscript writing and interpretation of the results. ZB and TS contributed to data collection, clinical examination, data analyses and final approval of the manuscript. MK, VD, VS contributed to data collection, immunological analysis, critical revision and final approval of the manuscript. All authors participated in the interpretation of the results, editing, and approved the final version of the manuscript. All co-authors are fully responsible for the integrity of the study and the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest concerning this article.
Ethical approval
The study was approved by ethics committee of Medical University of Plovdiv (protocol № 3/31.05.2018). All study procedures were performed according to the ethical principles of the Declaration of Helsinki and Good Clinical Practice.
Informed consent
Voluntarily signed informed consent was obtained from all participants before entering the study in accordance with the ethical recommendations of the Helsinki Declaration.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Karalilova, R., Kazakova, M., Sapundzhieva, T. et al. Serum YKL-40 and IL-6 levels correlate with ultrasound findings of articular and periarticular involvement in patients with systemic sclerosis. Rheumatol Int 39, 1841–1848 (2019). https://doi.org/10.1007/s00296-019-04402-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-019-04402-9