Abstract
Background
Papillary thyroid cancer (PTC) metastasizes to central lymph node (CLN). CLN metastasis is associated with high risk of locoregional recurrence and distant metastasis. The significance of the number of metastatic CLN has not been addressed. This study was designed to evaluate the clinical implication of the number of metastatic CLN in PTC.
Methods
We reviewed the patients who underwent total thyroidectomy and CLN dissection with or without lateral neck dissection due to PTC, from March 2008 to June 2009. The relationships between the number of CLN and risk factors, including age, gender, tumor size, extrathyroidal extension, and lateral lymph node metastasis, were assessed. Patients were divided into three groups according to the number of CLN: group A = 0; group B = 1–2; and group C = ≥3.
Results
Of 258 patients enrolled in this study, 113 were in group A, 73 in group B, and 72 in group C. Extrathyroidal extension and lateral neck lymph node metastasis were related to increased rate of CLN metastasis (P < 0.05). Tumor size increased as the number of CLN increased; group C had the largest tumor size (P < 0.05). When evaluating the distribution of patients with extrathyroidal extension, group C had a significant odds ratio (4.213, P < 0.05). When evaluating the distribution of lateral neck lymph node metastasis, groups B and C had significant odds ratio (14.353, 75.403, respectively, P < 0.05).
Conclusions
The number of CLN metastasis correlated with the negative prognostic factors, including tumor size, extrathyroidal extension, and lateral neck lymph node metastasis. This suggests that the number of CLN has prognostic implication.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Papillary thyroid cancer (PTC) metastasizes primarily to central lymph node (CLN). The necessity for central lymph node dissection (CLND) is controversial [1–3]. CLND is generally recommended in high-risk patients with old age, a large tumor, extrathyroidal extension, and so on [4, 5]. It is currently presumed that CLN metastasis is associated with high risk of locoregional recurrence and distant metastasis but do not directly correlate with survival rate [1, 6, 7]. Although nodal metastasis or recurrence does not affect survival rate directly, it can affect increased morbidity and mortality rate due to repeated surgery [8–10]. According to TNM classification established by the International Union Against Cancer (UICC) and the American Joint Commission on Cancer (AJCC; 2002, 6th edition), the number of lymph node metastasis is one of the cardinal prognostic factors in head and neck squamous cell carcinomas [11]. N stage in PTC is divided into N1a and N1b according to the location of metastatic nodes. However, clinical implications of the number of metastatic lymph nodes in PTC has not been well understood. In addition, the prognostic significance of the number of CLN in PTC has not been determined and has been overlooked. Therefore, the direction of treatment according to the number of the CLN has not been issued well.
The slow and insidious disease progression of PTC disturbs a large, prospective, randomized study and entails controversy as to the ideal therapeutic modality [4, 12]. These problems make it difficult to clarify the prognostic function of some factors when they are analyzed with survival and local control rates. Male gender, older age, large tumor size, and massive extrathyroidal extension are known poor prognostic factors [5, 13–15]. These factors have been anticipated to be relevant to local control rate rather than survival rate.
This study was designed to evaluate the prognostic value of the number of CLN metastasis in PTC. We investigated the relationship between the number of CLN metastasis and previously known prognostic factors, such as age, sex, tumor size, extrathyroidal extension, and lateral neck lymph node metastasis.
Methods
Study populations
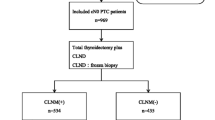
A retrospective medical chart review of patients who were diagnosed with papillary thyroid cancer and treated at a Pusan National University Hospital from March 2008 to June 2009 was performed. This study was approved by the institutional review board. Preoperative assessment for cervical lymph node metastasis included a high-resolution ultrasound scan, fine needle aspiration cytology, and a computerized tomography (CT) scan. Based on this preoperative data, enrolled patients underwent total thyroidectomy with CLND, with or without lateral neck dissection (LND). Procedures for follicular, medullary, and anaplastic thyroid carcinomas were excluded from this analysis. Patients who underwent CLND for recurrent PTC after thyroidectomy also were excluded.
Surgical technique
Bilateral CLND was performed routinely accompanied with total thyroidectomy. This dissection was performed completely according to our techniques, previously described by Lee et al. [16].
The surgical technique for CLND is as follows. The recurrent laryngeal nerve is usually identified distally near the lower border of the cricothyroid muscle. After the thyroid is removed, the nerve is dissected retrograde to the point where it courses under the clavicles, the innominate artery, or the right carotid artery. The fibrofatty tissues are dissected off the nerve and usually reflected medially, and then dissected off the trachea. If possible, the specimen is removed en bloc. However, additional nodal tissues that are deep to the right recurrent laryngeal nerve may be removed separately. Particular attention is given to identify the parathyroid glands, and autotransplantation of the parathyroid glands was performed only when their vascularity was compromised. If there was a positive or suspicious finding in the lateral neck lymph node on radiographic finding, LND was accompanied. When multiple metastatic CLN were identified from frozen biopsy after ipsilateral CLND to tumor side, LND was performed even if it was clinically negative for lateral neck lymph nodes. LND comprehended the level II, III, and IV lymph nodes. Tumor size, extrathyroidal extension, lateral neck lymph node metastasis and the number of CLN metastasis were verified postoperatively with histopathology reports.
Relationship between the number of CLN metastasis and prognostic factors
Patients were divided into three groups—A, B, C—according to the number of metastatic lymph nodes when defining the relationship between tumor size, extrathyroidal extension, or lateral neck lymph nodes and the number of metastatic CLN. Group A had no CLN metastasis, group B had one or two nodal metastases, and group C had more than two nodal metastases. Instead of survival rate, we analyzed the relationship between generally accepted prognostic factors and the number of CLN metastasis due to short follow-up period. Age, gender, tumor size, extrathyroidal extension, and lateral neck lymph node metastasis were included. At first we analyzed whether risk factors were related to the rate of CLN metastasis. The rate of metastasis was defined as proportion of the patients with CLN metastasis in each group (patients with metastatic CLN/patients with or without risk factor) without considering the number of CLN metastasis. After finding significant risk factors, we analyzed the relationship between the risk factors and the number of CLN metastasis.
Statistical analysis
Statistical analysis was performed using SPSS version 15.0 software (Chicago, IL). When analyzing correlation between risk factors, which contains age group (<45, ≥45 years), gender, extrathyroidal extension, and lateral neck lymph node metastasis and the rate of CLN metastasis, a chi-square test was applied. The relationship between gender and the number of CLN metastasis was analyzed with a t test. The correlation between tumor size and the number of CLN metastasis was estimated with linear regression tests and Tukey tests. A logistic regression test was used to evaluate the relationship between the number of CLN metastasis and some risk factors, containing extrathyroidal extension and lateral neck lymph node metastasis.
Results
Demographic findings
A total of 258 patients were eligible for this study (222 women and 36 men; mean age, 49.8 years). Mean ages of men and women were 50.7 (±12.51) and 49.6 (±14.32) years, respectively. Mean follow-up period was 13.4 (±7.11) months. Most patients underwent total thyroidectomy with CLND (n = 182). Based on the indication mentioned above, LND combined with total thyroidectomy and CLND was performed in the remaining 76 patients. Group A had 113 patients, group B had 73 patients, and group C had 72 patients. Extrathyroidal extension was observed in 140 patients and lateral neck lymph node metastasis was found in 58 patients; and 18 patients presented only suspicious findings. Most patients with metastasis in the lateral neck also had metastatic node in CLN (56/58) except two patients, who presented skipped metastatic nodes in the lateral neck.
Age and gender
Seventy-one patients were younger than age 45 years, and 43 of these patients had positive CLN metastasis (60.6%). It did not differ from the rate of CLN metastasis in older patients (54.5%). The rate of CLN metastasis did not change according to gender (Table 1). Age and gender did not affect the rate of CLN metastasis. It was also found that age was not significantly related to the number of CLN metastasis (Pearson correlation = −0.073, P = 0.246). The average number of CLN metastasis in men and women was 2.47 (±0.48) and 1.86 (±1.76), respectively. There was no significant difference (P = 0.251). Age and gender seemed to have no effect on the rate or the number of CLN metastasis.
Tumor size
We used to two methods to investigate the relationship between the number of CLN metastasis and tumor size. With the linear regression method, the size of primary tumors increased as the number of CLN metastasis increased (correlation coefficient = 0.277, P < 0.05; Fig. 1). Although the positive correlation between the number of metastatic CLN and tumor size was found, there were still many micropapillary thyroid cancer (<1 cm) that presented multiple CLN metastasis. The mean size of groups A, B, and C was 0.92 cm (±0.73), 1.17 cm (±0.93), and 1.41 cm (±0.84), respectively. Then, we compared the mean size of each group using the Tukey methods. Group C had significantly larger tumors than group A (P < 0.001), but there was no significant difference between groups A and B or between groups B and C in average tumor size (Fig. 2).
Extrathyroidal extension
The rate of CLN metastasis was higher in patients with extrathyroidal extension than in patients without that (Table 1). The rate of extrathyroidal extension was 41.6% (47/113), 53.4% (39/73), and 75% (54/72) in groups A, B, and C, respectively. The odds ratio was 1.1611 when comparing group A with group B but it was not statistically significant (P = 0.115). The rate of extrathyroidal extension increased when comparing group C with group A and it was statistically significant (odds ratio = 4.213, P < 0.001; Table 2). There was a difference in the rate of extrathyroidal extension in each group and the possibility of extrathyroidal extension increased, especially in group C.
Lateral neck lymph node metastasis
Lateral neck lymph node metastasis was related to a higher rate of CLN metastasis compared with negative lateral neck lymph node (Table 1). We investigated the relationship between the number of CLN metastasis and lateral neck lymph node metastasis. Lateral neck lymph node metastasis was identified in 58 patients and it was detected in level III (n = 46), IV (n = 41), and II (n = 29), consecutively. Group A presented 1.8% (2/113), group B 20.5% (15/73), and group C 56.9% (41/72) proportion of lateral neck lymph node metastasis. When comparing group A with B or group A with C, probability of lateral neck lymph node metastasis was found to increase according to the number of CLN metastasis. The odds ratio of group B was 14.353 (P < 0.001) and that of group C was 73.403 (P < 0.001; Table 3).
Discussion
The number of regional lymph node metastasis is regarded as a prognostic factor and it guides treatment modality, especially in breast and gastric cancer [17–19]. Also, in head and neck cancer, except papillary thyroid cancer, more than 1 metastatic lymph node is classified as N2 in the AJCC cancer staging system. It was found to be relevant to treatment modality and prognosis [20, 21]. But the number of CLN metastasis in PTC has not been considered as a prognostic factor. Even though most of the patients with PTC have a good survival rate, substantial recurrence rate and morbidity remain to be a problem. Recurrence is relevant to mortality especially in high risk group [22]. Cervical recurrence in PTC after the primary operation holds up to 30% [23, 24]. Repeated surgery after cervical recurrence may increase the risk of recurrent laryngeal nerve injury, hypocalcemia, and cosmetic problems. The prognostic value of the number of cervical (central or lateral) neck lymph nodes in PTC were confined to cervical recurrence [25]. Consequently, reducing the risk of cervical recurrence may contribute to the better prognosis. Although the prognostic implication of central lymph node has been studied, there are few articles regarding relevance of the number of CLN metastasis to the prognosis.
Various factors and the staging system have been used to predict the prognosis [5]. Age, sex, tumor size, extrathyroidal extension, and lateral neck lymph node metastasis are generally accepted variables that were related to survival and local control rates [14]. Besides this, family history, multifocality, and incomplete excision were regarded as prognostic parameters [13, 15, 26]. One of the useful staging systems is TNM staging defined by the AJCC (2002, 6th edition); it reflects the importance of lymph node metastasis [14]. It simply divides N stages into N1a and N1b, and this may be due to a good prognosis of PTC or relatively lower significance of the lymph node metastasis. Moreover, the number of metastatic CLN in TNM staging defined by the AJCC was not considered [14, 27]. This study was initiated from the hypothesis that the number of CLN has prognostic validity. Previously, receiver operating characteristic (ROC) curve test was performed to find cutoff number of CLN metastasis when dividing patients. The cutoff number was 1.5 or 2.5, and we divided the patients into three groups (patients with 0, 1 or 2, more than 3 metastatic lymph nodes; data not shown).
Some studies have shown that the metastasis to the CLN increased when tumor size increased [28–30]. Tumor size greater than 3 cm is one of the risk factors for recurrence in the lymph node [13]. Tumor size exceeding 1 cm presented a high risk of ipsilateral CLN metastasis, which can be another risk factor for contralateral CLN metastasis, simultaneously [31]. We found that the number of CLN metastasis has positive correlation with tumor size. Because there were patients with small tumors with multiple metastatic CLN, correlation coefficient was not high. It seems to have reflected the molecular or pathologic characteristics of the tumors [25, 28]. More than two (group C) CLN metastases were significantly associated with larger tumor size than a lower number of nodal metastasis. This infers that the number of CLN is relevant to prognosis as well as only existence of the CLN by itself.
Many authors found that patients with extrathyroidal extension seemed to have higher mortality and recurrence rates than patients without that. The extrathyroidal extension itself was presumed to have a negative effect on survival and local control rates [29, 32–36]. The evident extrathyroidal extension increased the rate of CLN metastasis and had an effect on the survival rate [25, 26, 37]. Distributions of patients with extrathyroidal extension in each group, A, B, and C, were different and the odds ratio increased as the number of CLN metastasis increased. Throughout this finding, we can suggest that the thyroid cancer and perithyroidal tissues should be removed more carefully if the number of CLN metastasis was at least three (group C).
Many reports dealing with metastatic patterns of PTC revealed that the first echelon of the thyroid is the central lymph node and it extends laterally through levels II, III, and IV [38, 39]. A positive node in CLN may hint probability of lateral neck lymph node metastasis. Lateral neck lymph node extension is associated with high rates of recurrence and distant metastasis [40] and needs to be managed more aggressively when it is suspicious. Our findings showed that patients with lateral neck lymph node extension had a higher rate of CLN metastasis than patients without that. We found that the higher number of CLN metastasis was involved in higher risk of lateral neck lymph node extension. In summary, it is important that further treatment for the lateral neck should be emerged if the number of metastatic CLN is more than 2 (group C).
We presumed that the number of CLN metastasis was ignored because the number of nodes was not adequately categorized and compared, in addition to insidious growth and a relatively good prognosis of PTC. Due to a short follow-up period, we could not analyze the relationship between the number of CLN metastasis and the survival or local control rates. This study was designed to be a cross-sectional study and we could not use the hazard model. To solve this problem, we used age, gender, tumor size, extrathyroidal extension, and lateral neck extension as prognostic indicators; this is a main limitation of our study.
Conclusions
The existence of CLN metastasis was related to negative prognostic factors, including extrathyroidal and lateral neck lymph node extensions. The number of CLN metastasis also was related to the poor prognostic factors, including tumor size, extrathyroidal extension, and lateral neck lymph node extension. Therefore, the number of CLN may suggest prognostic significance. Notably, lateral neck lymph node metastasis was strongly correlated with the number of CLN metastasis. Further study for longer period is needed to clarify the effect of the number of CLN on treatment results.
References
Machens A, Hinze R, Thomusch O et al (2002) Pattern of nodal metastasis for primary and reoperative thyroid cancer. World J Surg 26:22–28
Roh JL, Kim JM, Park CI (2008) Central cervical nodal metastasis from papillary thyroid microcarcinoma: pattern and factors predictive of nodal metastasis. Ann Surg Oncol 15:2482–2486
Shaha AR (1998) Management of the neck in thyroid cancer. Otolaryngol Clin North Am 31:823–831
Cooper DS, Doherty GM, Haugen BR et al (2006) Management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 16:109–142
Lundgren CI, Hall P, Dickman PW et al (2006) Clinically significant prognostic factors for differentiated thyroid carcinoma: a population-based, nested case-control study. Cancer 106:524–531
Choi SJ, Kim TY, Lee JC et al (2008) Is routine central neck dissection necessary for the treatment of papillary thyroid microcarcinoma? Clin Exp Otorhinolaryngol 1:41–45
Hughes CJ, Shaha AR, Shah JP et al (1996) Impact of lymph node metastasis in differentiated carcinoma of the thyroid: a matched-pair analysis. Head Neck 18:127–132
White ML, Doherty GM (2007) Level VI lymph node dissection for papillary thyroid cancer. Minerva Chir 62:383–393
Mazzaferri EL, Doherty GM, Steward DL (2009) The pros and cons of prophylactic central compartment lymph node dissection for papillary thyroid carcinoma. Thyroid 19:683–689
Mazzaferri EL (2009) A vision for the surgical management of papillary thyroid carcinoma: extensive lymph node compartmental dissections and selective use of radioiodine. J Clin Endocrinol Metab 94:1086–1088
Greene F (2002) AJCC cancer staging manual. Springer-Verlag, New York
Davidson HC, Park BJ, Johnson JT (2008) Papillary thyroid cancer: controversies in the management of neck metastasis. Laryngoscope 118:2161–2165
Ito Y, Higashiyama T, Takamura Y et al (2007) Risk factors for recurrence to the lymph node in papillary thyroid carcinoma patients without preoperatively detectable lateral node metastasis: validity of prophylactic modified radical neck dissection. World J Surg 31:2085–2091
Ito Y, Miyauchi A, Jikuzono T et al (2007) Risk factors contributing to a poor prognosis of papillary thyroid carcinoma: validity of UICC/AJCC TNM classification and stage grouping. World J Surg 31:838–848
Kim TY, Hong SJ, Kim JM et al (2008) Prognostic parameters for recurrence of papillary thyroid microcarcinoma. BMC Cancer 8:296
Lee BJ, Lee JC, Wang SG et al (2009) Metastasis of right upper para-esophageal lymph nodes in central compartment lymph node dissection of papillary thyroid cancer. World J Surg 33:2094–2098
Camp RL, Rimm EB, Rimm DL (2000) A high number of tumor-free axillary lymph nodes from patients with lymph node negative breast carcinoma is associated with poor outcome. Cancer 88:108–113
Huang CM, Lin BJ, Lu HS et al (2008) Prognostic impact of metastatic lymph node ratio in advanced gastric cancer from cardia and fundus. World J Gastroenterol 14:4383–4388
Nemoto T, Vana J, Bedwani RN et al (1980) Management and survival of female breast cancer: results of a national survey by the American College of Surgeons. Cancer 45:2917–2924
Chatni SS, Thankappan K (2008) Lymph node status cannot be ignored in survival analysis of laryngeal cancer. Arch Otolaryngol Head Neck Surg 134:786 (author reply 786–787)
Schmitz S, Machiels JP, Weynand B et al (2009) Results of selective neck dissection in the primary management of head and neck squamous cell carcinoma. Eur Arch Otorhinolaryngol 266:437–443
Palme CE, Waseem Z, Raza SN et al (2004) Management and outcome of recurrent well-differentiated thyroid carcinoma. Arch Otolaryngol Head Neck Surg 130:819–824
Mazzaferri EL, Jhiang SM (1994) Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am J Med 97:418–428
Simon D, Goretzki PE, Witte J et al (1996) Incidence of regional recurrence guiding radicality in differentiated thyroid carcinoma. World J Surg 20:860–866 (discussion 866)
Noguchi S, Yamashita H, Uchino S et al (2008) Papillary microcarcinoma. World J Surg 32:747–753
Cushing SL, Palme CE, Audet N et al (2004) Prognostic factors in well-differentiated thyroid carcinoma. Laryngoscope 114:2110–2115
Wittekind C, Compton CC, Greene FL et al (2002) TNM residual tumor classification revisited. Cancer 94:2511–2516
Mercante G, Frasoldati A, Pedroni C et al (2009) Prognostic factors affecting neck lymph node recurrence and distant metastasis in papillary microcarcinoma of the thyroid: results of a study in 445 patients. Thyroid 19:707–716
Moo TA, Umunna B, Kato M et al (2009) Ipsilateral versus bilateral central neck lymph node dissection in papillary thyroid carcinoma. Ann Surg 250:403–408
Sadowski BM, Snyder SK, Lairmore TC (2009) Routine bilateral central lymph node clearance for papillary thyroid cancer. Surgery 146:696–703 (discussion 703–695)
Koo BS, Choi EC, Yoon YH et al (2009) Predictive factors for ipsilateral or contralateral central lymph node metastasis in unilateral papillary thyroid carcinoma. Ann Surg 249:840–844
Lin JD, Chao TC, Weng HF et al (1999) Prognostic variables of papillary thyroid carcinomas with local invasion. Endocr J 46:91–98
Andersen PE, Kinsella J, Loree TR et al (1995) Differentiated carcinoma of the thyroid with extrathyroidal extension. Am J Surg 170:467–470
Arora N, Turbendian HK, Scognamiglio T et al (2008) Extrathyroidal extension is not all equal: Implications of macroscopic versus microscopic extent in papillary thyroid carcinoma. Surgery 144:942–947 (discussion 947–948)
Bellantone R, Lombardi CP, Boscherini M et al (1998) Prognostic factors in differentiated thyroid carcinoma: a multivariate analysis of 234 consecutive patients. J Surg Oncol 68:237–241
Ortiz S, Rodriguez JM, Soria T et al (2001) Extrathyroid spread in papillary carcinoma of the thyroid: clinicopathological and prognostic study. Otolaryngol Head Neck Surg 124:261–265
Ito Y, Tomoda C, Uruno T et al (2006) Prognostic significance of extrathyroid extension of papillary thyroid carcinoma: massive but not minimal extension affects the relapse-free survival. World J Surg 30:780–786
Sivanandan R, Soo KC (2001) Pattern of cervical lymph node metastases from papillary carcinoma of the thyroid. Br J Surg 88:1241–1244
Kupferman ME, Patterson M, Mandel SJ et al (2004) Patterns of lateral neck metastasis in papillary thyroid carcinoma. Arch Otolaryngol Head Neck Surg 130:857–860
Noguchi S, Murakami N, Yamashita H et al (1998) Papillary thyroid carcinoma: modified radical neck dissection improves prognosis. Arch Surg 133:276–280
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, Y.S., Lim, Y.S., Lee, JC. et al. Clinical Implication of the Number of Central Lymph Node Metastasis in Papillary Thyroid Carcinoma: Preliminary Report. World J Surg 34, 2558–2563 (2010). https://doi.org/10.1007/s00268-010-0749-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-010-0749-0