Abstract
Purpose
The purpose was to evaluate the impact of intra-operative administration of tranexamic acid (TXA) and pre-operative discontinuation of prophylactic chemoprophylaxis in patients undergoing internal fixation of pelvic or acetabular fractures on the need for subsequent blood transfusion. Operative time and the incidence of deep vein thrombosis (DVT) and pulmonary embolism (PE) were also assessed.
Methods
Data from a single level one trauma centre was retrospectively reviewed from January 2014 to December 2017 to identify pelvic ring or acetabular fractures managed operatively. Patients who did not receive their scheduled dose of chemoprophylaxis prior to surgery but who did receive intra-operative TXA were identified as the treatment group. Due to the interaction of VTE prophylaxis and TXA, the variables were analyzed using an interaction effect to account for administration of both individually and concomitantly.
Results
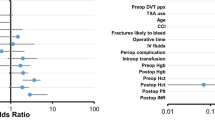
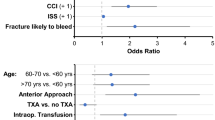
One hundred fifty-nine patients were included. The treatment group experienced a 20.7% reduction in blood product transfusion (regression coefficient (RC): − 0.207, p = 0.047, 95%CI: − 0.412 to − 0.003) and an average of 36 minutes (RC): − 36.90, p = 0.045, 95%CI: − 72.943 to − 0.841) reduction in surgical time as compared to controls. The treatment group did not experience differential rates of PE or DVT (RC: 1.302, p = 0.749, 95%CI: 0.259–6.546) or PE (RC: 1.024, p = 0.983, 95%CI: 0.114–9.208).
Conclusions
In the study population, the combination of holding pre-operative chemoprophylaxis and administering intra-operative TXA is a safe and effective combination in reducing operative time and blood product transfusions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pelvic and acetabular traumas are known risk factors for venous thromboembolism (VTE) [1]. This increased risk may be related to disruption of the pelvic vasculature at the time of the trauma, manipulation of the vasculature during subsequent operative fixation or secondary to the prolonged period of protected weight bearing required thereafter [2]. The estimated incidence of deep venous thrombosis (DVT) and pulmonary embolism (PE) has ranged from 0.51 to 61% [2,3,4,5,6,7] and 0.2 to 10% [4, 6, 8], respectively. Subsequent mortality resulting from PE is estimated to occur in 0.5% to 4% of patients [8]. The use of chemo-thromboprophylaxis such as with low molecular weight heparin (LMWH) has proven effective in decreasing VTE rates in this high-risk population [9]. To the authors’ knowledge, the optimal timing of initiation, discontinuation, and resumption of chemoprophylaxis in regard to operative reconstruction of pelvis or acetabulum fractures are not clearly defined in the literature.
Due to the lack of data regarding exact protocols for VTE prevention in patients with pelvic and acetabulum trauma, there are no official guidelines, and most surgeons use guidelines developed for total joint replacement and hip fracture management to help guide practice. There is much more information regarding VTE prevention protocols in patients undergoing total hip and knee replacement as well as hip fracture management. Despite the greater amount of available literature, there is controversy and conflicting guidelines for VTE prevention exist. The American Academy of Orthopaedic Surgeons (AAOS) recommends the use of VTE prophylaxis in hip fracture patients but does not specify type or duration [10]. Their recommendations recognize the need for a multi-armed randomized controlled study to answer many questions that remain [10]. Similarly, the AAOS recommends pharmacologic and/or mechanical prophylaxis for patients undergoing elective total hip or knee arthroplasty; however, there is insufficient evidence to recommend for or against a specific strategy [11]. In contrast, the American College of Chest Physicians (ACCP) recommends administration of chemoprophylaxis 12 hours or more prior to and following surgery rather than within four hours pre- or post-operatively in patients undergoing hip and knee arthroplasty or hip fracture surgery [12]. The ACCP goes on to list recommended medications which include low molecular weight heparin and aspirin. Beyond organizational recommendations, many authors have commented on the timing of chemoprophylaxis in cases of hip and knee arthroplasty and hip fracture surgery [13,14,15,16,17,18,19,20,21].
In regard to minimizing intra-operative blood loss, tranexamic acid (TXA) (Cyklokapron, Pfizer, New York, New York) is an increasingly popularized synthetic derivative of lysine which competitively inhibits the conversion of plasminogen to plasmin. This prohibits fibrin degradation and dissolution of formed clots [22]. The use of TXA as a safe means to control intra-operative blood loss and need for transfusion has been incorporated into many elective orthopaedic procedures, especially in joint arthroplasty [23,24,25,26,27]. Many studies have also found TXA to be both safe and effective in controlling bleeding within spine surgery [28, 29]. Within orthopaedic trauma literature, routine use of TXA for fracture fixation has been suggested given safety and efficacy; however, the majority of these studies focus on hip fractures and do not address pelvic or acetabular injuries [30, 31]. One randomized clinical trial specifically looking at the use of TXA in acetabular 88 patients failed to find show a difference in transfusion rates and average blood loss [32]. To the authors’ knowledge, this is the only study directly assessing TXA use in pelvic or acetabular fracture patients.
Given the paucity of literature regarding peri-operative chemoprophylaxis and the use of TXA in a subset of trauma patients with pelvic and acetabulum fractures, the purpose of this study was to evaluate the impact of pre-operative discontinuation of prophylactic chemoprophylaxis and intra-operative administration of TXA in polytrauma patients undergoing internal fixation on the need for subsequent blood transfusion. The authors hypothesized that the combined effects of pre-operative discontinuation of VTE chemical prophylaxis in addition to intra-operative administration of TXA would reduce operative time and the need for blood transfusion. Secondary measures included an assessment of operative time and the safety of these measures by evaluating the frequency of DVT and PE.
Patients and methods
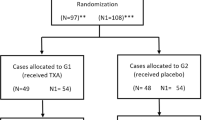
After institutional review board approval, patients either admitted to or followed by the orthopaedic trauma service between January 2014 and December 2017 at a single, level 1 trauma centre were retrospectively reviewed. Patients were included if they had pelvic ring or acetabular fractures managed operatively. From that cohort, the authors identified those patients whose fractures required an open approach and internal fixation. Thus, patients were then excluded if their pelvic fractures were managed with external fixation, INFIX, trans-sacral or sacroiliac screw placement in isolation or in combination so long as there was no open approach performed. The above listed procedures are often minimally invasive with minimal dissection and expected blood loss thus obviating the indication for TXA administration. Additionally, the authors felt this helped bring uniformity to the cohort. No other exclusion criteria were applied.
Demographic information including age and body mass index (BMI) were recorded. Risk factors for VTE such as a history of malignancy and history of previous DVT or PE were documented based on patient history recorded in the electronic medical record. Other orthopaedic and non-orthopaedic injuries necessitating surgical intervention were recorded. The presence of an intra-cranial haemorrhage was recorded based on the attending radiologists’ impression of the admission-computed tomography brain scan, if performed. Embolization of intra-pelvic vessels by the interventional radiologists was similarly recorded. The AO/Orthopaedic Trauma Association fracture classification system was used to catalog both pelvic ring and acetabulum fractures.
Operative data, operative time, estimated blood loss, and peri-operative changes in haemoglobin/haematocrit were then collected for all patients. Intra-operative Cell Saver (Braintree, MA, USA) transfusion records were included. For the purposes of the study, the authors defined a blood transfusion event if a patient received an autologous transfusion from the use of Cell Saver or an allogeneic transfusion intra-operatively or within 48 hours of surgery. At the authors’ institution, there were no set protocols for transfusion of blood products and are largely performed at the discretion of the primary team—whether that be the orthopaedic or trauma surgery team. Clinical evaluation of the patient, including but not limited to their vital signs, age, and medical comorbidities, was paramount when considering transfusing blood products. However, patients were typically transfused to maintain a haemoglobin above 7.0 g/dL or a haematocrit above 21%. Intra-operative transfusions are administered at the discretion of the anesthesiologist at the time of surgery. Administration of tranexamic acid (TXA) and discontinuation of prophylactic anti-coagulation prior to surgery were recorded. Type of prophylactic anti-coagulation was recorded as either unfractionated heparin, or low molecular weight heparin (LMWH), with LMWH being the standard of care at this institution. Most patients received 30 mg LMWH subcutaneously twice daily for DVT prophylaxis. Upon discharge, most patients were converted to 40 mg LMWH subcutaneously daily for 14 days. If the patients had bilateral injuries such that they could not weight bear on either extremity, the prophylaxis was continued until they were allowed to weight bear. The use of intra-operative Cell Saver and TXA was at the discretion of the attending surgeon and anesthesiologist at the time. No institutional protocols were in place. All TXA was administered intravenously; however, the rate of administration varied among patients and was not recorded in the medical record.
Screening tests for deep venous thrombosis (DVT) and pulmonary embolism (PE) were performed at the discretion of the trauma or orthopaedic teams. DVT screening was performed with ultrasonography, while PE screening used computed tomography angiography (CTA) or ventilation-perfusion (VQ) scan. Patients treated empirically for pulmonary embolism without radiographic confirmation based on clinician judgment were treated as having a PE for the purposes of the chart review.
Patients who received their pre-operative dose of prophylactic anticoagulation prior to surgery and did not receive intra-operative TXA served as the control group. Statistical analysis was performed using Stata 15 Software (College Station, Texas, USA). Due to the interaction of prophylactic anticoagulation and TXA, the variables were analyzed using an interaction effect to account for administration of both individually and concomitantly. A logistic regression model using the interaction effect condition was used to test for significance. Coefficients from the model are reported along with standard error (SE) and 95% confidence interval (95%CI) for binary variables only. Significance was defined as p < 0.05.
No funding was received for this study.
Results
A total of 195 patients with pelvic ring or acetabulum fractures managed operatively were identified. After exclusion criteria were applied, 159 patients (109 male, 50 female) with ages ranging from nine to 86 years (mean = 39.18 years, standard deviation (SD) = 16) were included for analysis. Two patients expired due to their injuries. Neither mortality occurred during pelvic nor acetabular fixation. Other patient characteristics of the cohort are summarized in Table 1.
Of the 159 patients, 50 patients had isolated pelvic ring injuries while 92 had isolated acetabulum fractures. Seventeen patients had both pelvic ring and acetabulum fractures. Table 2 summarizes the AO/OTA fracture classifications treated in this cohort.
During the study period, 102 patients had their chemical DVT prophylaxis discontinued prior to surgery. Forty-six patients received an average of 1.33 g of TXA (range 1–3 g) intra-operatively. Variability in the amount of TXA administered was secondary to the anaesthesia department’s initial hesitation and unfamiliarity administering TXA in trauma patients. The preferred dosage schedule was one dose of 1 g prior to incision and an additional gram at the time of wound closure. Initially, 1 g just prior to incision was administered. However, as the institution has become more comfortable with the use of TXA, the preferred dosing regimen was allowed. The two patients who received 3 g of TXA had a mean operative time of 363.5 minutes. The retrospective nature of the study does not allow for evaluation of the indication or justification for administration of a third dose. Over the three year period, 26 patients received TXA intra-operatively and had their chemoprophylaxis discontinued prior to surgery.
Eighty-two patients (51.6%) received blood product transfusions within 48 hours of their pelvic or acetabulum fracture surgery. Of those, 56 (68.2%) transfusions were intra-operative. These included 36 patients who received autologous transfusions using a cell salvage system.
Patients whose chemoprophylaxis was held pre-operatively and who received intra-operative TXA experienced a 20.7% reduction in blood product transfusion (regression coefficient (RC): − 0.207, p = 0.047, 95%CI: − 0.412 to − 0.003) and an average reduction in operative time by 36 min (RC): − 36.90, p = 0.045, 95%CI: − 72.943 to − 0.841) as compared to the remainder of the patients in the cohort.
Six and ten patients, respectively, experienced non-lethal pulmonary emboli and deep venous thromboses. Table 3 summarizes the frequency of PE and DVT as it pertains to intra-operative TXA administration and discontinuation of peri-operative DVT prophylaxis. In the logistic regression model, the combination of TXA and holding pre-operative chemoprophylaxis did not result in statistically significant increases in the rate of DVT (RC: 1.302, p = 0.749, SE: 1.073, 95%CI: 0.259–6.546) or PE (RC: 1.024, p = 0.983, SE: 1.147, 95%CI: 0.114–9.208) as compared to the remainder of the cohort.
Discussion
The data presented above demonstrate that holding pre-operative chemoprophylaxis in combination with intra-operative administration of TXA decreased operative times and reduced requirement for blood product transfusions without increasing the rate of DVT or PE in patients with operative fractures of the pelvis and acetabulum. To the authors’ knowledge, this is the first study addressing the utility and safety of both peri-operative DVT prophylaxis and TXA in this population.
Reduction of operative time and blood product transfusion are important considerations in preventing morbidity and mortality. Prolonged operative times have been shown to result in increased rates of infection in extremity fracture surgery [33]. Similarly, recipients of blood products are at risk for several complications including acute lung injury, circulatory overload, HIV, and hepatitis C [34, 35]. Moreover, transfusions have been previously shown to have detrimental immunomodulating effects through impaired T cell-mediated immunity [36, 37]. Clinically, these immunologic effects manifest as increased rates of infection in trauma [38,39,40], spine [41], and joint arthroplasty patients [42, 43]. Blood product transfusion has also been shown to be an independent predictor of mortality in trauma patients after controlling for severity of shock [44]. Conflicting evidence exists regarding the impact of leukoreduction of blood products as it pertains to the transfusion-related immunomodulation (TRIM). Some have shown that leukoreduction reduces the frequency of post-operative infections in orthopaedic procedures [45]; however, other studies failed to demonstrate benefits as it pertains to infection [46] and mortality [47]. Nevertheless, avoidance of blood product transfusion when possible precludes the possibility of these undesirable outcomes and therefore should be the goal.
The literature specifically focusing on patient’s undergoing acetabular fracture surgery echoes that cited above. Blood product transfusion, estimated blood loss, and operative time were identified as risk factors for surgical site infection by both Suzuki et al. and Li et al. [48, 49].
In efforts to mitigate intra-operative blood loss, TXA has emerged as a reliable tool. Among the most referenced studies in the trauma literature involving TXA is the CRASH-2 trial. In this randomized, controlled study, early administration of TXA within three hours of injury reduced mortality from haemorrhage without increasing the risk of thromboembolic events. It is important to note that there was no reduction in the frequency of blood product transfusion in TXA group as compared to the placebo group (relative risk 0.98, 95% confidence interval 0.96–1.01, p = 0.21) and that administration of TXA between three and eight hours after injury increased the risk of death from bleeding [50].
The role of TXA in orthopaedic trauma surgery has yet to be elucidated [27]. Two recent meta-analyses evaluating TXA usage in orthopaedic trauma surgery concluded that its use is associated with decreased rates of transfusion and lower estimated blood loss with no associated increase in VTE risk [30, 31]. Farrow et al. reached similar conclusions regarding decreased rates of transfusion without concomitant increase in VTE in a review of hip fractures [51]. In contrast, Zufferey et al. reported a non-significant trend towards increased DVT in a randomized controlled study evaluating TXA use in hip fracture surgery [52]. TXA has repeatedly demonstrated its strong therapeutic potential and safety profile by attenuating blood loss and therefore decreasing the need for transfusions while avoiding increased VTE rates in total knee arthroplasty (TKA) [53,54,55,56] and total hip arthroplasty (THA) [53]. It is worth noting that as compared to total joint arthroplasty patients, orthopedic trauma patients have relatively increased risk of fatal pulmonary embolism [57].
As discussed above, there is limited literature specifically evaluating TXA in pelvic ring and acetabular fractures. Lack et al. failed to demonstrate a difference in transfusion, estimated blood loss, DVT, and PE in a randomized controlled study evaluating TXA use in acetabular fracture surgery [32]. In contrast to the current study, no mention of peri-operative DVT prophylaxis was included.
There are important limitations to this study through which the results should be interpreted. The absence of a treatment protocol during the study period and the retrospective nature of the study generate inherent variability in treatment exposures. For instance, patients received between 1 and 3 g of intravenous TXA at the discretion of the operating surgeon and the anaesthesia provider. Additionally, there was no uniformity if the type (unfractionated heparin versus low molecular weight heparin) or timing of DVT prophylaxis administered. As such, for patients who did not receive a pre-operative dose, there is the potential for variability in the time from last dose to surgery. However, standardization of chemoprophylaxis administration in this setting would be logistically difficult due to varying surgical start times as well as the unpredictable nature of a level 1 trauma centre. Lastly, while this patient cohort is at high risk for VTE, its incidence remains low enough that the current study is underpowered to accurately detect a difference in PE and DVT. Further investigation of this topic with a prospective, randomized trial is needed as demonstrated by the results herein. Strengths of this study include the number of patients included relative to the available studies.
Conclusion
In summary, in patients with operative pelvic ring and acetabular fractures, the combination of holding pre-operative chemoprophylaxis and administration of intra-operative TXA is a safe and effective combination in reducing operative time and blood product transfusion. Further prospective studies are needed to better evaluate the treatment effects; however, the findings herein contribute to addressing a gap in the available literature specific to pelvic and acetabular fractures.
References
Sharma OP, Oswanski MF, Joseph RJ, Tonui P, Westrick L, Raj SS, Tatchell T, Waite PJ, Gandaio A (2007) Venous thromboembolism in trauma patients. Am Surg 73:1173–1180
Kim JW, Oh CW, Oh JK, Baek SG, Lee BJ, Hong HP, Min WK (2014) The incidence and the risk factors of venous thromboembolism in Korean patients with pelvic or acetabular fractures. J Orthop Sci 19:471–477. https://doi.org/10.1007/s00776-014-0553-z
Godoy Monzon D, Iserson KV, Cid A, Vazquez JA (2012) Oral thromboprophylaxis in pelvic trauma: a standardized protocol. J Emerg Med 43:612–617. https://doi.org/10.1016/j.jemermed.2011.09.006
Dwyer EP, Moed BR (2019) Venous thromboembolism after hospital discharge in pelvic and acetabular fracture patients treated operatively. J Orthop Surg (Hong Kong) 27:2309499019832815. https://doi.org/10.1177/2309499019832815
Wang P, Kandemir U, Zhang B, Wang B, Li J, Zhuang Y, Wang H, Zhang H, Liu P, Zhang K (2019) Incidence and risk factors of deep vein thrombosis in patients with pelvic and acetabular fractures. Clin Appl Thromb Hemost 25:1076029619845066. https://doi.org/10.1177/1076029619845066
Cohen-Levy WB, Liu J, Sen M, Teperman SH, Stone ME Jr (2019) Prophylactic inferior vena cava filters for operative pelvic fractures: a twelve year experience. Int Orthop 43:2831–2838. https://doi.org/10.1007/s00264-019-04384-0
Moed BR, Miller JR, Tabaie SA (2012) Sequential duplex ultrasound screening for proximal deep venous thrombosis in asymptomatic patients with acetabular and pelvic fractures treated operatively. J tRauma Acute Care Surg 72:443–447. https://doi.org/10.1097/TA.0b013e318241090d
Montgomery KD, Geerts WH, Potter HG, Helfet DL (1996) Thromboembolic complications in patients with pelvic trauma. Clin Orthopaedics Relat Res:68–87. https://doi.org/10.1097/00003086-199608000-00010
Knudson MM, Morabito D, Paiement GD, Shackleford S (1996) Use of low molecular weight heparin in preventing thromboembolism in trauma patients. J Trauma 41:446–459
Roberts KC, Brox WT (2015) AAOS clinical practice guideline: management of hip fractures in the elderly. The Journal of the American Academy of Orthopaedic Surgeons 23:138–140. https://doi.org/10.5435/JAAOS-D-14-00433
Mont MA, Jacobs JJ (2011) AAOS clinical practice guideline: preventing venous thromboembolic disease in patients undergoing elective hip and knee arthroplasty. The Journal of the American Academy of Orthopaedic Surgeons 19:777–778
Falck-Ytter Y, Francis CW, Johanson NA, Curley C, Dahl OE, Schulman S, Ortel TL, Pauker SG, Colwell CW, Jr. (2012) Prevention of VTE in orthopedic surgery patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 141:e278S-e325S. https://doi.org/10.1378/chest.11-2404
Perka C (2011) Preoperative versus postoperative initiation of thromboprophylaxis following major orthopedic surgery: safety and efficacy of postoperative administration supported by recent trials of new oral anticoagulants. Thromb J 9:17. https://doi.org/10.1186/1477-9560-9-17
Randelli F, Biggi F, Della Rocca G, Grossi P, Imberti D, Landolfi R, Palareti G, Prisco D (2011) Italian intersociety consensus statement on antithrombotic prophylaxis in hip and knee replacement and in femoral neck fracture surgery. J Orthop Traumatol 12:69–76. https://doi.org/10.1007/s10195-010-0125-8
Raskob GE, Hirsh J (2003) Controversies in timing of the first dose of anticoagulant prophylaxis against venous thromboembolism after major orthopedic surgery. Chest 124:379S–385S
Hull RD, Pineo GF (1999) Extended prophylaxis against venous thromboembolism following total hip and knee replacement. Haemostasis 29(Suppl S1):23–31. https://doi.org/10.1159/000054109
Hull RD, Pineo GF, Francis C, Bergqvist D, Fellenius C, Soderberg K, Holmqvist A, Mant M, Dear R, Baylis B, Mah A, Brant R (2000) Low-molecular-weight heparin prophylaxis using dalteparin extended out-of-hospital vs in-hospital warfarin/out-of-hospital placebo in hip arthroplasty patients: a double-blind, randomized comparison. North American Fragmin Trial Investigators. Arch Intern Med 160:2208–2215
Hull RD, Pineo GF, Stein PD, Mah AF, MacIsaac SM, Dahl OE, Butcher M, Brant RF, Ghali WA, Bergqvist D, Raskob GE (2001) Extended out-of-hospital low-molecular-weight heparin prophylaxis against deep venous thrombosis in patients after elective hip arthroplasty: a systematic review. Ann Intern Med 135:858–869
Hull RD, Pineo GF, Stein PD, Mah AF, MacIsaac SM, Dahl OE, Ghali WA, Butcher MS, Brant RF, Bergqvist D, Hamulyak K, Francis CW, Marder VJ, Raskob GE (2001) Timing of initial administration of low-molecular-weight heparin prophylaxis against deep vein thrombosis in patients following elective hip arthroplasty: a systematic review. Arch Intern Med 161:1952–1960
Ktistakis I, Giannoudis V, Giannoudis PV (2016) Anticoagulation therapy and proximal femoral fracture treatment: an update. EFORT Open Rev 1:310–315. https://doi.org/10.1302/2058-5241.1.160034
Borgen PO, Dahl OE, Reikeras O (2012) Blood loss in cemented THA is not reduced with postoperative versus preoperative start of thromboprophylaxis. Clin Orthop Relat Res 470:2591–2598. https://doi.org/10.1007/s11999-012-2320-9
McCormack PL (2012) Tranexamic acid: a review of its use in the treatment of hyperfibrinolysis. Drugs 72:585–617. https://doi.org/10.2165/11209070-000000000-00000
Poeran J, Rasul R, Suzuki S, Danninger T, Mazumdar M, Opperer M, Boettner F, Memtsoudis SG (2014) Tranexamic acid use and postoperative outcomes in patients undergoing total hip or knee arthroplasty in the United States: retrospective analysis of effectiveness and safety. BMJ 349:g4829. https://doi.org/10.1136/bmj.g4829
Ho KM, Ismail H (2003) Use of intravenous tranexamic acid to reduce allogeneic blood transfusion in total hip and knee arthroplasty: a meta-analysis. Anaesth Intensive Care 31:529–537
Li JF, Li H, Zhao H, Wang J, Liu S, Song Y, Wu HF (2017) Combined use of intravenous and topical versus intravenous tranexamic acid in primary total knee and hip arthroplasty: a meta-analysis of randomised controlled trials. J Orthop Surg Res 12:22. https://doi.org/10.1186/s13018-017-0520-4
Peng Zhang MM, Jifeng Li MM, Xiao Wang MM (2017) Combined versus single application of tranexamic acid in total knee and hip arthroplasty: a meta-analysis of randomized controlled trials. Int J Surg 43:171–180. https://doi.org/10.1016/j.ijsu.2017.05.065
Jennings JD, Solarz MK, Haydel C (2016) Application of tranexamic acid in trauma and orthopedic surgery. Orthopedic Clinics North Am 47:137–143. https://doi.org/10.1016/j.ocl.2015.08.014
Luo W, Sun RX, Jiang H, Ma XL (2018) The efficacy and safety of topical administration of tranexamic acid in spine surgery: a meta-analysis. J Orthop Surg Res 13:96. https://doi.org/10.1186/s13018-018-0815-0
Yuan QM, Zhao ZH, Xu BS (2017) Efficacy and safety of tranexamic acid in reducing blood loss in scoliosis surgery: a systematic review and meta-analysis. Eur Spine J 26:131–139. https://doi.org/10.1007/s00586-016-4899-0
Amer KM, Rehman S, Amer K, Haydel C (2017) Efficacy and safety of tranexamic acid in orthopaedic fracture surgery: a meta-analysis and systematic literature review. J Orthop Trauma 31:520–525. https://doi.org/10.1097/BOT.0000000000000919
Gausden EB, Qudsi R, Boone MD, O'Gara B, Ruzbarsky JJ, Lorich DG (2017) Tranexamic acid in orthopaedic trauma surgery: a meta-analysis. J Orthop Trauma 31:513–519. https://doi.org/10.1097/BOT.0000000000000913
Lack WD, Crist BD, Seymour RB, Harvin W, Karunakar MA, Group TXAS (2017) Effect of tranexamic acid on transfusion: a randomized clinical trial in acetabular fracture surgery. J Orthop Trauma 31:526–530. https://doi.org/10.1097/BOT.0000000000000968
Colman M, Wright A, Gruen G, Siska P, Pape HC, Tarkin I (2013) Prolonged operative time increases infection rate in tibial plateau fractures. Injury 44:249–252. https://doi.org/10.1016/j.injury.2012.10.032
Viberg B, Gundtoft PH, Schonnemann J, Pedersen L, Andersen LR, Titlestad K, Madsen CF, Lauritsen J, Overgaard S (2018) Introduction of national guidelines for restrictive blood transfusion threshold for hip fracture patients--a consecutive cohort study based on complete follow-up in national databases. J Orthop Surg Res 13:116. https://doi.org/10.1186/s13018-018-0828-8
Sward-Nordmo M, Paulsen BS, Wold JK, Wehler T, Jansson PE (1991) Further structural studies of the carbohydrate moiety of the allergen Ag-54 (Cla h II) from the mould Cladosporium herbarum. Carbohydr Res 214:267–279
Dzik S, Blajchman MA, Blumberg N, Kirkley SA, Heal JM, Wood K (1996) Current research on the immunomodulatory effect of allogeneic blood transfusion. Vox Sang 70:187–194
Innerhofer P, Luz G, Spotl L, Hobisch-Hagen P, Schobersberger W, Fischer M, Nussbaumer W, Lochs A, Irschick E (1999) Immunologic changes after transfusion of autologous or allogeneic buffy coat-poor versus white cell-reduced blood to patients undergoing arthroplasty. I Proliferative T-cell responses and the balance of helper and suppressor T cells. Transfusion (Paris) 39:1089–1096
Dunne JR, Riddle MS, Danko J, Hayden R, Petersen K (2006) Blood transfusion is associated with infection and increased resource utilization in combat casualties. Am Surg 72:619–625 discussion 625-616
Bochicchio GV, Napolitano L, Joshi M, Bochicchio K, Meyer W, Scalea TM (2008) Outcome analysis of blood product transfusion in trauma patients: a prospective, risk-adjusted study. World J Surg 32:2185–2189. https://doi.org/10.1007/s00268-008-9655-0
Carson JL, Altman DG, Duff A, Noveck H, Weinstein MP, Sonnenberg FA, Hudson JI, Provenzano G (1999) Risk of bacterial infection associated with allogeneic blood transfusion among patients undergoing hip fracture repair. Transfusion 39:694–700
Fisahn C, Jeyamohan S, Norvell DC, Tubbs RS, Moisi M, Chapman JR, Page J, Oskouian RJ (2017) Association between allogeneic blood transfusion and postoperative infection in major spine surgery. Clin Spine Surg 30:E988–E992. https://doi.org/10.1097/BSD.0000000000000539
Everhart JS, Sojka JH, Mayerson JL, Glassman AH, Scharschmidt TJ (2018) Perioperative allogeneic red blood-cell transfusion associated with surgical site infection after total hip and knee arthroplasty. J Bone Joint Surg Am 100:288–294. https://doi.org/10.2106/JBJS.17.00237
Koyanagi I, Iwasaki Y, Isu T, Akino M, Abe H (1989) Significance of spinal cord swelling in the prognosis of acute cervical spinal cord injury. Paraplegia 27:190–197. https://doi.org/10.1038/sc.1989.28
Malone DL, Dunne J, Tracy JK, Putnam AT, Scalea TM, Napolitano LM (2003) Blood transfusion, independent of shock severity, is associated with worse outcome in trauma. J Trauma 54:898–905; discussion 905-897. https://doi.org/10.1097/01.TA.0000060261.10597.5C
Llewelyn CA, Taylor RS, Todd AA, Stevens W, Murphy MF, Williamson LM, Leucodepletion Study G (2004) The effect of universal leukoreduction on postoperative infections and length of hospital stay in elective orthopedic and cardiac surgery. Transfusion (Paris) 44:489–500. https://doi.org/10.1111/j.1537-2995.2004.03325.x
Frietsch T, Karger R, Scholer M, Huber D, Bruckner T, Kretschmer V, Schmidt S, Leidinger W, Weiler-Lorentz A (2008) Leukodepletion of autologous whole blood has no impact on perioperative infection rate and length of hospital stay. Transfusion (Paris) 48:2133–2142. https://doi.org/10.1111/j.1537-2995.2008.01804.x
Kim DG, Ha JK, Hwang CJ, Lee DH, Lee CS, Cho JH (2016) Is one-stage posterior corpectomy more favorable compared to decompression with fusion to control thoracic cord compression by metastasis? Clin Spine Surg. https://doi.org/10.1097/BSD.0000000000000267
Suzuki T, Morgan SJ, Smith WR, Stahel PF, Gillani SA, Hak DJ (2010) Postoperative surgical site infection following acetabular fracture fixation. Injury 41:396–399. https://doi.org/10.1016/j.injury.2009.11.005
Li Q, Liu P, Wang G, Yang Y, Dong J, Wang Y, Zhou D (2015) Risk factors of surgical site infection after acetabular fracture surgery. Surg Infect 16:577–582. https://doi.org/10.1089/sur.2014.134
Roberts I, Shakur H, Coats T, Hunt B, Balogun E, Barnetson L, Cook L, Kawahara T, Perel P, Prieto-Merino D, Ramos M, Cairns J, Guerriero C (2013) The CRASH-2 trial: a randomised controlled trial and economic evaluation of the effects of tranexamic acid on death, vascular occlusive events and transfusion requirement in bleeding trauma patients. Health Technol Assess 17:1–79. https://doi.org/10.3310/hta17100
Farrow LS, Smith TO, Ashcroft GP, Myint PK (2016) A systematic review of tranexamic acid in hip fracture surgery. Br J Clin Pharmacol 82:1458–1470. https://doi.org/10.1111/bcp.13079
Zufferey PJ, Miquet M, Quenet S, Martin P, Adam P, Albaladejo P, Mismetti P, Molliex S, tranexamic acid in hip-fracture surgery s (2010) Tranexamic acid in hip fracture surgery: a randomized controlled trial. Br J Anaesth 104:23–30. https://doi.org/10.1093/bja/aep314
Abdel MP, Chalmers BP, Taunton MJ, Pagnano MW, Trousdale RT, Sierra RJ, Lee YY, Boettner F, Su EP, Haas SB, Figgie MP, Mayman DJ (2018) Intravenous versus topical tranexamic acid in total knee arthroplasty: both effective in a randomized clinical trial of 640 patients. J Bone Joint Surg Am 100:1023–1029. https://doi.org/10.2106/JBJS.17.00908
Guo P, He Z, Wang Y, Gao F, Sun W, Guo W, Li Z, Cheng L (2018) Efficacy and safety of oral tranexamic acid in total knee arthroplasty: a systematic review and meta-analysis. Medicine 97:e0587. https://doi.org/10.1097/MD.0000000000010587
Wang S, Gao X, An Y (2017) Topical versus intravenous tranexamic acid in total knee arthroplasty: a meta-analysis of randomized controlled trials. Int Orthop 41:739–748. https://doi.org/10.1007/s00264-016-3296-y
White CC, Eichinger JK, Friedman RJ (2018) Minimizing blood loss and transfusions in total knee arthroplasty. J Knee Surg 31:594–599. https://doi.org/10.1055/s-0038-1648223
Geerts WH, Bergqvist D, Pineo GF, Heit JA, Samama CM, Lassen MR, Colwell CW (2008) Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 133:381S–453S. https://doi.org/10.1378/chest.08-0656
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Investigation Performed at Jackson Memorial Hospital, Miami, FL
The University of Miami Institutional Review Board approved this study. This research was previously presented at the American Academy of Orthopaedic Surgeons Annual Meeting, Mar 12–16, 2019.
Conflict of interest
Each author certifies that he or she has no commercial associations (e.g., consultancies, stock ownership, equity interest, patent/licensing arrangements, etc) that might pose a conflict of interest in connection with the submitted article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(XLSX 106 kb)
Rights and permissions
About this article
Cite this article
Cohen-Levy, W.B., Rush, A.J., Goldstein, J.P. et al. Tranexamic acid with a pre-operative suspension of anticoagulation decreases operative time and blood transfusion in the treatment of pelvic and acetabulum fractures. International Orthopaedics (SICOT) 44, 1815–1822 (2020). https://doi.org/10.1007/s00264-020-04595-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-020-04595-w